Abstract
The frequency of severe antithrombin deficiency (SAD) was examined in the hematopoietic disorder-, infectious-, and basic-types of the disseminated intravascular coagulation (DIC). A posthoc analysis of 3008 DIC patients (infectious-type, 1794; hematological disorder-type, 813; and basic-type, 401) from post-marketing surveillance data of thrombomodulin alfa was performed. The clinical features of patients and outcomes were compared between patients with and without SAD, using an antithrombin cutoff value of 50%. Patients with SAD accounted for 40.4% of infectious-type DIC, 8.0% of hematopoietic disorder-type DIC, and 26.7% of basic-type DIC. There was no significant difference in thrombin–antithrombin complex levels between patients with and without SAD. The decreased fibrinogen level and differences in clinical features were significantly greater but the increases in fibrinolytic markers were significantly lower in patients with SAD than in those without. The 28-day survival rate was significantly lower in patients with SAD than in those without. Severe antithrombin deficiency was observed in all types of DIC, including hematopoietic disorders. Both hypofibrinolysis and hypercoagulability in patients with SAD may cause multiple organ failure and poor outcomes.
Keywords
Introduction
Disseminated intravascular coagulation (DIC) is a potentially fatal disease due to severe bleeding or multiple organ failure (MOF), which is associated with sepsis, hematological malignancy, solid cancer, trauma, inflammation, and aneurysms, showing various symptoms. 1 –3 The definition of DIC is systemic activation of the hemostatic system as shown by increased thrombin or fibrin generation and consumption of hemostatic factors, such as hypofibrinogenemia or thrombocytopenia, resulting in life-threatening bleeding. 4,5 A decreased level of antithrombin (AT), which inhibits thrombin and activated coagulation factor X, is frequently observed in infectious-type DIC patients, suggesting it to be a risk factor for poor outcomes in infectious-type DIC. 6,7 However, in the hematopoietic disorder-type of DIC in the diagnostic criteria for DIC established by the Japanese Society of Thrombosis and Hemostasis (JSTH), 8 the AT level is not usually measured for diagnosing DIC.
Several randomized, clinical trials of AT, activated protein C, tissue factor pathway inhibitor, and thrombomodulin alpha (TM-α) for severe sepsis did not show improvement of the outcomes of patients with severe sepsis. 9 –12 A phase 3 study 13 and several retrospective studies 14 –16 including post-marketing surveillance (PMS) have reported the efficacy and safety of TM-α for DIC patients with infectious and hematological diseases. In Japan, DIC patients with AT <70% are usually treated with AT or TM-α. 17
Excessive thrombin generation causes the inhibition of fibrinolysis via thrombin-activatable fibrinolytic inhibitor (TAFI) activation, increasing fibrin formation and microvascular thrombosis, leading to MOF as one of the pathogenetic mechanisms of DIC. 18,19 However, there is little clinical evidence for the relationship between excessive thrombin generation and suppression of fibrinolysis related to the pathogenesis of DIC, and thrombin generation is suppressed in severe AT deficiency (SAD). 20,21
In the present study, the frequency of SAD (AT level ≤ 50%) was examined in infectious-, hematopoietic disorder-, and basic-types of DIC of the DIC diagnostic criteria of the JSTH, 8 and the characteristics of SAD in the 3 types of DIC were analyzed. Furthermore, the relationship between SAD and suppression of fibrinolysis in DIC patients was examined.
Materials and Methods
Study Design and Data Collection
The original PMS study was an open-label, multicenter, noninterventional, prospective, observational cohort study of patients with DIC who received recombinant soluble TM (TM-α; 2008-2010). 14 The PMS for TM-α was conducted in accordance with the Japanese Society on Thrombosis and Hemostasis Post-Marketing Surveillance Committee for Recomodulin Injection and the guidelines for Good Post-Marketing Surveillance Practices, as required by the Japanese Ministry of Health, Labour, and Welfare (JMHLW). Existing data without personally identifiable information were used throughout the study. The original PMS study was therefore exempted from local institutional review and formal approval as well as the requirement for informed consent. All patients who received TM-α were consecutively registered on initiation of the treatment by documenting the patient demographics using a central registration system. The patients were prospectively observed until 28 days after administration of TM-α. The standard dose of TM-α was 380 U/kg, and the adjusted dose of 130 U/kg was used for patients with renal dysfunction. All patients were treated according to the attending physician’s decisions, and there was no limitation on the concomitant use of other anticoagulants or medicine for the treatment of underlying diseases and complications.
A posthoc analysis of PMS data of TM-α was conducted. Of the 4342 patients, the 4056 patients who underwent first TM-α administration were divided into 3 groups (hematopoietic disorder-type, infectious-type, and basic-type) on the basis of the underlying disease in accordance with the JSTH DIC definition. 4 In each group, the clinical features of DIC and the outcome by AT level at baseline were investigated.
Thrombin generation and activity are physiologically regulated by AT and TM. Because TM is a membrane-bound protein on endothelial cells and AT is a soluble factor, TM cannot be monitored, but AT can be monitored by blood samples. Antithrombin activity was measured as anti-Xa or antithrombin activity using chromogenic substrate in each institute. Disseminated intravascular coagulation patients were defined as having normal and low AT levels, reflecting the conditions where thrombin generation can and cannot be efficiently inhibited, respectively. Cutoff values of the AT level were set at 50% (SAD) or 70%.
Evaluation
The posthoc analyses included 3008 DIC patients (infectious-type, 1794; hematological disorder-type, 813; and basic-type, 401) from the PMS of TM-α. The primary objective of the present study was to clarify the clinical features of DIC patients with and without SAD. The secondary objective was to investigate the outcomes, including DIC resolution rates, 28-day survival rates, clinical course of bleeding symptoms, and the rates of adverse drug reactions and bleeding-related adverse drug reactions, between the DIC patients with and without SAD. The degree of coagulopathy was evaluated by calculating the DIC score according to the DIC diagnostic criteria of the JMHLW, 22 of the Japanese Association for Acute Medicine (JAAM) 23 for infectious-type, and of the International Society of Thrombosis Haemostasis (ISTH) 4 for hematological disorder-, infectious-, and basic-types. After treatment with TM-α, resolution of DIC was defined as a score ≤3 points using the diagnostic criteria of the JAAM, of ≤2 points using those of the JMHLW for DIC with hematological diseases, ≤5 points using those of the JMHW for DIC with infectious- and basic-types, and of ≤4 points using those of the ISTH for all types. Survival at 28 days from the beginning of TM-α treatment was calculated.
In DIC with infectious- or basic-type, the severity of organ failure was assessed by the sequential organ failure assessment (SOFA) score.
24
Laboratory tests such as white blood cell counts, platelet counts, hemoglobin, albumin, lactate dehydrogenase, total bilirubin (T-Bil), creatine, choline esterase (ChE), and C-reactive protein (CRP) and hemostatic tests such as prothrombin time (PT)-international ratio (INR), activated partial thromboplastin time (APTT), fibrinogen, fibrinogen and fibrin degradation products (FDP),
The rates of adverse drug reactions and bleeding-related adverse drug reactions were evaluated from the start of TM-α administration to day 28 after the end of TM-α administration. Safety data were coded with the preferred terms from the Medical Dictionary for Regulatory Activities.
Statistical Analysis
In the descriptive analysis of baseline characteristics, numerical data are expressed as medians (Q1, Q3; interquartile range). Statistical analysis was performed to compare values using the χ2 test and the Wilcoxon signed-rank test. A value of P < .05 was considered significant. All analyses were performed using SAS version 9.4 (SAS Institute, Co Ltd) by EPS Corporation according to the statistical analysis plan.
Results
Patients’ Characteristics
A flow chart of patient selection for the analyses is shown in Figure 1. Plasma AT levels were measured in these patients. Patients with SAD for 40.4% of infectious-type DIC, 8.0% of hematopoietic disorder-type DIC, and 26.7% of basic-type DIC (Table 1).
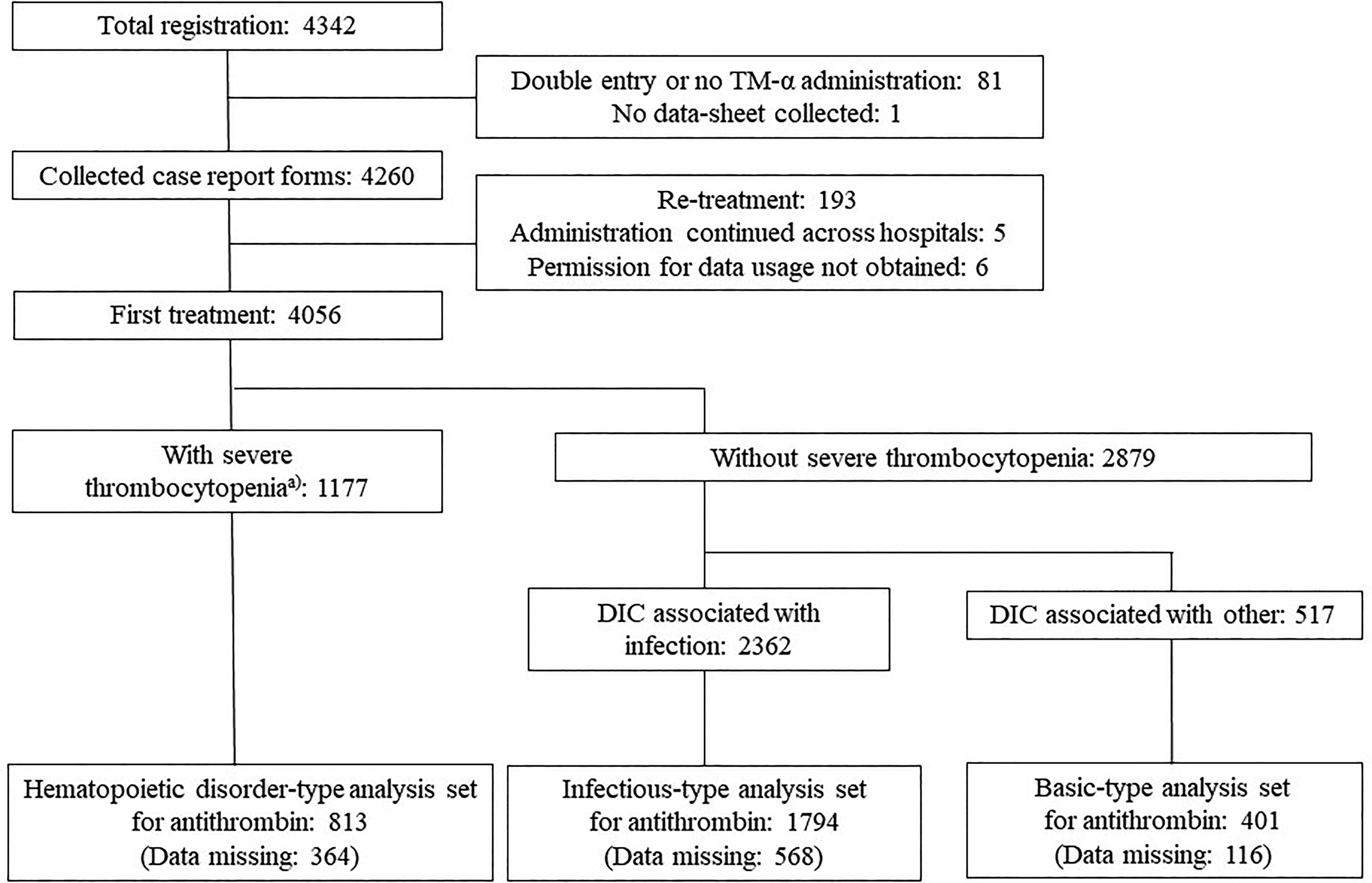
Patient disposition. a The leukemia group according to JMHLW DIC criteria (the number of megakaryocytes markedly decreases due to hematologic malignancy, aplastic anemia, or antitumor agent administration). DIC indicates disseminated intravascular coagulation; JMHLW, Japanese Ministry of Health, Labour, and Welfare.
Demographic Characteristics of Patients With Each Type of DIC With and Without Severe Antithrombin Deficiency (≤50%).a

Abbreviations: AT, antithrombin; DIC, disseminated intravascular coagulation; LMWH, low-molecular-weight heparin.
a Data are given as n (%) or medians (IQR).
Age was significantly higher in infectious- and hematopoietic disorder-types of DIC with SAD than in those without. Although there was no significant difference in body weight between infectious- and hematopoietic disorder-type DICs with and without SAD, body weight was significantly lower in basic-type DIC with SAD than in those without. Before registration, the rate of administration of AT concentrate was significantly lower in infectious-type DIC patients with SAD than in those without (Table 1). There were no significant differences in bleeding and organ failure symptoms between patients with and without SAD, except for those without bleeding symptoms of hematopoietic disorder-type DIC. Patients with an AT level ≤ 70% accounted for 75.9% of infectious-type DIC, 29.4% of hematopoietic disorder-type DIC, and 56.6% of basic-type DIC (Supplemental Table 1). The differences in demographic characteristics between patients with and without a AT level ≤ 70% tended to be smaller compared with the differences between the patients with and without SAD (Table 1 and Supplemental Table 1).
Clinical Features at Baseline
The median SOFA and DIC (JMHLW, ISTH, and JAAM) scores were significantly higher in the infectious-type DIC patients with SAD than in those without, and there was no significant difference between the patients with and without SAD in hematopoietic disorder-type DIC. The median SOFA and DIC (ISTH and JAAM) scores were significantly higher in basic-type DIC patients with SAD than in those without (Table 2).
Sequential organ failure assessment and DIC Scores in Patients With Each Type of DIC With and Without Severe Antithrombin Deficiency (≤50%).a

aData are given as medians (IQR).
Abbreviations: DIC, disseminated intravascular coagulation; ISTH, International Society of Thrombosis Haemostasis; JAAM, Japanese Association for Acute Medicine; JMHLW, Japanese Ministry of Health, Labor and Welfare; SOFA, sequential organ failure assessment.
In all types of DIC, the decrease of albumin and ChE levels and the increase in total bilirubin and creatinine levels were more significant in patients with SAD than in those without (Table 3). The decrease of platelet counts in infectious-type DIC and increase in CRP levels in hematopoietic disorder-type DIC were more significant in patients with SAD than in those without.
Laboratory Characteristics of Patients With Each Type of DIC With and Without Severe Antithrombin Deficiency (≤ 50%).a

a Data are given as medians (IQR).
Abbreviations: AT, antithrombin; ChE, choline esterase; CRP, C-reactive protein; DIC, disseminated intravascular coagulation; LDH, lactate dehydrogenase; T-Bil, total bilirubin; WBC, white blood cell.
Of the hemostatic characteristics, PT-INR and APTT were significantly prolonged, and fibrinogen, FDP, AT, PC, and PIC levels were significantly lower in all types of DIC patients with SAD than in those without (Table 4). There was no significant difference in TAT levels between patients with and without SAD (Table 4).
Hemostatic Characteristics of Patients With Each Type of DIC With and Without Severe Antithrombin Deficiency (≤ 50%).

a Data are given as medians (IQR).
Abbreviations: APTT, activated partial thromboplastin time; AT, antithrombin; FDP, fibrin and fibrinogen degradation products; DIC, disseminated intravascular coagulation; PIC, plasmin-plasmin inhibitor complex; PT-INR, prothrombin time-international normalized ratio; TAT, thrombin-antithrombin complex.
In the treatment of DIC, there were no significant differences in dose and period of TM-α administration between patients with and without SAD (Table 5). The concomitant frequencies of AT concentrate and fresh frozen plasma administration were significantly higher in patients with SAD than in those without. In addition, gabexate mesilate, nafamostat mesilate, unfractionated heparin, low-molecular-weight heparin, danaparoid sodium, and platelet concentrates, and red blood cells were administered in those patients, as shown in Table 5. The differences in SOFA and DIC scores, laboratory characteristics, hemostatic characteristics, and treatment of DIC between patients with and without a AT level ≤ 70% tended to be smaller compared with the difference between the patients with and without SAD (Supplemental Tables 2-5).
Treatments for DIC in Patients With Each Type of DIC With and Without Severe Antithrombin Deficiency (≤ 50%).a
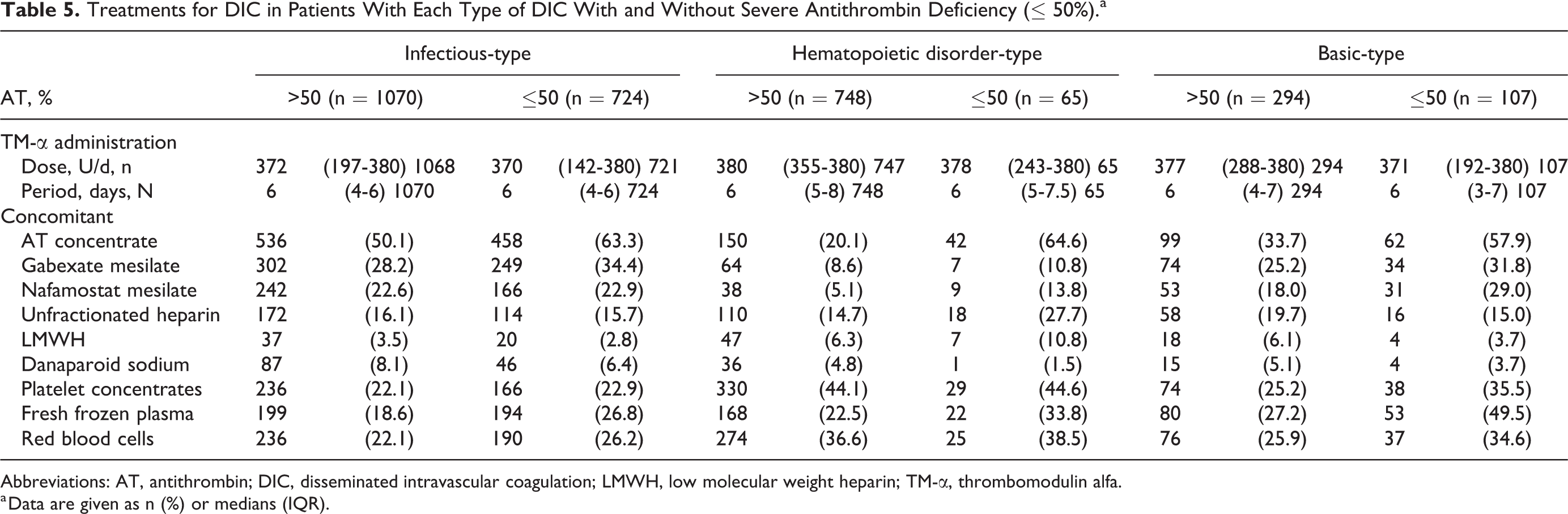
Abbreviations: AT, antithrombin; DIC, disseminated intravascular coagulation; LMWH, low molecular weight heparin; TM-α, thrombomodulin alfa.
a Data are given as n (%) or medians (IQR).
Outcomes
The 28-day survival rate was lower in all types of DIC patients with SAD than in those without (Table 6). The resolution rate by JMHLW, ISTH, or JAAM DIC diagnostic criteria was lower in infectious- and hematopoietic disorder-type DIC patients with SAD than in those without, but there was no difference in the resolution rate by JMHLW, ISTH, or JAAM DIC diagnostic criteria in basic-type DIC patients between those with and without SAD. There were no differences in the clinical courses of bleeding symptoms between all types of DIC patients with and without SAD (≤ 50%; Table 6). The differences in outcomes between patients with and without a AT level ≤ 70% tended to be smaller than between patients with and without SAD (Supplemental Table 6). The rates of adverse drug reactions (ADRs), serious ADRs, bleeding ADRs, and serious bleeding ADRs were similar (from 3.1%-8.6%) between all types of DIC patients with and without SAD or a low AT level ≤70%, and these did not increase in patients with SAD (Table 7 and Supplemental Table 7).
Outcomes of Patients With Each Type of DIC With and Without Severe Antithrombin Deficiency (≤50%).a

a Data are given as n (%).
Abbreviations: AT, antithrombin; DIC, disseminated intravascular coagulation; ISTH, International Society of Thrombosis Haemostasis; JAAM, Japanese Association for Acute Medicine; JMHLW, Japanese Ministry of Health, Labor and Welfare.
Adverse Drug Reactions (ADRs) of Patients With Each Type of DIC With and Without Severe Antithrombin Deficiency (≤50%).

Abbreviation: DIC, disseminated intravascular coagulation.
a Data are given as n (%).
Discussion
An acquired AT deficiency is frequently observed in DIC patients with sepsis. 25,26 Since the mechanism for the onset of DIC is different in hematopoietic diseases from sepsis, 27,28 many researchers and physicians have considered that acquired AT deficiency is rare in hematopoietic disorder-type DIC. Indeed, the SOFA and DIC scores were markedly higher in infectious-type DIC with SAD, but DIC scores were not high in hematopoietic disorder-type DIC with SAD. However, the present findings showed that hematopoietic disorder-type DIC patients with SAD or a AT level ≤70% accounted for 8.0% or 29.4%, respectively, in the analysis of PMS data. Since CRP levels are higher in hematopoietic disorder-type DIC patients with SAD than in those without, these patients often develop DIC induced by infection.
The reduction of AT levels may be caused by decreased production due to liver dysfunction, consumption of AT, or leak to the third space by increased permeability in vascular endothelial cells. 28 Although a high DIC score indicates the existence of consumption coagulopathy in DIC patients with SAD, decreased albumin and ChE levels and increased T-Bil and creatinine levels suggest that liver dysfunction, plasma leakage, or renal dysfunction might play some role in AT deficiency in patients with DIC.
Although plasma fibrinogen levels are increased in infectious-type DIC, 25 the fibrinogen levels were significantly lower in all types of DIC patients with SAD than in those without. The present findings show that DIC scores were higher in DIC patients with SAD than in those without, suggesting that the progress of DIC may increase fibrinolysis in hematopoietic disorder-type DIC with AT deficiency. However, in the present study, the FDP and PIC levels were significantly lower in patients with SAD than in those without, indicating that fibrinolysis is suppressed in DIC patients with SAD. Indeed, the frequency of bleeding symptoms was also not high in hematopoietic disorder-type DIC patients with SAD.
Plasma levels of TAT, which is related to thrombin generation and AT concentration, are elevated in all types of DIC, but plasma TAT levels are slightly lower in DIC patients with SAD than in those without, suggesting that DIC patients with SAD might be unable to inhibit the excess generated thrombin. The excess generated thrombin and TM may activate TAFI, which plays an important role at the interface between thrombin generation and the suppression of fibrinolysis. 29 The present findings indicate that hypofibrinolysis exists in DIC patients with SAD. Decreased or increased levels of TAFI have been reported in patients with DIC or MOF. 30 –32 Plasminogen activator inhibitor-1 (PAI-1) is also reported in patients with sepsis to cause hypofibrinolysis. 33,34 Elevated TAFI and PAI levels have also been reported in patients with sepsis with poor outcomes. 32,35 Thrombin-activatable fibrinolytic inhibitor and PAI-1 were not measured in the present study because data from PMS in daily clinical practice did not include the special coagulation tests used for clinical research. Hemostatic data showed a consumptive coagulopathy with low anticoagulant and low fibrinolytic activities in all types of DIC with SAD, indicating a thrombogenic state instead of a bleeding tendency. Therefore, in the treatment of DIC, the frequencies of AT concentrate and fresh frozen plasma administration were markedly higher in DIC patients with SAD in all types of DIC in this PMS.
The 28-day survival rate was very low in all types of DIC with SAD, suggesting that decreased AT may reflect poor outcomes in DIC patients. It has also been reported that 28-day survival was markedly lower in septic patients with AT ≤50%. 26 In infectious-type DIC patients with SAD, clinical features such as SOFA and DIC scores reflected severe disease, and outcomes such as the 28-day survival rate and DIC resolution rate were poor. The SOFA and DIC scores and AT levels have been reported to be useful as prognostic factors. 3 In infectious- and hematopoietic disorder-types of DIC, both the 28-day survival and resolution rates from DIC were significantly lower in patients with SAD than in those without. However, in basic-type DIC patients, 28-day survival was significantly lower in patients with SAD than in those without, but there were no significant differences in the resolution rate from DIC between the 2 groups. These finding suggest that DIC might cause poor outcomes in infectious- and hematopoietic disorder-types of DIC, but not in the basic-type of DIC. Although there are recently much progress in the therapy of sepsis and intensive care unit–based care, 36 the issues of diagnosis and anticoagulant therapy for DIC have still been remained.
In conclusion, all types of DIC, including hematopoietic disorder-type DIC, are associated with SAD. Suppression of fibrinolysis and poor outcomes are observed in all types of DIC with SAD. Therefore, the monitoring of AT levels is important for the management of the underlying diseases of DIC.
Supplemental Material
Supplemental Material, Wada_H_supplTable_rev - Severe Antithrombin Deficiency May be Associated With a High Risk of Pathological Progression of DIC With Suppressed Fibrinolysis
Supplemental Material, Wada_H_supplTable_rev for Severe Antithrombin Deficiency May be Associated With a High Risk of Pathological Progression of DIC With Suppressed Fibrinolysis by Hideo Wada, Goichi Honda, Noriaki Kawano, Toshimasa Uchiyama, Kazuo Kawasugi, Seiji Madoiwa, Naoki Takezako, Kei Suzuki, Yoshinobu Seki, Takayuki Ikezoe, Toshiaki Iba and Kohji Okamoto in Clinical and Applied Thrombosis/Hemostasis
Footnotes
Acknowledgments
The authors would like to thank all investigators who participated in this PMS study.
Authors’ Note
HW planned the study design, interpreted the data, and wrote the manuscript. HW provided oversight and supervised the current study. NK, TU, KK, SM, NT, KS, YS, TI, TI, and KO advised on study design and interpretation of the data and reviewed the manuscript. GH planned the study design. The data that support the findings of this study are available from Asahi Kasei Pharma Corporation, but restrictions apply to the availability of these data, which were used under license for the current study, and so are not publicly available. Approval for this study was obtained from the appropriate ethics committees and institutional review boards of Mie Prefectural General Medical Center.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article. GH is an employee of Asahi Kasei Pharma Corporation.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Asahi Kasei Pharma Corporation, which funded the analysis and the English editing of the manuscript. HW received grants and personal fees from Asahi Kasei Pharma Corporation and Japan Blood Products Organization outside the present work. TU, SM, NT, KS, YS, and KO received personal fees from Asahi Kasei Pharma Corporation outside the present work. KK and TI received grants and personal fees from Asahi Kasei Pharma Corporation outside the present work. TI received personal fees from Asahi Kasei Pharma Corporation and received research grants from Japan Blood Products Organization and Japan Immunoresearch Laboratories outside the present work.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
