Abstract
Thrombomodulin (THBD) serves as a cofactor for thrombin-mediated activation of anticoagulant protein C pathway. Genetic aberrations in THBD have been studied in arterial and venous thrombosis. However, genetic changes in THBD and their role in the risk assessment of recurrent venous thromboembolism (VTE) are not well understood. The aim of the present study was to identify the genetic aberrations in THBD and their association with the risk of VTE recurrence in a prospective population-based study. We sequenced the entire THBD gene, first in selected patients with VTE (n = 95) by Sanger sequencing and later validated those polymorphisms with minor allele frequency (MAF) ≥5% in the whole study population (n = 1465 with the follow-up period of 1998-2008) by Taqman polymerase chain reaction. In total, we identified 8 polymorphisms in THBD, and 3 polymorphisms with MAF ≥5% were further validated. No significant association between THBD polymorphisms and risk of VTE recurrence on univariate or multivariate Cox regression analysis was found (hazard ratio [HR] = 0.89, 95% confidence interval [CI] = 0.62-1.28, HR = 1.27, 95% CI = 0.88-1.85, and HR = 1.15, 95% CI = 0.80-1.66 for THBD rs1962, rs1042580, and rs3176123 polymorphisms, respectively), adjusted for family history, acquired risk factors for VTE, location of deep vein thrombosis, and risk of thrombophilia. Subanalysis of patients with unprovoked first VTE also showed no significant association of identified THBD polymorphisms with the risk of VTE recurrence. Our results show that aberrations in the THBD gene may not be useful for the assessment of VTE recurrence; however, further studies with large sample size are needed to confirm these findings.
Introduction
Venous thromboembolism (VTE), which includes deep vein thrombosis (DVT) and pulmonary embolism (PE), is a frequent, potentially lethal disease with an incidence rate of 1 to 2 cases per 1000 persons-years. 1,2 Patients with the first episode of VTE are at increased risk of new events of VTE. The risk of recurrence varies with time after the first event, it is higher in the first 6 to 12 months and never becomes zero. 3 The rate of VTE recurrence is increasing with time and is reported as follows: after 2 years as 17.5%, after 5 years as 24.6%, and it becomes 30.3% after 8 years of the first diagnosis with primary VTE. 4 Previous studies show that the rate of recurrence is higher in unprovoked VTE (without identified acquired risk factors for VTE, eg, older age, trauma, major surgery, immobilization, female hormone therapy, pregnancy) as compared to those patients with provoked VTE. 5 Standard treatment regimen of acute VTE is the usage of anticoagulant drugs for several months. Anticoagulant therapy is a “double-edged sword” that is efficient to prevent from recurrent VTE, albeit at the cost of severe bleeding with a case-fatality rate of 11.3%. 6,7 Irrespective of the number of known risk factors such as sex, residual thrombosis, and D-dimers level, the probability of VTE recurrence after stopping the anticoagulation treatment could not be predicted precisely. 6 –8 From the clinical perspective, it might be very useful to possess new biomarkers that allow early identification of patients at low or high chance of recurrent VTE and permit therapy assessment accordingly.
The risk of venous thrombosis is increased when the hemostatic balance between procoagulant and anticoagulant forces is shifted in favor of coagulation. When this is caused by an inherited defect, the resulting hypercoagulable state is a lifelong risk factor for thrombosis. Familial and twin studies suggest that the heritability of VTE is as high as 50% to 60% that could be explained by genetic factors and most of which is still unknown. 9,10 A study by Germain et al suggests that 7% of total genetic variation involved in VTE susceptibility might be explained by the chromosome 20 because it may harbor unidentified genetic variants. 11
Even though a number of genes involved in VTE have been identified, the major part of the heritability for VTE remains unknown. The role of thrombomodulin (THBD) in coagulation is well defined in preclinical models, that is, it has an indirect anticoagulant function. Animal model data suggest that transgenic mice with THBD mutations (targeted point mutation that substitute the glutamic acid 404 with proline) possess a prothrombic disorder. 12 Another study showed that mice with ablated THBD died right after the birth because of the consumptive coagulopathy. 13 Thrombomodulin is localized on chromosome 20, which transcribes a transmembrane protein that is present on the surface of vascular endothelial cells. Thrombomodulin has at least 3 independent activities involved in the anticoagulation pathway: inhibition of thrombin by antithrombin, activation of protein C, and inhibition of thrombin-mediated clotting (activation of procoagulant factors V, VIII, XI, XIII, and platelets) by altering the substrate specificity. 14,15
First mutation in THBD gene was identified by Ohlin and Marlar, which was a missense mutation predicting a change in amino acid from Asp468 to Tyr. 16 Clinical studies have shown numerous functional genetic alterations in THBD that lead to impaired function of THBD in arterial and venous thrombosis. 17 –20 Several studies have analyzed the genetic aberrations in THBD and their association with the risk of primary VTE with controversial results. 20 –23,25,26 However, there is not much information available on the role of genetic aberrations in THBD gene in recurrent VTE. To investigate genetic aberrations related to VTE recurrence, we in this study sequenced the whole THBD gene including promotor region −1600 base pair (bp), exon, and 5′ and 3′ untranslated regions (UTRs) in a well-established recurrent VTE cohort. To our knowledge, this is the first study in which the whole THBD gene is sequenced in a prospective, follow-up study of patients with recurrent VTE.
Materials and Methods
Study Participants
Patients with VTE (n = 1465) were selected from Malmö Thrombophilia Study (MATS), a prospective population-based study of consecutive unselected patients with VTE performed at Skåne University Hospital. Malmö Thrombophilia Study is a well-characterized cohort in which patients with VTE were included and followed from 1998 until VTE recurrence or death or at the end of the study (December 2008). 27,28 The inclusion criteria in MATS were an objective diagnosis of DVT, PE, or recurrence performed by duplex ultrasonography, phlebography, computed tomography, lung scintigraphy, or magnetic resonance imaging; age >18 years; and patients’ ability to communicate in Swedish. For the screening of hospital records of patients with VTE, a research nurse was assigned. The rate of consensual participation in MATS was 70%. The remaining 30% of patients with VTE were excluded because they did not participate in questionnaire, language problems, the presence of other severe diseases, and in a few cases, dementia and unwillingness to participate in MATS. Immobilization and cast therapy, hospitalization, surgical intervention, malignancies that were diagnosed previously or at diagnosis of VTE, hormonal therapy, use of contraceptive pills, pregnancy and postpartum period (first 6 weeks after delivery), family history of VTE (history of VTE in first-degree relatives), VTE events before inclusion into the study, VTE recurrence during follow-up period, and location of DVT at inclusion were recorded.
Malmö University Hospital standard protocol was used to treat all patients with VTE, that is, with low-molecular-weight heparin or unfractionated heparin and then with warfarin as an oral anticoagulants (OACs). Malmö University Hospital treatment protocol recommends 3 to 6 months of OAC therapy for first-time VTE with the consideration of extension of treatment if VTE recurrence occurs. Thrombophilia was defined as the presence of the factor II G20210A mutation (rs1799963), factor V Leiden (FVL) mutation (rs6025), or a level below the laboratory reference range of protein C (<0.7 kilo international unit [kIU]/L), antithrombin (<0.82 kIU/L), or free protein S (women <0.5 kIU/L, men <0.65 kIU/L) in patients without warfarin treatment.
After stopping the anticoagulant treatment, follow-up period (mean ± standard deviation [SD], 3.9 ± 2.5) was counted in years until the diagnosis of VTE recurrence or the end of study (December 2008).
All the participants provided written permission before their inclusion into the study, according to the Declaration of Helsinki. Ethical approval for this study was obtained from the Lund University ethical committee.
Laboratory Methods
DNA Extraction and Primer Designing
Whole blood was used to extract the DNA by using the QiAmp 96 DNA Blood Kit (Qiagen, Hilden, Germany). Online software, Primer-Blast, by National Center for Biotechnology Information (NCBI) was used to design the polymerase chain reaction (PCR) primers for the THBD gene including promotor region (−1600 bp), exon, and 5′ and 3′ UTR. 29 Thrombomodulin gene sequence was obtained from the publicly available NCBI database (NCBI reference sequence: NC_000020.11, GI: 568815578). M13-tailed primer sequences (M13 forward [F] = TGTAAAACGACGGCCAGT, M13 reverse [R] = CAGGAAACAGCTATGACC) were added to each PCR primer before primer synthesis to get better sequencing results. To minimize the chances of sequencing errors at the beginning and end point of the amplicon, during the primer designing, it was kept in mind that each primer should overlap the amplicon of the next primer for at least 100 bp. Primers were subjected to PCR for optimization at different temperatures. A total of 10 primer pairs were designed to sequence the above-mentioned THBD gene regions. Primer sequences (F and R primers) along with their amplicon lengths are provided in Table 1.
Forward and Reverse Primers for THBD Promotor, Exon, and 5′ and 3′ UTR Along With Their Amplicon Length.a

Abbreviation: THBD, thrombomodulin.
aM13-tailed sequence was added to all primers before synthesis.
Patient Selection and THBD Sequencing
From 1465 patients with VTE, initial screening was performed on age at first VTE and sex-matched patients with nonrecurrent (n = 60) and recurrent VTE (n = 35) samples. Sanger sequencing was used to sequence the THBD gene in selected samples. 30 Polymerase chain reaction amplification of THBD was performed by using BigDye Direct Cycle Sequencing Kit (Applied Biosystems, Life Technologies Corporation, Carlsbad, California), according to the manufacturer’s protocol. Briefly, for each sample, 2.5 μL of deionized water, 5.0 μL of BigDye Direct Master Mix (MM), 1.5 μL of M13-tailed PCR primers (0.8 μM), and 1.0 μL genomic DNA (5 ng/μL) were used for the first PCR amplification. Following thermal cycling conditions were used to perform PCR, denaturation for 5 minutes at 95°C for 1 cycle, followed by 35 cycles of 30 seconds at 94°C, 45 seconds at 62°C, and 45 seconds at 68°C. The last cycle was performed at 72°C that lasts for 2 minutes. In second PCR, amplified PCR product from the first PCR was added with BigDye sequencing MM (2 μL) and BigDye Direct M13 F/R primers (1.0 μL) to run second PCR with following thermal conditions; start with 37°C for 15 minutes, 80°C for 2 minutes, 96°C for 1 minute followed by 25 cycles of 10 seconds at 96°C, 5 seconds at 50°C, and 4 minutes at 60°C. All PCR reactions were conducted in T100 Thermal Cycler (Bio-Rad Laboratories, Marnes-la-Coquette, France). DyeEx 96 Kit (Qiagen) was used to purify the PCR product, according to the manufacturer’s instructions. Purified DNA samples were sent to Eurofins Genomics (Eurofins Genomics, Ebersberg, Germany) for Sanger sequencing analysis. Lasergene sequence analysis software (DNA Star software; DNASTAR, Madison, Wisconsin) was used to align and analyze the Sanger sequencing data.
Genotyping of selected polymorphisms in the whole population was performed by TaqMan SNP Genotyping Assays (Applied Biosystems, Life Technologies Corporation). For THBD polymorphisms, predesigned Taqman genotyping assays were available that were used according to the manufacturer’s instructions (Applied Biosystems, Life Technologies Corporation).
Briefly, for each sample, a PCR MM was prepared by adding 2.5 μL Taqman MM, 0.25 μL Taqman gene-specific assay probes complimentary to wild-type and the mutant allele (VIC and FAM probes), and 0.25 μL deionized water. A 384 PCR plate was used to run the assay and 3 μL of MM was added to each well followed by addition of 10 ng (2 μL) genomic DNA. Polymerase chain reaction plates were vortexed and centrifuged at 1000 rpm for 30 seconds. BioRad CFX384 real-time PCR (Hercules, California) was used for polymorphism analysis with the following temperature conditions, 95°C for 10 minutes followed by ×40 (92°C for 15 seconds, 60°C for 1 minute). Different alleles of the polymorphisms were determined by BioRad CFX manager software 3.1.
Analysis of Known Thrombophilic Variants
Taqman allele discrimination assays (Applied Biosystems, Life Technologies Corporation) were used for DNA mutation analysis in FVL and factor II G20210A as described previously. 31 Protein C levels were analyzed by a chromogenic method using the Berichrom Protein C reagent (Siemens Healthcare Diagnostics, Upplands Väsby, Sweden). 32 Latex immunoassay with Coamatic protein S-free (Chromogenix; Haemochrom Diagnostica AB, Gothenburg, Sweden) was used for free protein S analysis. 33 Antithrombin analysis was performed by thrombin-based method using Berichrom Antithrombin (Siemens Healthcare Diagnostics). 34 For all analysis, BCS-XP coagulation analyzer (Siemens Healthcare Diagnostics) was used.
Statistical Analysis
SPSS version 21 (IBM, Armonk, New York) was used for statistical analysis performance. Continuous variable were compared by Mann-Whitney U test, and dichotomous variables were compared by χ2 test or Fisher exact test where appropriate. Univariate and multivariate Cox regression analyses, after adjusting for family history and location of VTE, mild and severe thrombophilia, and acquired risk factors for VTE, were performed using Cox proportional hazards models. Hazard ratios (HRs) with 95% confidence intervals (CIs) were calculated for each group of patients. Multivariate Cox regression analyses were performed as sensitivity analyses by including all patients with VTE except those who had had thrombotic events before inclusion. Follow-up period for sensitivity analyses was calculated from the time of inclusion of patients and was adjusted for the duration of anticoagulation treatment, family history, location of VTE, mild and severe thrombophilia, and acquired risk factors for VTE. Hardy-Weinberg equilibrium analysis was performed to see the genotypic distribution.
Results
Clinical Data of the Patients
Of 1465 patients, 154 had thrombotic events before inclusion, therefore, they were excluded from further analysis. For the remaining patients with VTE (n = 1311), 148 (11.3%) had recurrence during the follow-up period. Of the patients with recurrent VTE, 40.8% had FVL mutations as compared to 28.5% in nonrecurrent VTE (P = .002). Frequency of family history for VTE was significantly lower in patients with nonrecurrent VTE (23.5%) as compared to patients with recurrent VTE (32.4%; P = .024), whereas no significant difference in age, gender, body mass index, factor II mutations, protein C, protein S, and antithrombin deficiency was found between patients with recurrent and nonrecurrent VTE (P > .05; Table 2).
Characteristics of Studied Population Including the Distribution of THBD Genotypes Stratified by Recurrent and Nonrecurrent Status.a
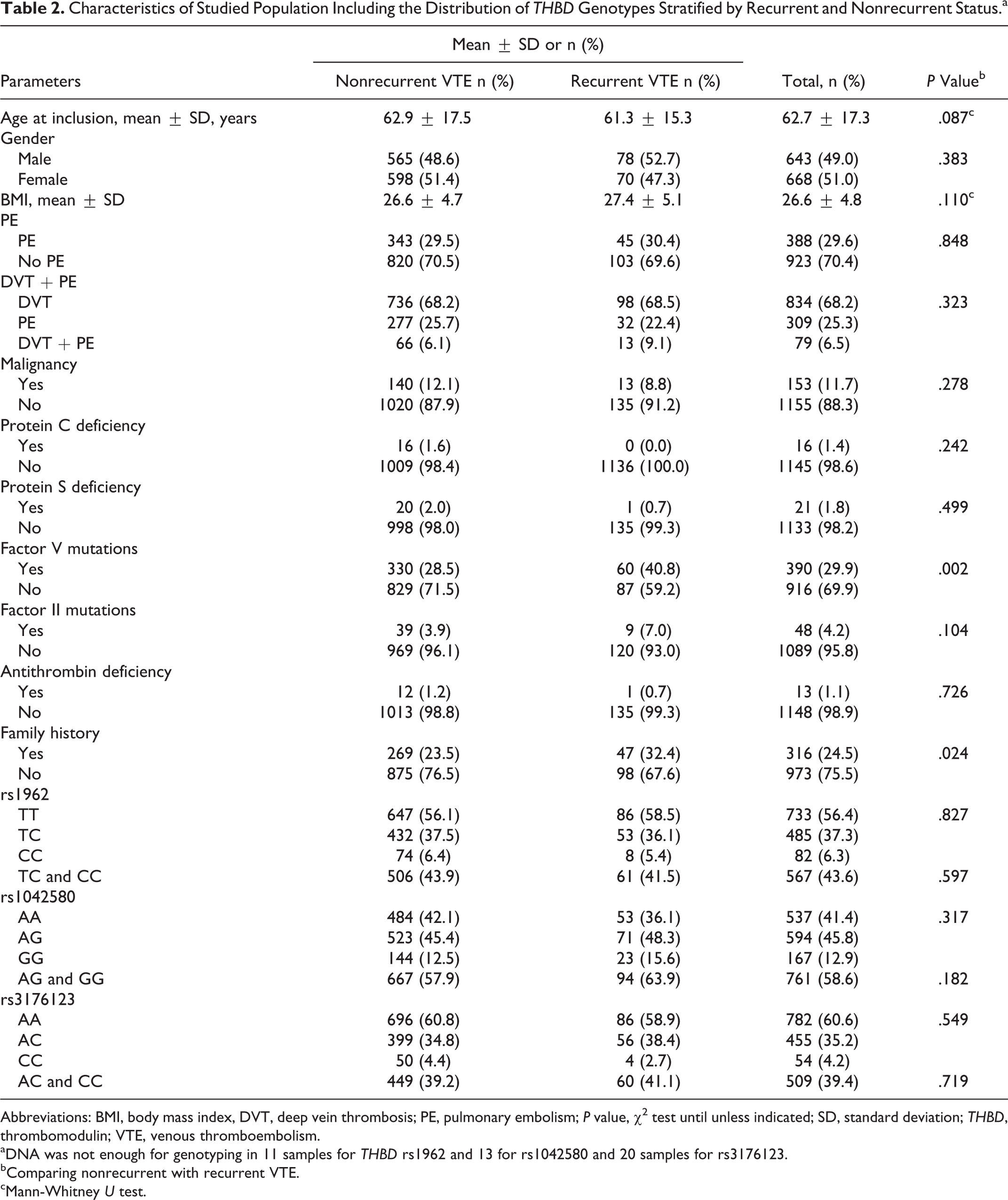
Abbreviations: BMI, body mass index, DVT, deep vein thrombosis; PE, pulmonary embolism; P value, χ2 test until unless indicated; SD, standard deviation; THBD, thrombomodulin; VTE, venous thromboembolism.
aDNA was not enough for genotyping in 11 samples for THBD rs1962 and 13 for rs1042580 and 20 samples for rs3176123.
bComparing nonrecurrent with recurrent VTE.
cMann-Whitney U test.
Screening of THBD Genetic Aberrations in VTE
During initial screening on age- and sex-matched samples (n = 95), we identified 8 polymorphisms (THBD rs3176117 [2%], rs3176130 [1%], rs369909901 [1%], rs41282276 [1%], rs1042579 [22%], rs1042580 [36%], rs1962 [25%], and rs3176123 [22%]) in THBD gene. Of them, 4 polymorphisms (THBD rs1042579, rs1042580, rs1962, and rs3176123 polymorphisms) were identified with minor allele frequency (MAF) >5% in patients with VTE. Thrombomodulin rs1042579 (1418C>T) polymorphism has been previously studied for its association with the risk of VTE recurrence in the same cohort, 24 and the remaining 3 polymorphisms, that is, THBD rs1962 (3646T>C), rs1042580 (2521A>G), and rs3176123 (2729A>C) polymorphisms, were analyzed in all MATS samples (n = 1311) by TaqMan PCR. No deletions or insertions in the THBD gene were identified in our cohort.
All the THBD polymorphisms have 3 different genotypic forms, homozygous wild type, heterozygous-, and homozygous-mutated form. In data analysis, all 3 genotypic forms (for each polymorphism) were analyzed separately as well as by combining homozygous-mutated and heterozygous-mutated form. As the results were similar after combining the homozygous-mutated and heterozygous-mutated form, in unprovoked VTE and sensitivity analyses, we only presented the results for combined homozygous-mutated and heterozygous-mutated forms. The distribution of identified THBD polymorphisms was not significantly different between recurrent and nonrecurrent VTE (Table 2). Genotypic distributions in THBD polymorphisms did not deviate significantly (P > .05) in Hardy-Weinberg equilibrium analyses.
Thrombomodulin Polymorphisms and Risk of VTE Recurrence
Of 1311 patients with VTE, those who died, who had VTE recurrence during anticoagulant treatment, or in whom complete information was missing were excluded for the risk analysis of recurrent VTE (n = 261). Among the remaining 1050 patients with VTE, 126 (12%) had recurrent VTE during follow-up period. On univariate Cox regression analysis, we found no significant association between risk of VTE recurrence and THBD polymorphisms identified in this study, and the HRs and CIs were as follows: HR = 0.90, CI = 0.63 to 1.29, HR = 1.30, CI = 0.90 to 1.87, and HR = 1.14, CI = 0.80 to 1.62 for THBD rs1962, rs1042580, and rs3176123 polymorphisms, respectively. Similar results were found on multivariate Cox regression analysis (HR = 0.89, CI = 0.62-1.28, HR = 1.27, CI = 0.88-1.85, and HR = 1.15, CI = 0.80-1.66 for THBD rs1962, rs1042580, and rs3176123 polymorphisms, respectively) adjusted for acquired risk factors of VTE, mild (heterozygous prothrombin G20210A or FVL) and severe thrombophilia (homozygous carriers of FVL or those patients who had natural anticoagulant deficiencies, eg, protein C, antithrombin, protein S deficiency, and/or carriers of multiple abnormalities), family history of VTE, and location of DVT. We also analyzed the association between identified polymorphisms and risk of VTE recurrence according to gender and, however, found no significant associations between identified polymorphism and the risk of VTE recurrence in either sex (Table 3).
Uni- and Multivariate Analyses of THBD rs1962, rs1042580, and rs3176123 Polymorphisms in Patients With Recurrent VTE.
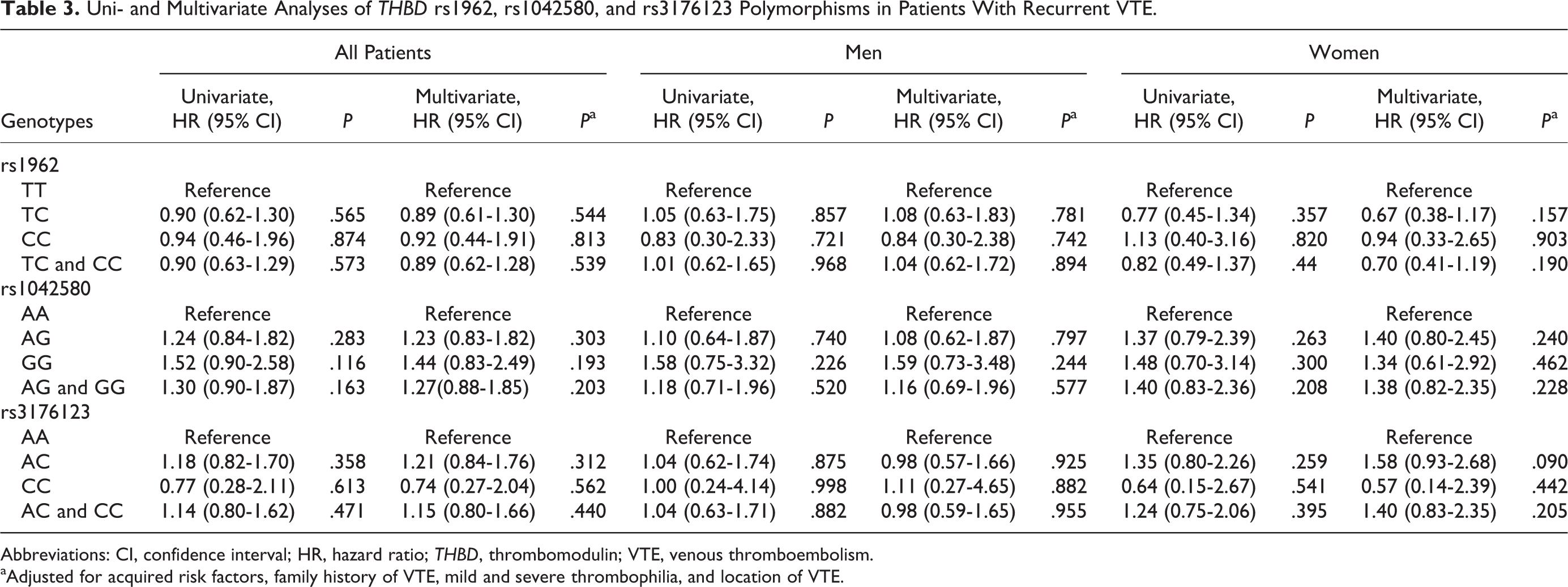
Abbreviations: CI, confidence interval; HR, hazard ratio; THBD, thrombomodulin; VTE, venous thromboembolism.
aAdjusted for acquired risk factors, family history of VTE, mild and severe thrombophilia, and location of VTE.
Thrombomodulin Polymorphisms and Risk of VTE Recurrence in Patients With Unprovoked VTE
We further performed a subanalysis on patients with unprovoked VTE. Patients with a recorded acquired risk factor for VTE, that is, immobilization or cast therapy within the last month, surgical intervention, use of contraceptives pills, malignancies diagnosed prior to or at diagnosis of the first VTE event, current pregnancy and postpartum period (first 6 weeks after delivery), and female hormone therapy were excluded during this analysis. Patients with unprovoked VTE (n = 618) with 81 (13.1%) patients with recurrent VTE were selected to investigate the association between THBD polymorphisms and the risk of VTE recurrence as a whole and according to gender. No significant association was found between THBD polymorphisms and risk of VTE recurrence in univariate Cox regression analysis (HR = 0.68, CI = 0.43-1.07; HR = 1.43, CI = 0.90-2.26; and HR = 1.25, CI = 0.80-1.95 for THBD rs1962, rs1042580, and rs3176123 polymorphisms, respectively; Table 4). On stratification of data according to gender, we found that THBD rs1962 polymorphism was associated with the risk of VTE recurrence in female patients; however, results did not reach the significant level (HR = 0.53, CI = 0.27-1.04, P = .065) on univariate Cox regression analysis (Table 4). On multivariate Cox regression analysis, after adjusting for mild and severe thrombophilia, family history of VTE, and location of DVT, THBD 1962 (T>C) polymorphism was significantly associated with lower risk of VTE recurrence in female patients (HR = 0.41, CI = 0.21-0.82, P = .012; Table 4).
Uni- and Multivariate Analyses of THBD rs1962, rs1042580, and rs3176123 Polymorphisms in Patients With Unprovoked Recurrent VTE.

Abbreviations: CI, confidence interval; HR, hazard ratio; THBD, thrombomodulin; VTE, venous thromboembolism.
aAdjusted for family history of VTE, mild and severe thrombophilia, and location of VTE.
We also performed sensitivity analyses on all MATS patients except those who were diagnosed with VTE before inclusion (n = 154). Multivariate analyses were performed for the remaining patients (n = 1311 including 148 patients with recurrent VTE) with follow-up time from the time of inclusion for this study (adjusted for duration of anticoagulant treatment, mild and severe thrombophilia, location of DVT, family history, and acquired risk factors for VTE) and similarly found no association between any of the identified THBD polymorphism and risk of VTE recurrence (Supplemental Table 1).
Discussion
In the present study, we have sequenced the whole THBD gene (promotor region −1600 bp, exon, and 5′ and 3′ UTRs) in a well-defined prospective cohort of patients with recurrent VTE to identify the genetic aberrations in THBD associated with VTE recurrence. We first performed an initial screening by Sanger sequencing and in total identified 8 single-nucleotide polymorphisms in THBD (rs3176117, rs3176130, rs369909901, rs41282276, rs1042579, rs1042580, rs1962, and rs3176123). For validation, polymorphisms with MAF more than 5% (rs1042580, rs1962, and rs3176123) were analyzed in the whole population.
Our results show that none of these 3 polymorphisms (rs1042580, rs1962, and rs3176123) were significantly associated with the risk of VTE recurrence (P > .05) in all patients. We could not find any other study showing the role of THBD rs1042580, rs1962, and rs3176123 polymorphisms in recurrent VTE; however, these polymorphisms have been analyzed in primary VTE and other cardiovascular diseases. Similar findings for THBD rs1042580 polymorphism were reported by Sugiyama et al and Arellano et al showing that this polymorphism is not associated with risk of primary venous thrombosis in Japanese population and in Multiple Environmental and Genetic Assessment (MEGA) study, respectively. 25,35 Furthermore, Auro et al have shown that THBD rs1042580, rs1962, and rs3176123 polymorphisms did not contribute significantly to the risk of cardiovascular events. 23 Another study shows that THBD rs1042580 polymorphism alone may not be a risk factor for cardiovascular events, however, in association with another gene polymorphisms involving coagulation pathway (F5 gene), is a risk factor for cardiovascular events in female patients. 36 Therefore, there is a possibility that THBD polymorphisms alone do not play any significant role in recurrent VTE also and may be influenced by other genes present in the coagulation pathway. Thrombomodulin rs3176123 and rs1962 polymorphisms have also been studied in patients with primary deep venous thrombosis. Thrombomodulin rs3176123 polymorphism was significantly associated with a higher risk of DVT in male patients and levels of the soluble THBD. 25 Furthermore, THBD rs3176123 polymorphism was associated with increased mortality after coronary artery bypass surgery. 37 However, in our study, THBD rs3176123 polymorphism was not associated with the risk of VTE recurrence. A possible explanation for these results could be due to the fact that the risk factors that are associated with primary VTE may not be the same as for the VTE recurrence. 8,38
It remains a challenge to predict the individual risk of VTE recurrence in patients with unprovoked VTE. We further investigated the role of THBD polymorphisms in patients with unprovoked VTE; none of the identified polymorphisms were significantly associated with the risk of VTE recurrence. However, on multivariate Cox regression analysis (adjusted for family history of VTE, mild and severe thrombophilia, and location of DVT), after stratification of data according to gender, THBD rs1962 polymorphism was significantly associated with lower risk of VTE recurrence in female patients and found that family history was a confounding factor. These results show that THBD rs1962 polymorphism may be a risk factor for VTE recurrence in female patients with a family history of VTE. However, due to the lower number of patients (total 320 female patients with 41 having recurrent VTE during follow-up) in this analysis, these results should be interpreted with caution. We suggest further studies with a larger number of female patients to confirm the association between THBD rs1962 polymorphism and risk of recurrent VTE in patients with unprovoked VTE.
Thrombomodulin is an integral part of protein C pathway which plays an important role in coagulation inhibition. It is present on the endothelium throughout the vascular system. However, its concentration is higher (approximately 500 nmol/L) in the capillaries as compared to the large vessels (approximately 0.1-0.2 nmol/L). This scarcity of THBD in large vessels is compensated by another receptor called endothelial protein C receptor (EPCR), which binds to protein C and activates the protein C complex. 39 –41 Our results show that THBD polymorphisms may not play a role in VTE recurrence. It is therefore possible that THBD polymorphisms may be important for thrombosis events in microvasculature/capillaries where there is no other receptor to bind with protein C and activate protein C pathway, while in large vessels, the lack of THBD is counterbalanced by EPCR. Thus, polymorphisms in THBD might be relevant in diseases with microthrombotic mechanisms, for example, purpura fulminans rather than large-vessel thrombosis.
There are several rare mutations/polymorphisms in THBD that were reported previously in other diseases (eg, Asp468Tyr, Ala25Thr, del791-801, etc) but not identified in our cohort. 16,19,21,22 The reasons for this discrepancy could be the lower frequency of those polymorphisms, different diseases, and different ethnic populations involved in previous studies. 42,43
Minor allele frequencies for THBD rs3176117, rs3176130, rs369909901, and rs41282276 polymorphisms were found to be very low (1% for each polymorphism except rs3176117 with 2% MAF) and therefore were not further analyzed in this study. Thrombomodulin rs3176117, rs369909901, and rs41282276 polymorphisms were not studied previously in any other disease, most probably due to very low MAFs (0.4%, 0.06%, and 0.08%, respectively, as are reported in NCBI). 44 Thrombomodulin rs3176130 polymorphism was studied previously in only 1 study on acute respiratory distress syndrome; however, it was not associated with the risk of disease. 45
One of the potential limitations of our study is that we could not analyze THBD rs3176117, rs3176130, rs369909901, and rs41282276 polymorphisms due to lower MAFs.
In conclusion, for the first time, the whole THBD gene is sequenced in patients with recurrent VTE. We found 8 polymorphisms in the THBD gene in Swedish population. None of these polymorphisms was found to be significantly associated with the risk of VTE recurrence. Our results indicate that THBD polymorphisms may not be a risk factor for VTE recurrence, although larger studies may be needed to rule out THBD polymorphisms as useful predictors for recurrent VTE.
Footnotes
Authors’ Note
K.S., B.Z., J.S., and A.A.M. conceived and designed the study. AAM and AA performed the experiments. All authors performed the data analysis and interpretation and drafted and revised the article and approved the final version.
Acknowledgments
The authors would like to thank biobank services at Biobank, Lab medicine Skåne, Sweden.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants awarded to Dr Bengt Zöller by the Swedish Heart-Lung Foundation, ALF funding from Region Skåne awarded to Dr Bengt Zöller and Dr Kristina Sundquist, grants awarded to Dr Bengt Zöller and Dr Kristina Sundquist by the Swedish Research Council and grants awarded to Dr Jan Sundquist by King Gustaf V and Queen Victoria’s Foundation of Freemasons. The funders played no role in study design; in the collection, analysis, and interpretation of data; in the writing of the report; and in the decision to submit the article for publication.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
