Abstract
We evaluated the effectiveness of a novel hemostatic powder called Tranexamic Acid-loaded Porous Starch (TAPS) developed recently on blood clotting activity and hemostasis. The effectiveness of TAPS was evaluated by comparing hemostatic properties with those of Quick-acting Styptic Powder (QSP) and Compound Microporous Polysaccharide Haemostatic powder (CMPHP). The blood clotting activities of human blood were analyzed by thromboela-stogram (TEG) assays in vitro. The hemostatic effectiveness in vivo was evaluated using a rat model with hepatic traumatic hemorrhage. The blood loss and standardized bleeding score, which reflects the degree of bleeding after treatment with styptic powder, were used to evaluate hemostatic efficacy. In vitro, the values of TEG parameters in TAPS group were significantly different, compared with untreated controls or CMPHP group (p < 0.05). In vivo, the application of QSP, CMPHP and TAPS led to significantly decreased post-treatment blood loss than in the control group (p < 0.01). The scores of the groups treated with QSP, CMPHP and TAPS (0, 0.2±0.422, 0.3±0.483, respectively) were significant lower than with gauze control (1.6±0.516) which success hemostatic was achieved at 5 minutes (p < 0.01). Hemostasis was achieved successfully within approximately 4 minutes after the application of TAPS. TAPS could help blood to form an artificial scab on a wound and to seal injuries for hemostasis to reduce blood loss in rats with hepatic trauma and hemorrhage. It was safe to use with no impact on blood clotting function or other apparent side effects.
Background
Life-threatening hemorrhage is the leading cause of death (50%) among combat casualties and is the second most common cause of death in civilian trauma patients. 1,2 In some urgent conditions, hemorrhage could have potentially been ameliorated by effective hemostasis. With the development of surgery and the aging of the population, the demand for hemostasis has increased. For example, styptic powder is usually applied in the surgical therapy of patients with hip fractures caused by osteoporosis. 3 Thus, the search for more effective hemostatic material continues to have high priority in the fields of trauma surgery and combat wound salvaging.
Currently, there are several forms of hemostatic products, such as tourniquets, hemostatic dressings, hemostatic powders, and injections. Comparatively speaking, the powder agents are advantageous for treating complex, irregular wounds because they can be spread and tightly packed in the wound, covering all of the bleeding sites that can be missed when small-size, inflexible dressings are used. For example, Yunnan Baiyao has been widely used by traditional Chinese medicine practitioners and civilians to stop bleeding caused by traumatic injuries and surgery. 4 In addition, we recently identified 2 types of new hemostatic powders that are more effective than the current hemostatic products, including a powder based on zeolite granules and a powder based on microporous polysaccharide hemospheres (MPHs). These products constitute a type of hemostatic that stops bleeding based on the mechanism of a molecular sieve, which was introduced to the field of hemostasis by Hursey and Dechene. 5 When the powder is applied directly to an actively bleeding wound, it works by accelerating the intrinsic clotting cascade by extracting fluid from the blood and concentrating coagulation factors and platelets, thus promoting the formation of a fibrin clot.
Although QuikClot and MPH have become available for hemorrhage in trauma surgery and on the battlefield, adverse effects, such as inflammation and tissue injury caused by exothermic reactions, and high costs have imposed restrictions on their clinical application. To address these challenges, US researchers outlined the following ideal qualities of hemostatic agents for prehospital/battlefield use: (1) capability to stop large vessel arterial and venous bleeding within 2 minutes of application when applied to an actively bleeding wound through a pool of blood; (2) no requirement for mixing or preapplication preparation; (3) simplicity of application by a wounded victim, companion, or medic; (4) lightweight and durability; (5) long shelf-life in extreme environments; (6) safety in use with no risk of injury to tissues or transmission of infection; and (7) low cost. 6
We found that tranexamic acid (TA) was an antifibrinolytic agent that reduced or prevented bleeding 7 and porous starch had good adsorption. Therefore, our laboratory developed a novel hemostatic powder called TA-loaded porous starch (TAPS) in an attempt to attain the qualities of ideal hemostatic agents. 8 Tranexamic acid and porous starch play roles in forming and maintaining a stable clot at the bleeding site. The goal of this study was to examine the coagulation effects of TAPS in vivo and in vitro via animal experiments and thromboelastography (TEG).
Methods
For the TEG assay, informed consent was obtained from volunteers. The animal experiment protocol was approved by the Animal Research Committee of the Chinese PLA Medical College. All of the animals received care and were used in strict compliance with the Guide for the Care and Use of Laboratory Animals. They were maintained on a 12-hour light/dark cycle at a temperature of 20°C to 22°C, with water and food freely available.
Reagents and Materials
Brief descriptions of each test agent are as follows. Tranexamic acid–loaded porous starch was supplied by our laboratory (Chinese PLA Clinic Blood Transfusion Center, China). Compound microporous polysaccharide hemostatic powder (CMPHP) was purchased from Shandong Success Pharmaceutical Technology Company (Shandong, China). Quick-acting styptic powder (Flashclot) was supplied by Guangdong Honghua Pharmaceutical Technology Co, Ltd (Dongguan, China). The disposable cups and pins of the TEG Hemostasis System and TEG Hemostasis System Kaolin were supplied by Haemoscope Corporation (Niles, Illinois; www.haemonetics.com). Male Wistar rats with age between 9 and 11 weeks were purchased from Peking Vital River Laboratory Animals Technology Co, Ltd (a Charles River Company, Beijing, China). Conventional coagulation tests were detected by a STA-R Evolution auto-blood coagulation analytical system (STAGO Diagnostic Technique Co, Ltd, Paris, France).
In Vitro Methods
The blood clotting activities of human blood exposed to hemostatic powder were analyzed by TEG assays in vitro. 9,10 Conventional coagulation tests, TEG assays, and blood cell counts were performed in the clinical laboratory department of our hospital. The TEG machine (TEG Hemostasis Analyzer 5000; Haemoscope Corporation) was calibrated before use with quality control standards obtained from Haemoscope. The TEG assays were performed within 2 hours of sample collection, in accordance with the manufacturer’s instructions. In order to study the effect of different doses of TAPS on coagulation function, 5 mL of whole blood was collected from each of 10 healthy blood donor with a tube containing citrate (3.2%). Ten, 20, and 30 mg TAPS were placed in a small plastic vial, respectively; then, 1 mL of blood from 1 blood donor was added, and the vial was capped. The vials were gently inverted 8 times, and 340 µL blood samples were obtained and placed in TEG cups for analysis. Twenty microliters of calcium chloride (0.2 mol/L) were added to the cups before adding the blood samples to overcome the anticoagulant effect. The coagulation effects of the powders were compared with a vial of standard kaolin, which is a known activator of the intrinsic clotting pathway. A recalcified blood sample without further treatment was tested as a negative control. Thromboelastography samples were tested at 37°C until all of the parameters were reported, and the following variables were recorded: reaction time (R, minutes), clotting time (K, minutes), angle (α, degrees), and maximum amplitude (MA, millimeters). Based on the statistical analysis (Table 1), the dose of TAPS in our study was determined to be 20 mg. Then we compared the coagulation performance of TAPS with CMPHP by TEG assays. A volume of 5.4 mL of whole blood was collected from other 10 healthy blood donor with 2 tubes (2.7 mL per tube) containing citrate (3.2%), and 2 mL of whole blood was collected in a tube containing EDTA-k2. Conventional coagulation tests, TEG assays, and blood cell counts were performed, and 20 mg of each powder was used for TEG assays.
TEG Assays of Human Blood Exposed to Different Dose of TAPS.

Abbreviations: K, clotting time, minutes, the speed of clot formation and is the time from the R time until a clot with a fixed firmness is formed; MA, maximum amplitude, millimeter, and the maximum strength or firmness of the developed clot; R, reaction time, minutes, the time that the initial fibrin formation is detected; TEG, thromboelastography; TAPS, tranexamic acid–loaded porous starch; α, angle, degree, and the kinetics of clot development.
In Vivo Methods and Surgical Procedures
In our preexperiment, 27 rats were randomized to 3 treatment groups (9 rats each, group 1: 200 mg TAPS, group 2: 300 mg TAPS, and group 3: 400 mg TAPS; Figure 1). Anesthesia was induced with 3% pentobarbital sodium and was maintained at a dosage of 45 mg/kg, delivered by peritoneal injection. Two milliliter blood samples were collected from heart to confirm that each animal exhibited normal coagulation function including prothrombin time (PT), international normalized ratio (INR), fibrinogen (Fg), and prothrombin time activity (PTA). Three days after blood collection, liver injuries were induced according to a previous report. 11,12 Briefly, anesthetized rats underwent standard midline laparotomy with a 2-cm incision to expose the anterior tip of the left external lobes. Then the left lobe was wrapped with a saline-moistened sterile gauze pad and was gently lifted by manual elevation to allow for adequate exposure. Next, a sharp-edged razor blade (with a length of 1.5 cm) was positioned over the right side of the liver, approximately 1 cm from the intersection of the left external and left medial lobes. The incision was approximately 1.5 cm × 0.4 cm, with one or more of the left external lobar veins lacerated, thus achieving an active, severe condition of hepatic trauma with bleeding. Then the wound was immediately covered with a gauze pad to suction excess blood. Thirty seconds after the injury, different doses of styptic powders were poured directly into the bleeding site, respectively, when the excess blood was removed, and we began to record the time with a stopwatch. Then a piece of filter paper (2 cm × 2 cm) and a preweighed gauze pad were placed over the powder, and a 50-g scale weight was placed on the gauze pad for 90 seconds. The scale weight was removed every 30 seconds, and we immediately identified whether the hemorrhage from the incision stopped with the naked eye. Hemostasis was defined as no obvious blood oozing from the incision without stress. 13 As soon as the hemorrhage stopped, we recorded the time to hemostasis (seconds). If there was still bleeding or oozing, we continued pressure for an additional 30 seconds in the same manner until the hemorrhage stopped. The experiments confirmed that we only need 200 mg of TAPS to achieve hemostasis (Figure 2). Other 40 rats were randomized to 4 treatment groups (10 rats each, group 1: TAPS, group 2: CMPHP, group 3: Flashclot, and group 4: control group). After induction of the same bleeding injury, the wound was immediately covered with a gauze pad to suction excess blood until application of the powder (30 seconds after the injury), and the shed blood was collected by suction, weighed, and recorded as pretreatment blood loss. The incision was treated with 200 mg of each styptic powders, then recorded the time to hemostasis (seconds). In the hemostatic stage, blood loss was collected by preweighed gauze, which was placed over the powder, and then reweighed and recorded as posttreatment blood loss. Grams of blood loss were then converted to milliliters per kilogram (weight of rat), where 1 g was equal to 1 mL.
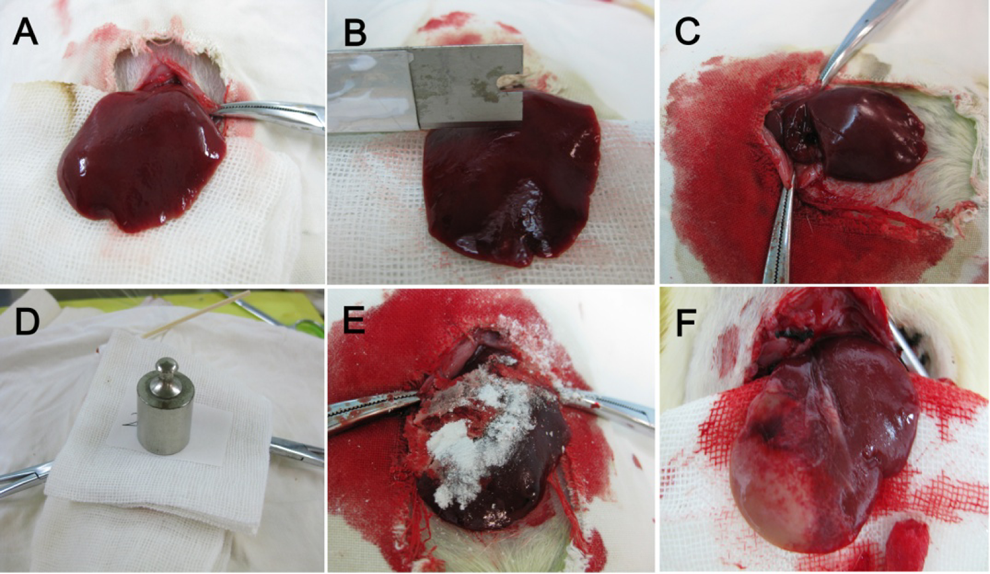
The surgical procedures in the liver of a rat. (A) The left external liver lobes; (B) sharp-edged razor blade (with length: 1.5 cm); (C) creation of liver injury (approximately 1.5 cm × 0.4 cm), resulting in the left external lobar vein being lacerated; (D) compression of a 50 g scale weight; (E) 200 mg of styptic powder; (F) degradation of styptic powder within 24 hours.
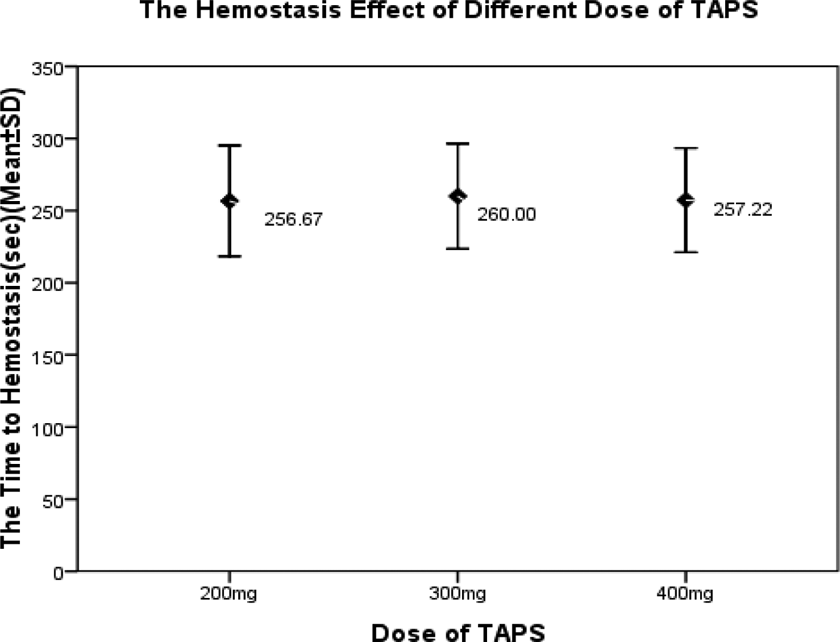
In the animal model of Sprague Dawley (SD) rats with hepatic trauma, hemorrhage was treated with 200, 300, and 400 mg TAPS, respectively. Effective hemostasis was defined as no obvious blood oozing from the incision without stress. As soon as the hemorrhage stopped, we recorded the time to hemostasis (seconds). n = 9. The time to hemostasis (seconds) to 200, 300, and 400 mg TAPS was 256.67 ± 50.000, 260 ± 47.434, and 257.22 ± 46.912, respectively. Statistical significance was based on the analysis of variance, and the hemostatic effect has no significant difference among different doses of TAPS. TAPS indicates tranexamic acid–loaded porous starch.
The same experimental procedures were repeated for the control group without the application of styptic powder, using only a gauze pad and standard pressure to stop bleeding. After completion of the treatment application, the abdomen was closed with sutures, and the animal was monitored for 36 hours. At the end of the animal experiment, 2 mL blood samples were collected from the abdominal aorta to evaluate the impact of TAPS on blood physiological parameters (WBC, RBC, Hb, Hct, and PLT) and conventional coagulation parameters (PT, INR, Fg, and PTA). All of the animals were killed by bloodletting from the abdominal aorta.
Data Analysis
Statistical analysis was performed using the Statistical Package for the Social Sciences (SPSS), version 17.0 for Windows (SPSS Inc, Chicago, Illinois). All of the continuous variables are reported as the mean ± standard deviation (SD) and were compared by analysis of variance and the LSD (L) test for comparisons between groups. Data with high variance were log transformed for analysis of variance. The nonparametric data were analyzed using the Kruskal-Wallis H (K) test. The data of TEG were analyzed by the paired t test. P < .05 was accepted as significant.
Results
Thromboelastography Profiles
The hematological characteristics of the donors showed normal coagulation function (Table 2). Among the most valuable parameters examined in the TEG assays, the R value and K value of the TAPS group were significantly decreased compared to the control group (5.390 ± 0.285 vs 12.320 ± 5.783, P < .05; 2.380 ± 0.489 vs 4.270 ± 2.722, P < .05, respectively) and the CMPHP group (5.390 ± 0.285 vs 8.030 ± 2.250, P < .05; 2.380 ± 0.489 vs 3.880 ± 1.353, P < .05, respectively), and the α angle and MA value of the TAPS group were much higher than in the control group (59.280 ± 4.667 vs 45.220 ± 10.835, P < .05; 58.970 ± 2.838 vs 54.580 ± 5.035, P < .05, respectively) or the CMPHP group (59.280 ± 4.667 vs 46.520 ± 9.332, P < .05; 58.970 ± 2.838 vs 53.040 ± 5.707, P < .05, respectively). There was no significant difference between the kaolin group and the TAPS group (P > .05). There was no significant difference between the untreated group and the CMPHP group in clotting parameters (K, α, and MA). The clotting profiles (TEG tracings) of blood samples treated with hemostatic powder or kaolin are shown in Figure 3. The traces represent the average measurements of venous blood samples. The analysis of TEG parameters (R, K, α, MA) for CMPHP and TAPS is summarized in Table 3. Treatment of blood with TAPS resulted in a shorter R time, a shortened K time, a larger angle, and a greater MA than treatment with CMPHP.
Baseline Hematological Measurements in the Healthy Volunteers.a,b
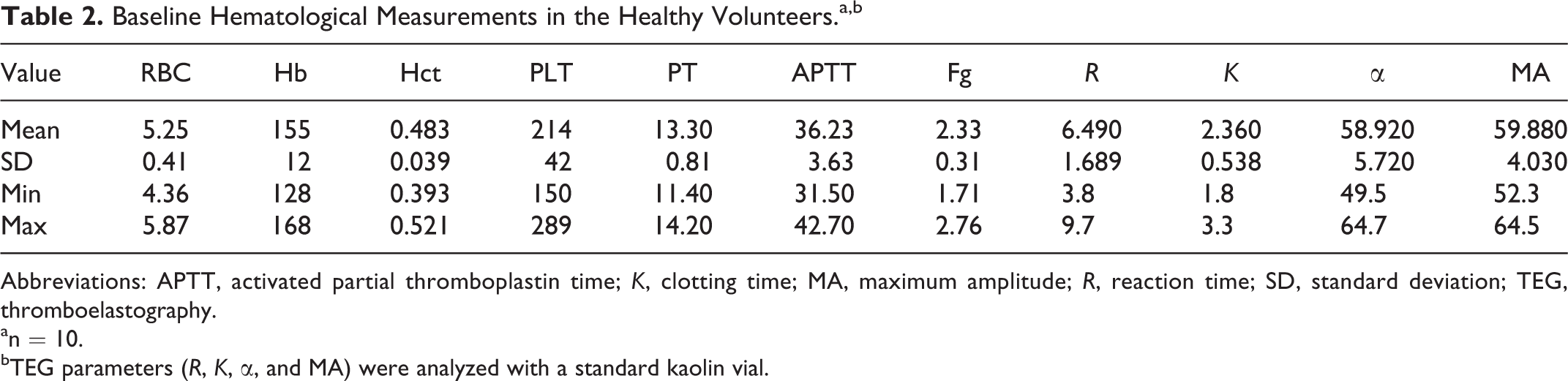
Abbreviations: APTT, activated partial thromboplastin time; K, clotting time; MA, maximum amplitude; R, reaction time; SD, standard deviation; TEG, thromboelastography.
an = 10.
bTEG parameters (R, K, α, and MA) were analyzed with a standard kaolin vial.
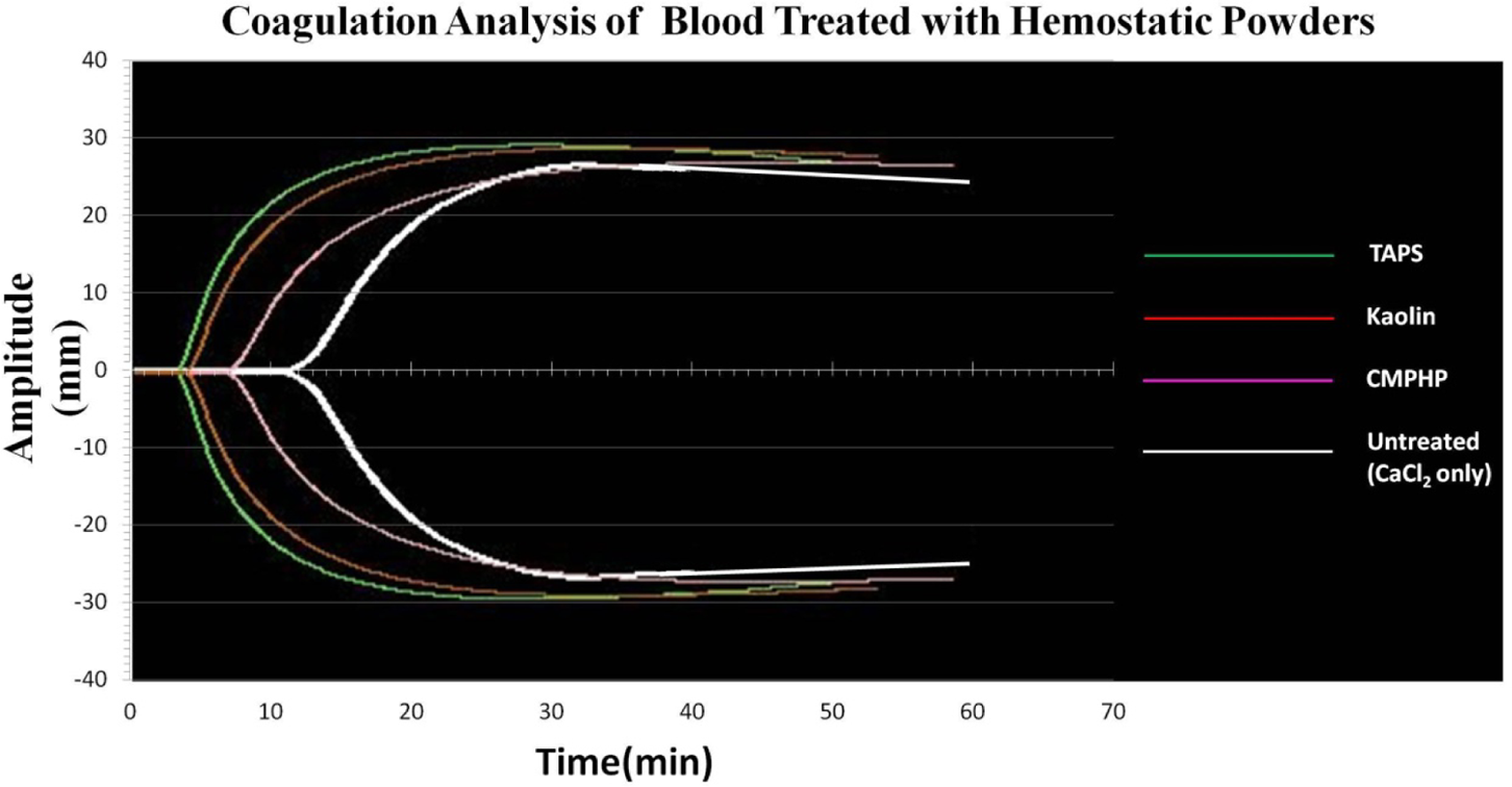
The average TEG of a healthy human blood donor after blood exposure to hemostatic powders in vitro. Venous blood samples were collected from the ulnar vein with citrated tubes. One milliliter blood samples were mixed with 20 mg of each powder and were assayed for coagulation by the TEG method after addition of calcium chloride. The tracings were compared with kaolin and with untreated blood. CMPHP indicates compound microporous polysaccharide hemostatic powder; TAPS, tranexamic acid–loaded porous starch; TEG, thromboelastography.
Blood Coagulation Measurements With TEG Method After Exposure to Hemostatic Powders.a,b
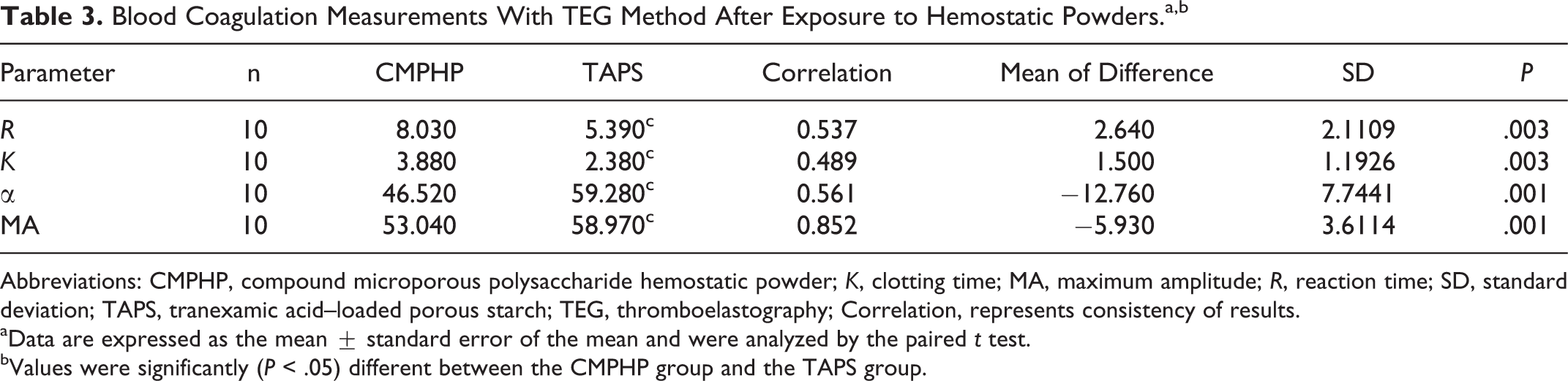
Abbreviations: CMPHP, compound microporous polysaccharide hemostatic powder; K, clotting time; MA, maximum amplitude; R, reaction time; SD, standard deviation; TAPS, tranexamic acid–loaded porous starch; TEG, thromboelastography; Correlation, represents consistency of results.
aData are expressed as the mean ± standard error of the mean and were analyzed by the paired t test.
bValues were significantly (P < .05) different between the CMPHP group and the TAPS group.
Hemostasis in Surgical Procedures
Forty rats were procured in this study to reproduce a severe hemorrhage model. All of the rats were male and weighted between 300 and 350 g. There were no significant differences in baseline physiological and hematological parameters among the 4 groups (P > .05; Table 4). The pretreatment blood loss was approximately 1.4 mL, and there was no difference among the 4 groups (P = .068). In the control group, 2 animals died after procedures due to postoperative hemorrhage. Based on the comparison of posttreatment blood loss, Flashclot, CMPHP, and TAPS decreased blood loss dramatically more than in the control group (P < .01), but there were no significant differences between TAPS and the CMPHP and Flashclot groups (P > .05). During the procedure, we found that CMPHP showed better adhesion to hepatic tissue, and zeolite particles were difficult to remove from wound surfaces. Both CMPHP and TAPS degraded between 36 and 48 hours after treatment. The time to hemostasis (seconds) of each group is shown in Figure 4. Flashclot resulted in a significantly shorter time to hemostasis, compared to Gauze, CMPHP, or TAPS (P < .01). In the groups treated with CMPHP or TAPS, the hemostatic success was achieved at 222 ± 63.561 and 249 ± 51.088, respectively. The hemostatic effect has no significant difference between CMPHP and TAPS (P > .05). In the rat model with hepatic trauma and hemorrhage, hemostasis was achieved within approximately 4 minutes after the application of TAPS. The hematological, physiological, and coagulation parameters after injury and hemorrhage were not different among the groups.
Baseline Physiological and Hematological Measurements in SD Rats.
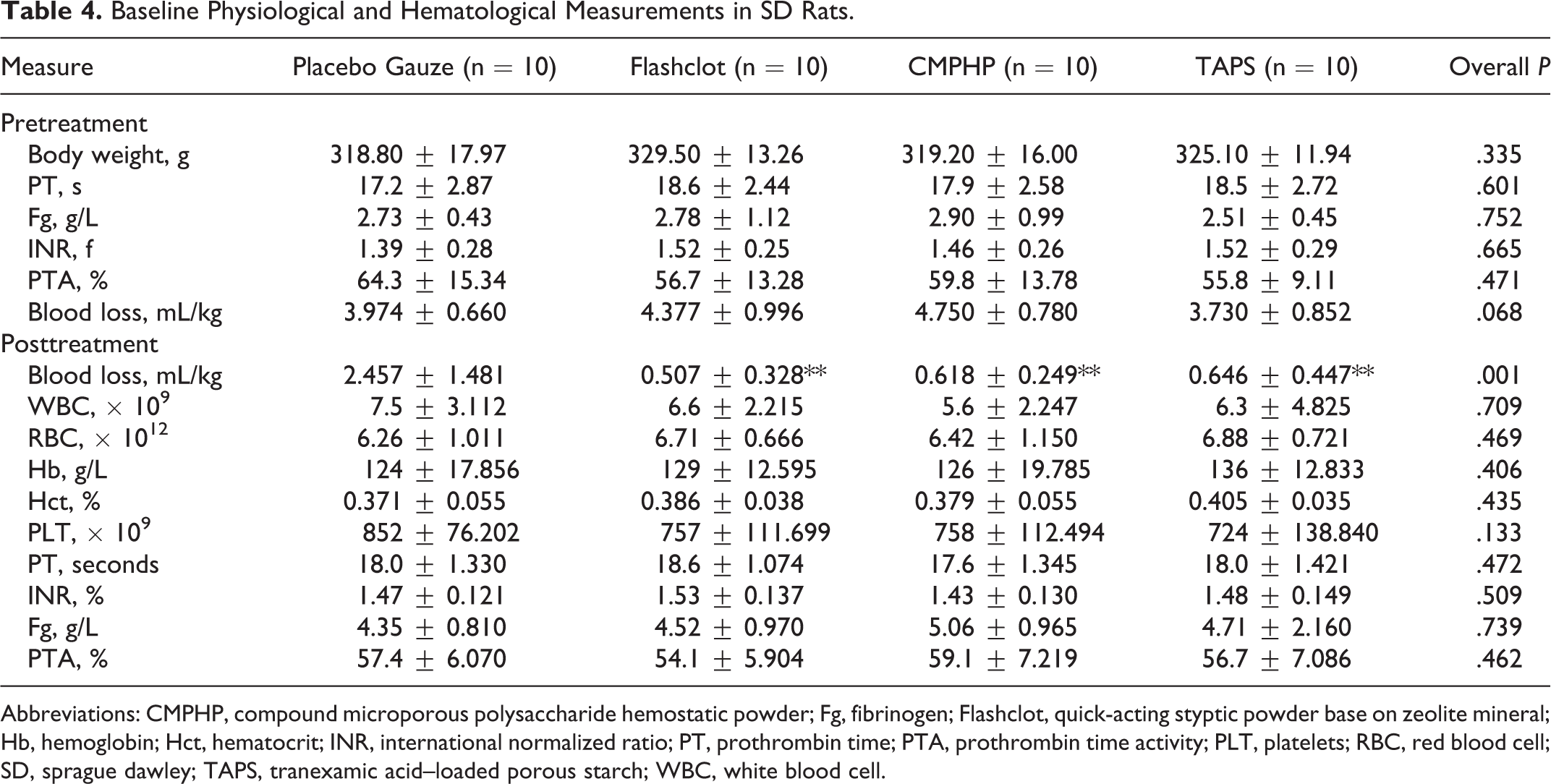
Abbreviations: CMPHP, compound microporous polysaccharide hemostatic powder; Fg, fibrinogen; Flashclot, quick-acting styptic powder base on zeolite mineral; Hb, hemoglobin; Hct, hematocrit; INR, international normalized ratio; PT, prothrombin time; PTA, prothrombin time activity; PLT, platelets; RBC, red blood cell; SD, sprague dawley; TAPS, tranexamic acid–loaded porous starch; WBC, white blood cell.
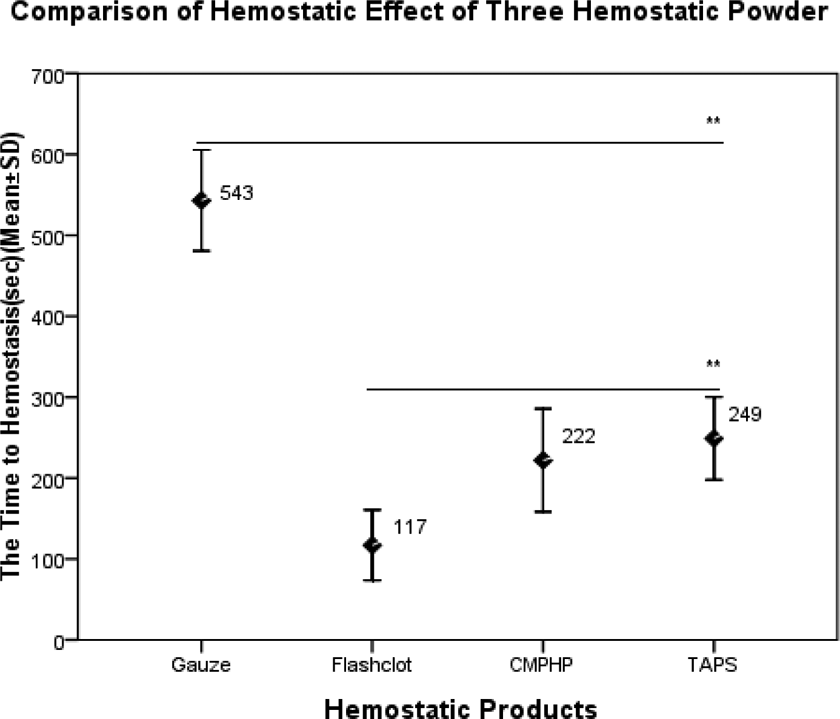
In the animal model of SD rats with hepatic trauma, hemorrhage was treated with placebo gauze and 200 mg Flashclot, CMPHP, and TAPS, respectively. Effective hemostasis was defined as no obvious blood oozing from the incision without stress. As soon as the hemorrhage stopped, we recorded the time to hemostasis (seconds). n = 10. The time to hemostasis (seconds) was 543 ± 62.370, 117 ± 43.474, 222 ± 63.561, and 249 ± 51.088, respectively. Statistical significance was based on the analysis of variance, and the hemostatic effect has no significant difference between CMPHP and TAPS (P > .05). CMPHP indicates compound microporous polysaccharide hemostatic powder; TAPS, tranexamic acid–loaded porous starch.
Discussion
Traditional hemostasis materials, such as tourniquets and hemostatic dressings, are usually insufficient or unsuitable to stop bleeding in an emergency, especially for penetrating wounds in the axillary or femoral region or in internal organs. Hemostatic powders can be sprayed over a large surface area, making them convenient for treating malignant bleeding tissue with multiple bleeding points. 14 In recent years, the literature has reported a series of novel styptic powders that could achieve hemostasis based on the molecular sieve mechanism. However, none of these products could meet the criteria for ideal hemostatic powders. Our laboratory recently prepared a novel styptic powder called TAPS, with TA loaded into the micropores of porous starch. We confirmed the effects of hemostasis and controlled-release TA in our previous experiments, which were induced by the combined efficacy of porous starch and TA. Porous starch has been widely used as an adsorbent in the areas of food, medicine, and other fields; it is an MPH and was confirmed to have excellent adsorbability, a high rate of drug loading, and delayed drug release. 15,16 It has the characteristics of being bioinert, a degradation time of within 24 hours, no risk of tissue damage or infectious disease transmission, and a relatively low cost. A related study suggested that early administration of TA appeared to improve survival by reducing bleeding within 3 hours of injury, especially in bleeding trauma patients, and it offers a simple, safe, cost-effective therapeutic intervention for patients with acute traumatic coagulopathy. 17 –19 It was reported that the topical and canonical application of TA as a hemostatic could reduce postoperative bleeding and would not lead to an increase in complications. 20 –22 Thus, the second half of hemostatic function of TAPS was mainly due to TA being released from the micropores of porous starch. There was no significant increase in serious prothrombotic complications if TA was administered at an earlier time, and it can be administered safely to a wide range of patients with traumatic bleeding. 23,24 Now, TA has been added to the World Health Organization list of essential medicines and has been recommended to be added to the armamentarium for the treatment of bleeding trauma patients. 7,25 In our animal experiment, the coagulation function of the rats was also not affected by treatment with TAPS.
In our study, we compared the hemostatic properties of our product with CMPHP, which has physicochemical and hemostatic properties similar to Arista and Flashclot, the mechanism of which is similar to QuikClot. Compound microporous polysaccharide hemostatic powder, a type of novel biodegradable hemostatic material with a mechanism similar to Arista (essentially MPH), is made of starch polysaccharides and carboxymethyl chitosan mixed by emulsion and cross-linking. Starch polysaccharides also essentially are MPHs, which are 30- to 100-μm spheres with microporous surfaces and an extended shelf-life; they are bioinert and contain no human proteins. 26,27 Microporous spherical particles have the effect of a molecular sieve, and they can quickly start and strengthen the activation of endogenous clotting mechanisms. When blood contacts the powder, it forms a “hydrogel,” which provides mechanical vascular occlusion at the bleeding site. Therefore, this product is a relatively safe and simple agent and can be poured directly into a bleeding wound in a 1-step process. Flashclot is a zeolite mineral-based hemostatic powder that can reduce blood loss and mortality. The rapid absorption of blood by the product facilitates the concentration of red cells and clotting factors at the site of injury, and red cell aggregation and formation of a fibrin matrix caused by absorption are the major mechanisms of action. 1,28 Tranexamic acid–loaded porous starch has the same mechanism for hemostasis based on porous starch.
Thromboelastography assays showed that treatment of blood with TAPS resulted in a strong response and shifted the clotting curves to the left, indicating an earlier onset of clot formation and faster and stronger clot formation than with CMPHP. Thromboelastography is a dynamic tracing technology of coagulation, and generally 4 variables (R, K, α, and MA) are measured. R is related to clotting factor activity, K is time, α is mainly determined by the level of fibrinogen, and MA is mainly the quantity and function of platelets. Therefore, TAPS has excellent blood clotting activity. Tranexamic acid is an antifibrinolytic agent that acts by binding to plasminogen and blocking the interaction of plasminogen with fibrin, thereby preventing dissolution of the fibrin clot. Thus, TAPS could enhance the velocity of clot formation and the strength of the developed clot by maintaining the high level of fibrin. The clotting activity of TAPS was better than that of CMPHP in vitro, but the hemostatic effect of TAPS was not good as that of CMPHP. This phenomenon could be explained by the tissue adhesive properties of CMPHP due to the positive charge of carboxymethyl chitosan, which can promote erythrocyte and platelet aggregation. 9 Recent research has also demonstrated that the hemostasis of chitosan as a blood coagulant has no direct relationship with traditional intrinsic or extrinsic pathways, but it could promote the adhesion and aggregation of platelets, and it is related to positively charged groups that interact with negatively charged red blood cell membranes and the tissue surface. 29 Although the hemostatic effect of TAPS is similar to that of CMPHP, the cost of TAPS, which has an identical dosage, is only 2% that of CMPHP.
The rat model indicated that TAPS had biocompatibility and absorbability, and it could reduce blood loss equivalently to Flashclot. The immediate action of Flashclot might be related to its exothermic reactions, which promote vasoconstriction and the coagulation cascade, and the density of its particles. The immediate effect of TAPS is not good as that of Flashclot because of its weaker adhesion and limited adsorption, but the procoagulant effect is obvious when TA is released from porous starch. Tranexamic acid was also able to reverse fully major fibrinolysis and to normalize thromboelastographic measurements of coagulopathy. 30 Thus, the efficacy of TAPS mainly depends on TA, and it could be effective in trauma patients to prevent traumatic coagulopathy. Tranexamic acid–loaded porous starch seems to have many attributes of ideal hemostatic agents with ideal qualities for prehospital/battlefield use. For example, there is no requirement for mixing or preapplication preparation; it can be applied by wounded victims, companions, or medics; it is lightweight and durable; it is safe to use with no risk of injury to tissues or transmission of infection; and it is inexpensive.
Conclusion
Tranexamic acid–loaded porous starch had similar hemostatic effects as CMPHP and could help blood in forming an artificial scab on the wound and in sealing injuries for hemostasis. It had no apparent side effects and had no impact on the coagulation parameters of rats. Although application of TAPS might not stop bleeding immediately such as Flashclot, it will certainly reduce blood loss. The powder has the advantages of adsorbability, security, inexpensiveness, and ease of use to produce on a large scale. Thus, it could be utilized widely for civilian trauma patients with severe hemorrhage. In future studies, we intend to establish an animal model with traumatic coagulation disorders to validate the effects of TAPS for preventing traumatic coagulopathy.
Footnotes
Authors’ Note
Deqing Wang and Yuan Zhuang designed the study. Shufang Wang and Guixiang Sun performed the literature research and data collection. Yaqian Liu performed the data analysis. Chaoyun Xi and Liguo Zhu performed the experimental studies and manuscript writings. All authors contributed to data interpretation and critical revision of the manuscript.
Acknowledgments
The authors would like to thank the Department of Clinical Laboratory and the Medical Animal Experiment Center, Chinese PLA General Hospital for their technical assistances in this study. The authors also acknowledge the staff of the Department of Blood Transfusion, Chinese PLA General Hospital for their supports.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was sponsored by Military Major Special Project (AWS11J007-08).
