Abstract
Inhibitor risk in nonsevere hemophilia A increases with cumulative factor VIII (FVIII) exposure days and high-risk mutations. A standardized approach to minimize inhibitor risk is warranted. Following establishment of a systematic approach to reduce inhibitor risk in nonsevere hemophilia, we evaluated the uptake of these strategies into clinical practice. All adult males with nonsevere hemophilia A followed by British Columbia Adult Hemophilia Program from 2004 to 2016 were included in this retrospective audit. Quality-of-care indicators on inhibitor prevention were examined. Of 108 patients, 18 patients had high-risk FVIII mutations for inhibitor development. Rates of FVIII genotyping and 1-deamino-8-
Introduction
Hemophilia A is an inherited bleeding disorder due to the deficiency in coagulation factor VIII (FVIII) and affects approximately 1 in 10 000 people. Exposure to FVIII may lead to inhibitor development that poses a significant challenge for patients with hemophilia due to intractable bleeding. Patients with nonsevere hemophilia A (baseline FVIII activity 0.02-0.40 IU/mL) were historically thought to have a low cumulative incidence of inhibitor development ranging between 3% and 13%. 1 –3 In the few studies examining risk factors for inhibitors in nonsevere hemophilia A, age at first exposure, 4 age at peak treatment, 4,5 FVIII genotype, 4,6 intensive treatment episodes, 6,7 and immunological danger signals 6 have been implicated as risk factors.
The International Study on etiology of Inhibitors in patients with moderate or mild hemophilia A: influence of Immunogenetic and Hemophilia Treatment (INSIGHT) study is the largest cohort study on risk factors for inhibitors in nonsevere hemophilia. This study has shown that the risk of inhibitor development in nonsevere hemophilia A is much higher than previously thought for certain FVIII mutations and, unlike with severe hemophilia A, continues to rise with increasing number of FVIII exposure days. 8 However, there are no updated guidelines on management of inhibitor risk in nonsevere hemophilia.
It is unclear how the emerging data on inhibitor risk associated with exposure days and high-risk mutations have been translated into clinical practice in hemophilia treatment centers. The British Columbia Adult Hemophilia Program (BC Adult Program) developed a systematic approach to risk stratify and manage the risk of inhibitor development in patients with nonsevere hemophilia A. In this retrospective audit, we evaluated the uptake of these strategies in practice and identified areas for further quality improvement.
Materials and Methods
Setting
The BC Adult Program provides comprehensive care to adult patients with hemophilia over a large geographic catchment area (the province of BC and Yukon Territory) in Vancouver and outreach clinic in 4 other cities throughout the province. Our program typically books comprehensive reviews for patients with moderate hemophilia annually, while mild patients were historically seen intermittently every few years without prespecified frequency, limited by program capacity and patient preference. While we aim to obtain FVIII genotyping in all patients, there were no attempts to examine the specific mutation for risk stratification of inhibitor development prior to 2013. We do not have ongoing access to the number, doses, and dates of factor infusions in patients who are not on home prophylactic factor replacement, as factor concentrates may be prescribed by any physician in the province.
Development of Systematic Approach for Nonsevere Hemophilia A
In the aftermath of the INSIGHT study, we established a systematic approach to risk stratify and reduce the risk of inhibitor development in all patients with nonsevere hemophilia A, focusing on identification and closer surveillance of those with high-risk mutations for inhibitor development as identified by the INSIGHT study.
8
We aim to perform desmopressin (1-deamino-8-
Practice Audit
In this single-center retrospective audit, all adult males older than 19 years with mild (FVIII activity >0.05-0.40 IU/mL) and moderate (FVIII activity 0.01-0.05 IU/mL) congenital hemophilia A seen at least once by the BC Adult Program between January 2004 and November 2016 were included. Patients with a diagnosis of acquired hemophilia or another bleeding disorder were excluded. Hemophilia severity was ascertained based on 1-stage aPTT-based FVIII assay ± 2-stage chromogenic FVIII assay results, if available.
While practice audits aim to measure routine clinical practice against standards of care, there are no standardized guidelines on the optimal strategies to reduce inhibitor risk in nonsevere hemophilia A. As a result, we performed a literature review on current recommendations on inhibitor prevention in nonsevere hemophilia A and developed a list of quality-of-care indicators (Table 1). The audit focused on the processes of care, since the primary outcome of care (ie, inhibitor development) requires a longer follow-up period. We chose to evaluate the current state, instead of the change in performance indicators, before and after our systematic approach to inhibitor prevention. We chose this approach because (1) many of the indicators (eg, rate of documentation of high-risk mutations) were simply not relevant or not performed at all prior to the development of our standardized approach in response to the INSIGHT data and (2) a smaller number of patients were seen both before and after development of our standardized approach.
List of Quality-of-Care Indicators.

Abbreviations: DDAVP, 1-deamino-8-
Demographics, hemophilia and inhibitor history, and potential risk factors for inhibitor development including FVIII mutation, family history, cumulative FVIII exposure days, and the circumstances of treatment were collected from chart review and linkage with the Central Transfusion Registry. Central Transfusion Registry is the first population-based transfusion registry in Canada that contains detailed records of dates, FVIII product type, and dose dispensed in BC and Yukon Territory since 1999. There are no reliable data sources of factor utilization prior to 1999 or for patients who relocated from another province. Patients with either a family history of inhibitors or presence of a high-risk FVIII mutation associated with inhibitor development based on the INSIGHT study 8 and/or FVIII Variant Database 9 were classified as high risk for inhibitor development. Presence of an inhibitor was defined as ≥0.6 Bethesda units on at least 2 consecutive tests. Complete and partial DDAVP responses were defined as 1-hour post-DDAVP FVIII concentration ≥0.5 IU/mL and FVIII ≥ 0.3 IU/mL and at least 2-fold increase from baseline, respectively. 10
Descriptive analyses were performed for baseline characteristics and quality-of-care indicators. Differences in baseline characteristics and potential risk factors for inhibitor development between patients with high-risk mutations and non-high-risk mutations were tested using Mann-Whitney U test for continuous variables and Fisher exact test for categorical variables. Univariate logistic regression models were used to assess the association between potential risk factors for inhibitor development and history of inhibitor. Multivariate logistic regression was not performed due to a small number of events. Research ethics board approval from Providence Health Care Research Institute was obtained (H15-01514).
Results
Baseline Characteristics and Inhibitor Risk Factors
Overall, 108 adult males with nonsevere hemophilia A were included. Most patients (n = 91) attended at least 2 clinic visits. Baseline characteristics and putative risk factors for inhibitor development are summarized in Table 2. Overall, 75 patients had ≥1 exposure day to FVIII concentrates since 1999, and 24 patients had prior exposure to FVIII concentrates or cryoprecipitate, but none documented in the Central Transfusion Registry. Nine patients had never been exposed to cryoprecipitate or FVIII concentrates. Among the patients who had ≥1 exposure day to FVIII concentrates, the median cumulative number of FVIII exposure days from the registry was 16 days.
Baseline Characteristics and Potential Risk Factors for Inhibitor Development.
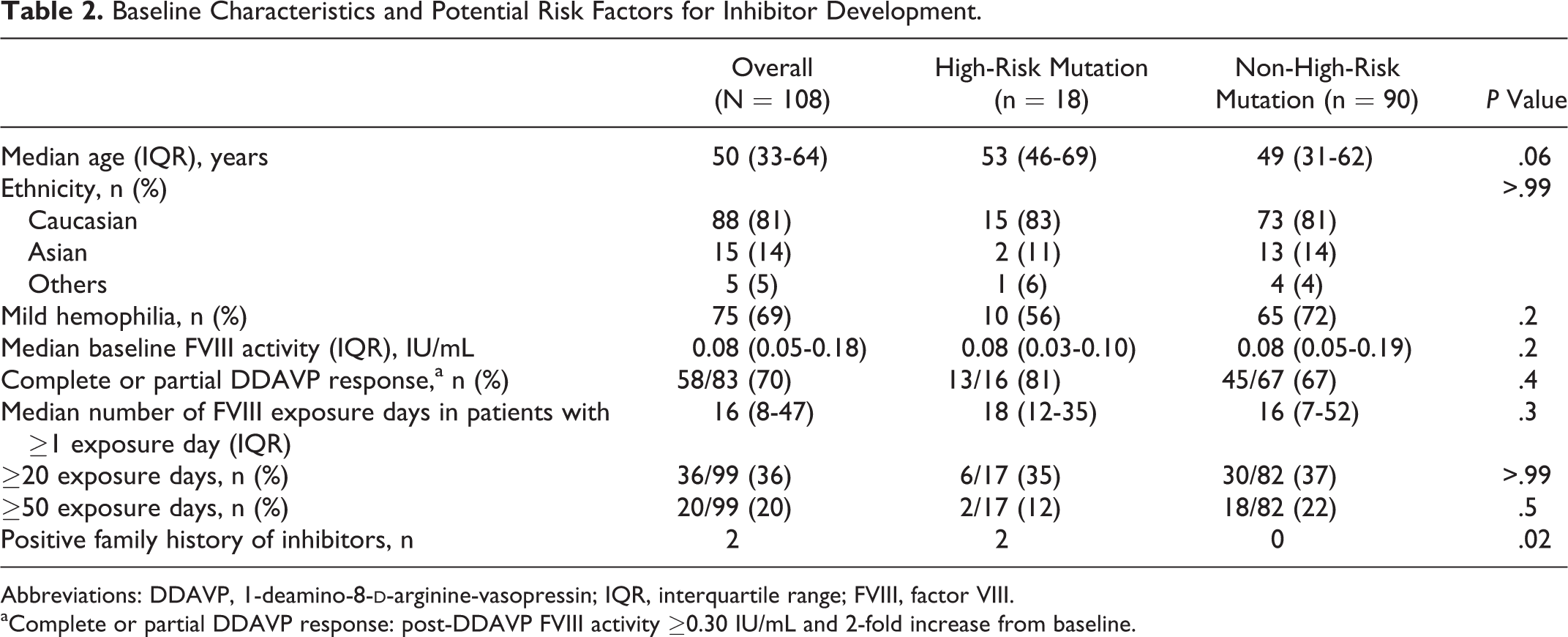
Abbreviations: DDAVP, 1-deamino-8-
aComplete or partial DDAVP response: post-DDAVP FVIII activity ≥0.30 IU/mL and 2-fold increase from baseline.
Among those with available FVIII genotype results (n = 100), 96 patients had missense mutations, 2 had splice site mutations, 1 had small deletion, and 1 had normal FVIII genotype including promoter region and no evidence of von Willebrand disease or FV and FVIII deficiency. Eighteen (18%) patients had FVIII mutations that have been reported to be high-risk mutations for inhibitor development, including Arg2169His (n = 7), Arg612Cys (n = 5), Arg2178Cys (n = 4), and Arg550Cys (n = 2) mutations (Human Genome Variation Society, HGVS number used). Two patients with Arg2178Cys mutation were related. Characteristics of these patients are summarized in Table 3. Of these 18 patients, 15 were seen in clinic within the past 36 months, 2 patients were lost to follow-up, and 1 died. A significant proportion of patients with high-risk mutations had reached ≥20 exposure days (35%) at the time of last follow-up. The majority of mutations were missense mutations (n = 96).
Characteristics of Patients With High-Risk Mutations for Inhibitor Development.
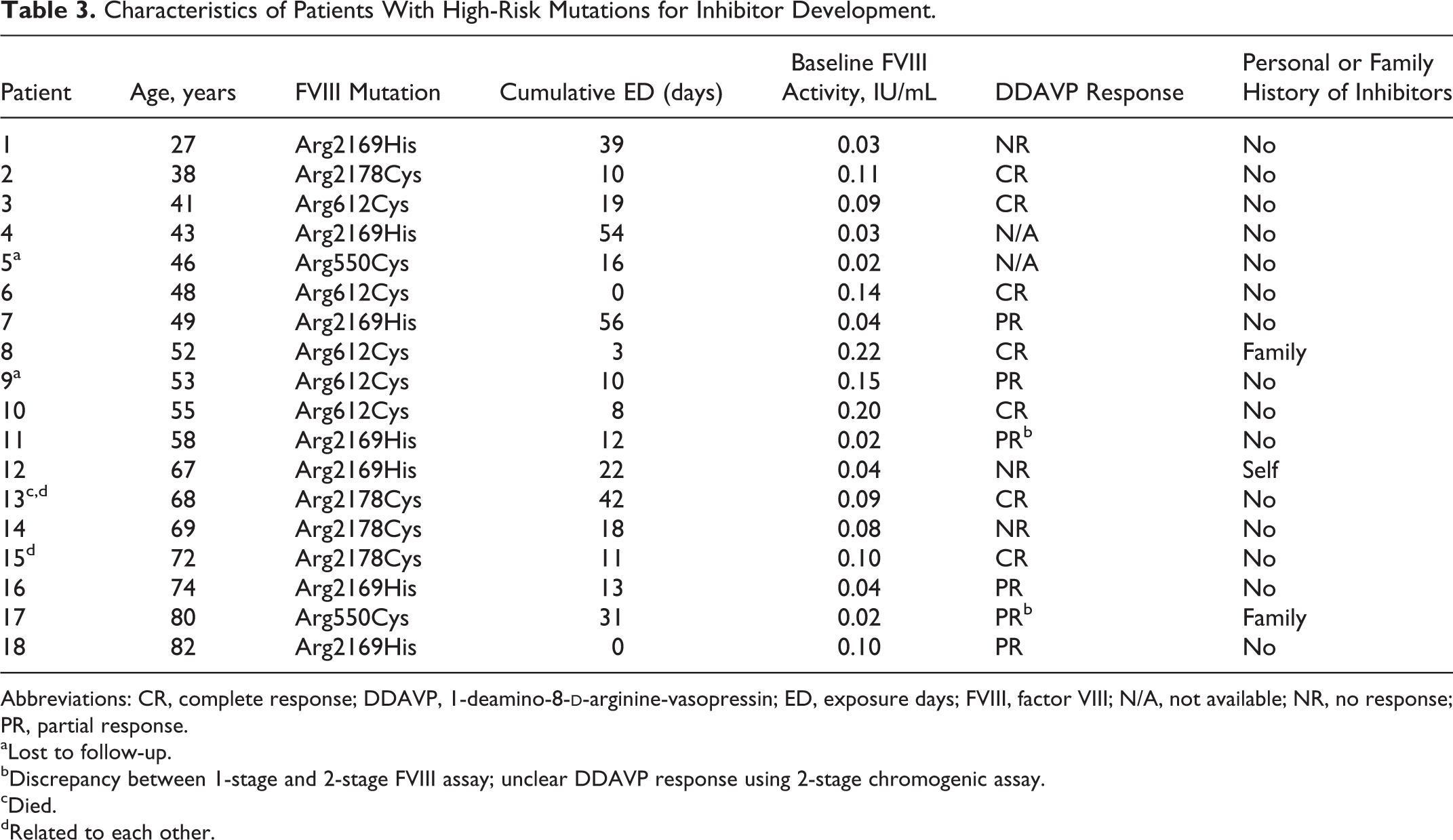
Abbreviations: CR, complete response; DDAVP, 1-deamino-8-
aLost to follow-up.
bDiscrepancy between 1-stage and 2-stage FVIII assay; unclear DDAVP response using 2-stage chromogenic assay.
cDied.
dRelated to each other.
The incidence of inhibitor development was 3.0% (3/99) among all patients who had prior exposure to FVIII products and/or cryoprecipitate. While 1 patient has a high-risk mutation (Arg2169His), the other 2 patients have mutations (C.787+3A>G and Arg1800Cys) that have not previously been associated with inhibitor risk. All 3 patients had high-titer inhibitors that developed in the context of intensive FVIII treatment, at a median age of 40 years (range: 38-55) and a median of 79 exposure days (range: 21-136). Two patients achieved spontaneous remission that was sustained after rechallenge with FVIII products, whereas the other patient developed an anamnestic response following FVIII reexposure.
On univariate logistic regression analysis, age (odds ratio [OR]: 1.03, P = .31), white ethnicity (OR: 5.12, P = .12), number of exposure days (OR: 1.01, P = .09), and high-risk FVIII mutation for inhibitor development (OR: 1.71, P = .65) were not associated with a history of inhibitor development. There was a trend toward higher odds of inhibitor development in those with 50 or more exposure days (OR: 4.77), although not statistically significant (P = .13).
Practice Audit
Table 4 summarizes the quality-of-care measures. The FVIII genotype was available in 93% of patients. Genotyping information was unavailable in 4 patients due to loss to follow-up and in 2 patients due to remoteness from the provincial coagulation laboratory. Of the 75 patients with mild hemophilia A, DDAVP was contraindicated in 7 due to documented history of atherosclerotic disease or multiple vascular risk factors. In the remainder without contraindications, the rate of testing for DDAVP responsiveness was 93%. Four potentially eligible patients did not undergo DDAVP testing due to geographic remoteness. Complete/partial DDAVP response was documented in 58 (70%) of 83 of all patients who underwent DDAVP testing. Of the 18 patients with high-risk mutations, 16 underwent DDAVP testing, of whom 13 (81%) demonstrated either complete (n = 7) or partial DDAVP response (n = 6; Table 3).
Quality-of-Care Indicators in Patients With Nonsevere Hemophilia A.

Abbreviations: DDAVP, 1-deamino-8-
aEither alone or in conjunction with FVIII concentrates.
Among patients with documented complete/partial DDAVP response, 27 patients had procedures since 2013, the year when a systematic approach to inhibitor prevention was initiated. The DDAVP was avoided due to interval development of atherosclerotic diseases since DDAVP testing (n = 4) and unclear chromogenic FVIII response to DDAVP in those with discrepancy between 1-stage and chromogenic FVIII activity (n = 2). The remaining 21 patients with known DDAVP responsiveness had a total of 27 procedures since 2013. The DDAVP was used either alone (n = 8) or in conjunction with FVIII (n = 11) in 19 (70%) procedures. Four procedures were covered with FVIII alone due to unknown or nonsustained DDAVP response at 4 hours, whereas 1 procedure did not require any hemostatic coverage.
Among the 15 patients with high-risk mutation with follow-up within the past 3 years, 87% had been identified to have a mutation associated with inhibitor risk in the last clinic note and 87% had FVIII genotype and inhibitor risk clearly highlighted under a prominent heading along with hemophilia diagnosis (Table 4). Patient education on their individualized inhibitor risk and counseling on judicious use of FVIII products were clearly documented in 12 (80%) patients. The updated information led to documented change in clinical management in 8 (53%) patients, including avoiding intensive FVIII exposure during major procedures and use of adjunctive DDAVP (n = 4), offering drop-in clinic visits to help patients distinguish between acute bleeding and musculoskeletal sources of pain (n = 2), modification of the Factor First card treatment recommendations (n = 2), and advocating surgery to be performed in a center with capacity for timely FVIII monitoring (n = 1).
Development of Systematic Approach to Reduce Inhibitor Risk
In follow-up from this retrospective practice audit, we have proposed systemic-level policies to facilitate optimal management of inhibitor risk in patients with nonsevere hemophilia. A systematic approach to reduce the risk of inhibitor development in nonsevere hemophilia A is outlined in Table 5, including strategies initially implemented by our program in 2013 as well as newly proposed strategies based on existing literature and our opinions. 8,11 –13
Strategies to Stratify and Reduce the Risk of Inhibitor Development.

Abbreviations: DDAVP, 1-deamino-8-
Discussion
While high-risk FVIII mutations and increased number of exposure days have been identified as risk factors for inhibitor development in nonsevere hemophilia A, it is unclear what is the best practice to reduce inhibitor risk on an individual and hemophilia program level. Our large provincial hemophilia program developed a systematic approach to stratify and reduce the risk of inhibitor development and audited our practice to determine whether the strategies have been adopted. Our audit showed a high level of uptake of the systematic approach, as over 80% of patients with high-risk FVIII mutations were identified during clinic visits, received clear documentation of inhibitor risk in the chart, and received individualized education on strategies to reduce inhibitor risk. However, this study is not powered to detect the impact of our systematic approach on the incidence of inhibitor development or to elucidate the predictors of inhibitor development.
Escalating inhibitor risk associated with cumulative FVIII exposure is highly relevant in the aging hemophilia population, as patients are likely to receive more intensive exposures during periods of immunological danger signals due to increased susceptibility for falls, injury, and procedures. In addition, candidacy for DDAVP may decrease with aging as a subset of the patients may develop cardiovascular risk factors and/or overt atherosclerotic diseases. Existing guidelines and expert opinions recommend regular monitoring at a hemophilia treatment center, genetic testing in all cases, DDAVP challenge in all mild patients without contraindications, preferential use of DDAVP in responders either alone or concomitantly to reduce cumulative exposure to FVIII, avoidance of high-risk situations that may lead to inhibitor development, regular inhibitor testing, and accurate record of cumulative FVIII exposure days. 8,11 –13
The availability of both FVIII genotype and detailed cumulative FVIII exposure days is crucial for inhibitor risk stratification in nonsevere hemophilia A. An accurate number of FVIII exposure days is not routinely available in all hemophilia treatment centers. Our study has the advantage of access to a province-wide transfusion registry that collected detailed information on all transfused factor products. The median FVIII cumulative exposure days of 16 days is comparable to the INSIGHT study, although it is underestimated given the age of the cohort and lack of exposure history before 1999. One would expect less exposure to FVIII in patients with high-risk mutations as a result of increased vigilance. This is not observed in our audit possibly due to the short follow-up period since identification of high-risk mutations and a higher proportion of moderate hemophilia in the high-risk mutation group. Furthermore, FVIII exposure is heavily influenced by the number and type of procedures and injuries patients undergo and may not be a good indicator of effective inhibitor prevention strategies.
Routine DDAVP responsiveness testing is important to optimize treatment regimens and potentially reduce inhibitor risk. Several studies reported good DDAVP responsiveness in most mutations associated with higher inhibitor risk. 10,14,15 In our institution, we avoid DDAVP exposure only in those with a documented history of atherosclerotic disease or multiple cardiovascular risk factors but do not use advanced age alone as a contraindication. Our audit demonstrated an excellent rate of DDAVP testing of 93% in patients with mild hemophilia A without contraindications and in 61% of those with moderate hemophilia A. Despite the high prevalence of DDAVP testing, DDAVP was used in only 70% of procedures either alone or in conjunction with FVIII concentrates. This may be assessed in a future audit. Given that the RISE study demonstrated a partial/complete DDAVP response in 21% of patients with moderate hemophilia A, DDAVP should not be completely discarded from the therapeutic armamentarium for moderate hemophilia A, especially as an adjunct agent to reduce FVIII exposure. 16
Implementation of any practice change requires regular clinic attendance, and we have observed suboptimal frequencies of clinic visits and inhibitor screening as per existing guidelines. For instance, the loss to follow-up rate is as high as 40% in young adults with mild hemophilia who transfer from the BC pediatric to adult program in the past 10 years. 17 The United Kingdom Haemophilia Clinic Directors Organization recommended inhibitor testing annually if exposed to FVIII concentrates, following intensive exposure or surgery, and after every exposure for patients with a high inhibitor risk mutation or family history of inhibitors. 13 In a review paper, Castaman and Fijnvandraat recommended inhibitor testing every 6 to 12 months if exposed to FVIII concentrates, preoperatively, and 4 to 6 weeks following intensive treatment. 12 Frequent clinic assessments are crucial, even among those with mild bleeding phenotype, to ensure regular inhibitor surveillance, keep abreast of upcoming procedures, and offer patient education.
There are several limitations to our study. First, our audit used a set of quality indicators that were developed for this study, as there are no validated quality indicators. We have established a systematic approach on the identification and management of patients with nonsevere hemophilia A at high risk of inhibitor development which may help guide future development of standardized performance indicators. Second, the quality of the data is limited by the retrospective nature of our data collection, such as underestimation of the cumulative number of exposure days and potential underdocumentation of certain quality indicators. Third, inhibitor screening was often performed opportunistically in the past, coinciding with the time of clinic visits. This may underestimate the prevalence of transient low-titer inhibitors, although the prevalence of clinically relevant inhibitors is likely not affected.
In conclusion, our study reports a standardized approach to stratify and reduce the risk of inhibitor development for nonsevere hemophilia A. We have demonstrated a high rate of adoption and have identified areas for quality improvement and the need for changes from a system level. Future prospective studies are required to evaluate the impact of a systematic strategy on performance indicators and the risk of inhibitor development.
Footnotes
Authors’ Note
H.S. contributed to the study design, data collection, data analysis, and data interpretation and wrote the first draft of the manuscript. S.C. contributed to study design and data collection and critically reviewed the manuscript. P.Y. contributed to data interpretation and critically reviewed the manuscript. S.J. contributed to the study conception and design, provided critical guidance during the project, and critically reviewed the manuscript.
Acknowledgments
The authors thank Karen Sims for reviewing the manuscript.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: P.Y. has received honoraria for speaking and consulting (advisory boards) from Octapharma, Alexion pharmaceuticals, and Pfizer.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: H.S. was supported by the University of British Columbia Clinician Investigator Programs. S.C. was supported by the Centre for Blood Research Summer Student Research Award.
