Abstract
Antithrombin (AT), a glycoprotein, plays a key role in anticoagulation by inhibiting coagulation proteases. It is also the primary mediator of heparin's anticoagulant effect, with heparin binding amplifying AT activity up to 1000-fold. Deficiency in AT contributes to a hypercoagulable state associated with adverse clinical outcomes, including morbidity and mortality. Hereditary AT deficiency (hATD) is a rare autosomal dominant disorder caused by mutations in the gene encoding AT (SERPINC1), resulting in decreased AT levels or activity. As the most thrombogenic inherited thrombophilia, hATD confers a lifetime venous thromboembolism (VTE) risk of up to 85%, with similar annual VTE incidence in both pediatric and adult patients. Management of hATD often includes AT concentrate (ATc) therapy in high-risk clinical settings. Although ATc has a long history of use in adults, pediatric use has been largely off-label and guided by extrapolation from adult data. In August 2025, the U.S. Food and Drug Administration (FDA) approved an expanded indication for antithrombin III concentrate (human) to include pediatric patients with hATD, representing the first product specifically approved for this population. In light of this recent shift in the treatment landscape, this review provides a timely synthesis of the current clinical understanding and evidence-informed strategies to optimize AT therapy in the management of pediatric hATD.
Keywords
Introduction
Hemostasis requires a fine-tuned balance within the coagulation pathway to prevent bleeding complications and mitigate the potential for thrombotic events.1,2 Antithrombin (AT) plays a crucial role in the regulation of the coagulation cascade by promoting anticoagulation through the formation of inhibitory complexes with coagulation proteases. 3 Importantly, a deficiency of circulating AT contributes to a hypercoagulable state, which may result in serious complications leading to morbidity and mortality.4,5 AT deficiency can occur due to a genetic deficiency (hereditary antithrombin deficiency, hATD) or physiological-induced states of decreased production or consumption of AT (acquired AT deficiency). The lifetime risk of thrombosis for most untreated individuals with hATD is approximately 50%–85%, even in the absence of additional thrombotic risk factors.6,7 AT concentrate (ATc) has been successfully used in adults with hATD for the treatment and prevention of thromboembolism for over three decades. 8 Until recently, the prescribing information for ATc therapies did not specify age ranges, and no AT replacement products were explicitly approved for pediatric patients with hATD.8–11 This changed on August 21, 2025, when the U.S. Food and Drug Administration (FDA) approved a label expansion for human AT III concentrate (Thrombate III, Grifols, S.A., Los Angeles, CA) to include adult and pediatric patients with hATD for the treatment and prevention of thromboembolism in general, perioperative and peripartum settings.12,13 Accordingly, this review aims to bridge existing knowledge gaps by summarizing current evidence on the pathophysiology, clinical presentation, and diagnosis of hATD in pediatric patients, and by consolidating available data on ATc use to support clinical decision-making following the recent FDA approval. In doing so, we also highlight key areas for future research and real-world evidence generation.
Antithrombin Physiology
Synthesized in the liver, AT is a glycoprotein that functions as a key endogenous inhibitor of blood coagulation proteases, including thrombin (factor IIa) and activated FX (FXa).14–16 By inactivating these enzymes, AT plays a crucial role in preventing excessive clot formation. Clinically, the anticoagulant activity of AT is frequently leveraged with the use of heparins, which enhance AT's inhibitory function by up to 1000-fold through conformational changes that accelerate protease binding.15,17,18 This heparin-AT interaction underlies the primary mechanism of action of both unfractionated heparin (UFH) and low-molecular-weight heparin (LMWH). In addition to its anticoagulation properties, AT also exerts anti-inflammatory functions through coagulation-dependent and independent pathways. These include the suppression of pro-inflammatory cytokine production and the inhibition of platelets and neutrophil adhesion to activated endothelial cells. 19 Additional details on the structure, regulation, and broader physiologic roles of AT have been described elsewhere. 20
Physiological Antithrombin Levels During Human Growth and Development
Hemostasis is a dynamic, tightly regulated, age-dependent process. 21 AT levels approach adult levels by ∼6 months of age, a delay that may be physiologically adaptive given AT's known antiangiogenic effects and the high angiogenic activity during early development.21–23 In healthy adults, the normal range for AT levels is between 80% and 120%, with 100% corresponding to 1 unit of AT per mL of pooled human reference plasma. 16 While these reference intervals are widely used for adult populations, age-specific reference ranges are necessary and are often set by the individual laboratories. 18 For instance, our institution utilizes age-appropriate pediatric and adult reference values for AT, as summarized in Supplementary Table 1. 24 Additional pediatric reference ranges have been published across other laboratories, and vary depending on the analyzer, assay reagents, and measurement methodologies used (Supplementary Table 2).23,25–35 Collectively these studies indicate that AT levels at birth are approximately 50% of those observed in healthy adults. AT levels gradually increase, reaching adult values by approximately 6 months of age and remaining stable throughout adulthood. Hence, it is important to consider potential age-dependent variations in hemostatic pathways, as differences in AT levels between adults and infants are typically physiological and not necessarily indicative of pathology (eg, acquired or inherited deficiency). 23
Hereditary Antithrombin Deficiency
In 1965, Olav Egeberg identified hATD as the first major thrombophilia by reporting its association with venous thrombosis. 36 Specifically, hATD is a rare autosomal dominant disease caused by mutations in the gene encoding AT, SERPINC1.37,38 The gene is located on chromosome 1q23-25.1 and comprises seven exons and six introns.4,16 The heparin binding site of AT is encoded by exons 2 and 3, and the carboxy-terminal reactive site is encoded by exon 7. 39 Notably, even seemingly minor nucleotide substitutions in the SERPINC1 gene can lead to substantial structural alterations in the AT protein, leading to loss of function effects and an impaired inhibition of coagulation proteases. 40 Currently, there are 546 SERPINC1 gene mutations documented in the Human Gene Mutation Database (HGMD).41,42 The reported prevalence of hATD among European and North American cohorts ranges from 1 in 500 to 1 in 5000 individuals.43,44 Diagnosis is typically based on persistently low functional AT activity levels (<70% of age-appropriate normal values) and can be confirmed by genetic testing.45,46
The key clinical manifestation of hATD is venous thromboembolism (VTE), with a lifetime risk estimated between 50% and 85%. 6 In this regard, VTE is the primary thrombotic concern in hATD, with the incidence of arterial thrombosis being rare. 16 Approximately 60% of VTE in patients with hATD occur spontaneously, and 40% occur in the presence of a known risk factor.47,48 A previous study showed that the risk of VTE linearly increases with decreasing plasma levels of AT, with lower levels associated with higher risks. For each 20 IU/dL reduction in plasma AT level, the VTE risk increases by 40%, with higher risk estimates observed for unprovoked VTE compared to VTE secondary to transient risk factors. 49
The type of hATD also impacts the risk of VTE. 16 Two types of hATD exist, differentiated by circulating antigen levels and functional AT activity. Type I is characterized by decreased synthesis of biologically normal AT, resulting in lower circulating AT (quantitative deficiency); consequently, measured AT activity is proportionally reduced due to decreased protein concentration, rather than impaired molecular function. Type I can occur from various mutation types (ie, nonsense, small deletions, insertions), resulting in diminished AT levels through mRNA instability, impaired protein folding, or intracellular retention or degradation. 4 While Type I is more prevalent among symptomatic patients who develop thrombosis, accounting for as many as 80% of all symptomatic cases, 16 Type II is more prevalent in the general population and is characterized by normal antigen levels accompanied by low AT activity due to dysfunctional protein (qualitative deficiency).16,50,51 Type II hATD exclusively occurs due to missense mutations and is divided into three subtypes based on the associated functional defect. 52 Type IIa occurs when mutations affect the binding of the substrate (eg, procoagulant proteins) to the reactive site within AT; this can result in mild to very severe manifestations of hATD.4,53 Type IIb describes mutations that affect the heparin binding site, which negatively impacts the ability of AT to interact with heparin. Type IIb mutations carry a low risk of thrombosis but can be problematic in clinical settings requiring heparin use. 4 Type IIc refers to mutations that result in pleiotropic effects that affect both the reactive site of AT as well as heparin binding affinity. Similar to Type IIa, Type IIc can result in mild to very severe manifestations of hATD. 4
Genotype-phenotype studies indicate that thrombotic risk varies significantly by SERPINC1 variant class (eg, null vs missense) and AT subtype, supporting more nuanced VTE risk stratification.38,50,54 Pediatric data remain limited, however available evidence suggests severe thrombosis in children with high-risk genotypes (eg, homozygous AT Budapest III [p.Leu131Phe]) and in those with null variants.52,55 While molecular identification shows promise for VTE prognostication, its clinical application remains complex; nevertheless, its utility is likely to improve as genomic sequencing and personalized medicine become more accessible and integrated into routine care. 20 Further details regarding genotype-phenotype correlations in AT deficiency have been described elsewhere.4,16,56,57
hATD Manifests Similarly in Pediatric Patients and Adults
Studies indicate that children with hATD exhibit a 300-fold greater risk of thrombosis compared to healthy pediatric populations (0.41%/year vs 0.0014%/year, respectively) and a 7.1-fold increased risk of ischemic stroke or cerebral sinovenous thrombosis (CSVT; OR 7.06, 95% CI, 2.44 to 22.42).53,58 However, prior understanding of hATD suggested that the condition primarily manifests in adulthood, with a lower risk of VTE observed in children.59–62 For instance, the American Thrombosis & Hemostasis Network's Hereditary Antithrombin Deficiency Registry (ATHN12), launched in 2020 to gather data on individuals of all ages with hATD across the U.S., reported that only 28% of patients with hATD treated at healthcare centers were under 18 years old. 63 Although a lower proportion of pediatric patients may be treated for hATD relative to adults, these findings do not account for the cumulative lifetime risk of thrombotic events.
Findings from multiple observational studies reveal that the annual incidence of VTE is comparable between adults (range: 0.08%–2.29%) and children (range: 0.43-3.80%) with hATD (Table 1).52,53,55,58,62,64,65,69,70,72,73 A recent prospective, long-term follow-up study by Tormene et al revealed that the risk of thrombosis in otherwise healthy children with inherited thrombophilia, including AT deficiency, was similar to that reported in adults under comparable circumstances. 70 Another study on pediatric patients with hATD, conducted by the International Society on Thrombosis and Haemostasis (ISTH), also found no significant differences in rates of VTE between age-based subgroups of infants (<1 year of age) and adolescents (≥13 years of age). 52 Collectively, these studies indicate that patients with hATD may manifest symptomatic events across the lifespan, conferring a similar risk of thrombotic events in both adult and pediatric populations.
Thrombotic Risk in Adult and Pediatric Patients with hATD.
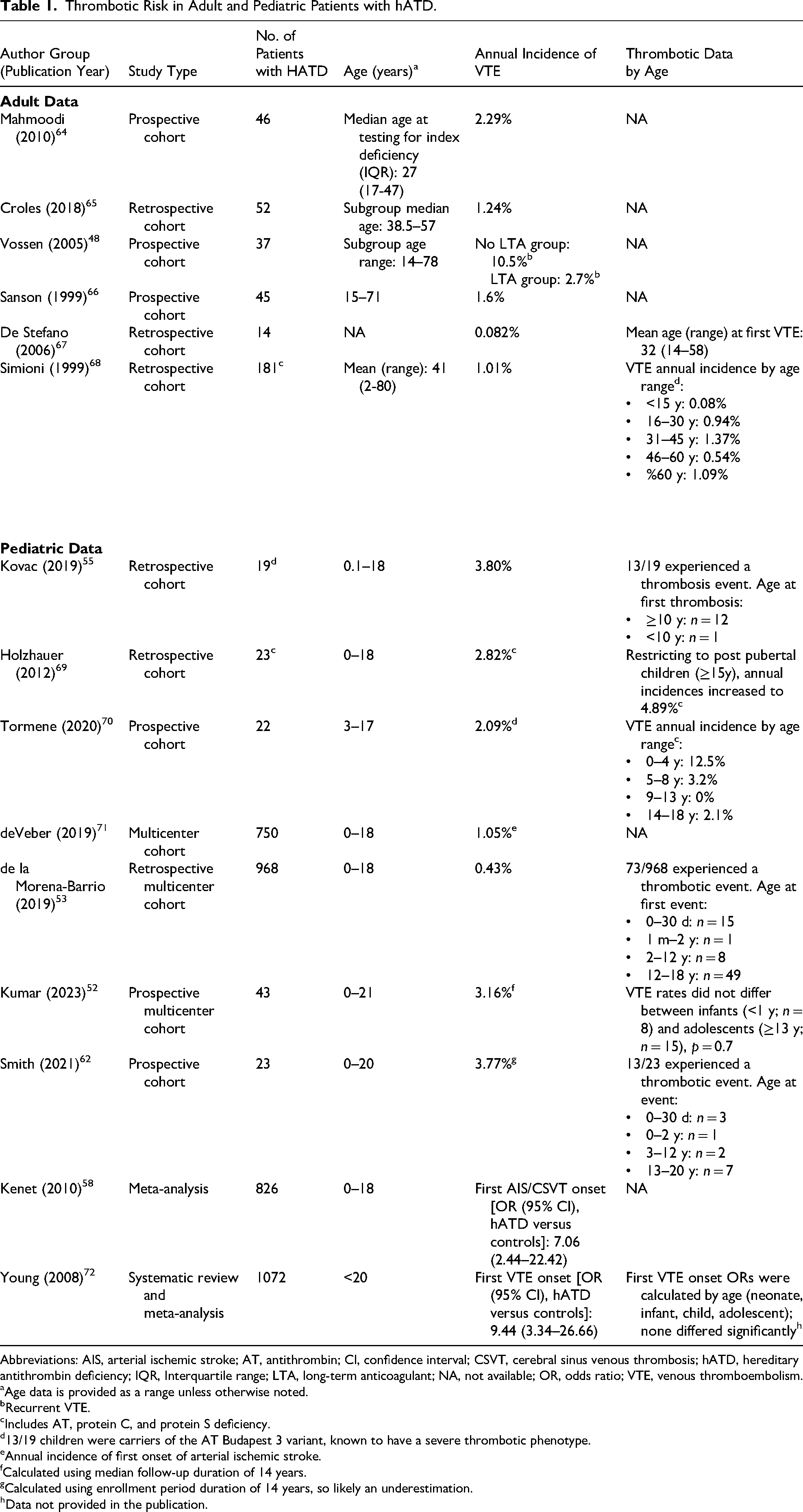
Abbreviations: AIS, arterial ischemic stroke; AT, antithrombin; CI, confidence interval; CSVT, cerebral sinus venous thrombosis; hATD, hereditary antithrombin deficiency; IQR, Interquartile range; LTA, long-term anticoagulant; NA, not available; OR, odds ratio; VTE, venous thromboembolism.
Age data is provided as a range unless otherwise noted.
Recurrent VTE.
Includes AT, protein C, and protein S deficiency.
13/19 children were carriers of the AT Budapest 3 variant, known to have a severe thrombotic phenotype.
Annual incidence of first onset of arterial ischemic stroke.
Calculated using median follow-up duration of 14 years.
Calculated using enrollment period duration of 14 years, so likely an underestimation.
Data not provided in the publication.
Testing and Diagnosis of Antithrombin Deficiency
Given the inherited nature of hATD and the high risk of recurrent thrombotic events, a comprehensive evaluation of personal and family history is essential in patients presenting with thrombosis.20,74 Upon clinical suspicion, diagnosis is typically made via a functional heparin cofactor activity assay, which reliably detects most clinically significant cases when performed under correct circumstances.75,76 However, functional assays may not be sensitive to certain mutations (eg, those causing type IIb or IIc deficiency) and these cases require detection through genetic testing.15,56,77 Functional tests rely on a chromogenic detection system to measure the capacity of AT in the patient's plasma sample to inhibit thrombin formation in the presence of heparin with excess thrombin or FXa. 15 Results are reported in IU/mL of AT or as percent of the AT activity level in a healthy normal adult. The thresholds for AT deficiency vary by assay and institutional guidance; however published literature is generally aligned that levels <80% (normal range) are suggestive of deficiency, with most AT-deficient patients being diagnosed with levels <70% and levels 70%–80% indicating suspected deficiency (Figure 1).20,46

An algorithm for the diagnosis of hereditary antithrombin deficiency. AT, antithrombin; DOAC, direct oral anticoagulant; HBS, heparin binding site; PE, pleiotropic effect; RS, reactive site; VTE, venous thromboembolism. *AT deficiency cutoff applies to individuals ≥6 months of age. aIf resources are available, genetic testing may be considered earlier in the diagnostic pathway for cases of suspected or probable hereditary antithrombin deficiency. bAcquired AT deficiency such as recent thrombosis, disseminated intravascular coagulation, surgery, extracorporeal membrane oxygenation, pregnancy, proteinuria, estrogen therapy, or L-asparaginase therapy. cConsider measuring AT antigen, especially when protein C and protein S levels are normal. dA similar reduction in either protein C or protein S levels typically indicates an acquired etiology. Testing should be repeated after these potential causes have resolved. The figure is inspired by and integrates concepts and data from several studies.4,18,39,46
When testing for AT deficiency in pediatric populations, it is critical to recognize the age-appropriate reference ranges, especially in patients younger than 6 months, and to consider paternal testing as an option if indicated.18,78–80 The timing of thrombophilia testing in relation to thrombotic events is important. To ensure accuracy, it is advisable to avoid testing during acute thrombotic events or while on anticoagulant medications,81,82 as these may impact the findings provided by functional AT tests. If a patient is receiving a direct oral anticoagulant (DOAC) that targets thrombin (eg, dabigatran) or FXa (eg, rivaroxaban, apixaban, edoxaban, betrixaban), AT activity measured by functional assays may appear falsely elevated depending on the type of functional AT assay used (eg, anti-IIa or anti-Xa-based assays).83,84 Therefore, repeat AT activity testing is recommended after DOAC discontinuation for these patients. 18 Acquired etiologies, being more frequent than hATD, should also be ruled out prior to testing for hATD. 18
Upon identification of AT deficiency through functional assays, patients with suspected or confirmed hATD can be further screened for type I and type II deficiencies through interpretation of both functional and antigenic assays (which quantify AT levels), with further confirmation via genetic testing.3,74,85 Type I is characterized by reduced function and antigenic levels, while type II is characterized as reduced function with normal antigenic levels. 85
According to White et al, genetic analysis should be considered as the gold standard method when testing for hATD, which allows for greater determination of risk stratification, family screening, and detection of type 2 defects that often go undetected by functional assays.15,56 A recent study by ISTH found an increased risk of thrombosis in children with null mutations in SERPINC1 compared to those with missense mutations, underscoring the importance of genetic testing. 52 Genetic testing is also strongly recommended to test for AT deficiencies in individuals who experience unprovoked thrombotic events despite having normal AT activity, as functional assays may fail to detect certain mutations resulting in hATD (eg, heparin binding site mutations). 77 However, the decision to pursue genetic testing must balance the potential clinical benefit against the crucial ethical factors of patient consent and the psychological and social implications of the test results. 86
Genetic analysis for hATD is typically performed using Next-Generation Sequencing technology and/or Bi-directional Sanger Sequence Analysis.87,88 Additionally, advanced proteomic techniques have emerged, utilizing liquid chromatography coupled to multiple-reaction-monitoring mass spectrometry (LC-MRM-MS) to enhance molecular characterization of plasma AT proteoforms, which has the potential to add further clinical value to hATD diagnosis and management. 89 Further information on emerging and alternative testing approaches are discussed elsewhere. 57
Current guidelines for thrombophilia testing generally do not recommend routine testing in pediatric populations unless certain criteria are met. The American Society of Hematology (ASH) guideline panel recommends AT deficiency testing in those with minor provoking risk factors who meet one of the following criteria: 1) a family history of VTE and AT deficiency, or 2) no family history with VTE but a first or second-degree relative diagnosed with AT deficiency. 90 Recommendations for inherited thrombophilia testing in children remain unclear, with ongoing debate among hematologists regarding appropriate candidates for testing. Many centers now recommend testing in children who experience idiopathic or unprovoked VTEs. 91 For both adults and pediatric populations with a family history of thrombophilia or hATD, testing for AT deficiency is generally advised, especially for those who have encountered severe arterial or venous thrombosis early in life or those who develop a thrombus located in an uncommon area such as the inferior vena cava, mesenteric, renal, or cerebral veins.20,85 Prior findings in pediatric patients with hATD found that 44% of participants were diagnosed given a personal history of VTE, whereas 56% were diagnosed in the setting of a positive family history. 52 The ongoing debate over the practice of routine testing for hereditary thrombophilia also includes considerations of costs. The decision to test for AT deficiency and other forms of thrombophilia should be individualized based on clinical presentation and VTE risk factors.74,92–96
Management of hATD
Treatment for AT deficiency varies based on the type of clinical presentation, which can broadly be categorized as patients receiving treatment for acute VTE, short-term thromboprophylaxis in high-risk settings (eg, surgery, pregnancy, and trauma), or long-term anticoagulant thromboprophylaxis for symptomatic patients. 20 For instance, the first line therapy for the treatment of acute VTE involves heparin or LMWH; however, their effectiveness depends on the availability of functional AT. Indeed, traditional anticoagulation therapy with heparin cannot exert its therapeutic effect without sufficient AT, as AT is the essential cofactor for heparin's anticoagulant activity. This inherent dependency necessitates careful management in patients with AT deficiency, as low AT levels can result in heparin resistance. 20 While DOACs have shown safety and efficacy for pediatric VTE management and are now approved for use in children, they do not correct the underlying AT deficiency and are not currently approved for hATD.20,46,97 AT replacement therapy is a critical consideration, as these treatments are essential to address the underlying pathophysiology of hATD. Accordingly, this review focuses on ATc use in pediatric hATD, and discussion of alternative therapies is beyond the scope of this article.
Antithrombin Replacement Therapies
In August, 2025, the FDA approved a label expansion for ATc to formally include pediatric patients with hATD, making this the first treatment specifically indicated for this patient population.12,13 Specifically, ATc is indicated in adults and pediatric patients with hATD for treatment and prevention of thromboembolism, and prevention of peri-operative and peri-partum thromboembolism. 13 The AT replacement therapies that have received approval by the FDA for use in patients with hATD include plasma-derived ATc (Thrombate antithrombin III (human®)) and recombinant human AT (rhAT).8,98 However, the availability of rhAT is subject to geographical restrictions, and was removed from authorization in the European Union in 2018 and from the US market in 2015 at the request of the marketing-authorization holder.99,100 Given the limited commercial availability of rhAT and the absence of studies assessing its use in pediatric patients with hATD, this review focuses exclusively on plasma-derived ATc and does not further elaborate on rhAT.
The standard approach for patients with hATD and VTE includes AT replacement with ATc, although fresh frozen plasma (FFP) has also been used in certain cases.101,102 FFP is prepared from whole blood or during plasmapheresis103,104 and contains all coagulation factors, including AT, fibrinogen, albumin, protein C, protein S, and tissue factor pathway inhibitor except platelets.105,106 It is stored at −30°C and requires thawing prior to administration; once thawed, it should be used within 1–6 h or stored at 1–6 °C if administration is delayed.105,107,108 While a standard dose of 10 to 20 mL/kg can increase clotting factor levels by about 20% in patients with active bleeding, the efficacy of FFP for hATD has not been well established, and its use is generally reserved for situations where ATc is unavailable, due to potential risks including volume overload, allergic reactions, transfusion-related acute lung injury (TRALI), infection transmission, and hemolysis.105,109–111
ATc is a sterile, nonpyrogenic, stable, lyophilized preparation supplied in single-dose vials containing ∼500 IU/vial. ATc is administered intravenously after reconstitution to return AT levels between 80% and 120% of normal on an individualized basis. The dose calculation for ATc is based on weight and baseline AT level. The initial loading dose is determined by the following formula: Dose (Units) =120%–baseline % × body weight (kg)/1.4%. Plasma AT levels are subsequently maintained between 80% to 120% by administering a maintenance dose of 60% of the loading dose, every 24 h. Administering a full dose of ATc typically takes 10 to 20 min and is generally well tolerated, though hypersensitivity reactions may occur. 8
Initial clinical trial findings on adverse events for ATc showed 29 adverse reactions during 17 of 389 infusions, with no serious adverse reactions and most being mild or moderate in severity. 8 The adverse reactions most frequently observed in at least 5% of subjects were dizziness, chest discomfort, nausea, dysgeusia, and pain (cramps). 8 As a product derived from human blood, ATc also hypothetically carries a risk of transmitting infectious agents (eg, viruses) and Creutzfeldt–Jakob agent; however, no cases of transmission have ever been reported. 8
Prior to 2025, prescribing information for ATc did not specify age ranges, and the existing literature indicated that pediatric patients with hATD had no approved AT replacement therapies.8–11 Consequently, clinical use of ATc in this population relied on published literature demonstrating congruent presentations of hATD in pediatric populations compared to adults (as discussed above) and similar exposure-response findings compared to adults. This paradigm has now shifted given the recent FDA's approval of a label expansion to include adult and pediatric patients with hATD for the treatment and prevention of thromboembolism and for the prevention of peri-operative and peri-partum thromboembolism.12,13
ATc use in Pediatric Populations with hATD
Although evidence exists supporting the use of AT in adult patients with hATD,112–114 its use in pediatric populations with hATD remains limited to only a handful of case reports and small patient cohort studies (Supplementary Table 3). The seminal study by Schwartz et al demonstrated the successful use of ATc in patients with hATD, revealing that hATD patients who received prophylactic ATc exhibited no signs of thromboembolism, and that those who received acute treatment of thrombosis and/or thromboembolism had no extension or recurrence. 114 Due to the limited clinical data available, treatment guidelines have largely relied on extrapolation of evidence from adult data to pediatrics. Current ASH guidelines for management of pediatric VTE suggests using AT replacement therapy in children with documented hATD failing to respond to anticoagulation therapy for VTE, as well as use in addition to standard anticoagulation in pediatric patients with deep vein thrombosis, CSVT, and pulmonary embolism who have clinically failed to achieve therapeutic anticoagulation with persistent low AT levels. 115
Notably, the rarity of hATD and the difficulties involved in detecting it early in life make it challenging (if not impossible) to conduct randomized clinical trials in pediatric populations. For instance, the Thrombo-PICS study by Smith et al at the University of Colorado Hemophilia and Thrombosis Center, enrolled only 20 children with hATD over 14 years, and only nine were symptomatic. 62 The study conducted by ISTH also only enrolled forty-three pediatric participants with hATD over 2 years from 4 participating centers, with only 19 (44%) having a history of VTE. 52 A retrospective, multicenter cohort study spanning 10 years with over 4000 patients <18 years of age who received ATc also found that only 3% received ATc for on-label conditions including hypercoagulable state/hATD, while the remaining 97% were off-label uses. 11
ATc use in pediatric populations has been ongoing for nearly as long as in adults and has increased significantly over time.11,115,116 Assessment of the FDA Adverse Events Reporting System (FAERS) for AT deficient pediatric patients treated with ATc reports only two adverse events: one event described a non-serious drug interaction in a 13-day old infant and another described a serious event of postpartum hemorrhage in a 17-year old female. 117 Due to its reliance on voluntary reporting, FAERS data may under-report and lack comprehensive exposure data, making it difficult to assess the true prevalence of adverse events, accurate incidence rates, or causality. Nonetheless, the low number of adverse events reported across three decades suggests a favorable safety profile for AT deficient pediatric patients receiving ATc. Moreover, findings from pediatric patients with pathophysiological consumption of ATc further support its use in pediatric patients with hATD.
ATc use in Pediatric Populations with Antithrombin Deficiency
Findings in pediatric patients experiencing pathophysiologically relevant conditions that result in AT deficiency provide useful insights for use of ATc in such cases (Supplementary Table 4). AT deficiency has been well documented to occur with the use of UFH for acute thrombosis and with the use of asparaginase (ASP) for acute lymphoblastic leukemia (ALL).118–123 Patient populations treated with UFH and ASP are clearly defined by their direct pathophysiological consumption of AT, which results in AT levels similar to that of patients aged six months to <18 years with hATD.
A study by Diaz et al assessed 51 children receiving ATc during UFH therapy for acute thrombosis and showed a significant increase in both AT and anti-FXa levels after the first dose of ATc. Only five patients experienced bleeding, with four minor cases and one case being a clinically relevant non-major event. 119 In a 5-year retrospective cohort study, Moffett et al identified 184 patients <19 years of age who received ATc with mean baseline AT activity levels of 59% of normal adult values, consistent with levels observed in individuals with hATD. 121 Utilizing a pharmacokinetic analysis, the study demonstrated the relationship between ATc dose and post-dose AT levels, which were influenced by weight and baseline AT levels in pediatric patients, aligning with current prescribing guidelines for adult ATc use. 121 A retrospective study by Alami et al assessed AT supplementation in 50 infants and neonates on a continuous heparin infusion and revealed improvements in the therapeutic effect of heparin in 90% of ATc treatment courses. 118 The authors noted that although 4 major bleeding events occurred, AT treatment itself showed no effect on the change in bleeding severity. 118 In pediatric patients with AT levels below 50% and a subtherapeutic UFH effect, Ryerson et al reported that administering ATc significantly increased AT and anti-FXa activity while reducing the UFH dose. There were no reports of hemorrhagic, thrombotic, or allergic events within 1 week following ATc administration. 122
Thrombotic events are among the most frequent adverse events associated with ASP treatment in ALL, occurring in ∼1% to 37% of patients.124,125 ASP treatment for ALL directly contributes to decreases in AT due to increased consumption and decreased hepatic secretion.120,126 Mitchell et al revealed that in children treated with ASP, ATc treatment reduced the incidence of thrombosis compared to those without ATc treatment; adverse events were unremarkable and only two minor bleeding events occurred. 120 More recently, findings from the THROMBOTECT study revealed that ATc significantly reduced the incidence of thromboembolism as compared to those receiving only UFH in pediatric patients treated with ASP. No statistical differences were found in bleeding events across groups. 123
Collectively, these studies demonstrate that administration of ATc increases AT levels114,119,121,122 and reduces the risk of thrombosis114,120 with an exposure-response118–122 and safety profile114,118–120,122 similar to that found in adults with hATD and patients <18 years of age with low levels of AT, which aligns with the recent U.S. FDA label expansion to include pediatric patients. Given that AT levels reach reference adult ranges by six months of age, these findings support current practices of dosing ATc in pediatric patients with hATD based on adult recommendations for those aged six months to <18 years.119,121,122,127,128
Notably, other sources exist for using ATc in indications outside of the conditions discussed here (eg, extracorporeal membrane oxygenation). 20 Although these sources provide interesting insights, the consumption and clearance of ATc in these conditions may be altered by various factors (eg, medications, changes in blood volume, extracorporeal circuit) that confound the interpretation of ATc use in these cases. Hence, an exhaustive discussion of acquired AT deficiency exceeds the scope of this review and presents an interesting area for future research.
Conclusion
Patients with hATD are predisposed to exponentially higher risks for developing thrombotic events, with similar risks present in both pediatric and adult populations. The clinical advantages of testing for AT deficiency, especially in individuals with a family history of thrombophilia or hATD, are becoming more recognized. Treatment of symptomatic or high-risk patients with hATD typically requires AT replacement or enrichment through AT products or FFP transfusion, with AT products being preferred due to potential safety issues and limited efficacy data associated with FFP. Although ATc has been successfully used for treatment in adults with hATD for over three decades, the rarity of this condition and challenges of detecting it early in life severely impact the feasibility of conducting randomized clinical trials in pediatric patients. Nonetheless, published literature demonstrates a similar disease presentation in adults and pediatric patients with hATD. Case reports/series from pediatric patients with hATD, as well as findings from ATc treatment in pediatrics with pathophysiologically relevant conditions, have indicated that ATc can be safely and effectively used in pediatrics. Consequently, this has led to FDA approval of a label expansion to include pediatric patients with hATD, supporting the long-standing pediatric use described in the literature. Future research is still warranted to better understand optimal approaches for testing and treating these patients, as well as understanding the potential clinical implications of specific mutations resulting in hATD. Novel approaches leveraging real-world evidence may be useful tools for gaining insights into this rare patient population.
Supplemental Material
sj-pdf-1-cat-10.1177_10760296261425036 - Supplemental material for Hereditary Antithrombin Deficiency in Pediatric Patients: Pathophysiology, Clinical Features, Diagnosis, and Antithrombin Replacement Therapy
Supplemental material, sj-pdf-1-cat-10.1177_10760296261425036 for Hereditary Antithrombin Deficiency in Pediatric Patients: Pathophysiology, Clinical Features, Diagnosis, and Antithrombin Replacement Therapy by George M. Rodgers and Wilton I. Rodriguez in Clinical and Applied Thrombosis/Hemostasis
Footnotes
Acknowledgments
The authors thank Hayden W. Hyatt, and Nisha S. Wadhwani, of Boston Strategic Partners for medical writing support. The authors thank Michael Calloway of Grifols, Inc. and Rahul Rajkumar of Boston Strategic Partners for their valuable insights and support throughout the development of this manuscript.
Ethical Approval and Informed Consent Statements
Not applicable
Author Contributions
Both authors contributed to the conceptualization of the study, drafting and critical revision of the manuscript for important intellectual content, and approved the final version for submission.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This review was funded by Grifols, Inc. in the form of medical writing and article processing charges.
Declaration of Conflicting Interest
The authors disclose support for the present manuscript in the form of medical writing, funded by Grifols, Inc. GMR discloses membership on a jury panel sponsored by Grifols, Inc., that reviews research grant proposals on antithrombin research. WR is an employee of Grifols, Inc.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
