Abstract

Gulec and coworkers recently disclosed in the Journal that Ankaferd hemostat (ABS)-derived iron is biologically active in terms of genomics and hemostasis. 1 Clin Appl Thromb Hemost had also previously published an important article indicating antithrombin mechanisms of the ABS elucidating prohemostatic but antithrombotic pharmacobiological actions of this unique hemostatic drug. 2 Akar and coworkers previously demonstrated the presence of Fe(III) ions inside ABS (ABS iron ion concentration were higher than 2000 ppm in their investigation). 3 We, therefore, investigated possible interrelationships between ABS-derived iron and the antithrombin effects of ABS. The aim of this letter is to report the results of those investigations.
Venous blood samples were collected in 2 mL 0.109 M trisodium citrate–containing tubes. The samples were immediately centrifuged for 15 minutes at 2500 g to obtain plasma. Thrombin generation was continuously measured in freshly isolated human platelet-poor plasma (PPP; with or without ABS) using the calibrated thrombinogram method. Diluted plasma samples were measured by Thrombin Generation Kits including Thrombin Calibrator, PPP-Reagent 5 pM, and FluCa-kit. One milliliter of distilled water was added to both the Thrombin Calibrator and PPP-reagent 5 pM, separately. Fluca kit comprised Fluo-Buffer, Fluo-Substrate solutions. Fluo buffer was kept in 37°C water bath before the initiation of the study. The Fluo-buffer and Fluo-Substrate solutions were together. Twenty milliliter PPP Reagent 5 pM was added to polypropylene 96-well microtiter plate. Then, citrated pool PPP or diluated pool PPP with ABS was added (80 µL), and the plate was warmed to 37°C for 5 minutes. For each of plasma samples, the fluorescent signal was converted to thrombin concentration by continuous comparison with the signal generated by a thrombin calibrator added to a separate sample of the test plasma (Thrombin Calibrator, Stago, France). The calibrator corrects for inner filter effects and quenching variation among individual plasmas. Each sample and the calibrator were assayed and analyzed via calibrated automatic thrombinogram (CAT) system (Stago, France). 1/8 diluted with water ABS was rediluted again with pooled fresh human plasma in order to study in vitro effects on ABS on several essential hemostatic parameters including prothrombin time (PT), activated partial thromboplastin time (aPTT), fibrinogen, thrombin time (TT), reptilase time, anti-Xa essay, antithrombin III activity, thrombin generation essay (CAT Assay, Stago, France). All of the hemostatic tests were carried out at 37°C, and the dilutions were left for no longer than 15 minutes before carrying out the tests. Each test was repeated twice and the mean was calculated. Pooled plasma was used as controls. These tests were performed using STA-R Evolution Haemostasis Device (Stago, France) according to manufacturer’s instructions. Iron sucrose (Venofer) was diluted to 1 mg/mL (2.79 mM) with water and rediluted again with 1/3 pooled PPP as a sample. Iron-chelating deferoxamine (Desferal, 500 mg/mL, 761.27 mM) was diluted to 7.61 mM with water and rediluted with ABS ½.
There was an increase in PT, aPTT, and TT in Pool PPP + ABS (1/8) group than the control group. Essential hemostatic parameters in Pool PPP (control) and Pool PPP + ABS groups demonstrated antithrombin effects of ABS (Table 1). Iron-chelating deferoxamine reversed ABS-induced antithrombin activity (Table 2).
Essential Hemostatic Parameters in Pool PPP (Control) and Pool PPP + ABS Groups, Demonstrating Antithrombin Effects of Ankaferd Hemostat.
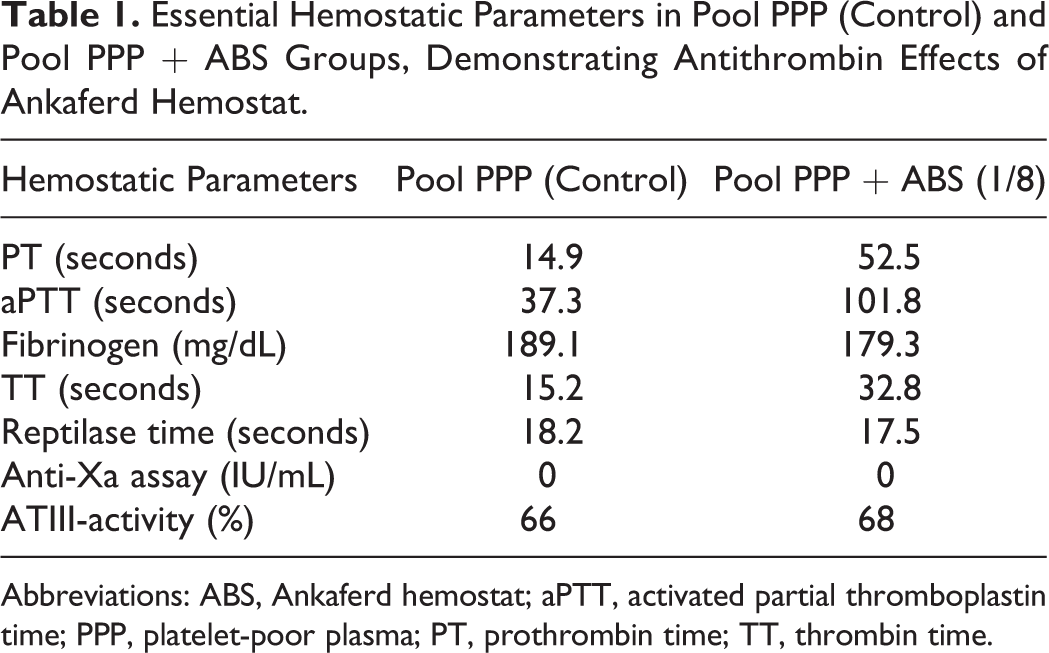
Abbreviations: ABS, Ankaferd hemostat; aPTT, activated partial thromboplastin time; PPP, platelet-poor plasma; PT, prothrombin time; TT, thrombin time.
The Impact of Iron-Chelating Deferoxamine on the ABS- Induced Thrombin Time, PT, aPTT Alterations.

Abbreviations: ABS, Ankaferd hemostat; aPTT, activated partial thromboplastin time; PPP, platelet-poor plasma; PT, prothrombin time.
The thrombin generation curve was assessed by examining the time of the lag phase, the thrombin peak, the time to reach the peak, the area under the curve (endogenous thrombin potential, ETP, and time to tail). There was a decrease in the thrombin peak and ETP in ABS group than in the control group. Moreover, there was a prolongation of the time of the lag phase and time to reach the peak in ABS group than control group.
Azizova et al had previously shown the kinetics of thrombin inhibition by iron ions. 4 They studied the kinetics and concentration characteristics for recovery of thrombin activity by deferoxamine that were evaluated at the various periods of thrombin incubation with iron ions. The effect of reversibility was shown to depend on the time of thrombin preincubation with iron in their study. Incomplete recovery of the thrombin activity after increasing the time of incubation with iron was ascribed to the oxidative modification of thrombin. 4 Our present study disclosed that antithrombin effect of ABS is linked to the high iron content of the drug. Ankaferd hemostat has already been tested in numerous controlled randomized clinical trials, such as in the fields of Cardiology and Cardiovascular Surgery, 5 –7 demonstrating its antihemorrhagic effects devoid of thrombosis. The results of our present investigation showed up the underlying ABS mechanism that the absence of clinical thrombosis is because of the antithrombin effects of the drug that could be ascribed to its high iron content.
