Abstract
Background. Low-molecular-weight heparins (LMWHs) are an alternative to unfractionated heparin (UFH) for anticoagulation during hemodialysis (HD). We performed a prospective randomized crossover study of the effect of enoxaparin, nadroparin, and dalteparin on some hemostatic factors, including tissue factor pathway inhibitor (TFPI), in patients with maintenance HD. Methods. Plasma levels (immunoassays) of total TFPI, platelet-derived growth factor-AB (PDGF-AB), and prothrombin fragment 1 + 2 (PF 1 + 2) were evaluated pre-HD, after 10 (T10) and 180 (T180) minutes of HD in 21 patients, who completed a 3-period (for 2 months each) crossover study in 6 groups (Latin-square design). Results. The baseline TFPI, PDGF-AB, and PF 1 + 2 levels were comparable under all LMWH treatments. Tissue factor pathway inhibitor levels, compared with the baseline, significantly increased (all P < 10−4), whereas PDGF-AB levels remained stable at each interval during enoxaparin, nadroparin, and dalteparin anticoagulated HD. Interestingly, TFPI increment at T10 was the highest, dose-dependent, and accompanied by PF 1 + 2 decrease under enoxaparin administration. Conclusion. The switch from enoxaparin to nadroparin and dalteparin used as anticoagulants had no long-term effect on the baseline total TFPI and PF 1 + 2 levels in chronically HD patients. Only short-term, overdialytic differences were noticed, indicating a single bolus of enoxaparin (0.75 mg/kg) as the most potent stimulus for endothelial TFPI
Keywords
Introduction
Low-molecular-weight heparins (LMWHs) comprise a variety of different molecular weight derivatives of unfractionated heparin (UFH). Major effects of the LMWHs are by potentiating antithrombin activity and exerting anti-Xa inhibition. They can also release tissue factor pathway inhibitor (TFPI), von Willebrand factor (vWF), modulate the activity of different adhesion molecules, and release many other profibrinolytic and antithrombotic mediators from blood vessel wall. 1
The LMWHs, with a mean molecular weight of 4.0 to 6.0 kd, exhibit specific molecular and structural attributes that are determined by the type of manufacturing process used. For example, enoxaparin, which is prepared by benzylation followed by alkaline hydrolysis of UFH, exhibits a double bond at the nonreducing end and the presence of a unique bicyclic structure namely 1,6 anhydromanno glucose or mannose, or both, at the reducing end. 2 Similarly, the other LMWHs, such as dalteparin, nadroparin, tinzaparin, and parnaparin, exhibit specific structural characteristics that may contribute to their own unique biochemical and pharmacological profiles. These unique features may not exhibit any major influence on the routinely determined anti-Xa and anti-IIa activities. 3,4 However, they may have an impact on the pharmacokinetics and other biological actions such as interaction with growth factors, blood components, and endothelial cells. Simultaneously, TFPI release contributes to the individuality of LMWHs.
Because the LMWHs represent product-specific molecular and structural attributes, the US Food and Drug Administration as well as European Medicines Agency consider each of these drugs as distinct pharmacological agents. Although differences between various commercially available products have been described in term of molecular weight profile and biologic properties, very limited information on the direct comparison of individual products in chronically HD patients is available. The current study was designed to comparatively investigate the influence of 3 LMWHs: enoxaparin (Clexane), dalteparin (Fragmin), and nadroparin (Fraxiparin) given for anticoagulation during HD on plasma levels of total TFPI, platelet-derived growth factor-AB (PDGF-AB), and prothrombin fragments (PF 1 + 2). Platelet-derived growth factor-AB was chosen among few widely used markers of platelet activation because it is also proven to have special importance in the progression of atherosclerosis which is flourishing in hemodialysis (HD) population. 5,6
Participants and Methods
Patients
We enrolled 21 patients who had been undergoing maintenance HD in the Department of Nephrology and Transplantation with Dialysis Unit of Medical University in Białystok. They were selected from a group of patients who had been receiving enoxaparin (Clexane, Bellon Rhône-Poulenc Rorer, Montrouge, France) as an anticoagulant during their HD session for at least 3 months prior to the study. The enrollment criteria excluded patients with diabetes requiring insulin, malignancy, HIV, active viral hepatitis B and/or C, other severe liver diseases, those receiving immunosuppressive, contraceptive, nonsteroidal anti-inflammatory drugs (except for acetylsalicylic acid administered because of cardiovascular disease), vitamin K antagonists, and those who had had any inflammatory or infectious diseases during the preceding month. The dose of dialysis delivered (Kt/V, calculated using a single-compartment model), reflecting the effectiveness of HD session was assigned in each case. Its value below 1.2, as well as incorrect basal whole blood-activated partial thromboplastin time determined exclusion from the study. If treated with recombinant human erythropoietin (rHuEPO), the patients had reached target hematocrit prior to enrollment and were on maintenance doses during the study. Other patients' characteristics are presented in Table 1 .
Clinical Characteristics of 21 Chronically Hemodialyzed Patients Enrolled in the Study a
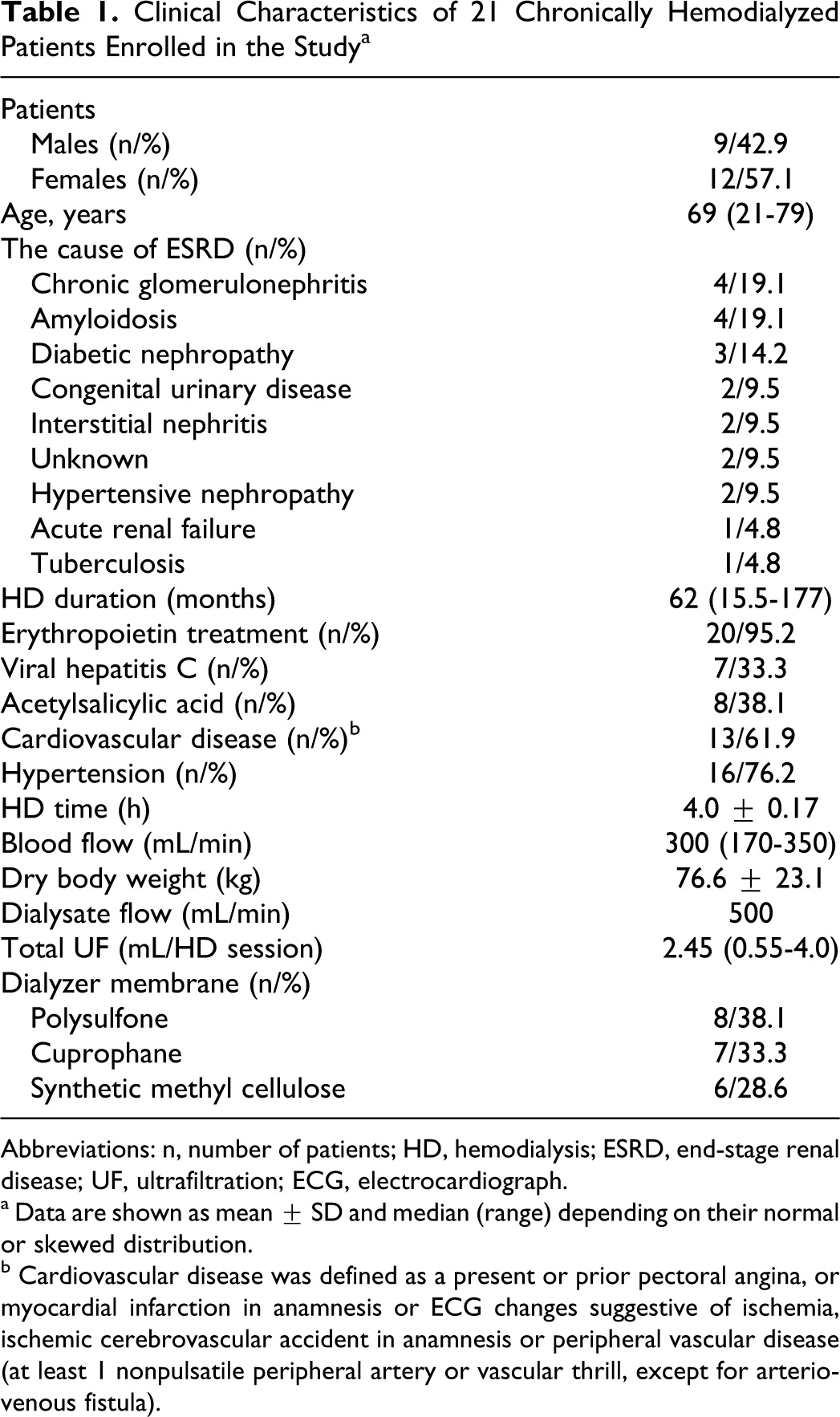
Abbreviations: n, number of patients; HD, hemodialysis; ESRD, end-stage renal disease; UF, ultrafiltration; ECG, electrocardiograph.
a Data are shown as mean ± SD and median (range) depending on their normal or skewed distribution.
b Cardiovascular disease was defined as a present or prior pectoral angina, or myocardial infarction in anamnesis or ECG changes suggestive of ischemia, ischemic cerebrovascular accident in anamnesis or peripheral vascular disease (at least 1 nonpulsatile peripheral artery or vascular thrill, except for arteriovenous fistula).
All the patients had a native arteriovenous fistula allowing double-needle dialysis at blood flow rates of at least 200 mL/min. The flow rate of a bicarbonate buffer was 500 mL/min.
Before each HD session, the extracorporeal circuit was rinsed with 1000 mL of 0.9% saline containing 2.0 IU/mL of UFH (Heparin, Biochemie, Kundl, Austria). No other drugs or blood products were given to patients during dialysis procedure.
Study Protocol
The study was performed in conformity with the Helsinki Declaration. Ethics committee approval was obtained and informed consent was sought from each patient.
This prospective, 3-period crossover study lasted 6 months. The patients were randomized (envelope method) to 6 groups. The sequence of treatments was determined according to Latin-square design (Figure 1 ). Such a protocol allowed avoiding random (carryover) and systemic (bias) error as well as decreasing the sample size at least 9 times. Each patient was tested against himself or herself in 3 different periods (2 months each) during HD under anticoagulation with 3 different LMWHs. The study was designed to keep the heparin doses, dialysis membrane, other HD prescriptions, and pharmacologic treatment stable during the entire 6 months.
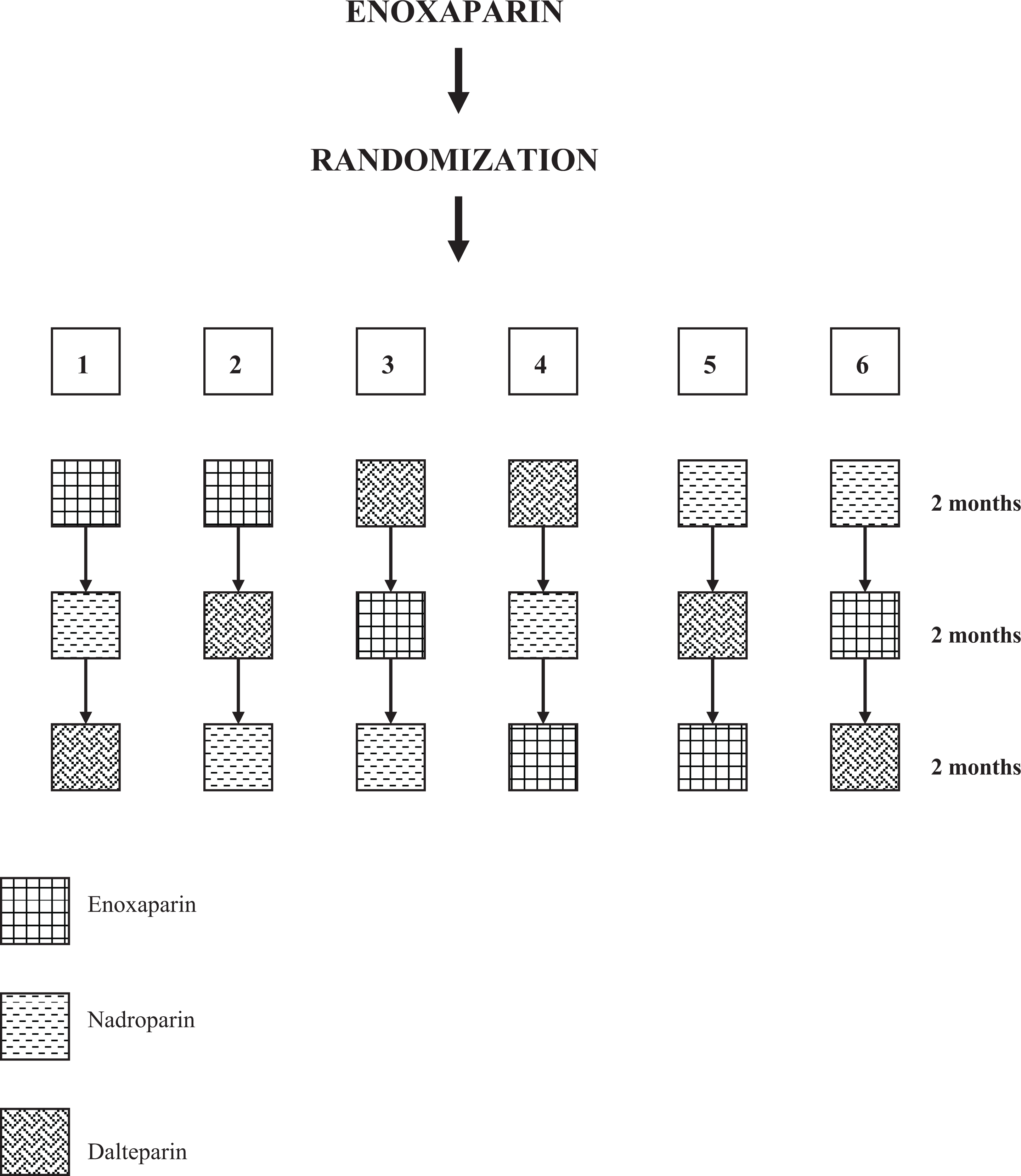
Crossover study design.
Initial LMWHs doses were administrated intravenously according to the manufacturer’s recommendation: 70 IU/kg of body weight in the case of nadroparin (Fraxiparin, GlaxoSmithKline, Brentford, UK) and dalteparin sodium (Fragmin, Pfizer, New York), and 0.7 mg/kg of body weight for enoxaparin. During the first 3 sessions after heparin change, the doses were modified on the basis of the following clinical guidelines: no visible fibrin clots in the arterial and venous bubble traps during HD, no clotted filters after HD, and no bleeding from the fistula puncture sites after compression. 7
Laboratory Procedures
After each 2-month period of the study, blood samples were obtained during a midweek morning HD session: before hemodialysis (T0) from the arterial needle (before heparinization), and at 10 and 180 minutes of dialysis (T10 and T180, respectively) from the predialyzer port after slowing the blood flow to 100 mL/min for 1 minute. The time range for collecting blood was adopted from previous studies. 8 –11
Fasting blood was drawn without the use of a tourniquet through a wide-gauge butterfly needle into (1) DiaTubesH (BD Vacutainer Systems, Plymouth, UK) containing a 0.5 mL solution consisting of citrate (0.109 mol/L), adenosine (3.7 mmol/L), theophylline (15 mmol/L), and dipyridamol (0.198 mmol/L) to minimize platelet degranulation; (2) Monovette vacutainers (Sarstedt, Nümbrecht, Germany) containing 0.129 mol/L trisodium citrate (0.5 vol anticoagulant and 4.5 vol whole blood). The first 1 mL of blood was discarded; then 4.5 mL of blood was collected. Tubes were placed in melting ice and centrifuged 15 minutes later at 2000g for 30 minutes. One third of the plasma was collected from the middle region of the supernatant, aliquoted, and immediately frozen at −70°C. Batch analyses were performed within 8 weeks.
Plasma PDGF-AB antigen levels were determined using solid-phase enzyme-linked immunosorbent assay (ELISA) kits purchased from R&D Systems, Inc, Minneapolis, Minnesota (Quantikine; cat no DHD00B); its minimum detectable levels was 1.7 pg/mL. Prothrombin fragment 1 + 2 was determined using enzyme immunoassays Enzygnost F 1 + 2 micro (cat no OWVV) from Dade Behring, Marburg, Germany; its minimum detectable level was 0.04 nmol/L. Plasma antigens of total TFPI were measured using commercial 2-antibody sandwich assay kit Imubind (cat no 849), American Diagnostica Inc Greenwich, Connecticut. The total TFPI assay detects both intact and truncated forms of the inhibitor as well as its complexes with TF/VIIa; its detection limit was 0.18 ng/mL.
All the measurements were performed in duplicate using a 400 SFC photometer (SLT-Labinstruments, Gröding/Salzburg, Austria), and calibrated using provided recombinant human reference samples and standards. For calculation of the results, a computer and curve-fitting software were used. Their intra- and inter-assay coefficients of variations were <9%. The values obtained after 180 minutes of HD were corrected for hemoconcentration according to plasma protein levels.
Statistical Analysis
All data were analyzed using Statistica software (version 6.0 PL, StatSoft, Tulsa, Oklahoma). Shapiro-Wilk W test of normality was used for data distribution analysis. The normally distributed data were presented as mean ± 1 SD, and the skewed data as median (full range). According to data distribution, repeated measures analysis of variances (RM ANOVA) or Friedman’s ANOVA test was performed for plasma levels of the markers studied within participants during 3 periods of different anticoagulation. For comparison between 2 groups nonparametric Wilcoxon single-rank or Mann-Whitney U tests were used when appropriate. Comparisons within 1 group were made by Kruskal-Wallis ANOVA and chi-square tests. Bivariate correlations were assessed using nonparametric Spearman regression test. All statistical tests were 2-sided and only P < .05 was considered significant.
Results
A total of 17 patients uneventfully completed the study; 2 patients died from complications not relevant to the study, 1 patient treated with nadroparin refused further cooperation because of repeated hemorrhage after HD despite minimal heparin dose, and 1 patient had to receive therapeutic doses of enoxaparin after surgery for femoral neck fracture.
Low-Molecular-Weight Heparin Treatment
The mean, minimal, and maximal doses of enoxaparin, nadroparin, and dalteparin were respectively, 0.75, 0.27, and 1.1 mg/kg for enoxaparin; 70.4, 61.3, and 78.6 IU/kg for nadroparin; 66.0, 55.6, and 75.2 IU/kg for dalteparin. Slight hemorrhagic complications, demanding dalteparin dose adjusting, appeared in only 1 case.
Total TFPI Levels During Different LMWHs HD
Plasma total TFPI levels significantly increased during HD under 3 LMWHs studied: enoxaparin (χ2 ANOVA = 24.47, P < 10−6), nadroparin (χ2 ANOVA = 20.12, P < 10−3) and dalteparin (χ2 ANOVA = 20.59, P < 10−3) as shown in Table 2 and Figure 2 . The baseline plasma total TFPI levels were comparable in whole group regardless of the heparin sort.
Overdialysis Plasma Total Tissue Factor Pathway Inhibitor (Total TFPI), Platelet-Derived Growth Factor-AB (PDGF-AB), and Prothrombin Fragment 1 + 2 (PF 1 + 2) Levels in Patients Anticoagulated With Enoxaparin, Nadroparin, and Dalteparin During Hemodialysis Procedure a
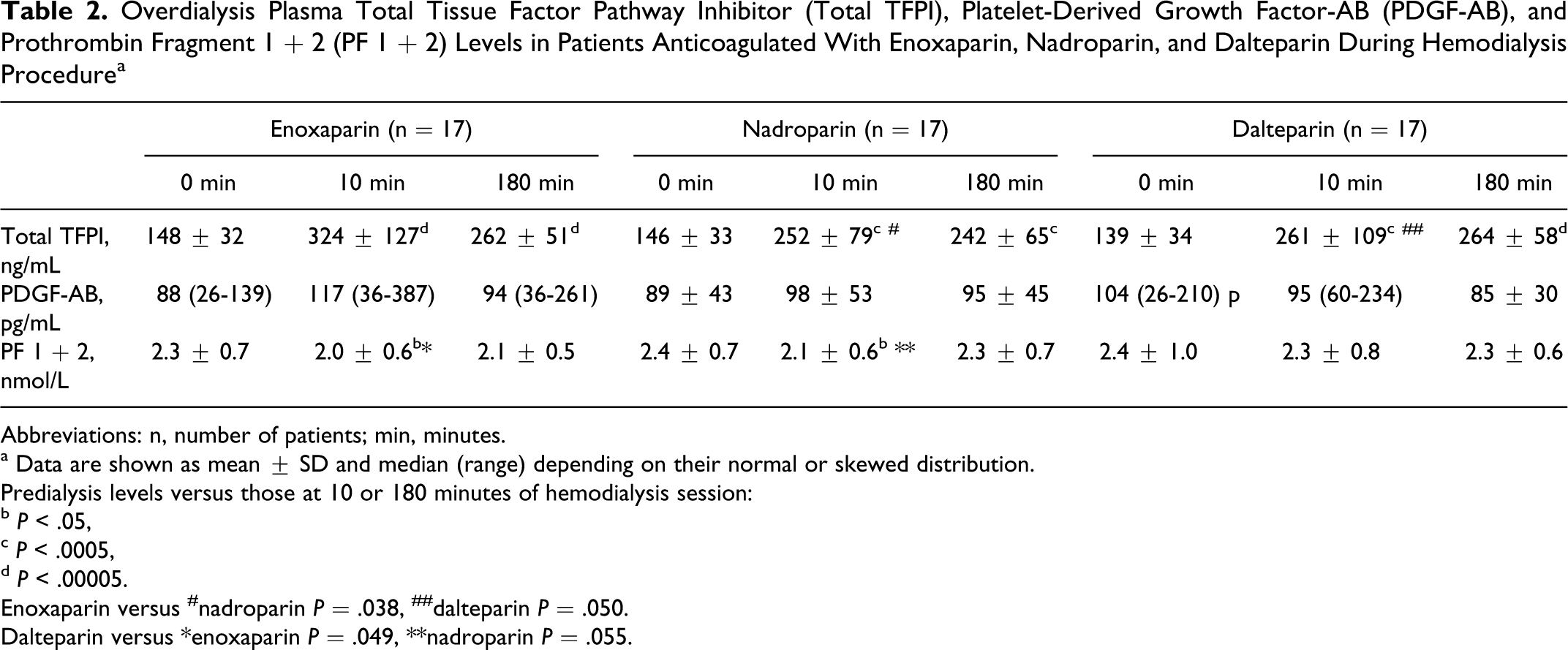
Abbreviations: n, number of patients; min, minutes.
a Data are shown as mean ± SD and median (range) depending on their normal or skewed distribution.
Predialysis levels versus those at 10 or 180 minutes of hemodialysis session:
b P < .05,
c P < .0005,
d P < .00005.
Enoxaparin versus #nadroparin P = .038, ##dalteparin P = .050.
Dalteparin versus *enoxaparin P = .049, **nadroparin P = .055.
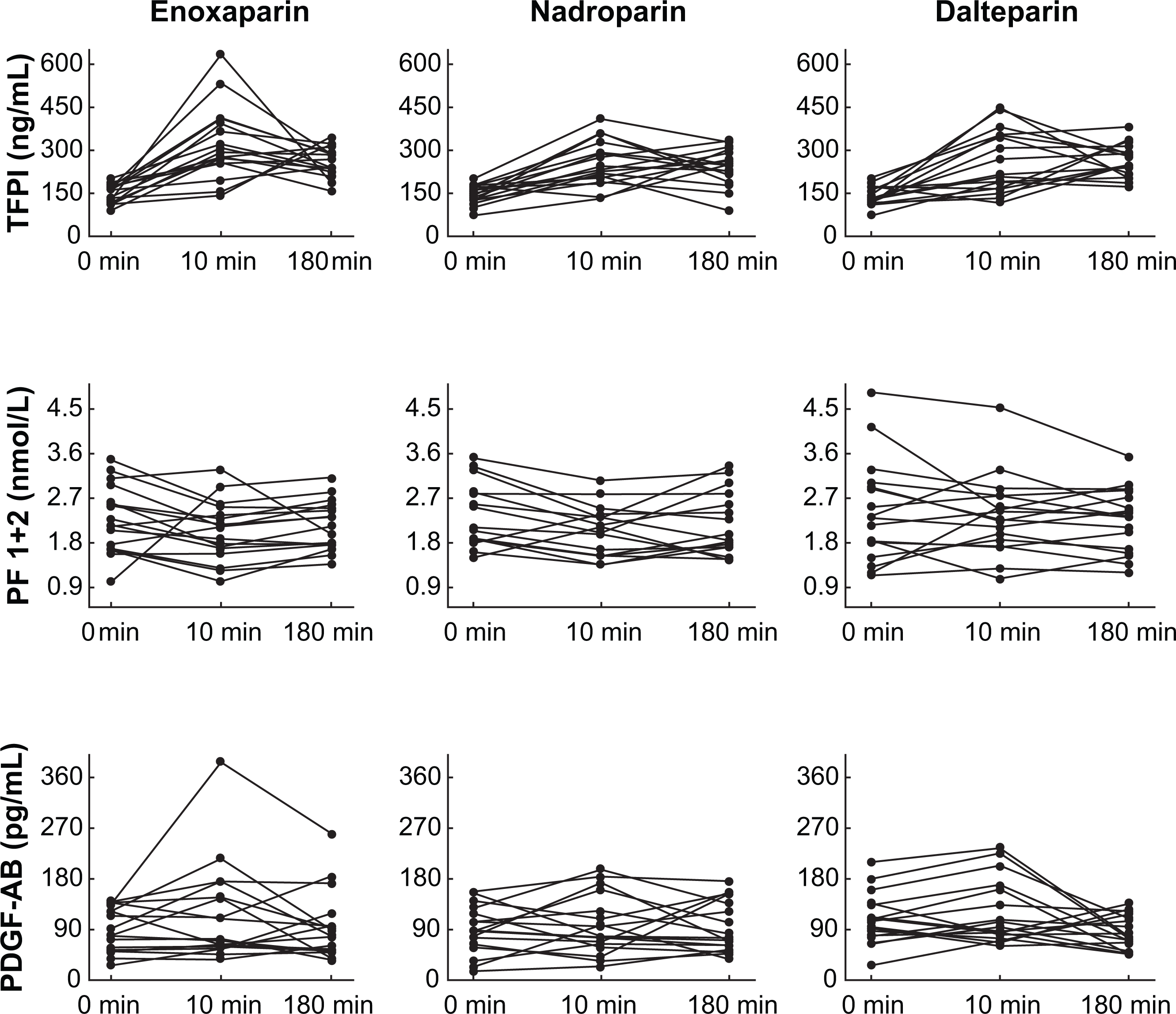
Plasma total TFPI, PF 1 + 2, and PDGF-AB case profiles during hemodialysis with 3 different LMWHs (enoxaparin, nadroparin, and dalteparin). TFPI indicates tissue factor pathway inhibitor; PF 1 + 2, prothrombin fragments 1 + 2; PDGF-AB, platelet-derived growth factor-AB; LMWHs, low-molecular-weight heparins; min, minutes.
During enoxaparin treatment, the total TFPI values increased by a median of 293% (P < 10−3) and remained still higher at T180 (P < 10−4) compared to pre-HD levels. During HD anticoagulated with nadroparin, they increased by a median of 233% (P < 10−3) and remained unchanged after further 180 minutes (P < 10−3) compared with the baseline levels. Under dalteparin treatment, the total TFPI increased by a median of 216% under the first 10 minutes (P < 10−2) and remained similar at T180 (P < 10−4) of HD session. The percentage total TFPI increment after 10 minutes of HD occurred to be dose-dependent (R = .576 P = .015) only under enoxaparin treatment.
The total TFPI increment after 180 minutes of HD procedure correlated inversely with its pre-HD values (Figure 3 ), and this association was independent of the type of anticoagulant.
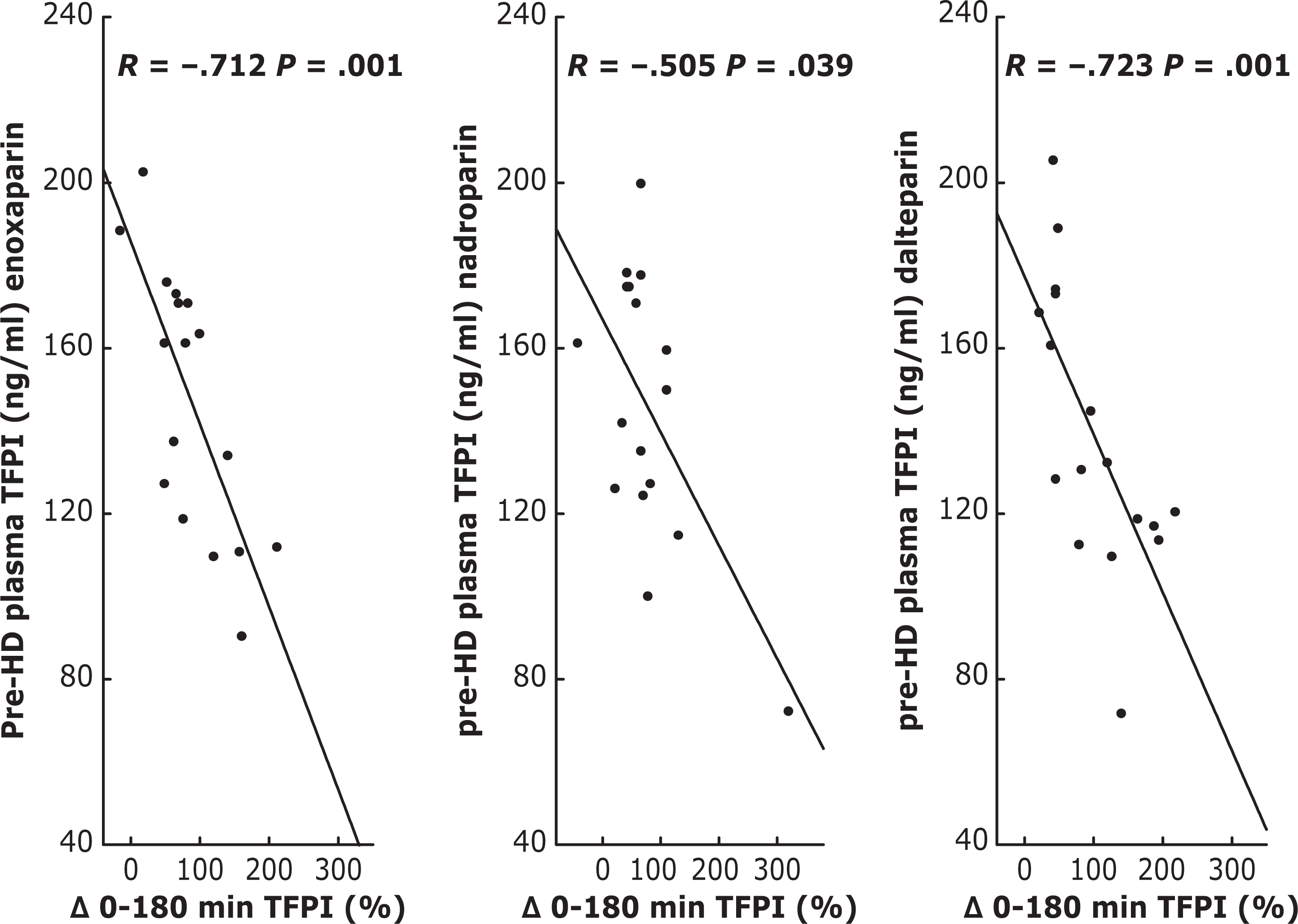
Spearman correlation between total TFPI percentage increase after 180 min of HD and its pre-HD levels under enoxaparin, nadroparin, and dalteparin anticoagulation. TFPI indicates tissue factor pathway inhibitor; min, minutes; HD, hemodialysis. The Δ symbol denotes the change in plasma TFPI levels at 180 minutes of hemodialysis session compared with baseline (T180 to T0).
Prothrombin Fragment 1+2 Levels During Different LMWHs HD
As shown in Table 2 and Figure 2, the plasma PF 1 + 2 levels significantly decreased at T10 during both enoxaparin (χ2 ANOVA = 6.12, P = .047) and nadroparin (χ2 ANOVA = 8.33, P = .015) anticoagulated HD and did not change during dalteparin treatment (χ2 ANOVA = 0.35, P = .838). The baseline plasma PF 1 + 2 levels were comparable under all LMWH treatments.
During enoxaparin treatment, PF 1 + 2 decreased by a median of 22% after 10 minutes (P = .015) and remained still lower after 180 minutes of the session compared to its pre-HD values (P = .031). Their decrement at T10 as well as the values obtained after 180 minutes of HD were associated with the enoxaparin dose/kg (r = −.431, P = .046; r = −.478, P = .049, respectively).
During nadroparin anticoagulated HD, plasma PF 1 + 2 decreased by a median of 18% after 10 minutes (P = .015) and returned to its baseline levels after 180 minutes of HD (P = 0.184). There were no associations between PF 1 + 2 and nadroparin dosage in any period of the study.
Hemodialysis with dalteparin did not affect plasma PF 1 + 2 levels (all Ps > .05). Simultaneously, there were no correlation between their levels and dalteparin dose after both 10 and 180 minutes of HD session.
Platelet-Derived Growth Factor-AB During LMWHs HD
Overdialysis plasma PDGF-AB levels remained stable under enoxaparin (χ2 ANOVA = 1.27, P = .363), nadroparin (χ2 ANOVA = 0.25, P = .899) and dalteparin (χ2 ANOVA = 1.78, P = .344) treatment (Table 2, Figure 2).
The Effect of Heparin Switch
The switch from enoxaparin to nadroparin and dalteparin used as anticoagulants during HD session had no long-range effect on the baseline total TFPI, PDGF-AB, and PF 1 + 2 levels. It was only connected with short-term, overdialytic different effects on TFPI release. Enoxaparin usage was associated with the highest TFPI increment after 10 minutes (χ2 ANOVA = 5.84, P = .048), whereas dalteparin treatment with the highest PF 1 + 2 levels after 10 minutes (χ2 ANOVA = 8.59, P = .014) of HD session (Table 2).
Discussion
Ever since the introduction of LMWHs for clinical use, one of the major questions relates to product interchangeability and differences between each of the individual LMWHs' clinical effects. The novel finding of our study is that a single bolus of enoxaparin (0.75 mg/kg) is the most potent stimuli of dose-dependent TFPI release during HD procedure. Simultaneously, we did not observe any long-term effect of different LMWHs on the baseline total TFPI, PDGF-AB, and PF 1+2 levels in chronically HD patients.
One of the most important actions of heparins is a substantial increase in plasma TFPI levels. 12 Tissue factor pathway inhibitor, the leading inhibitor of TF-initiated coagulation, is a circulating and endothelial-bound serine protease that inhibits factor VIIa/TF complexes in a factor Xa-dependent manner. The underlying mechanism and the vascular sites from which the molecule is released are not quite clear. Heparin has been found to displace TFPI from its endothelial membrane-bound complexes with glycosaminoglycans, that is heparan sulfate, 13 as well as to mobilize TFPI from intracellular stores. 14 However, the platelets can be the potential sources of plasma TFPI as well. We are aware of and have considered the possibility of ex vivo thrombocyte activation; first, because it inevitably occurs during the HD procedure (not fully biocompatible), second, due to the hardly avoidable platelet degranulation during blood sampling. 15 With the regard to the release of TFPI from platelets, there is a general agreement that PDGF-AB is a sensitive marker of platelet activation. 15 Even though it has been known that LMWH causes reduced platelet activation compared to UFH, 16 we have monitored plasma PDGF-AB levels during HD procedure in our patients. As we have already reported, 10 we did not observe any significant PDGF-AB increase during HD procedure, as well as its correlations with overdialytic total TFPI levels. This finding indirectly supports that platelets are not a meaningful source of plasma TFPI during LMWHs anticoagulated HD. Furthermore, once-daily subcutaneous injection of all 3 LMWHs during HD has been shown to result in transiently increased total TFPI levels, which present, however, no effect on freely available, heparin-releasable TFPI store in patients on maintenance HD. This conclusion is based on a strong inverse association between the post-heparin TFPI increments and the baseline levels of the inhibitor, which we have reported earlier in a study with UFH. 8
What is interesting in our study is that single enoxaparin bolus causes the highest plasma total TFPI increase and PF 1 + 2 depletion after 10 minutes of its administration during HD procedure compared with nadroparin and dalteparin. This effect is also dose-dependent. In contrast, during dalteparin treatment, plasma PF 1 + 2 level changes during HD were nearly unnoticeable. These discrepancies may depend on individual heparin properties such as pharmacokinetics. Enoxaparin (~4.2 kd), despite its lower mean molecular weight, offers longer biological half-life compared to nadroparin (~4.5 kd) and dalteparin (~5 kd). 12 The apparent total body clearance of enoxaparin is significantly smaller than those of nadroparin and dalteparin whereas dalteparin apparent clearance is about 1.5-fold greater than that of nadroparin (measured in people in whom prophylactic doses of those LMWHs were used). 12 In healthy people, renal excretion pattern, measured as the percentage of fragments exhibiting an anti-Xa activity which are recovered in urine after subcutaneous heparin administration, is the highest in the case of enoxaparin (6%-8.5%) and the lowest in the case of dalteparin (about 3.4%). Hence, apparent elimination half-life values amount to 4.1 hours for enoxaparin, 3.7 hours for nadroparin, and 2.8 hours for dalteparin. 12 We can only extrapolate these data to patients with maintenance HD, because there have been no such precise studies in patients with end-stage renal disease so far. There is no clear dose-adjustment conversion rate for the stage of chronic kidney disease. Currently, the dosage of heparins has been arbitrarily reduced up to 50% in chronically HD patients.
In conclusion, enoxaparin appears to cause greater TFPI release during HD procedure compared with nadroparin and dalteparin. This phenomenon needs further explanation but adds strongly to the discussion on LMWH interchangeability and proves once again that heparins are not created equal. The switch from enoxaparin to nadroparin and dalteparin used as anticoagulants during HD session has no long-range effect on the baseline total TFPI and PF 1 + 2 levels.
Footnotes
Acknowledgments
We are indebted to the patients of our dialysis unit for their cooperation and to Dr Krystyna Pawlak for her laboratory assistance.
The author(s) declared no conflicts of interest with respect to the authorship and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research and/or authorship of this article: a local grant No 3-54863L from the Medical University of Białystok, Poland. The authors were not financially supported by any manufacturer of LMWHs used.
