Abstract
A well-accepted test for monitoring anticoagulation by enoxaparin is not currently available. As inadequate dosing may result in thrombosis or bleeding, a clinical need exists for a suitable test. Previous
Keywords
Introduction
Thrombosis is the pathological formation of a blood clot within blood vessels. It occurs as a result of activation of the platelet-coagulation system and depending on the pathological process will lead to formation of an arterial or venous thrombus. 1 Thrombi are most commonly manifested clinically in the acute coronary syndrome (ACS) or as venous thromboembolism (VTE). Data produced by the Centers for Disease Control and Prevention in 2015 estimated the overall annual incidence of VTE in the United States to be between 1 and 2 per 1000 of the population. 2,3 Thrombosis is usually treated with anticoagulants (unfractionated heparin [UFH], low-molecular-weight heparins [LMWHs]), antiplatelet agents [aspirin, ticlopidine], or for selected conditions with high-morbidity fibrinolytics [streptokinase, alteplase]). 1 It is appropriately important to dose anticoagulants as bleeding may occur from excessive doses or thrombosis from inadequate doses.
Enoxaparin is an LMWH anticoagulant that increases the activity of a physiological inhibitor, antithrombin. 1,4 This results in increased inhibitory action of antithrombin on coagulation factors, factor Xa in particular, thus prolonging the time to form a fibrin clot. Despite claims that enoxaparin therapy does not need monitoring due to the reportedly predictable dose–response relationship; many studies have shown that enoxaparin has similar bleeding rates as UFH. 4 In a study carried out in 2005, out of 446 non-ST-elevated ACS admissions that received enoxaparin, major bleeding (eg, retroperitoneal bleeding, intracranial bleeding) occurred in 1.8% of these patients and minor bleeding in 8.7%. 5 Many studies have therefore stressed the point of monitoring enoxaparin therapy. 5,6
Prothrombin time (PT), usually reported as an international normalized ratio, and activated partial thromboplastin time (aPTT) tests are used to monitor the anticoagulant effects of warfarin and UFH, respectively. These clotting time (CT) tests have been shown to provide a good prediction of bleeding and thrombosis when used with warfarin and UFH. On the other hand, no well-accepted method exists for monitoring the anticoagulation effect of enoxaparin or other LMWHs. Both PT/INR and aPTT do not show significant dose–response changes with enoxaparin 4 and are therefore not suitable for measuring the effect of enoxaparin. Indirect measurement of anti-Xa activity using a chromogenic assay has been shown to correlate with the effect of enoxaparin, but a well-accepted target activity value is not available and dose adjustments are difficult to make. 4,7,8 In addition, these anti-Xa assays measure the activity of a single coagulation factor Xa which corresponds to enoxaparin concentration only, as compared to CT tests (such as PT and aPTT) that consider the activity of wider activity in the integrated coagulation network. Hence, a deficiency in a regulatory coagulation factor (eg, antithrombin) may affect clotting ability but would not have an effect in a standard anti-Xa test. We propose that a CT test that assesses wider coagulation activity would be of more value.
Previous attempts at developing CT tests for LMWHs include 3 Xa-based tests. However, they are not widely used clinically for LMWH testing and have limitations. StaClot (product of Diagnostica Stago, Asnières-sur-Seine, France) and HepTest (product of American Diagnostica Inc, Stamford, CT, USA) appear to have been designed to identify the anti-Xa concentration present in the sample, rather than determining the net anticoagulant effect which is likely to be clinically more relevant in terms of bleeding and clotting risk. The third Xa-based CT test that currently exists is the Xa-activated CT test which is a whole blood coagulation assay. Platelets play a major role in blood coagulation, and the main limitation of this test is the presence of platelets which may cause the behavior of the test to be significantly different compared to clinically used CT tests that use plasma samples.
Factor Xa was identified as a suitable activating agent for the development of a CT test for enoxaparin based on a recent
Specifically, this study was a proof-of-concept study designed to: explore the reproducibility of the TenaCT test and explore factors that could affect the performance of the TenaCT test
The factors chosen were (a) gender—to detect gender-specific differences in the CT using plasma from male and female healthy volunteers; (b) age—to detect age-specific differences in the CT using plasma from volunteers of different age groups; and (c) freezing the plasma samples—to explore if the freeze–thaw process affects the performance of the test. (d) The test was also performed using plasma obtained from the volunteers on 2 different occasions to explore between-sample test variability.
Materials and Methods
Materials
Factor Xa (catalytic activity = 71 nkat) was sourced from Abacus ALS Limited, Auckland, New Zealand, a supplier of Chromogenix products. Commercial enoxaparin was available as clexane injection (2000 IU/0.2 mL), manufactured by Sanofi Aventis, Paris, France. Cofactor Actin FS was purchased from Siemens Healthcare Diagnostics Ltd, Auckland, New Zealand.
Methods
Recruitment of Volunteers
Healthy adult volunteers (n = 20) aged between 18 and 50 years were recruited from staff and students at the University of Otago, Dunedin, New Zealand. The number of healthy volunteers was arbitrarily chosen for an initial evaluation. Future investigations will evaluate the test in a higher number of volunteers. Applicants were excluded if they had at the time of recruitment or in the previous month: An inflammatory condition as a result of physical injury or immunological disorder (eg, sprain, asthma, arthritis, etc) or an infection; Taken any prescription medicine that could suppress or treat an inflammatory response or that is known to affect CT or bleeding time; Had a recent visit to the dentist: fillings or scaling within 7 days, or extractions, gingival, and other invasive procedures within 21 days; and Been pregnant, breast-feeding, or were taking hormonal treatments including oral contraceptives.
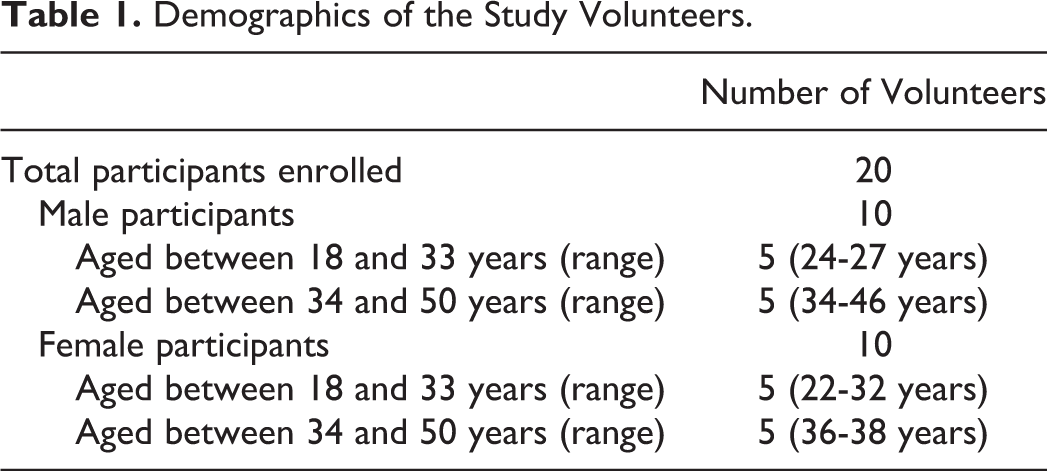
This study was approved by the University of Otago human ethics committee. Table 1 summarizes the demographics for the participants enrolled in the study.
Demographics of the Study Volunteers.
Blood Collection and Storage
Blood collection for this study was done by trained phlebotomists at the Blood Collection Centre, Dunedin Hospital, New Zealand. Blood was obtained from the volunteers on 2 separate occasions, at least 14 days apart. On each occasion, 30 mL of blood was obtained in citrated polystyrene tubes and was centrifuged at 2000
the first part (referred to as “fresh plasma”) was used to carry out the
the second part (referred to as “frozen plasma”) was snap frozen and stored at 80°C for repetition of the experiments after at least 24 hours in a single freeze–thaw cycle.
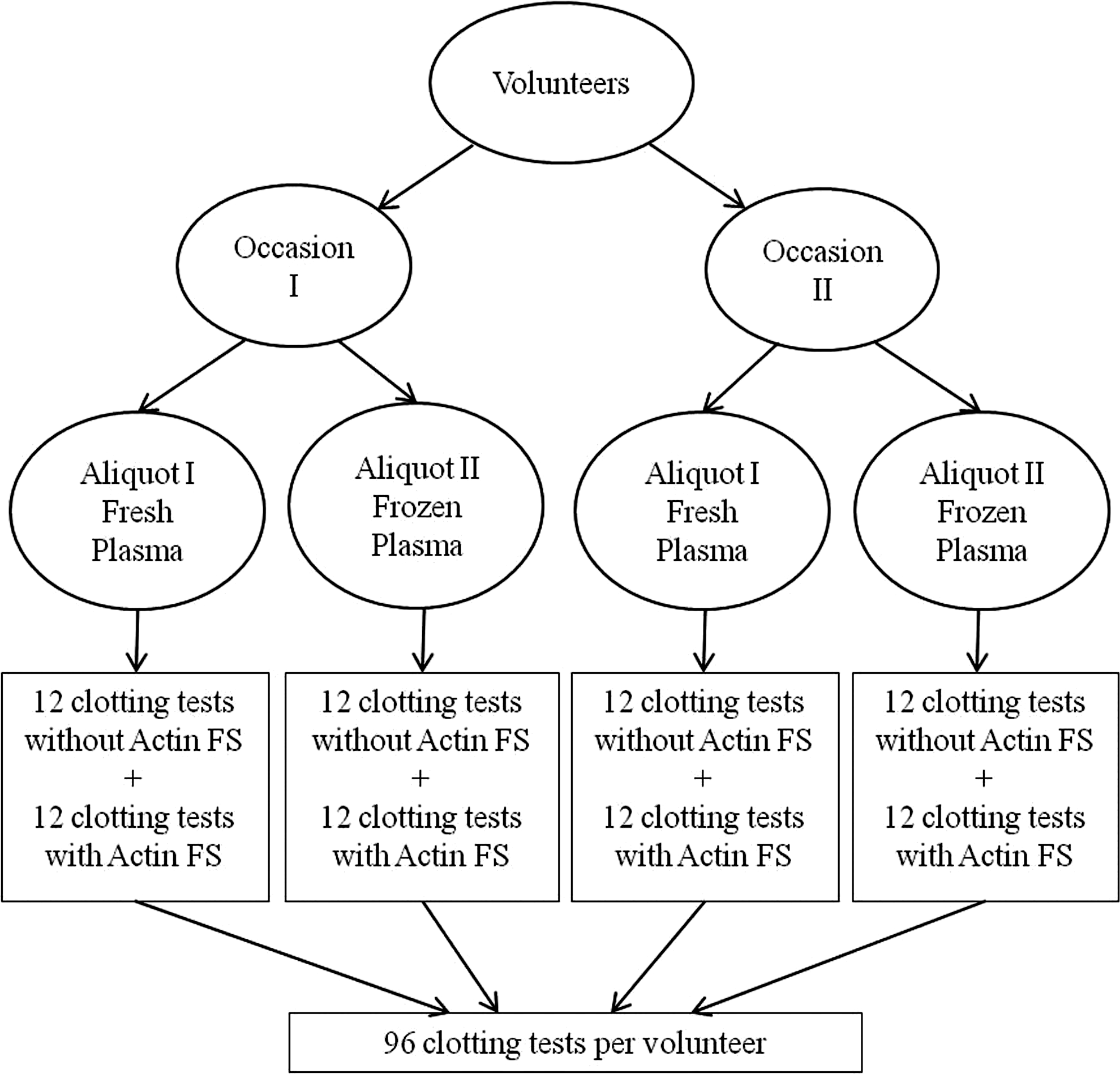
Schematic of the experimental design for the
In Vitro CT Test Experiments
The
A 200 µL mixture of equal volumes of Xa (20 nM) and CaCl2 (25 mM) was incubated at 37°C and added to 100 µL of plasma, which was prewarmed at 37°C. The time taken for the clot to form was recorded. The CT was then determined in the presence of various concentrations of enoxaparin (0 [control], 0.25, 0.50, and 1.0 IU/mL). The healthy volunteers were not being dosed with enoxaparin. Plasma samples were spiked with enoxaparin by adding 1 part of enoxaparin to 9 parts of plasma. The CT experiments were then carried out in the presence of the cofactor Actin FS. The activator solution in this case consisted of equal volumes of: a mixture of Xa (20 nM) and Actin FS (0.17%) and CaCl2 (25 mM). All CT measurements were performed in triplicate, and the results reported as mean (standard deviation [SD]). The increases in CTs were compared among the following groups to assess the factors that affect the performance of the Xa-based clotting time test: CTs of fresh versus frozen plasma samples; CTs of male versus female plasma; CTs of plasma from volunteers aged 18 to 33 years versus those 34 to 50 years; and CTs of plasma obtained on 2 different occasions at least 14 days apart.
The evaluation of gender and age as potential factors that could affect the performance of the test was initially carried out using a small sample size of n = 10, which was arbitrarily chosen. Future investigations will be carried out using a larger number of male and female healthy volunteers as well as using plasma from volunteers aged older than 50 years. Figure 1 shows the schematic of the experimental design for the
The most widely available test for monitoring of antifactor Xa activity of LMWHs is a chromogenic assay and is the test currently recommended by the College of American Pathologists. 11 Anti-Xa activity has been shown to be inversely related to thrombus propagation and the development of an occlusive thrombosis, 12,13 but the minimum effective level remains uncertain. In addition, a single factor inhibition assay will not detect the effect of other factors and inhibitors such as antithrombin. Currently, a therapeutic range for peak effect with twice daily administration of enoxaparin is 0.50 to 1.0 IU/mL and is used for patients being treated for acute VTE. 11,14 –16 The performance of the TenaCT test was therefore assessed in the presence of subtherapeutic and therapeutic enoxaparin concentrations. Among the concentrations of enoxaparin used in this study, control (0 IU/mL) and 0.25 IU/mL were considered as subtherapeutic concentrations and were compared to 0.50 and 1.0 IU/mL as therapeutic enoxaparin concentrations, respectively.
Statistical Analysis
Clotting times for individual volunteers were measured in seconds and data reported as mean (SD). The mean CT for the population of 20 volunteers was also reported along with the 95% confidence interval (CI) of the mean. A paired
Criterion for a Successful CT Test
A CT test in an individual volunteer at a single occasion was said to be successful if each of the following conditions were met: The CT for a control plasma sample was between 10 and 30 seconds. The CTs increased with increasing enoxaparin concentrations. The prolongation of CTs at enoxaparin concentration of 1.0 IU/mL should be greater than 3 times the control plasma sample. The CTs should not be more than 180 seconds to ensure rapid turnaround of test results.
Success in a volunteer was defined as when all of the conditions were true (ie, the criterion was met) with both plasma samples for each volunteer.
Results
The reproducibility of the Xa-based clotting time test was assessed in 20 healthy volunteers at various enoxaparin concentrations. In the presence of the cofactor Actin FS, the CTs for the majority of the volunteers were within the measurable range. Only 1 volunteer exceeded 200 seconds, the upper limit for STart Hemostasis Analyzer, at an enoxaparin concentration of 1.0 IU/mL. In the absence of Actin FS, however, CTs in majority of the volunteers were not measurable (>200 seconds) at the highest enoxaparin concentration of 1.0 IU/mL. There were only 4 occasions where the CT test met the criterion, and there was no volunteer for whom the test was deemed successful, that is, the criterion was met on both of the volunteer’s test samples. Table 2 summarizes the results with times less than 200 seconds in the absence or presence of Actin FS. The coefficient of variation (CV)% for all CT test experiments ranged between 0.14% and 3.38%, showing good reproducibility of test performance.
Number of Successes Observed for the TenaCT Test.a,b

aTotal number of volunteers were equal to 20, each for “without Actin FS” and “with Actin FS” groups.
bTotal number of occasions were equal to 40, each for “without Actin FS” and “with Actin FS” groups.
1. Evaluation of the performance of the TenaCT test based on the criterion for a successful CT test:
a. Condition 1 (CTs for control plasma should lie between 10 and 30 seconds)
Clotting times for both occasions for all patients were within the range of 10 to 30 seconds, except for 1 occasion where the mean CT slightly exceeded the upper limit at a value of 31 seconds.
b. Condition 2 (CTs should increase at increasing enoxaparin concentrations)
The mean CTs for the control plasma group (n = 20) at the first occasion was 17 ± 4.2 seconds (95% CI: 15-19 seconds). The mean CTs significantly increased (
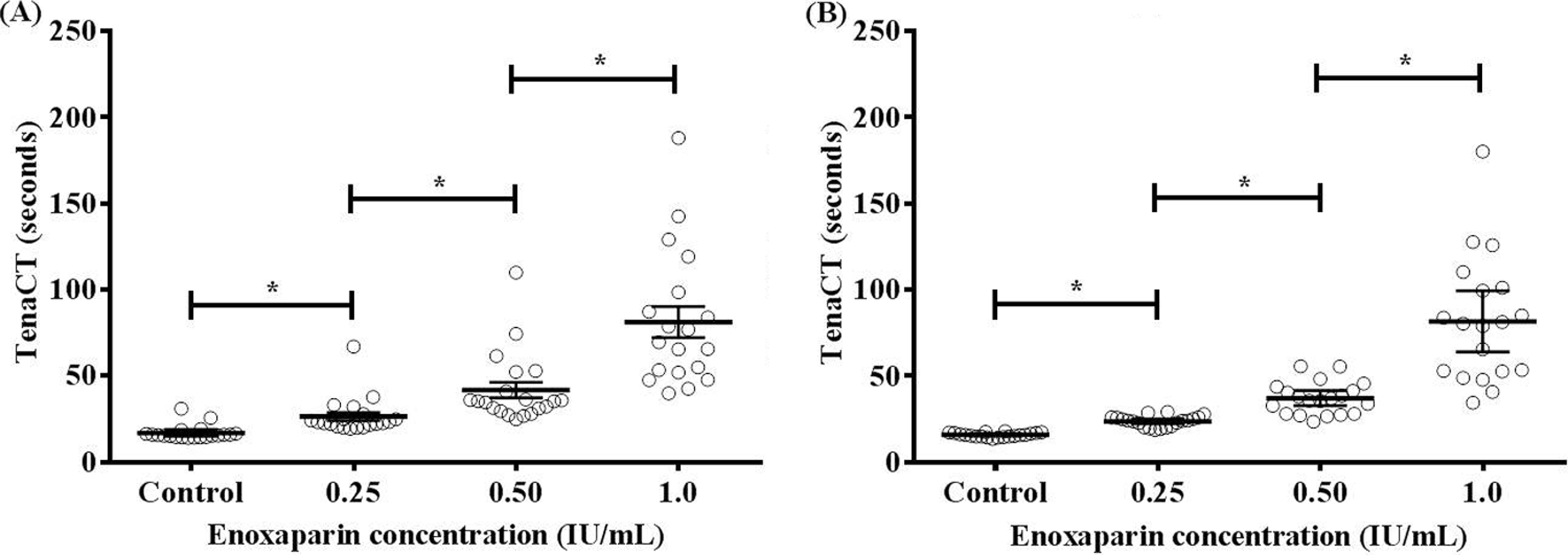
TenaCTs (in seconds) in the absence (control) or presence of enoxaparin concentrations of 0.25, 0.50, and 1.0 IU/mL at the (A) first and (B) second occasions. Mean TenaCT for each individual volunteer is represented by a circle. The bars represent the mean TenaCT and the 95% confidence interval for the population of 20 volunteers. * denotes the level of significance at
Similar trends were observed with the plasma samples collected from the volunteers at the second occasion. The mean CTs for the control plasma group (n = 20) was 16 ± 1.3 seconds (95% CI: 15-16 seconds) which increased to 24 ± 3.2 seconds (95% CI: 22-25 seconds, n = 20) at an enoxaparin concentration of 0.25 IU/mL, 37 ± 9.3 seconds (95% CI: 33-41 seconds, n = 20) at an enoxaparin concentration of 0.50 IU/mL, and 82 ± 37 seconds (95% CI: 64-99 seconds, n = 19) at an enoxaparin concentration of 1.0 IU/mL. These differences in the CTs at different enoxaparin concentrations were significant (
c. Condition 3 (prolongation of CTs in the presence of 1 IU/mL enoxaparin should be at least 3 times higher than the control group)
Mean prolongation of TenaCT at an enoxaparin concentration of 1.0 IU/mL compared to the control was greater than 3 times at both the occasions (n = 18 for each occasion). Mean fold prolongation in TenaCT at the first occasion was 4.8 ± 1.7 (95% CI: 3.9-5.6) compared to 5.1 ± 2.1 (95% CI: 4.1-6.1) at the second occasion. The CTs for the 2 occasions were not statistically different (
d. Condition 4 (all CTs should be <180 seconds)
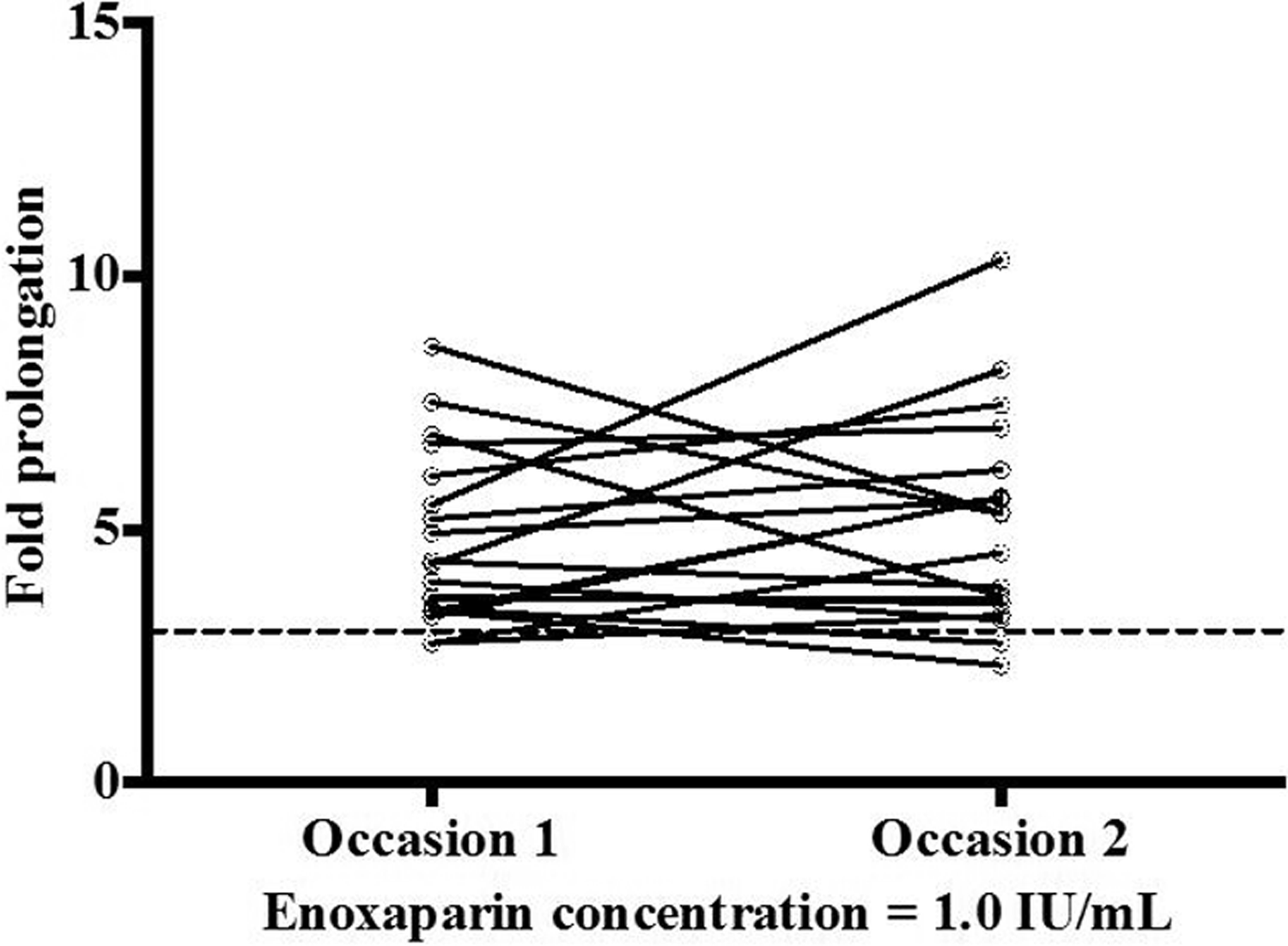
Fold prolongations in TenaCT in the presence of enoxaparin concentration of 1.0 IU/mL compared to the control (enoxaparin absent) at first versus the second occasions. Solid lines connect the fold prolongations for each individual volunteer at occasions 1 and 2. Dashed line represents the 3-fold prolongation criterion.
TenaCT at all enoxaparin concentrations was less than 180 seconds on all but 4 samples. The upper limit for CTs on the STart Hemostasis Analyzer can be adjusted and for the period of this study was set at 200 seconds.
2. Evaluation of the performance of the TenaCT test at subtherapeutic versus the therapeutic enoxaparin concentrations:
The reported therapeutic concentration for enoxaparin is 0.50 to 1.0 IU/mL. Prolongations in CTs of each sample at each concentration of enoxaparin were compared with the control samples. The first set of samples from volunteers at an enoxaparin concentration of 0.25 IU/mL, resulted in a mean fold prolongation in TenaCT of 1.5 ± 0.19 (95% CI: 1.4-1.6, n = 20). This prolongation was significantly different (
3. Evaluation of the factors that could affect the performance of the TenaCT test:
a. Clotting times in fresh versus frozen plasma samples
Significant differences in CTs were observed between fresh and frozen plasma samples at all concentrations of enoxaparin (
b. Clotting times in male versus female volunteers
The fold prolongations in Xa CTs in the absence and presence of enoxaparin were not different between male and female volunteers (
c. Clotting times in volunteers aged between 18 and 33 years versus those between 34 and 50 years
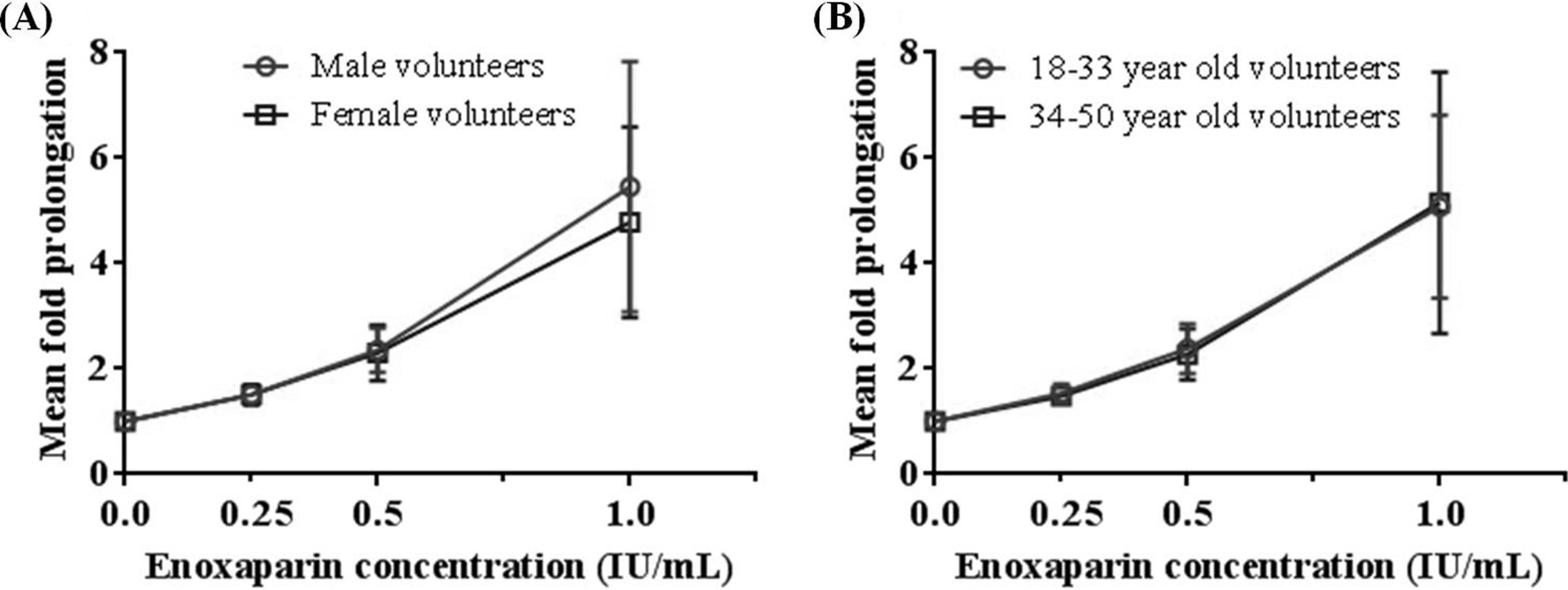
A, Mean fold prolongation in TenaCT using fresh unfrozen plasma of healthy male (red circles) and female (blue squares) volunteers in the absence or presence of enoxaparin concentrations of 0.25, 0.50, and 1.0 IU/mL. B, Mean fold prolongation in TenaCT using fresh unfrozen plasma of volunteers aged between 18 and 33 years (red circles) versus those between 34 and 50 years (blue squares) in the absence or presence of enoxaparin concentrations of 0.25, 0.50, and 1.0 IU/mL.
The age groups of volunteers evaluated in this study were 18 to 33 years and 34 to 50 years (Figure 4B). No significant difference were observed between the mean fold prolongations of Xa CTs of the 2 age groups (
Discussion
The Xa clotting time (TenaCT) test has been designed to provide a means of assessing the effect of therapeutic LMWH anticoagulants in individual patients. The impetus for developing the test is that the variability in the sensitivity of individual patients to LMWHs is potentially an important clinical variable that affects both the therapeutic effect and the risk of bleeding. These variables are known to affect the CTs obtained with other tests that measure overall coagulation activity. 7,17 The differences in CTs observed within individuals seen in Figure 3 are likely to reflect differences in anticoagulation that create differences in hemostatic effect. We believe that it is desirable that the tests used to evaluate dose–response of LMWHs in general, and enoxaparin in particular, should assess the effect within the context of plasma samples from individual patients and not as a standardized measure of the amount of LMWH present in the plasma sample. The TenaCT achieves this goal and has been designed to provide CTs that meet the requirements of hematology laboratories for rapid, high-volume CTs tests.
This proof-of-concept study for a Xa-based clotting time test for enoxaparin was carried out to explore the test reproducibility and to identify factors that may affect its performance.
Experiments with plasma samples, freeze-thawed once, resulted in significantly shorter CTs compared to the fresh (unfrozen) plasma samples and were not completed for all 20 volunteers. The shorter CTs may be due to the presence of microvesicles from trace numbers of frozen-thawed platelets, or activation of 1 or more clotting factors at low temperature. The experiments with frozen plasma samples suggest that the TenaCT test would not work with frozen plasma at the Xa and Actin FS concentrations used in this study. Additional experiments are needed out to identify the reasons for the differences and there is a possibility that an entirely different set of Xa Actin FS concentrations may have to be used for frozen plasma. Significant differences (almost 2-fold) in the prolongation of CTs were also observed between the 2 plasma samples obtained from some individuals (n = 4). Biological factors such as differences in the concentrations of CTs or inhibitors may have contributed to these differences. Further studies are needed to identify the cause(s).
Through the
Currently, none of the alternative factor Xa-activated tests are used routinely to monitor anticoagulation with LMWHs. This is an important omission and we believe that the variance in dose levels and exposure–response of these agents is likely to be associated with adverse outcomes in some patients. We propose that patients should be monitored at the beginning of treatment and where important changes in health status occur that may impact the level of anticoagulation (eg, significant changes in renal or liver function, cardiac failure, inflammatory processes, etc). The magnitude of the differences observed in the current study provides scope for challenging the view that monitoring is not required. While patients with renal impairment or liver disease may be obvious candidates for monitoring, confirmation and dose adjustment to standardize anticoagulation dose have the potential to reduce bleeding in patients with greater sensitivity to anticoagulation by LMWH (and indeed potentially other Xa antagonists) or thrombosis as a result of relative resistance to anticoagulation. Advancement of this concept of monitoring will require data from further evaluation of the prototype test to explore the correlation between the TenaCT, the dose of LMWH, and more extensive testing of the TenaCT on a larger number of normal human patients to evaluate the variance in TenaCT test result with LMWH spiked samples; evaluation of TenaCT test in a range of patient samples: samples from patients who have received LMWH at prophylactic and therapeutic dose levels will be needed to evaluate whether the TenaCT shows values that are consistent with the intended range of prophylactic or therapeutic effects, or whether the TenaCT may provide a useful guide on dose adjustment. This set of studies may provide evidence for using the TenaCT to control management by providing a guide for dose adjustment, rather than using the standard weight-based dosing for LMWH. It will be particularly relevant in patients with renal disease, liver disease, inflammatory disorders, and in cancers and other prothrombotic disorders.
Overall, the proof-of-concept study was considered successful, based on the predefined criteria. This study provided proof-of-concept for a Xa-based clotting time test that can be used to detect enoxaparin exposure–response. However, the test is currently not at a stage to be used clinically. Additional studies are needed to develop the test for clinical use, such as evaluation with a higher number of healthy volunteers over a wider age range, with whole blood, and with frozen plasma samples.
Supplemental Material
Supplementary_material - An Evaluation of a Factor Xa-Based Clotting Time Test for Enoxaparin: A Proof-of-Concept Study
Supplementary_material for An Evaluation of a Factor Xa-Based Clotting Time Test for Enoxaparin: A Proof-of-Concept Study by Deborah P. J., Stephen B. Duffull, James M. Faed, Geoffrey K. Isbister, and Abhishek Gulati in Clinical and Applied Thrombosis/Hemostasis
Footnotes
Acknowledgment
The authors acknowledge the support of University of Otago Postgraduate Scholarship and the University of Otago Research Grant, 2011.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially supported by University of Otago Research Grant 2011.
Supplemental Material
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
