Abstract
Evidence suggests that there is platelet activation in obstructive sleep apnea syndrome (OSAS) and chronic obstructive pulmonary disease (COPD). Our objective is to evaluate mean platelet volume (MPV) and platelet distribution width (PDW) in patients with overlap syndrome (OS), that is, concurrent COPD with OSAS. Mean platelet volume and PDW were assessed in consecutive patients who had undergone polysomnography and pulmonary function testing. They were divided into the following groups: controls (apnea–hypopnea index [AHI] <5/hour, and forced expiratory volume in 1st second [FEV1]/forced vital capacity [FVC] >70%), OSAS group (AHI ≥5/hour and FEV1/FVC >70%), and OS group (AHI ≥5/hour and FEV1/FVC <70%). A total of 485 patients (360 males and 125 females) were included. Mean platelet volume in controls was lower compared with the other groups: 10 ± 0.9 fL for controls versus 10.3 ± 1.2 fL for OSAS (
Keywords
Introduction
The overlap syndrome (OS) is characterized by the coexistence of chronic obstructive pulmonary disease (COPD) and obstructive sleep apnea syndrome (OSAS). 1 Its prevalence has been estimated at 0.5% to 1% among patients over 40 years old, with future estimates indicating a higher prevalence due to the increasing obesity and tobacco smoking. 1 Patients with OS are more prone to daytime hypoxia compared with patients with OSAS. 2,3 They develop more severe nocturnal oxygen desaturation in comparison with COPD patients with equal degree of airflow obstruction. They also carry an increased risk of developing pulmonary hypertension and cardiovascular disease compared with patients with COPD or OSAS alone. 2,3 Optimal treatment refers to continuous positive airway pressure or noninvasive positive pressure ventilation with the intent to correct upper airway obstruction and hypoxia during sleep. 4
Platelets play a pivotal role in the formation of atherosclerotic plaques and therefore in the progression of atherosclerotic lesions and thrombosis. They express and secrete substances that participate in the process of coagulation, inflammation, thrombosis, and atherosclerosis. 5 Larger platelets are characterized by increased enzymatic and metabolic activity and demonstrate an increased prothrombotic status. 6 Moreover, platelet activation has been linked with cardiovascular morbidity. 7 Accumulating data suggest that platelet activation is increased in patients with OSAS and COPD. 8,9
Both mean platelet volume (MPV) and platelet distribution width (PDW) are markers of platelet activation. 10,11 Increased MPV is predictive of cardiovascular disease, including peripheral, cerebrovascular, and coronary artery disease, and has been associated with the presence of obesity, diabetes mellitus, and metabolic syndrome. 12,13 Elevated MPV values in patients with OSAS and COPD have been reported compared with controls. 14,15
However, there is still no study in the literature regarding the role of platelet activity in patients with OS. Therefore, we assessed MPV and PDW values in patients with OS and compared them with those in healthy controls and patients with OSAS.
Patients and Methods
Patients
Patients referred to the sleep laboratory of our institution with symptoms suggestive of sleep-disordered breathing were prospectively enrolled. The study was conducted in accordance with the Helsinki Declaration of Human Rights and was approved by the institutional ethics committee. 16 All participants gave their informed consent.
Exclusion criteria were: history of hematological disorders or malignancies, abnormal hematocrit and/or abnormal white blood cell count, and/or abnormal platelet count.
At baseline, detailed data on medical history, current medication use, and tobacco smoking were obtained. Clinical examination, including measurement of anthropometric characteristics, was performed. Height, weight, neck circumference, hip, and waist circumference and waist/hip circumference ratio were measured using a standardized protocol. Body mass index (BMI) was calculated using the following formula: weight (kg)/height2 (m)2. Physical examination comprised of pulmonary function testing using a spirometry device (Chest Co, Tokyo, Japan) and of arterial blood gas analysis during wakefulness. Arterial blood samples were collected from the radial artery and analyzed using an ABL3000 autoanalyzer (Radiometer Co, Tokyo, Japan).
Chronic obstructive pulmonary disease was diagnosed on the basis of medical history (chronic and progressive dyspnea, cough, and sputum production) and clinical examination and confirmed by the spirometry results (forced expiratory volume in 1st second [FEV1]/forced vital capacity [FVC] <0.70 after bronchodilation). 17 Sleepiness was assessed using the Greek version of the Epworth Sleepiness Scale (ESS), 18 a self-administered questionnaire evaluating the possibility of falling asleep in a variety of situations (maximum score: 24; score >10: excessive daytime sleepiness).
Polysomnography
Overnight polysomnography (PSG), attended by an experienced sleep technician, was performed from 22:00 to 06:00 hours, and variables were recorded on a computer system (Alice 4; Philips Respironics, Murrysville, Pennsylvania). A standard montage of electroencephalogram, electroocculogram, electromyogram, and electrocardiogram signals together with pulse oximetry was used. Airflow was detected using combined oronasal thermistors. Thoracic cage and abdominal motion were recorded by inductive plethysmography. Apneas, hypopneas, and electroencephalogram recordings were manually scored according to standard criteria. 19 Apnea was defined as a complete cessation of airflow for at least 10 seconds. Hypopnea was defined as a 50% reduction in airflow for at least 10 seconds in combination with oxyhemoglobin desaturation of at least 4% or an arousal registered by the electroencephalogram. The average number of apneas and hypopneas per hour of PSG-recorded sleep time was calculated as the apnea–hypopnea index (AHI). The respiratory disturbance index was defined as the average number of respiratory disturbances (apneas, hypopneas, and respiratory event-related arousals) per hour of PSG-recorded sleep time. Obstructive sleep apnea syndrome was defined as AHI ≥5/hour accompanied by symptoms and was graded as mild (AHI: 5-14.9/hour), moderate (AHI: 15-29.9/hour), and severe (AHI ≥ 30/hour). 20
Biochemical Analysis of Blood Samples
Blood samples were collected the morning after PSG examination, following an overnight fast. To avoid platelet swelling, 21 MPV and PDW were measured in EDTA tubes within 15 to 30 minutes after sampling. An automated blood cell counter was used for these measurements (Sysmex XE 2100, Kobe, Japan). All samples were run in duplicate, and the mean values were used for analysis. Biochemical analysis including fasting glucose levels and lipid profile were also performed.
Statistical Analysis
All analyses were performed using version 17.0 of the IBM Statistical Package for Social Sciences (SPSS Inc, Released 2008, SPSS Statistics for Windows, version 17.0, Chicago). Continuous variables were tested for normality of distribution by the Kolmogorov-Smirnov test. For normally distributed values, descriptive results are expressed as mean (standard deviation). Variables with skewed distribution are expressed as median (25th-75th percentile). The χ2 test was used for comparison of percentages between groups. Correlations were analyzed with Pearson correlation coefficient, while comparisons between means were explored with the Student's
Results
A total of 485 consecutive patients (360 males and 125 females), examined between December 2015 and December 2017, were included. According to their AHI and postbronchodilator FEV1/FVC, patients were divided into 3 groups: control group (AHI <5/hour and FEV1/FVC > 70%) including 82 patients (60 males and 22 females), OSAS group (AHI ≥5/hour and FEV1/FVC >70%) including 324 patients (234 males and 90 females), and OS group (AHI ≥5/hour and FEV1/FVC <70%) including 79 patients. The control group significantly differed from the other groups in terms of age (47.2 ± 13.3 years for controls vs 53.7 ± 12.5 years for OSAS vs 60.5 ± 11.3 years for OS,
Characteristics of the Study Population.
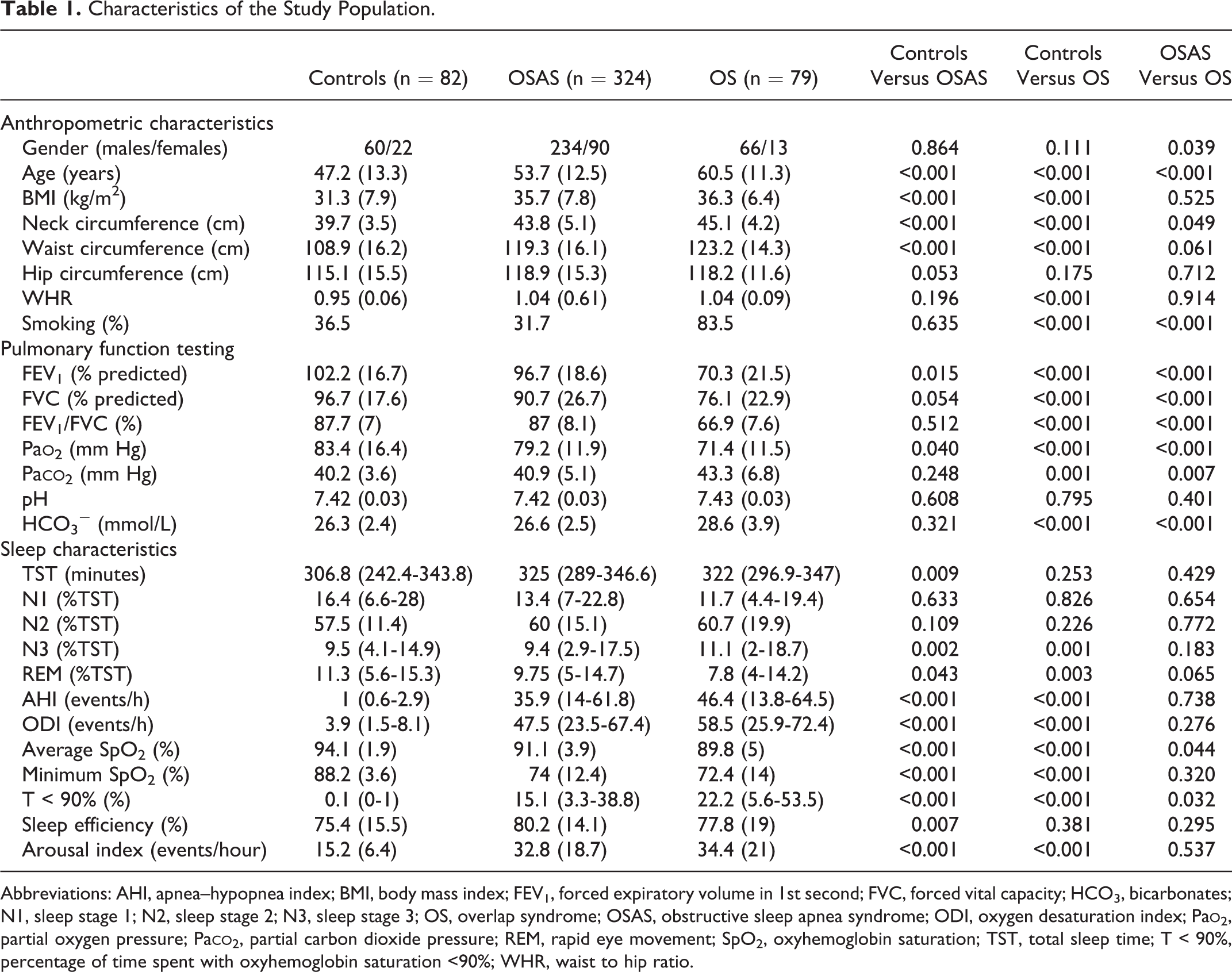
Abbreviations: AHI, apnea–hypopnea index; BMI, body mass index; FEV1, forced expiratory volume in 1st second; FVC, forced vital capacity; HCO3, bicarbonates; N1, sleep stage 1; N2, sleep stage 2; N3, sleep stage 3; OS, overlap syndrome; OSAS, obstructive sleep apnea syndrome; ODI, oxygen desaturation index; Pa
The distribution of diabetes among groups was as follows: 15 (18.3%) patients in the control group, 47 (14.5%) patients in the OSAS group, and 12 (15.2%) patients in the OS group. There was no difference regarding the prevalence of diabetes between controls and patients with OSAS (
The groups differed also in terms of fasting glucose levels and lipid profile. Mean platelet volume was significantly decreased in controls compared with the other groups (10 ± 0.9 fL for controls vs 10.3 ± 1.2 fL for OSAS,
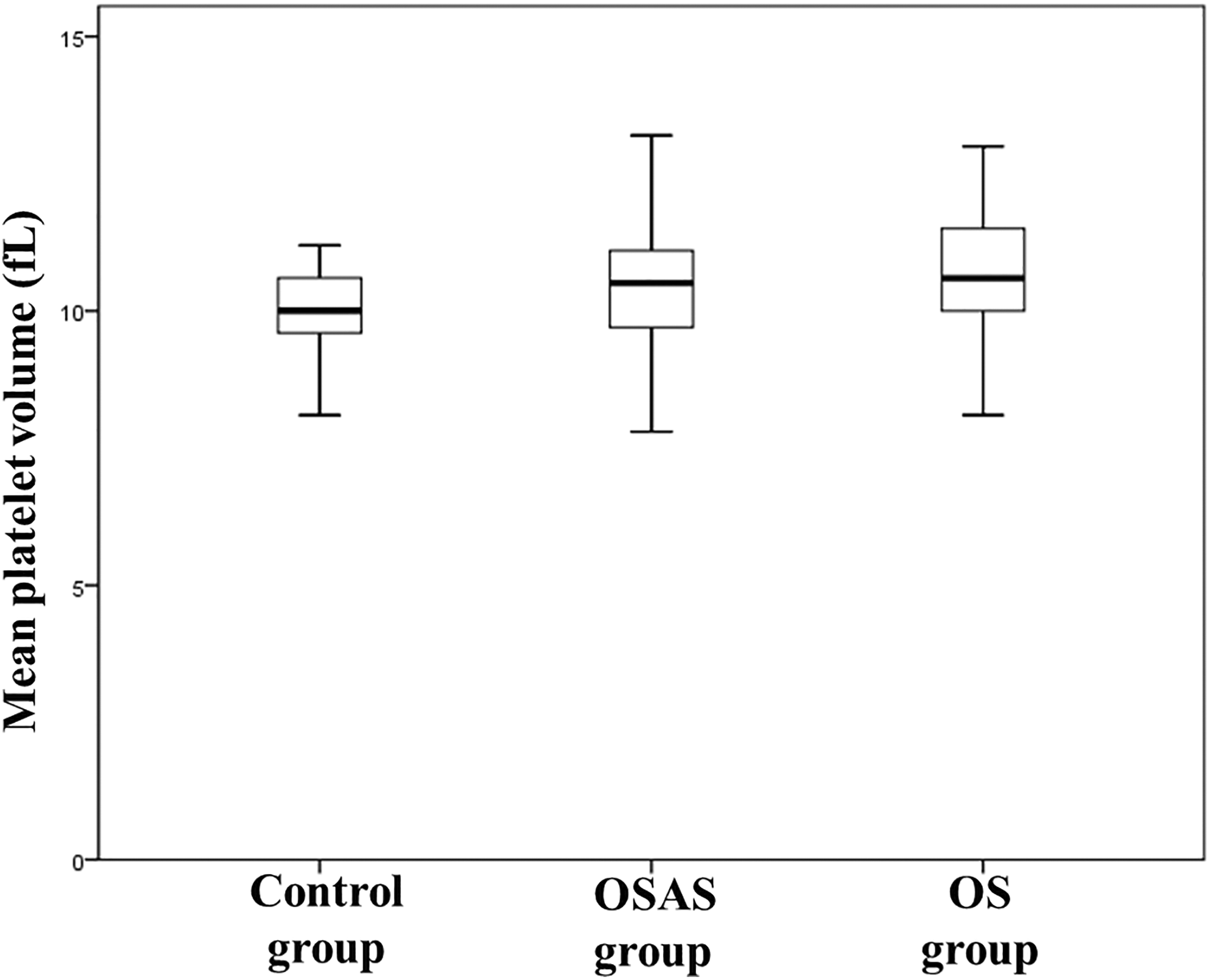
Comparison of mean platelet volume values between groups.
Platelet distribution width was lower in the control group compared with all other groups (12.9 ± 2 fL for controls vs 13.6 ± 1.9 fL for OSAS,
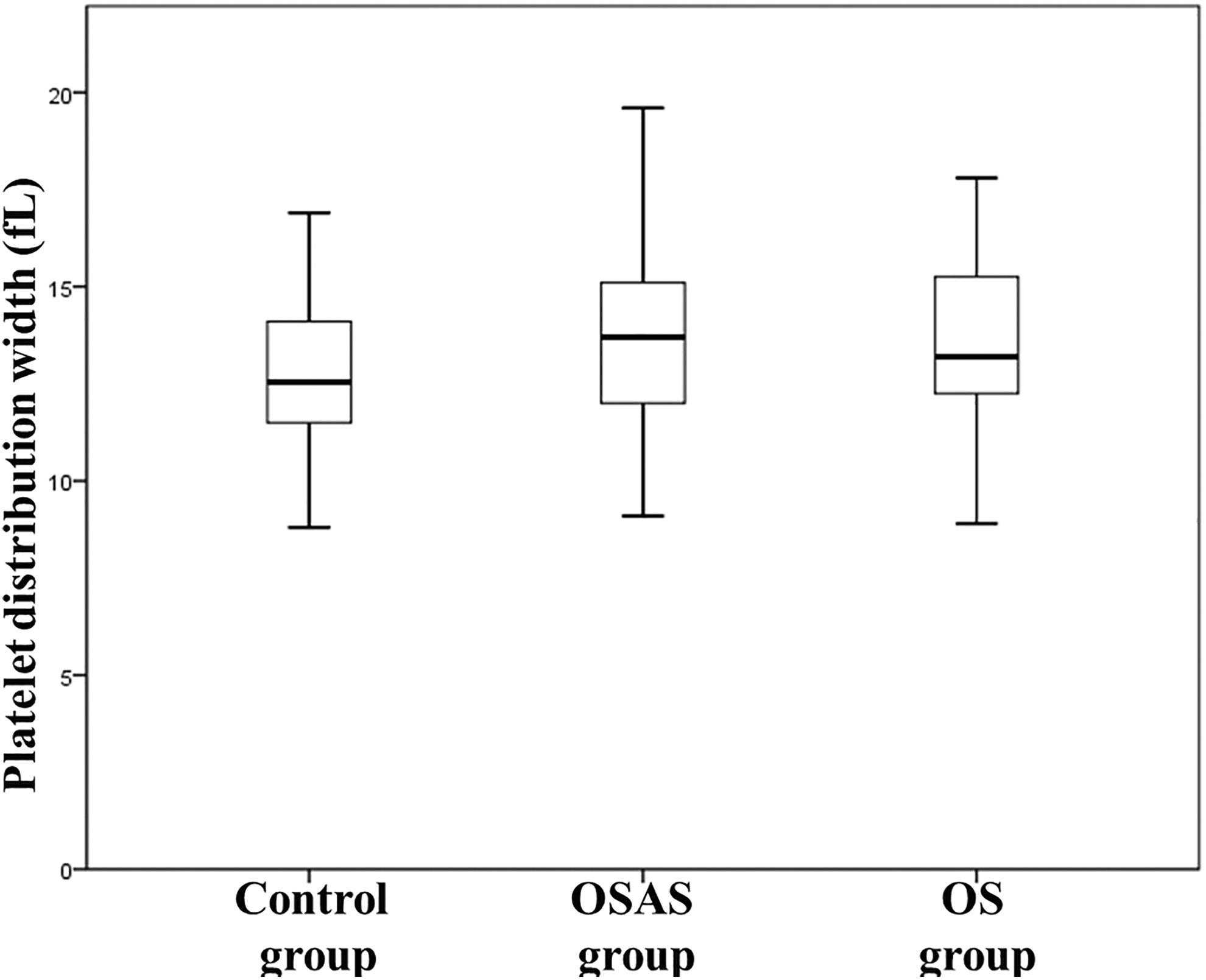
Comparison of platelet distribution width values between groups.
Results from blood counts and biochemical tests are presented in Table 2. In the OS group, MPV had a positive correlation with total sleep time (
Study Population Blood Counts and Biochemical Tests.
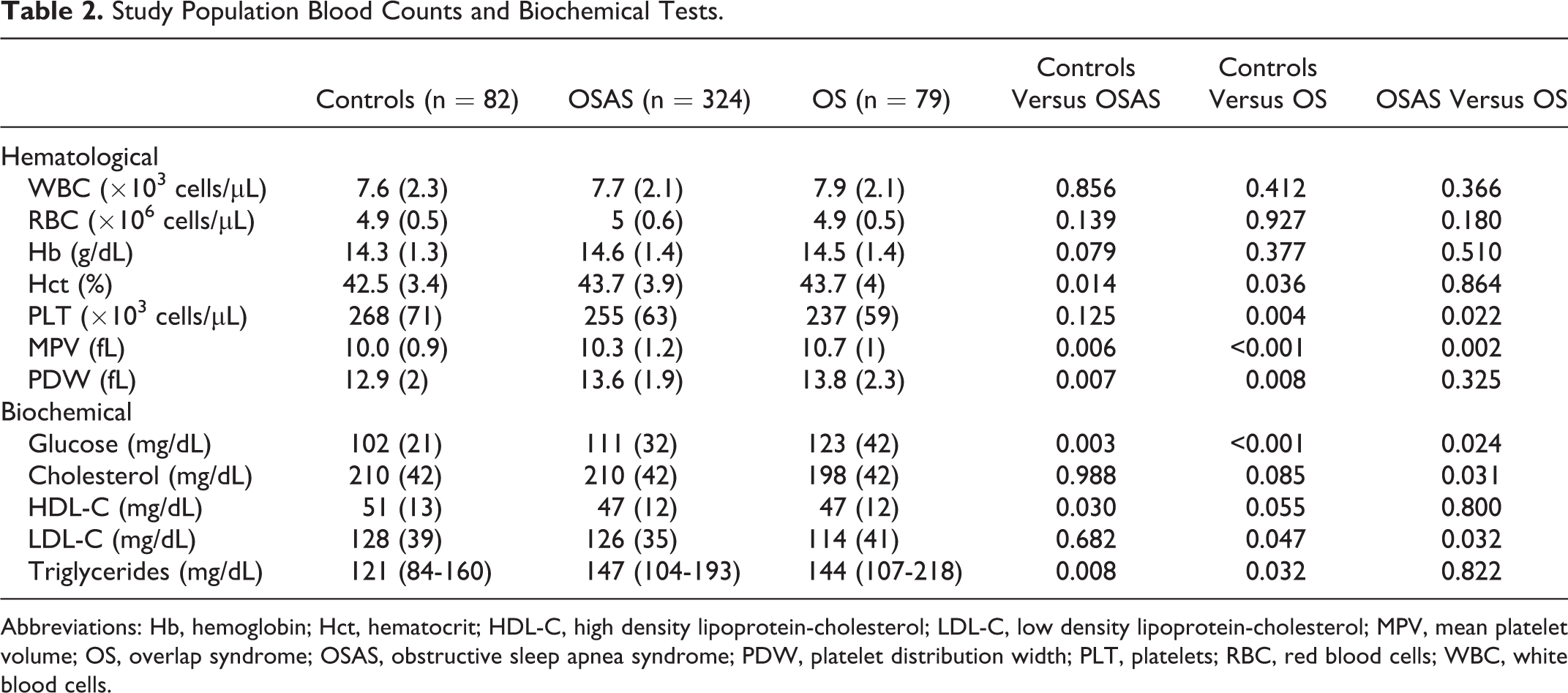
Abbreviations: Hb, hemoglobin; Hct, hematocrit; HDL-C, high density lipoprotein-cholesterol; LDL-C, low density lipoprotein-cholesterol; MPV, mean platelet volume; OS, overlap syndrome; OSAS, obstructive sleep apnea syndrome; PDW, platelet distribution width; PLT, platelets; RBC, red blood cells; WBC, white blood cells.
Discussion
This study has demonstrated a significant increase in MPV and PDW (markers of platelet activation) in patients with OS compared with controls. Furthermore, MPV was higher in patients with OS compared with those of the OSAS group. To the best of our knowledge, this is the first study examining MPV and PDW and their associations with sleep characteristics and demographics in patients with OS versus controls and patients with OSAS alone.
Mean platelet volume and PDW have been previously evaluated separately in both OSAS and COPD, which are the components of OS. In a study including 610 nondiabetic patients with suspected OSAS,
15
MPV was higher in patients with severe OSAS compared with mild and moderate apneic patients and healthy controls (12.1 ± 1.3 fL vs 9.8 ± 1.6 fL vs 11.5 ± 1.3 fL vs 9.8 ± 1.1 fL, respectively,
In a study of 119 patients,
14
patients with stable COPD exhibited higher MPV compared with smoker controls (10.69 + 1.0 fL vs 9.96 + 1.10 fL,
An additional finding in our study was that patients with OS exhibited higher MPV compared with patients with OSAS. Patients with OS exhibited decreased FEV1, FVC, and lower daytime partial oxygen pressure compared with the other groups. Previous evidence has demonstrated an association between respiratory function and increased MPV in COPD. 9 Moreover, hypoxemia plays a central role in platelet activation. 26 Consequently, reduced FEV1 seems to be the main determinant of increased MPV values in OS.
In OS, several mechanisms may be implicated in platelet activation. Indeed, increased sympathetic activation, a basic feature of OSAS, increases circulating catecholamines, which in turn activate platelets in a dose-dependent manner. 27 Additionally, increased arterial shear stress, due to hypertension peaks during apneas, may also lead to platelet activation. 27 Acute and chronic intermittent hypoxia, accompanying apneic events, a feature which is also present in patients with COPD, directly triggers platelet activation. 26,28 In patients with COPD, local platelet activation in pulmonary vessels and local activation of the coagulation cascade and fibrinolysis system may also play a significant role for the systemic effects on platelet dysfunction. 29,30 Cigarette smoking, the main cause of COPD, has been shown to directly impact the size of platelets, resulting in increased MPV values compared with nonsmokers. 31 Moreover, decreased lung function is associated with a persistent inflammatory status, which leads to impaired endothelial function and promotes increased atherosclerotic risk and overall cardiovascular events. 29,30
Chronic inflammation and hypoxia may induce platelet activation. 28,32 Both OSAS and COPD are now appreciated to increase systemic inflammation. 33 –35 A common denominator in both conditions may be an increased secretion of pro-inflammatory cytokines, like interleukin-6, or an overexpression of adhesion molecules playing an important role in platelet activation. 33,36 –39 Other mechanisms responsible for increased thrombotic risk among patients with COPD, which should not be overlooked, are obesity and concurrent reduced exercise capacity. 40,41 Indeed, it has been found that visceral obesity and adipose tissue-derived inflammation are inducing platelet activation leading to greater cardiovascular risk. 42 Along with COPD, obesity constitutes a common feature and a risk factor for OSAS development. 43
Thus, the mechanisms responsible for platelet activation in OS appear to be complex, interacting, or additive and still inadequately explained. Eventually, patients with OS, either due to features of COPD or due to characteristics of OSAS, are predisposed to a greater cardiovascular risk as a consequence of enhanced platelet activity.
Interestingly, there are no data assessing cutoff values for MPV and PDW that would suggest increased platelet activation in patients with OS. Regarding PDW, Bulbul et al
44
suggested that the optimal cutoff value, distinguishing patients with OSAS from nonapneic controls, was 16.62 (sensitivity 57.8%, specificity 58.1%, AUROC = 0.60,
The present study has some limitations. First, the groups were not age- and BMI-matched. However, this reflects everyday clinical practice, given that patients with OS and OSAS, indeed, tend to be older and more obese. Secondly, there was a larger representation of patients with OSAS compared with the other groups. Again, this reflects the common distribution of consecutively examined patients who had been referred to a specialized sleep laboratory. Moreover, our study lacks follow-up data, and especially the effect of continuous positive airway pressure therapy on the measured indices, but this was beyond the scope of our study. In addition, blood samples were collected in tubes containing EDTA, but we were careful in measuring MPV between 15 to 30 minutes after sampling and carried out each measurement twice using mean values for analysis. Finally, we did not consider the amount of young platelets or protein disulfide isomerase. The latter has been implicated in platelet function, thrombus formation, vascular inflammation, and especially in cardiovascular disease 46,47 ; however, such considerations were beyond the scope of this study.
The practical implications of our findings can be summarized as follows. Both COPD and OSAS are characterized by increased platelet activation and therefore by a high risk of thrombosis. 9,15 In OS, patients may carry an even higher risk of thrombosis, partly due to the concurrence of OSAS and COPD. Platelet activation (as expressed by MPV and PDW) should be taken into account as an index of potential future cardiovascular disease in patients with OS. Given the simplicity and the relatively limited cost of MPV and PDW measurements, these platelet indices might contribute toward timely identification of high-risk patients, suggesting further assessment and identification of vascular risk factors. 10,11 Further research focusing on the implementation of this knowledge in the overall assessment of vascular risk in patients with OS is now desirable.
In conclusion, MPV and PDW are higher in patients with OS compared with healthy individuals. Respiratory function appears to play a fundamental role towards platelet activation either in OS or as a single disorder. The current study is the first to evaluate platelet activation in OS and suggests that MPV and PDW merit further evaluation as surrogate markers of increased cardiovascular risk in such patients. Further prospective data on platelet indices in OS may be proven useful in prognosis of cardiovascular risk.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Nikolaos Papanas has been an advisory board member of Astra-Zeneca, Boehringer Ingelheim, MSD, Novo Nordisk, Pfizer, Takeda, and TrigoCare International; has participated in sponsored studies by Astra-Zeneca, Eli-Lilly, GSK, MSD, Novo Nordisk, Novartis, and Sanofi-Aventis; has received honoraria as a speaker for Astra-Zeneca, Boehringer Ingelheim, Eli-Lilly, Elpen, MSD, Mylan, Novo Nordisk, Pfizer, Sanofi-Aventis and Vianex; and has attended conferences sponsored by TrigoCare International, Eli-Lilly, Galenica, Novo Nordisk, Pfizer, and Sanofi-Aventis. Paschalis Steiropoulos has been an advisory board member of AstraZeneca, Boehringer Ingelheim, Novartis, and Menarini; has participated in sponsored studies by Angelini and Elpen; has received honoraria as a speaker for Boehringer Ingelheim, Chiesi, Elpen, and Novartis; and has attended conferences sponsored by Boehringer Ingelheim, Elpen, Novartis and Roche. Dimitri P. Mikhailidis has given talks and attended conferences sponsored by MSD, AstraZeneca, and Libytec.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
