Abstract
Anti-beta-2-glycoprotein 1 (anti-β2GP1) antibodies are associated with increased thrombotic risk in patients with autoimmune disease. There is conflicting evidence on the effects of anti-β2GP1 antibodies on platelets, with both enhanced and inhibited aggregation previously reported. However, previous studies did not include isotype antibodies to ensure the observed effects were due to anti-β2GP1 antibodies. The aims of this study were to (1) investigate the effects of anti-β2GP1 antibodies on collagen-induced platelet aggregation in parallel with negative control (buffer normal saline) and isotype control antibodies and (2) determine the lupus anticoagulant (LA) activity of anti-β2GP1 antibodies used. Three animal-derived anti-human-β2GP1 antibodies (1.25, 2.5, and 5 μg/mL) incubated with healthy platelet-rich plasma were activated by collagen (2.5 μg/mL). Each anti-β2GP1 antibody demonstrated the inhibition of aggregation compared to negative control, but not to isotype control. No anti-β2GP1 antibody demonstrated LA activity, suggesting they were probably nonpathological. This study highlights both negative and isotype control markers are important to validate the effects of anti-β2GP1 antibodies. Assays to measure anti-domain I-β2GP1 antibodies are recommended to be used in conjunction with functional measures to further investigate the effects of anti-β2GP1 antibodies.
Keywords
Introduction
Anti-beta-2-glycoprotein 1 (anti-β2GP1) antibodies are a type of antiphospholipid antibody that include a group of heterogeneous immunoglobulins (Igs) such as anticardiolipin antibodies and lupus anticoagulants (LAs). 1 –3 The presence of anti-β2GP1 antibodies is associated with an increased risk of thrombosis and/or pregnancy-related complications in patients with autoimmune conditions, for example, antiphospholipid syndrome (APS) and systemic lupus erythematosus (SLE). The risks are even higher in patients with persistently high titers of antibodies and/or positivity for LA activity, 4 –6 although the precise mechanism(s) remain to be determined.
Anti-β2GP1 antibodies have been suggested to activate platelets in the presence of β2GP1 via various platelet membrane receptors, including apolipoprotein E receptor 2′ (apoER2′), 7 glycoprotein Ibα (GPIbα), 8 and the Fc Receptor for IgG, (FcγRIIa). 9 However, there are conflicting reports on the effects of anti-β2GP1 antibodies on platelet activation and function using adenosine diphosphate (ADP) and collagen as agonists in in vitro systems. Both murine monoclonal 10 and purified human anti-β2GP1 antibodies from patients with SLE 1 have been demonstrated to enhance ADP-induced platelet aggregation, suggesting a prothrombotic role for anti-β2GP1 antibodies. In contrast, affinity-purified rabbit anti-β2GP1 antibodies significantly inhibited ADP-induced platelet aggregation, in a concentration-dependent manner. 1,3 Furthermore, anti-β2GP1 IgG antibodies in serum from patients with APS have also demonstrated the inhibition of ADP-induced in vitro platelet aggregation. 11 Using collagen as an agonist in vitro platelet aggregation was not affected by human serum containing anti-β2GP1 IgG antibodies 11 but enhanced by rabbit-derived anti-β2GP1 IgG antibodies. 3 These discrepant results may be due to the heterogeneous properties of anti-β2GP1 antibodies, varying avidity, and/or different binding specificities.
Previous studies investigating the effects of anti-β2GP1 antibodies on platelets 1,3,11 have not reported isotype controls used in parallel to ensure that the inhibitory or pro-aggregatory effects were due to the antibodies. Furthermore, they did not determine whether the anti-β2GP1 antibodies demonstrated LA activity, an established laboratory marker associated with a higher risk of thrombosis in patients with APS and SLE. 4,5,12 The aims of this study were to therefore (1) investigate the effects of 3 anti-β2GP1 antibodies (and paired isotype control antibodies) on collagen-induced in vitro platelet aggregation and (2) determine the LA activity of anti-β2GP1 antibodies.
Methods
Ethics
This study was approved by the Tasmanian Health and Medical Human Research Ethics Committee (Reference number H0015749). All participants completed an informed written consent prior to taking part in the study.
Participants
Healthy donors (n = 24; age, mean ± SD = 30 ± 10 years) without any known hemostatic abnormality were recruited for the study. Participants avoided aspirin and other antiplatelet medications for 10 days prior to blood collection. They also avoided food and beverage known to affect platelet function, for example, garlic, coffee, and alcohol 13,14 for at least 1 day before blood donation.
Antibodies
Commercially available animal-derived IgG antibodies were used in this study (Table 1). Anti-human-β2GP1 IgG antibodies used included goat polyclonal (Bethyl Laboratories Inc, Montgomery, Texas), rabbit polyclonal (Hyphen-BioMed, Neuville-sur-Oise, France), and rabbit monoclonal (Abcam, Cambridge, United Kingdom) antibodies. Isotype control IgG antibodies including goat polyclonal (Bethyl Laboratories Inc), rabbit polyclonal (Thermo Scientific, Waltham, Massachusetts), and rabbit monoclonal (Abcam) antibodies, respectively, were used in parallel.
Commercially Available IgG Antibodies Used in the Current Study.
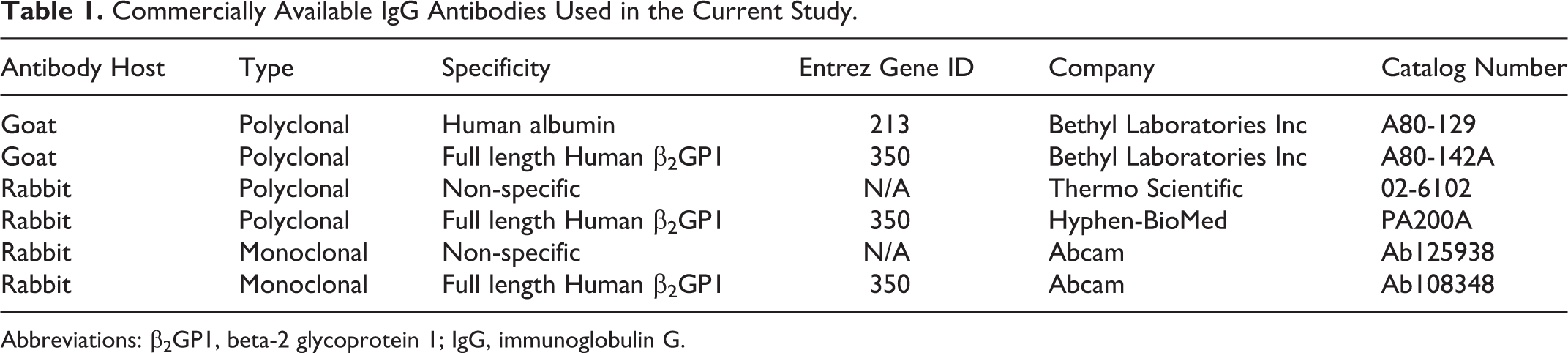
Abbreviations: β2GP1, beta-2 glycoprotein 1; IgG, immunoglobulin G.
Platelet Aggregation
Whole blood samples from healthy donors were collected into 3.8% sodium citrate vacutainer (Vacuette, Greiner Bio-One, Kremsmünster) with minimal stasis and centrifuged at 150g for 10 minutes at room temperature (RT). After separating platelet-rich plasma (PRP), the remaining sample was centrifuged at 2000g for 20 minutes at RT to collect platelet-poor plasma (PPP). For all tests, the platelet count of PRP was adjusted to 250 × 109/L using PPP from the same donor (measured by Sysmex XS 1000i; Sysmex, Lincolnshire, Illinois). Experimental testing was completed within 3 hours of blood collection.
Platelet aggregation was performed using a 4-channel AggRAM module light transmission aggregometer (Helena Laboratories, Beaumont, Texas). Aggregation was induced by 2.5 μg/mL type I equine tendon collagen (25 μL; Helena Laboratories). Two hundred microliters of PRP was first incubated with 25 μL of commercially available anti-β2GP1 antibody or isotype control antibody (final concentrations 5, 2.5, and 1.25 μg/mL) for 10 minutes at 37°C prior to activation by collagen. Each concentration of antibody was tested in triplicate (PRP + antibody), and a baseline control (PRP + buffer normal saline, pH 7.1) was included in each run (4 channels: 3 tests + 1 control). Each experiment was repeated using PRP from 4 different healthy donors. Aggregation software (HemoRAM 1.1.0; Helena Laboratories) was used to generate aggregation parameters including percentage maximum aggregation (%MAX), percentage area under curve (%AUC), and lag time. Results for baseline control were normalized to 0. Differences between the results from antibodies and baseline were then determined.
Lupus Anticoagulant Activity
STA-Coag lyophilized control citrated normal human plasma (NP; Diagnostica Stago, Paris, France) was reconstituted with distilled water and allowed to stand at RT for 30 minutes before testing. The LA 1 screening and LA 2 confirmation reagents (Siemens Healthcare Pty Ltd, Victoria, Australia) were reconstituted with distilled water, left at RT for 15 minutes, and then prewarmed at 37°C prior to testing. The LA testing was performed according to the manufacturer’s instructions (Siemens Healthcare Pty Ltd). Briefly, NP was incubated for 10 minutes with different commercially available anti-β2GP1 antibodies (final concentrations 5, 2.5, and 1.25 μg/mL). Siemens LA 1 screening reagent (100 μL) containing Russell’s viper venom and phospholipids was added to 100 μL of plasma spiked with antibodies. Antibody-free NP and high and low LA controls (100 μL) were also assessed. All clotting times were measured using a STart coagulation analyzer (Diagnostica Stago, Paris) in triplicate. Samples with a “screen” (S) clotting time greater than 44 seconds were retested with LA 2 confirmation reagent (containing higher amounts of phospholipid) to obtain a “confirm” (C) clotting time. The normal ratio of S/C was 0.8 to 1.2. The ratios for weak, moderate, and strong LA activities are 1.2 to 1.5, 1.5 to 2.0, and greater than 2, respectively.
Statistical Analysis
All figures were constructed using GraphPad Prism 7 (GraphPad Software, Inc, La Jolla, California), with platelet aggregation parameters (%MAX, %AUC, and lag time) expressed as the mean of difference from baseline (normal buffered saline) ± standard error of the mean. Data were analyzed using mixed-effects general linear modeling adjusted for repeated measures (STATA version 14; StataCorp, College Station, Texas). Results were tested for assumptions of linear regression. If violated, data were analyzed again with repeated-measures ordinal logistic regression. All P values were adjusted using Holm post hoc test. P values less than .05 were considered as statistically significant. Data from anti-β2GP1 antibodies were first compared against baseline. If statistically significant, anti-β2GP1 antibodies results were compared to their isotype control antibody to ensure those effects were specific for anti-β2GP1 antibodies.
Results
Effects of Goat Polyclonal Anti-β2GP1 IgG Antibodies on 2.5 μg/mL Collagen-Induced Platelet Aggregation Against Baseline and Isotype Control Antibodies
At all concentrations (1.25, 2.5, and 5 μg/mL) tested, goat polyclonal anti-β2GP1 antibodies inhibited 2.5 μg/mL collagen-induced platelet aggregation (%MAX and %AUC) compared to baseline control (Figure 1A, B). Stronger inhibition of aggregation was demonstrated with increasing concentrations of anti-β2GP1 antibodies (eg, %MAX for 1.25, 2.5, and 5 μg/mL was −4.98% ± 0.99%, P < .01; −5.81% ± 1.00%, P < .01; and −6.94% ± 1.01%, P < .01, respectively). The effects of anti-β2GP1 antibodies on lag time were the same as baseline control (Figure 1C). Although goat polyclonal anti-β2GP1 antibodies showed inhibitory effects compared to baseline, these were not significantly different to the isotype control antibodies (Figure 1A to C).
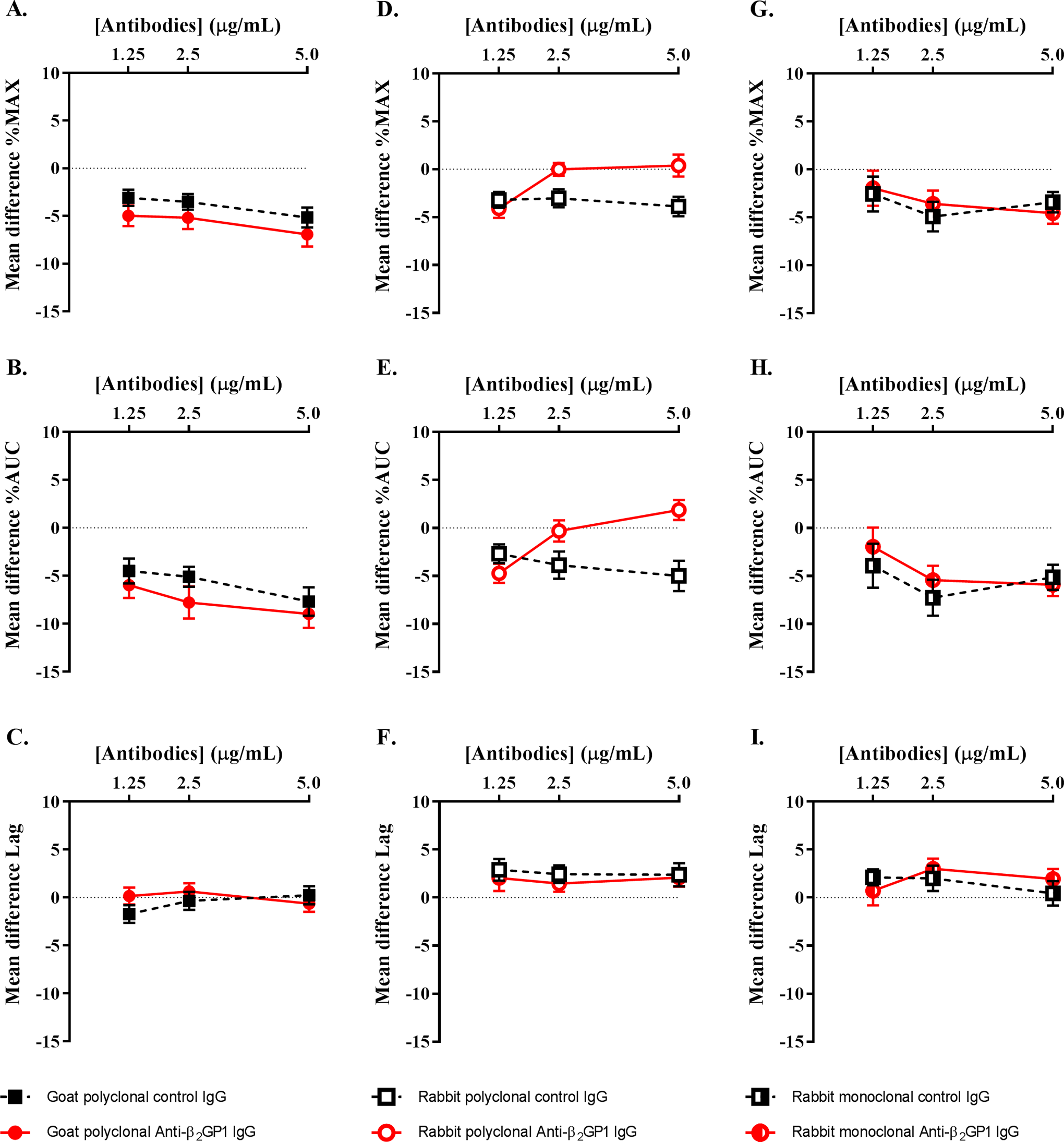
Effects of commercially available animal-derived polyclonal and monoclonal IgG antibodies on 2.5 μg/mL collagen-induced platelet aggregation. Horizontal dotted lines represent the baseline (PRP + buffered normal saline only). For each antibody, results are presented as the mean difference against baseline ± standard error of the mean from 4 separate experiments. All 3 animal-derived anti-human-β2GP1 antibodies had inhibitory effects compared to baseline control on 2.5 μg/mL collagen-induced platelet aggregation. However, these effects parallel with their isotype control antibodies. β2GP1 indicates beta-2-glycoprotein 1; IgG, immunoglobulin G; PRP, platelet-rich plasma.
Effects of Rabbit Polyclonal Anti-β2GP1 IgG Antibodies on 2.5 μg/mL Collagen-Induced Platelet Aggregation Against Baseline and Isotype Control Antibodies
Rabbit polyclonal anti-β2GP1 antibodies (1.25 μg/mL) significantly inhibited %MAX (−4.08% ± 0.93%, P < .01) and %AUC (−4.71% ± 1.22%, P < .01) compared to baseline control; however, their effects on lag time were the same as baseline (Figure 1D to F). There was also no significant difference between the inhibitory effects of rabbit polyclonal anti-β2GP1 and the isotype control antibodies (Figure 1D to F).
Effects of Rabbit Monoclonal Anti-β2GP1 IgG Antibodies on 2.5 μg/mL Collagen-Induced Platelet Aggregation Against Baseline and Isotype Control Antibodies
Rabbit monoclonal anti-β2GP1 antibodies (1.25 μg/mL) did not show a significant difference compared to baseline; however, 2.5 and 5 μg/mL concentrations of anti-β2GP1 antibodies significantly inhibited %MAX (−3.57% ± 1.27%, P < .01 and −4.56% ± 1.27%, P < .01, respectively) and %AUC (−5.43% ± 1.56%, P < .01 and −5.91% ± 1.56%, P < .01, respectively; Figure 1G, H). Furthermore, 2.5 μg/mL of rabbit monoclonal anti-β2GP1 antibodies prolonged the lag time of aggregation (3.01 ± 1.35 seconds, P = .03; Figure 1I). The effect of rabbit monoclonal anti-β2GP1 antibodies on platelet aggregation was similar to their corresponding isotype antibodies (Figure 1G to I).
Effects of Anti-β2GP1 Antibodies on LA Activity of NP
Goat polyclonal, rabbit polyclonal, and rabbit monoclonal anti-β2GP1 antibodies did not demonstrate the LA activity (Table 2).
Lupus Anticoagulant (LA) Activity of Commercially Available Anti-β2GP1 Antibodies, Lupus Patients’ IgG Fractions and Plasma.
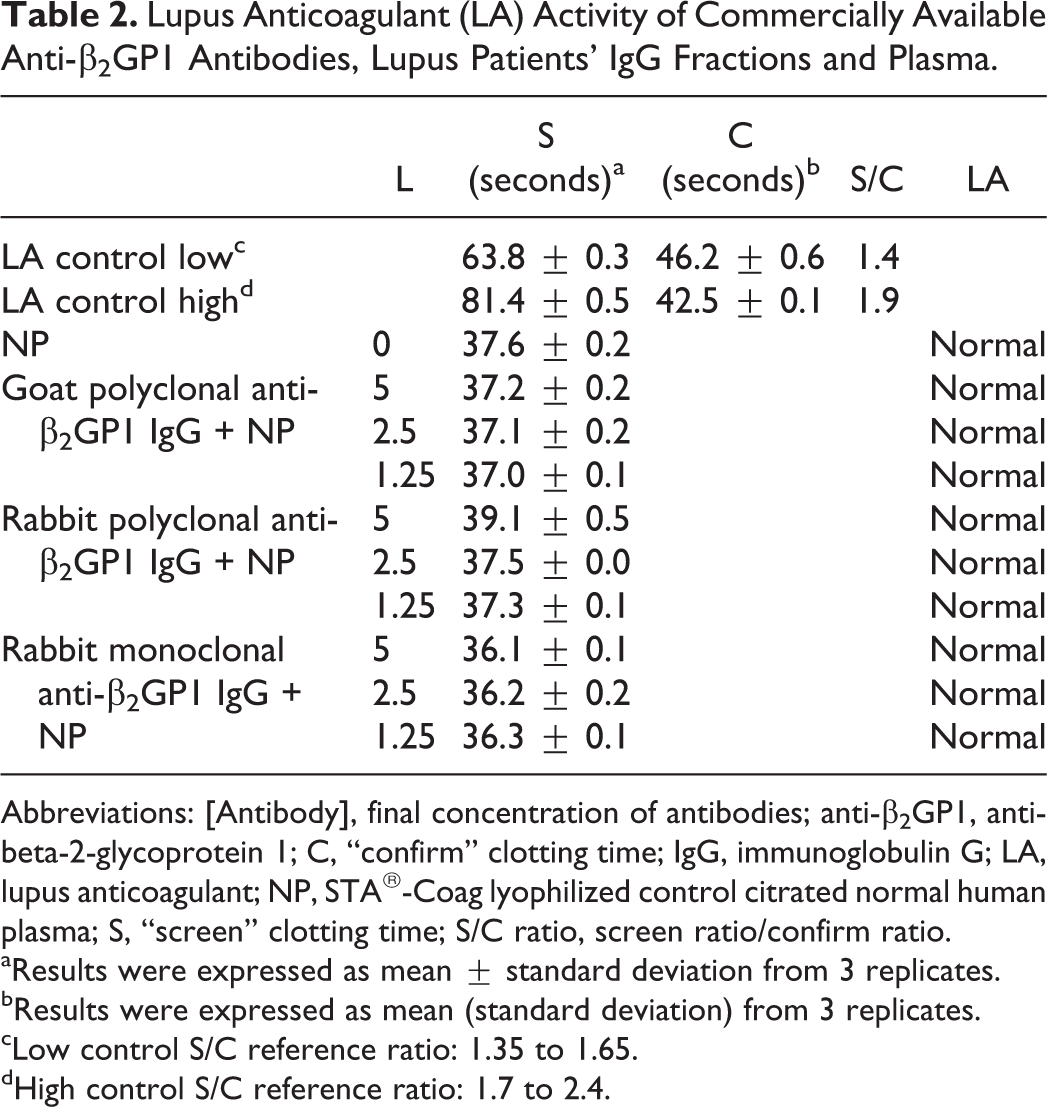
Abbreviations: [Antibody], final concentration of antibodies; anti-β2GP1, anti-beta-2-glycoprotein 1; C, “confirm” clotting time; IgG, immunoglobulin G; LA, lupus anticoagulant; NP, STA®-Coag lyophilized control citrated normal human plasma; S, “screen” clotting time; S/C ratio, screen ratio/confirm ratio.
aResults were expressed as mean ± standard deviation from 3 replicates.
bResults were expressed as mean (standard deviation) from 3 replicates.
cLow control S/C reference ratio: 1.35 to 1.65.
dHigh control S/C reference ratio: 1.7 to 2.4.
Discussion
This study has demonstrated that the 3 commercially available anti-β2GP1 antibodies inhibited in vitro collagen-induced (2.5 μg/mL) platelet aggregation, compared to baseline control (4% to 9% inhibition; %MAX and %AUC). These antibodies, however, produced similar effects in comparison with the isotype control antibodies investigated in parallel. Furthermore, the 3 anti-β2GP1 antibodies did not demonstrate the LA activity. Taken together, these results suggest that the anti-β2GP1 antibodies used in the current study may not be suitable for the in vitro investigation of mechanisms of diseases where anti-β2GP1 antibodies are implicated in the pathophysiology, such as APS and SLE.
To our knowledge, only 2 published studies have previously investigated the effects of anti-β2GP1 antibodies on collagen-induced platelet aggregation. 3,11 The relatively small amount of inhibition of aggregation by anti-β2GP1 antibodies compared to baseline demonstrated in the current study is similar to a previous study that investigated human serum containing anti-β2GP1 IgG antibodies. 11 However, this is in contrast to a previous study by our group 3 that demonstrated increasing concentrations of rabbit polyclonal anti-β2GP1 IgG antibodies enhanced collagen-induced platelet aggregation (%AUC) compared to baseline control. As the changes to aggregation in the presence of anti-β2GP1 antibodies compared to baseline control are relatively small, this suggests that there may be some variability between sources of antibodies, particularly binding strengths and/or specificities. Furthermore, the very similar effects demonstrated by the anti-β2GP1 antibodies used in the current study and their paired isotype controls provide further evidence that these commercially available anti-β2GP1 antibodies probably do not have a significant effect on collagen-induced in vitro platelet aggregation.
It was also interesting to observe that none of the 3 commercial anti-β2GP1 antibodies demonstrated the LA activity, as determined by the dilute Russell viper venom coagulation test. 5 This suggests that the antibodies used in the current study had the characteristics of nonpathological “type B” anti-β2GP1 antibodies, as opposed to “type A” pathological antidomain (D) I-β2GP1 antibodies that have been implicated in patients with APS and SLE. 15,16 Furthermore, the variable effects of patient (SLE)-derived anti-β2GP1 and rabbit anti-β2GP1 antibodies are consistent with data from a previous study that investigated their effects on ADP-induced platelet aggregation. 1 Anti-β2GP1 antibodies purified from 2 patients with SLE demonstrated the concentration-dependent (1.25 to 10 µg/mL) increases in platelet aggregation (%MAX, %AUC) in the presence of 2.5 and 5 µM ADP, compared to the inhibitory effects of 10 µg/mL rabbit anti-β2GP1. 1
The data presented in this study raise doubt about the suitability of using animal-derived anti-β2GP1 for research studies into the mechanisms of action of anti-β2GP1 antibodies. The characteristics of animal-derived anti-β2GP1 antibodies may vary as they are dependent on the structure of β2GP1 used to immunize animals. In healthy human plasma, most β2GP1 is present in a circular conformation. 17 Animals immunized with circular β2GP1 are less likely to generate type A anti-DI-β2GP1 antibodies as the cryptic epitope in DI is hidden by DV. 17 This is in contrast to when the cryptic epitope is/remains exposed when circular-shaped β2GP1 transforms into the J-shaped conformation. Yet, transformation is only favored in β2GP1 with increased oxidation 18 or sialylation of glycan structure, 19 normally found in patients with APS and SLE. Alkaline pH, 17 high salts, 17 and β2GP1 concentration 20 also promote transformation, but these conditions can only be generated in vitro.
Anti-β2GP1 IgG antibody subclasses have varying pathogenicities. 21 Anti-β2GP1 IgG2 and IgG3 antibodies are found in patients with APS and healthy children, respectively. Yet, IgG3 antibodies activate C3c to enhance the clearance of anti-β2GP1–β2GP1 complexes and protect children from complications. 21,22 Therefore, it is possible that commercially available animal-derived anti-β2GP1 antibodies are nonpathological type B IgG3 antibodies. The targeted epitope and subclasses of animal-derived antibodies used in future studies need to be clarified to better correlate with in vivo pathological manifestations.
Similarly, the source of collagen used to investigate the effects of anti-β2GP1 antibodies on platelets may affect results. The type I equine tendon collagen (Helena Laboratories) used in this study to activate platelets in the presence of anti-β2GP1 antibodies contained a substantial amount of von Willebrand factor (vWF). 23 The presence of vWF in this reagent acts synergistically with collagen to increase the potency of collagen in supporting platelet adhesion and aggregation. 23 We attempted to use 2 other sources of collagen (calf skin type I acid soluble collagens, C3511 and C9791, Sigma-Aldrich, St Louis, Missouri) in the current study, but neither was able to induce platelet aggregation in a range of healthy donors (data not shown). Whether the results in the current study were due to pathways mediated by vWF, collagen, or both was therefore unable to be determined.
Finally, it is possible that the PRP-based in vitro aggregation system used in the study is not suitable to investigate the effects of anti-β2GP1 antibodies on platelets. 10,24 In vivo, platelets expose negatively charged phospholipid after adherence to collagen, allowing transformation of circular β2GP1 to J-shaped and formation of complexes. Using a PRP-based in vitro aggregation system, collagen and platelets in a fluid phase are not exposed to damaged endothelial cells. Thus, exposure of negatively charged platelet phospholipids only occurs to a minor extent at the end of the aggregation process. 24 The anti-β2GP1–β2GP1 complexes might not be formed and therefore have no effect on platelet aggregation. It may be that anti-β2GP1–β2GP1 complexes only promote platelet interaction with collagen under more physiological flow conditions. 25 Moreover, anti-β2GP1 antibodies may require additional exogenous β2GP1 to activate platelets. 26
Conclusion
This study demonstrated that the 3 commercially available animal-derived anti-human-β2GP1 antibodies did not affect collagen-induced in vitro platelet aggregation, relative to their respective isotype controls. The antibodies used were probably nonpathological, type B anti-β2GP1 antibodies as they had no significant effect on collagen-induced in vitro platelet aggregation and did not demonstrate LA activity. This study highlights the importance of using both isotype antibodies and negative controls in parallel to validate the inhibitory or pro-aggregatory effects of anti-β2GP1 antibodies. This study has also highlighted that caution is required when interpreting the effects of anti-β2GP1 antibodies on platelets in clinical conditions, as the effects are dependent on their targeted domain epitope. Further studies are required to use anti-β2GP1 antibodies with known targeted epitopes, for example, immunoassays, to measure anti-DI-β2GP1 antibodies to better correlate in vitro results with clinical manifestations.
Footnotes
Acknowledgments
The authors would like to thank Jane Pittaway and Merrilyn Johnson for their assistance with blood collection and processing, Malena Hyde for her technical support, and all study participants for their time and blood donations.
Declaration of Conflicting Interests
The author(s) declared no potential conflict of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Lupus Association of Tasmania and the Clifford Craig Medical Research Trust, Launceston, Tasmania, Australia.
