Abstract
When patients on antiplatelet therapy (APT) require minor invasive surgery, APT is usually continued to limit the risk of thrombosis. However, the possibility of hemostatic difficulties necessitates the monitoring of platelet aggregation to prevent unexpected bleeding. We examined whether whole blood aggregometry as a point-of-care testing (POCT) could be useful as a tool for predicting hemostatic difficulties. Sixty-five patients receiving APT and 15 patients who were not receiving APT were enrolled in the present study; all patients were scheduled to undergo a tooth extraction. Whole blood samples were obtained and were examined using multiple electrode aggregometry. The aggregometry was performed using arachidonic acid (AA), adenosine diphosphate (ADP), and thrombin receptor activating peptide. Hemostatic difficulty was defined as a need for more than 10 minutes of compression to achieve hemostasis. The AA test results were significantly lower in patients treated with aspirin (control: 97.7 [29.0] U, aspirin: 14.5 [7.2] U, P < .001). The ADP test results were also significantly lower in patients treated with a P2Y12 inhibitor (control: 77.7 [21.7] U, P2Y12 inhibitor: 37.3 [20.4] U, P < .01). Six of the examined cases exhibited hemostatic difficulties. The cutoff values for the prediction of hemostatic difficulty were 16.5 U for the AA test (sensitivity, 0.833; specificity, 0.508) and 21 U for the ADP test (sensitivity, 0.847; specificity, 0.500). Our study showed that whole blood aggregometry was useful as a POCT for the prediction of hemostatic difficulties after tooth extraction in patients receiving APT.
Introduction
Regular prescriptions for antiplatelet drugs are not usually suspended before dental extraction because hemostasis can normally be achieved in most cases. 1 In rare cases, however, hemostasis can be difficult after extraction. 2 Since antiplatelet function is not routinely monitored for dose titration, the dose is usually fixed; occasionally, this situation can lead to an overdose. Sensitivity to antiplatelet therapy (APT) varies individually 3,4 and is genetically determined. 5 In a certain fraction of patients, platelet function is severely inhibited. 6 The popularity of coronary stent placement has led to an increasing number of individuals who are being treated with APT; consequently, the incidence of hemostasis difficulty has also been increasing. The use of point-of-care testing (POCT) for the safe management of outpatients has been attracting attention. 7 Platelet aggregation testing is typically performed in laboratories using a light transmission aggregation test. The use of an aggregometer, which utilizes the whole blood impedance method, as a POCT is expected to help predict hemostasis difficulties and to contribute to the safe management of patients requiring invasive treatments. 8 Here, we performed platelet aggregometry in patients who were or were not receiving APT and examined the relationship between hemostasis difficulty after tooth extraction and the pre-extraction aggregometry value.
Materials and Methods
This study protocol was approved by the ethical committee of Showa University (2011-044) and registered with University Hospital Medical Information Network (UMIN) (UMIN000007480). Sixty-five patients receiving APT were enrolled between April 2012 and March 2015; all patients were informed of the nature of this study and provided written consent. These patients were taking the recommended doses of each drug (aspirin, 100 mg; clopidogrel, 75 mg; ticlopidine, 100 mg; cilostazol, 100 or 200 mg), and exceptional doses were not given to any of the patients. Fifteen patients who were not receiving APT and who provided informed consent were assigned to the control group. No congenital or acquired coagulation disorders were present in either the control group or the patients.
A whole blood sample (5 mL) was obtained from each of the enrolled patients prior to tooth extraction. Hirudin (45 μg for 3 mL sample; Verum Diagnostica GmbH, Munich, Germany) was used as an anticoagulant because this thrombin inhibitor enables anticoagulation without influencing the physiological calcium levels. The blood samples were then processed using multiple electrode aggregometry (MEA, Multiplate Analyzer; Roche Diagnostics International Ltd, Risch-Rotkreuz, Switzerland). Multiple electrode aggregometry was induced using arachidonic acid (AA; ASPI test; Dynabyte Informationssysteme GmbH, Munich, Germany), adenosine diphosphate (ADP test; Triolab AS, Brøndby, Denmark), or thrombin receptor activating peptide (TRAP). The final concentration of each agent was 0.5 mM for the AA test, 6.5 μM for the ADP test, and 32 μM for the TRAP-6 test. Since aspirin acts on cyclooxygenase, its activity can be monitored using the AA test. Meanwhile, clopidogrel and ticlopidine antagonize the P2Y12 ADP receptor; thus, their activities can be monitored using the ADP test. The TRAP test theoretically shares a pathway with the AA test and the ADP test. We used U (Area Under the Curve [AUC]/10) as a quantitative unit to express the AUC of each test, as per the manufacturer’s recommendation.
Tooth extraction was performed under local anesthesia by an oral surgeon at Showa University Dental Hospital. Two percent lidocaine with 1:80 000 epinephrine was used as an infiltration anesthesia in all the control and APT treatment group patients, except in 5 patients in the APT treatment group who were treated with 3% prilocaine with 0.54 μg/mL of felypressin. The surgeons were asked to apply compression to the area of extraction for 10 minutes; if bleeding was subsequently confirmed, compression was again applied, and so on. Hemostasis difficulty was defined as a need for compression hemostasis lasting more than 10 minutes.
Several other parameters were also recorded to explore their influence on hemostatic difficulty, such as systemic and diastolic blood pressure, tooth mobility, radiolucency of root apex, swelling, pus discharge, use of forceps, use of an elevator, and need for sutures.
Statistical Analysis
Differences in the AA test or ADP test results between the control group and the AA-sensitive or ADP-sensitive group were statistically analyzed using a t test. A multivariate logistic regression analysis was used to extract significant predictors of hemostatic difficulty. The statistical software SPSS version 17 (SPSS Inc, Chicago, USA) was used for all calculations and to draw Receiver Operating Characteristic (ROC) curves. The cutoff values for the use of each test as a predictor of hemostatic difficulty were then explored based on the resulting ROC curves that were calculated according to the Youden index.
Statistical significance was defined as P < .05. All the values were expressed as mean (SD).
Results
Fifteen control patients and 65 APT patients were enrolled. No significant differences in background parameters were seen between the 2 groups (Table 1). Aspirin was the most frequently prescribed APT, followed by clopidogrel (Table 2). Fifteen cases were receiving dual antiplatelet therapy (DAPT).
Patient Characteristics.
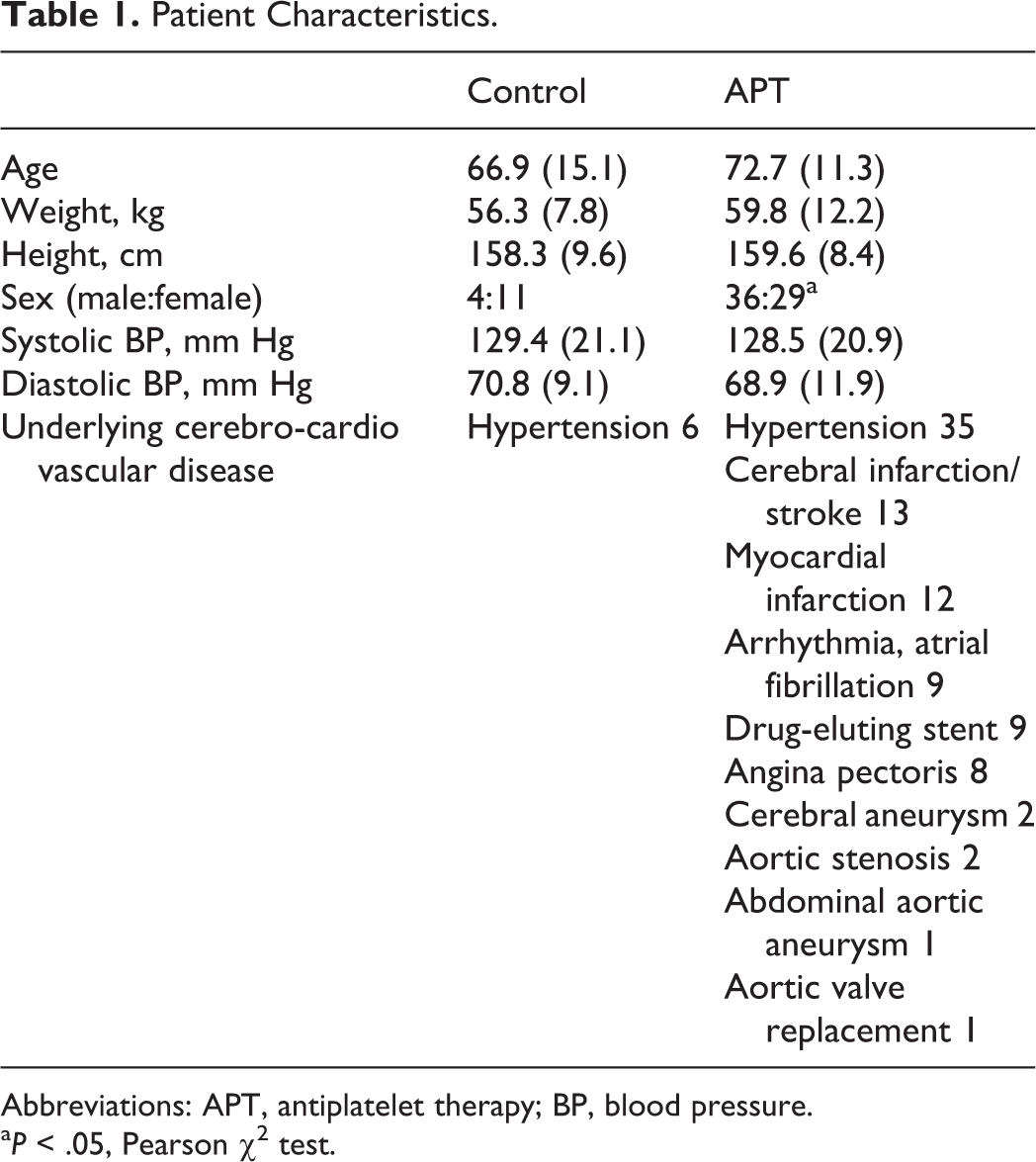
Abbreviations: APT, antiplatelet therapy; BP, blood pressure.
a P < .05, Pearson χ2 test.
Antiplatelet Therapies.

Abbreviations: APT, antiplatelet therapy; DAPT, dual antiplatelet therapy.
The control value at our institute was 97.7 (29.0) U for the AA test and 77.7 (21.7) U for the ADP test; these values fell within the ranges of the manufacturer’s reference values (79-141 U for the AA test, 55-117 U for the ADP test). Our control TRAP data (131.7 [26.7] U) were somewhat higher than the manufacturer’s reference values (87-147 U).
The average AA test result was significantly lower among the patients who were taking aspirin, compared with the results in the control group (control, 97.7 [29.0] U [n = 15]; aspirin, 14.5 [7.2] U [including DAPT patients, n = 46]; P < .001; Figure 1A). The ADP test result was also significantly lower among patients who were taking ADP-monitorable drugs, including P2Y12 inhibitor and cyclic AMP (cAMP) inhibitor (control, 77.7 [21.7] U [n = 15]; ADP-monitorable drugs, 39.0 [21.1] U [including DAPT patients, n = 28]; P < .05; Figure 1B). The ADP test result, not including cAMP inhibitor, was 37.3 (20.4) U (P < .05, compared with the control). The AUC distributions for the TRAP results were not significantly different between the control patients (n = 15) and the patients who were receiving APT (n = 65; control, 122.9 [43.2] U; APT, 111.7 [29.8] U; P > .05; Figure 1C).
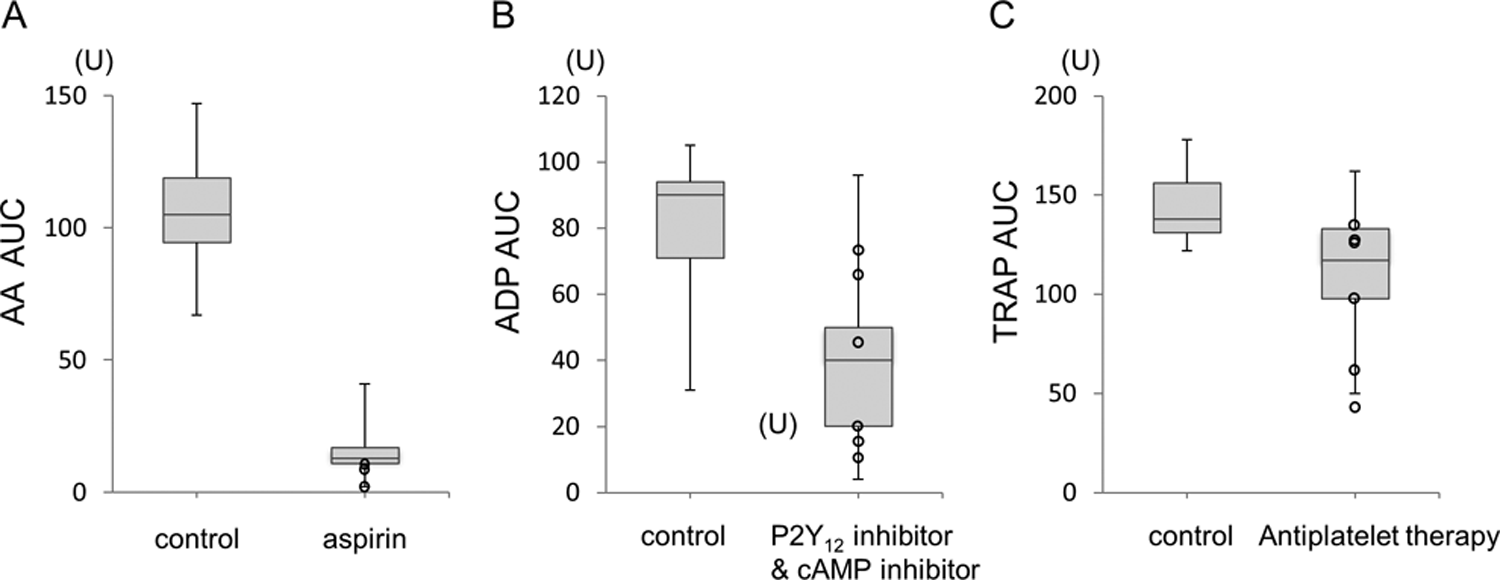
Box-and-whisker plots of each platelet functions evaluated using Multiplate aggregometry. A, Arachidonic acid–induced platelet function in the control (n = 15) and aspirin-treated groups (n = 46). A significant difference was observed between the 2 groups. B, Adenosine diphosphate (ADP)–induced platelet function in the control (n = 15) and P2Y12 inhibitor and cAMP inhibitor-treated groups (n = 28). Although the values in the treated group varied considerably, a significant difference was observed between the 2 groups. C, Thrombin receptor activating peptide (TRAP)–induced platelet function in the control (n = 15) and antiplatelet therapy groups (n = 65). The AUC values were not significantly different between the control and APT groups (P > .05). Thus, this parameter cannot differentiate the efficacy of APT. The open circles indicate cases with hemostatic difficulties.
Six cases in the APT group exhibited hemostatic difficulties requiring more than 10 minutes of compression hemostasis (Table 3), and no patient in the control group exhibited hemostatic difficulty. None of the patients reported offsite rebleeding. All cases with hemostatic difficulties had AA test values that were less than 2 SD of the value in the control group. Three cases with hemostatic difficulties had ADP test values that were less than 2 SD of the value for the control group (Table 3). Two cases with hemostatic difficulties had TRAP test values that were less than 2 SD of the value for the control group (Table 3).
Cases With Hemostatic Difficulty.a
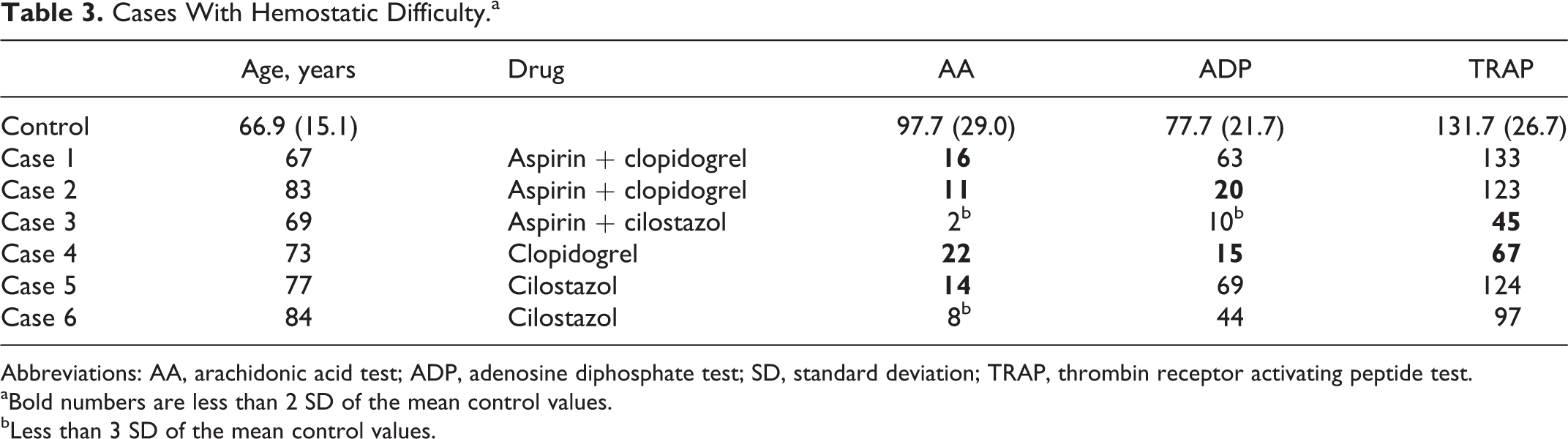
Abbreviations: AA, arachidonic acid test; ADP, adenosine diphosphate test; SD, standard deviation; TRAP, thrombin receptor activating peptide test.
aBold numbers are less than 2 SD of the mean control values.
bLess than 3 SD of the mean control values.
ROC curves were drawn to detect the optimal cutoff value for predicting hemostatic difficulties (Figure 2). The cutoff value for the prediction of hemostatic difficulty was 16.5 U for the AA test, with a sensitivity and specificity of 0.833 and 0.508, respectively. The AUC of the ROC was 0.691. The cutoff value for the prediction of hemostatic difficulty was 21 U for the ADP test, with a sensitivity and specificity of 0.847 and 0.500, respectively. The AUC of the ROC was 0.685.
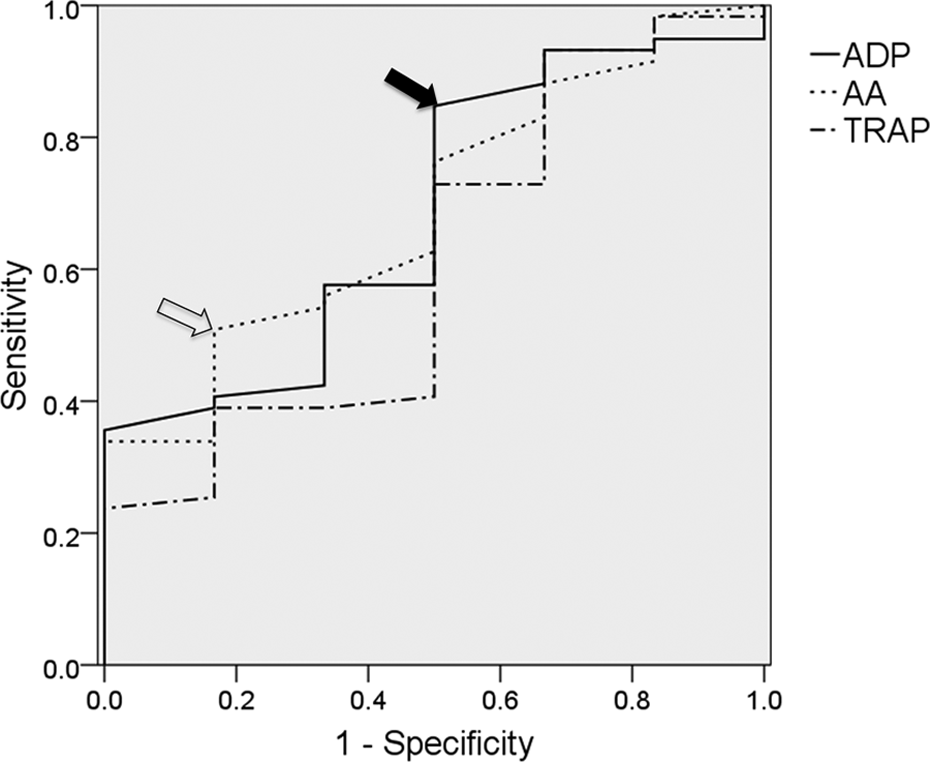
ROC analysis of hemostatic difficulties after tooth extraction in patients receiving antiplatelet therapy. Each plot shows the results for the arachidonic acid (AA) test, adenosine diphosphate (ADP) test, and thrombin receptor activating peptide (TRAP) test. The optimal cutoff values for the AA test and the ADP test are shown by the open and closed arrows, respectively. The open arrow corresponds to an AA test result of 11.5 U, and the closed arrow corresponds to an ADP test result of 21 U.
The amount of local anesthetic (mL) was selected as a significant factor (odds ratio, 2.71; confidence interval [CI], 1.22-6.00) for the prediction of hemostatic difficulty using a logistic regression analysis. The existence of a radicular defect of the apex was also a significant predictor of hemostatic difficulty (odds ratio, 0.078; CI, 0.009-0.709). None of the aggregometry tests were selected as significant predictors of hemostatic difficulty.
The statistical power of the ROC curves was examined using free software “R” (version 3.3.1 (2016-06-21)—“Bug in Your Hair” Copyright (C) 2016 The R Foundation for Statistical Computing Platform: i386-w64-mingw32/i386 [32-bit]) with the “pROC” library. To obtain an AUC of 0.9 with a power of 0.95 and a significance level of 0.05, the required numbers of cases were 54.6 without hemostatic difficulty and 5.56 with hemostatic difficulty.
Discussion
Hemostasis after dental extraction can typically be achieved within 5 minutes, even among patients taking aspirin. 9,10 Although hemostasis can be achieved in most outpatients, a few cases have reportedly required more than 30 minutes to achieve hemostasis. 11 Previous articles and reviews discussing the risk of bleeding in patients receiving APT have not suggested any indications for altering or stopping APT prior to dental extraction. 10 –12 The incidence of bleeding complications after invasive dental treatment in patients receiving APT has been reported to be low to negligible. 13 However, the end points of these studies were bleeding for more than 24 hours or continuing for 12 hours after treatment. 10,13 Since platelets are involved in primary hemostasis, platelet inhibition might not influence rebleeding after treatment. Therefore, we focused on hemostatic difficulties occurring immediately after tooth extraction. Hemostatic difficulties requiring suturing have been previously reported, with incidences of 6.8% for DAPT and 1.6% for single APT. 13 In addition, a few cases have reportedly required more than 30 minutes to achieve hemostasis. 11 We encountered 6 cases requiring more than 10 minutes to achieve hemostasis, and these cases were considered to have exhibited abnormal hemostasis, even though all the cases were safely managed at an outpatient clinic. The formation of a platelet plug is the second stage of hemostasis, followed by the coagulation cascade. Antiplatelet therapy inhibits platelet aggregation prior to plug formation. Hemostasis is usually monitored by compression at the bleeding site and presumably depends on the efficacy of platelet aggregation and plug formation prior to clot formation.
Whole blood aggregometry can evaluate only platelet aggregation because coagulation is inhibited using hirudin. Our measured values for whole blood aggregometry were almost in agreement with data reported in previous studies. 14,15 In the present study, very low AA results were observed prior to tooth extraction in some of the cases that subsequently required prolonged hemostasis compression. Although the AA test results varied individually over a considerably wide range, the cases that experienced hemostatic difficulties had exhibited very low AA test values, including 2 cases among the patients who had been taking aspirin with results that were less than 3 SD from the mean control value and 4 cases with results that were less than 2 SD from the mean control value. Therefore, the AA test might be useful for predicting a bleeding tendency. Our results suggest that even aspirin alone can cause hemostatic difficulty. A switch from DAPT to aspirin alone might be beneficial before surgery to avoid excessive surgical bleeding. However, the validity of such changes in treatment will require confirmation.
Platelet aggregometry has been previously used to predict thrombosis rather than to predict a bleeding tendency or to monitor the efficacy of the inhibition of platelet aggregation. A reduced antiplatelet effect of aspirin is an indicator of a risk of myocardial infarction. 16 –18 The Intracoronary Stenting and Antithrombotic Regimen-Aspirin and Platelet Inhibition registry, which contains information on 7090 percutaneous coronary intervention (PCI)-treated patients, suggests that AA test values are a good predictor of the risk of death or stent thrombosis. They reported that an AA test result of more than 203 could be defined as high on-aspirin treatment reactivity. Thus, this parameter can be clinically used as a quantitative index to evaluate the inhibition of platelet aggregation. 16
Regarding evaluations of hemorrhagic tendency, the AA test has been applied to predict the risk of postoperative platelet transfusion after coronary artery surgery. The report stated that an AA test result of 15 U after aortic declamping could be used as a cutoff value with an 80% sensitivity and a 54% specificity. 19 Petricevic et al also tried to define a therapeutic window for aspirin using aggregometry and reported that the lowest AA value to avoid bleeding was 20 U. 20 Our study showed that an AA value of 16.5 U exhibited an 83.3% specificity and a 50.8% sensitivity. Thus, our cutoff value is similar to those of previous reports. An AA value of less than 10 to 20 U seems to suggest that a patient may be at risk for a bleeding tendency.
Three cases treated with cilostazol experienced hemostatic difficulty. Cilostazol is categorized as a cAMP phosphodiesterase inhibitor. This pathway is closely linked to the ADP pathway. 21,22 The ADP test value for the cilostazol-treated patients was 43.9 (22.1), which was lower than the control value of 77.7 (21.7; P < .05) and was almost identical to that of the P2Y12 inhibitor-treated patients (37.3 [20.4]; P > .05). Therefore, we grouped the cilostazol-treated patients together with the P2Y12 inhibitor-treated patients.
Cilostazol itself might cause hemostatic difficulties in some cases. Vasoconstriction is important for hemostasis at open wounds after tooth extraction, and supplemental adrenaline in the local anesthetic can be effective for stopping bleeding. However, cilostazol has a vasodilating effect, 23 which might have worsened hemostasis in the present study. We found that most of the 6 cases with hemostatic difficulties had a low AA test value, although some of the cases did not have a low ADP test value. These results suggest that the AA test might be influenced by the inhibition of pathways other than AA.
The ADP test has also been shown to be useful for predicting stent thrombosis in patients undergoing percutaneous coronary intervention. 24,25 Its usefulness for predicting bleeding has also been confirmed. Ranucci reported that an ADP test using MEA could be useful for predicting postoperative bleeding in patients receiving thienopyridine treatment. 26 They reported that an ADP cutoff value of 31 U had a 72% sensitivity, a 66% specificity, a 92% negative predictive value, and a 29% positive predictive value for postoperative bleeding. Postoperative bleeding is caused by multiple factors, not only by platelet inhibition. Agarwal et al reported that MEA aggregometry was not correlated with blood loss after cardiac surgery. 27 In a homogenous patient group, however, ADP test results were found to be correlated with blood loss. 28 Although their results varied considerably, they suggested an ADP cutoff value of 22 U, which had a negative predictive value of 94%. Sibbing et al also explored a cutoff value indicating an enhanced response to clopidogrel and proposed a value of 188 AU·min (corresponding to 18.8 U), which had a negative predictive value of 99%. 3 Woźniak also explored the cutoff value for predicting postoperative bleeding after coronary artery bypass graft surgery and reported a value of 26 U for the ADP test, with a 72% sensitivity and a 69% specificity. 29 According to these accumulated results, Kong et al issued the “Consensus recommendation of using the Multiplate for platelet function monitoring before surgery.” According to this statement, an ADP test value of less than 50 U should be interpreted as indicating some evidence of platelet inhibition, while a value of less than 30 U should be interpreted as indicating severe platelet inhibition likely requiring transfusion. 6 Our proposed cutoff value of 21 U is almost in agreement with these previous data, although the end points of the risk settings differed among the trials. Platelet inhibition by P2Y12 receptor antagonism resulting in an ADP test value of less than 20 to 30 U may indicate a high risk of operative bleeding.
Of note, the AA test is quite sensitive to APT and could be used as a predictor of hemostatic difficulty. Patients who are high responders could experience hemostatic difficulty even if they are only receiving aspirin alone. Further research is required to explore the risk of aspirin.
Limitations
To prevent arterial thrombosis, the use of DAPT is increasing in popularity. Dual antiplatelet therapy commonly includes aspirin and other APT. Therefore, hemostasis is likely to be influenced by multiple pathways of platelet aggregation. A substantial number of DAPT patients were included in this study, and determining whether the AA or ADP pathway contributed to the hemostatic difficulty on an individual basis was difficult in this study. Even though a larger number of patients is needed to explore an appropriate cutoff value for predicting hemostatic difficulty, differentiating the pathway contributing to the abnormal hemostasis might be difficult.
Conclusion
Our report is one of the first reports to show a correlation between hemostatic difficulty and aggregometry. The AA test might be useful for predicting hemostatic difficulties.
Footnotes
Authors’ Note
A part of this study was presented at the Annual Meeting of Japanese Society of Anesthesiologists in May 27, 2016, at Fukuoka, Japan.
Acknowledgment
The authors would like to thank Ms Myrna Harrod (British Columbia, Canada) for her assistance in editing the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
