Abstract
Background:
Poloxamer-188 (MST-188) is effective in the repair/recovery of damaged cell membranes. MST-188 is a promising agent for protecting blood cell viability. The aim of the study is to test the hypothesis that MST-188 can extend the duration of platelet function.
Materials and Methods:
Blood samples were collected from 20 healthy volunteers. MST-188 (10 or 2 mg/mL) containing platelet-rich plasma (PRP) was prepared with 2 procedures. First, PRP prepared from MST-188 added whole blood (WB); second, MST-188 was added to PRP. These were referred to MST-188-WB preparation (WBP) and MST-188-PRP preparation (PRPP), respectively. For control, saline was used in the same manner. Agonist-induced aggregation (AIA) studies were performed at 30, 180, and 300 minutes using Platelet Aggregation Profiler (PAP-8) aggregometer (Bio/Data Corporation, Horsham, Pennsylvania) and Adenosine diphosphate (ADP), arachidonic acid, collagen, and epinephrine as agonists at final concentration of 20 µM, 500 µg/mL, 0.19 mg/mL, and 100 µM, respectively.
Results:
There was a protective effect of MST-188 on ADP and collagen AIA. At 300 minutes, ADP AIA was found to be 50.2% higher than saline control in 2-mg WBP, 43% at 10-mg PRPP, and 10.4% at 2-mg PRPP. Protective effect of on collagen AIA was 65.9% in 2-mg WBP, 42.74% at 10-mg PRPP, and 11.42% at 2-mg PRPP. In comparison between 30 and 300 minutes, MST-188 showed significant protection in terms of ADP and collagen receptors and for both types of preparations (WBP and PRPP).
Conclusion:
The protective effects of MST-188 on ADP- and collagen-induced platelet aggregation may contribute to the preservation of platelet functionality upon storage in blood banks.
Introduction
MST-188 (Poloxamer-188, vepoloxamer) is an amphiphilic, nonionic, triblock copolymer surfactant. 1 MST-188 is effective in recovery of damaged cell membrane on muscle cells after electroshock, 2 neurons after trauma, 1 and fibroblasts 3 after heat shock. MST-188 also reduces the rigidity of sickled erythrocytes 4 and contributes to the resolution of symptoms of vaso-occlusive crisis 5 in patients with sickle cell disease.
The cell protective effect of MST-188 is not only related to its action on cell membrane integrity but perhaps to some other additional mechanisms that have been described. Antiapoptotic effect of MST-188 has been shown on neurons after trauma which is related to an inhibition of the activation of proapoptotic p38. 1 MST-188 prevents blood–brain barrier disruption through nuclear factor kappa B (NF-κB)–matrix metalloproteinases (MMPs)-mediated tight junction protein degradation by decreasing the level of MMP2, MMP9, and NF-KB p65. 6
MST-188 enhances survival of red blood cells (RBCs) by increasing the stability and decreasing the fragility of the membrane. 7 This protective effect has been used in the in vitro generation of RBCs from hematopoietic stem cells. 7 MST-188 is an attractive and promising agent for protecting blood cell viability and function during storage in blood banks and in vitro production of these cells in the future. Since platelets lose some part of their functionality during storage, 8 it was hypothesized that MST-188 may protect the ability of stored platelets to aggregate. The aim of this study was to test the protective effect of MST-188 on platelet function.
Materials and Methods
Blood was drawn from cubital vein of healthy male and female volunteers (n = 20) using a double-syringe technique and placed in 4 tubes containing 3.2% sodium citrate solution (1 part of citrate and 9 parts of whole blood [WB]). To facilitate the blood draw, a 21G 3/4 × 12′′ (0.8 mm × 1 9 mm × 304.8 mm) butterfly needle (Vacutainer Brand Safety-Lok Blood Collection Set) and 10 mL syringes manufactured by Becton Dickinson and Company, Franklin Lane, New Jersey, were used. To investigate the effects of MST-188 on platelet function, 2 arms (arm I and arm II) were established.
In arm I, the citrated WB was divided into 2 tubes (tube A and tube B). In the tube A, MST-188 (vepoloxamer, Mast Therapeutics, San Diego, California) was added to citrated WB (1 mL of MST-188 at 100 mg/mL to 9 mL of citrated WB) and mixed by inverting the tubes several times to achieve a final concentration of 10 mg/mL MST-188. In the tube B, saline was added in the same proportion and mixed by inverting the tubes several times. The MST-188 and saline containing tubes were centrifuged at 72g for 20 minutes and the supernatant platelet-rich plasma (PRP) was transferred to a clean tube and labeled as MST-188-WB preparation (MST-188-WBP) for tube A and saline-WB preparation (saline-WBP) for tube B. Following the separation of PRP, the blood samples were centrifuged at 1008g for 20 minutes and the supernatant platelet-poor plasma (PPP) was transferred to a clean tube.
In arm II, citrated WB was centrifuged as mentioned previously to obtain PRP and PPP. One milliliter of MST-188 at 100 mg/mL was added to PRP to achieve a final concentration of 10 mg/mL. For control, 1 mL of saline was added in the same manner. These samples were labeled as MST-188-PRP preparation (MST-188-PRPP) and saline-PRP preparation (saline-PRPP). The above procedures were repeated for MST-188 at a final concentration of 2 mg/mL. Thus, 4 groups (10 mg/mL WBP, 2 mg/mL WBP, 10 mg/mL PRPP, and 2 mg/mL PRPP) of samples were made. For the 4 groups, 5 donors each were studied. The samples were kept at room temperature for up to 300 minutes. During this period, Agonist-induced aggregation (AIA) studies were performed at 30, 180, and 300 minutes using a Platelet Aggregation Profiler (PAP-8) aggregometer (Bio/Data Corporation). Agonists were purchased from (Bio/Data Corporation). The agonists that were used included—Adenosine diphosphate (ADP) at final concentration of 20 µM, arachidonic acid (AA) at final concentration of 500 µg/mL, collagen at final concentration of 0.19 mg/mL, and epinephrine at final concentration of 100 µM.
Means and standard deviations for aggregation and slopes were calculated for 3 different time points. At 300 minutes, the percentage change of MST-188 preparations was calculated against the saline control counterparts.
The differences for aggregation and slope between 30 and 300 minutes are also calculated to detect the functionality lose during 300 minutes for all preparations. For statistical analysis (2 × 4 × 2 × 2), factorial experiment with 4 factors was used. Factors were additive solution (saline/MST-188), receptors (ADP/AA/collagen/epinephrine), final concentration of additive solution (2 or 10 mg/dL), and preparation (WBP/PRPP). A P value less than .05 was accepted as statistically significant.
Results
In the saline supplemented systems, a time-dependent decrease in platelet aggregation induced by all agonists was observed. At 30, 180, and 300 minutes, ADP-induced aggregation mean levels were 82.0%, 71.7%, and 49.1%, respectively; AA-induced aggregations were 66.6%, 40.7, and 27.6%, respectively; collagen-induced aggregations were 83.2%, 74.8%, and 56.7%, respectively; and the epinephrine-induced aggregations were 76.8%, 61.2%, and 33.7%, respectively.
In the analysis of the percentage change of MST-188 preparations compare to saline control counterparts at 300 minutes, the effect of MST-188 was found to be agonist dependent in the MST-188 supplemented systems. There was a protective effect on ADP- and collagen-induced aggregation. However, this protective effect for ADP and collagen was not observed at 10-mg WBP. A comparison of the 10-mg WBP and saline control at 300 minutes showed less aggregation in terms of percentage change in MST-188 system (13.9% less for ADP and 10.3% less for collagen).
At 300 minutes, the loss of functionality for AA and epinephrine was consistently observed in all preparations and concentrations. The functionality loss for AA and epinephrine was 71.2% and 74.6% at 10-mg WBP, 45.1% and 78.6% at 10-mg PRPP, 42.9% and 79.0% at 2-mg WBP, 19.3% and 23.2% at 2-mg PRPP, respectively.
At 300 minutes, the observed protective effect of MST-188 on ADP-induced aggregation was found to be 50.2% higher than saline control in 2-mg WBP. Platelets gave 50.2% more aggregation than saline control at 300 minutes. This protective effect in terms of percentage change was found to be 43.1% at 10-mg PRPP and 10.4% at 2-mg PRPP (Figures 1 and 2).
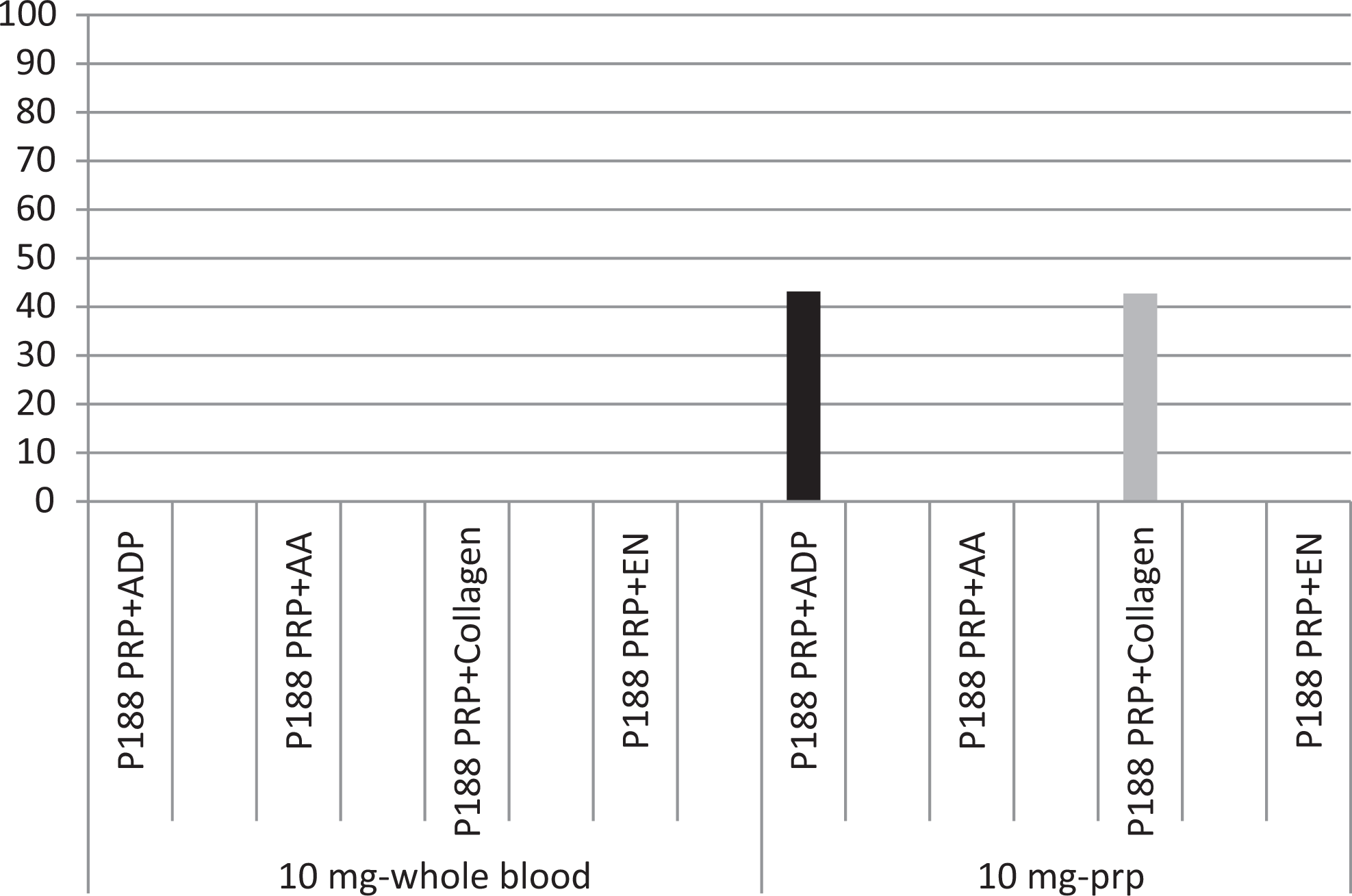
The protective effect of MST at 10 mg/mL final concentration.
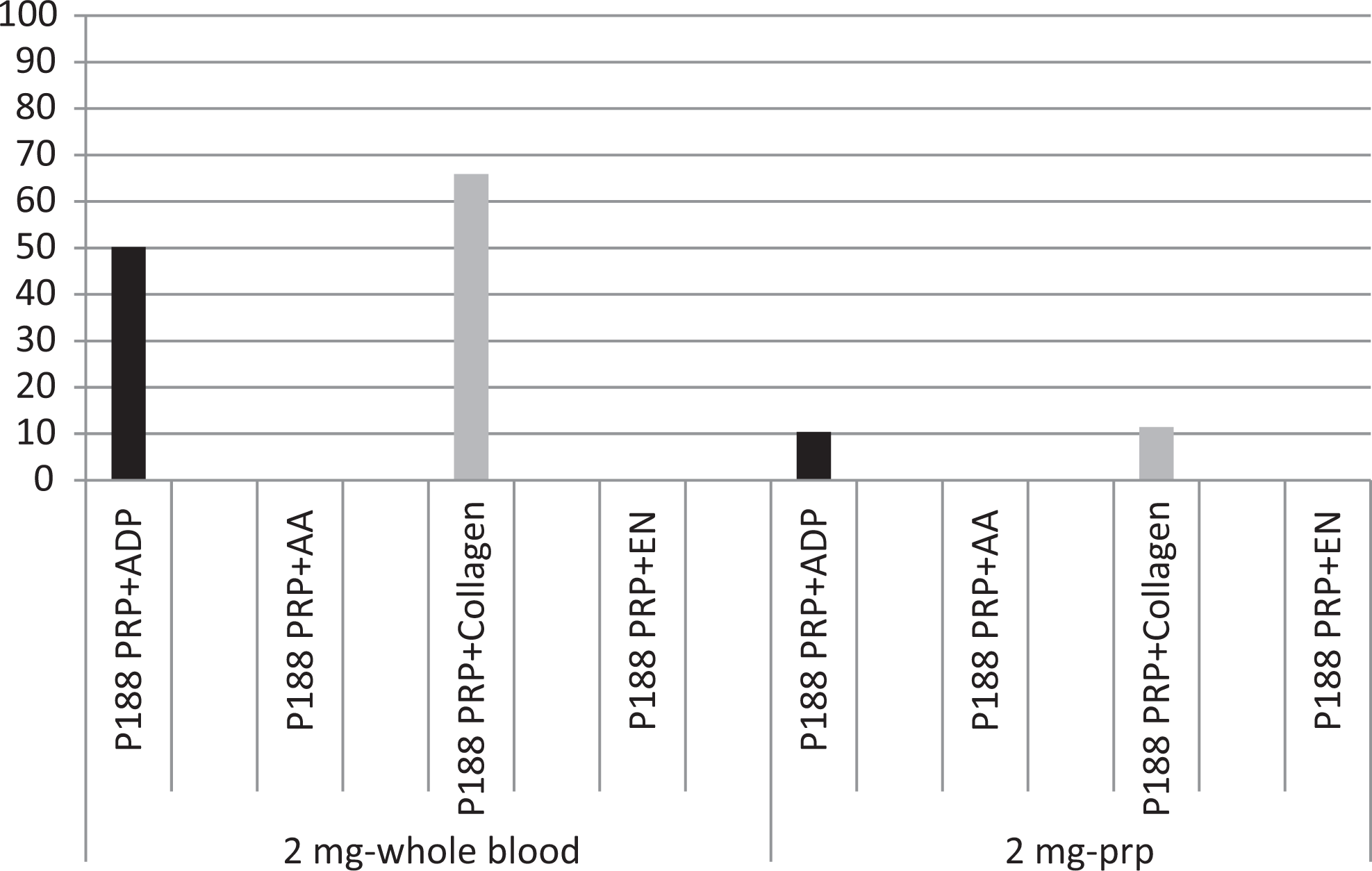
The protective effect of MST at 2 mg/mL final concentration.
At 300 minutes, the protective effect on collagen-induced aggregation in terms of percentage change was 65.9% compared to saline control in 2-mg WBP. Platelets gave 65.9% more aggregation than saline control in terms of percentage change at 300 minutes. At 10-mg PRPP and at 2-mg PRPP, this protective effect was 42.74% and 11.4%, respectively (Figures 1 and 2).
For the analysis of detection of losing the functionality of aggregation during 5 hours period from 30 to 300 minutes in terms of differences at mean values between 30 and 300 minutes, 4-factorial statistical experiment was done.
In the analysis of receptors and additive solution (saline/MST-188), ADP- and collagen-induced aggregation and slopes were statistically significantly protected at during 300 minutes in MST-188 preparations compare to saline preparations (Figure 3). P values for aggregation with ADP, AA, collagen, and epinephrine were .013, .383, .005, and .162, respectively. P values for slopes related with ADP, AA, collagen, and epinephrine were .017, .545, .009, and .646, respectively.
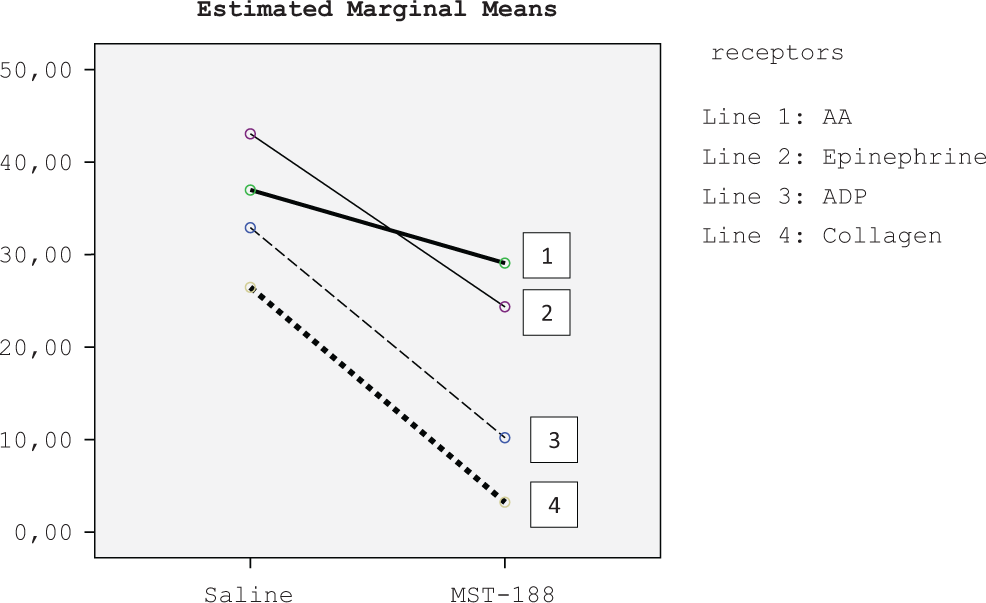
Decreasing of aggregation at the end of 5 hours according to receptors.
In the analysis of final concentration of additive solution (2 mg/dL or 10 mg/dL) and additive solution (saline/MST-188), aggregation and slopes were statistically significantly protected at during 300 minutes in 10 mg/mL final concentration of MST-188 preparation compare to saline preparation (Figure 4). P values of aggregation and slope for 10 mg/mL were <.0001 and .001, respectively. P values of aggregation and slope for 2 mg/mL were .24 and .162, respectively.
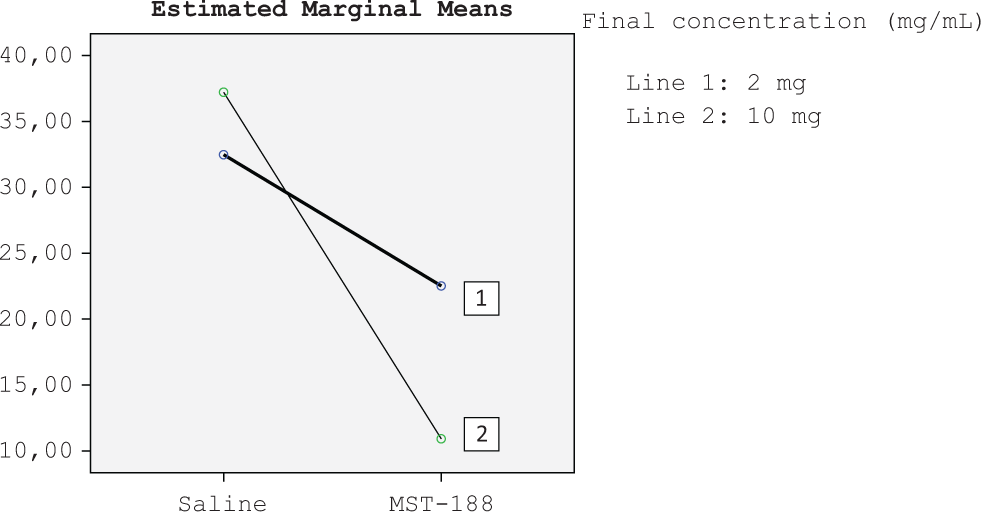
Decreasing of aggregation at the end of 5 hours according to amount of additive solutions.
In the analysis of preparation (WBP/PRPP) and additive solution (saline/MST-188), aggregation and slopes for both preparations with MST-188 were statistically significantly protected at during 300 minutes compare to saline counterparts (Figure 5). P values for aggregation in WBP and PRPP were .014 and .035, respectively. P values for slopes in WBP and PRPP were .016 and .025, respectively.
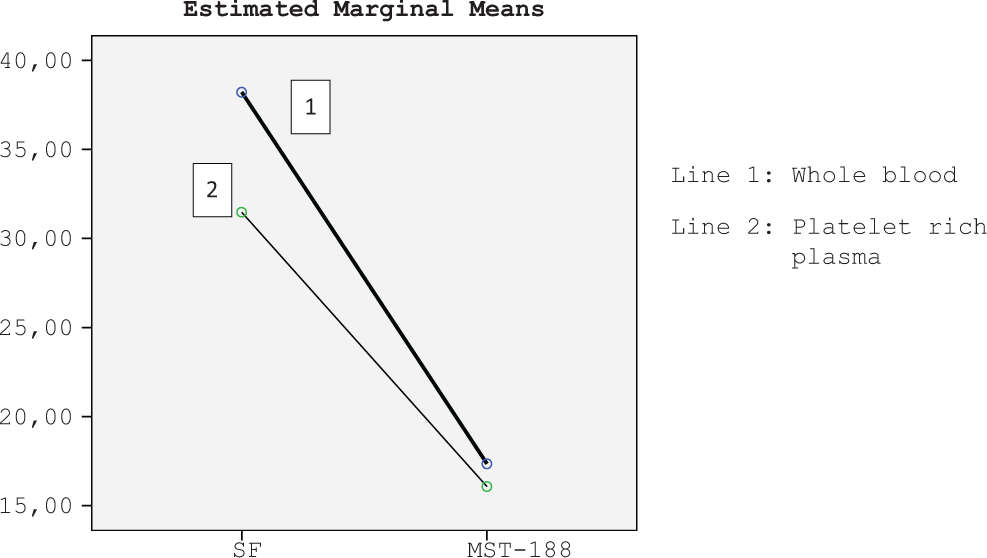
Decreasing of aggregation at the end of 5 hours according to preparations.
In the analysis of preparation (WBP/PRPP) and final concentration of additive solution (2 or 10 mg/dL), because of the high standard deviations, P values were not found significant. P values for aggregations for WBP and PRPP were .131 and .383, respectively. P values for slopes for WBP and PRPP were .066 and .138, respectively.
The aggregation values were lower in platelets recovered from MST-188-WBP in comparison to MST-188-PRPP with the exception of epinephrine-induced aggregation.
Discussion
Platelets are known to lose their functionality upon storage in the blood bank. Several approaches have so far been made to restore platelet functionality during storage such as decreasing the temperature 9 and pathogen 10 reduction. Despite these strategies, the functionality for ADP- and collagen-induced aggregation is decreased to 6% and 9%, respectively. 8
In this study, the MST-188-induced protection of the functionality of the platelets in terms of aggregation was detected. This protective effect was observed on ADP- and collagen-induced aggregation but not with AA- and epinephrine-induced aggregation. For ADP- and collagen-specific aggregation, the protective effect was prominent at 2-mg WBP and 10-mg PRPP.
Both preparations (WB or PRP) of MST-188 were protective compare to saline preparations.
A possible reason for decreased aggregation with some agonists may be because of the effects of MST-188 on cell–cell interactions. This effect has been shown on the RBCs. MST-188 reduced the adhesion rate of normal and phosphatidylserine-exposed RBCs to endothelial cells. MST-188 also increases the local viscosity inside the electrical double layer of RBC membrane. 11 These properties of MST-188 on blood cells may affect the AIA assays. The other reason may be related to the properties and molecular structures of each agonist receptor on the surface of the platelet and/or interaction of MST-188 with platelet surface.
The ADP-induced aggregation requires activation of P2Y1 and P2Y12 receptors. 12 In this study, aggregation induced by ADP and collagen was protected compared to saline controls. The major ADP receptor, P2Y12, is situated in lipid rafts. 13 Poloxamers are composed of a central hydrophobic chain of polyoxypropylene flanked by 2 hydrophilic chains of polyoxyethylene. 14 Probably the hydrophobic and hydrophilic properties of P188 and the placement of P2Y12 in the lipid rafts are ideal for the protection of the ADP-induced aggregation.
The AA is generated from platelet membrane phospholipids and metabolized to thromboxane2 (TxA2) that causes platelet aggregation. 15 The role of TxA2-mediated aggregation in the overall mechanism of platelet aggregation is moderate. 16 The loss of AA-induced platelet aggregation with MST-188 may be important in vivo for the risk of bleeding like the effect of aspirin. The reason of the loss of AA-induced aggregation may be due to the effects of P188 in bilayer phospholipid structure in which AA is metabolized to TxA2.
Epinephrine induces platelet aggregation by increasing TxA2 formation via phospholipase A2 activation and by facilitating TXA2-mediated signaling. 17,18 The reason of the observed decreased aggregation with MST-188 may be due to the interactions of MST-188 with membrane phospholipids and its probable effect on TXA2 synthesis. This can be the probable reason of decreased AIA with AA and epinephrine since their receptors are involved in TXA2 signaling system.
In this study, the mean aggregation values of agonists were found to be lower in platelets in MST-188-WBP systems than in MST-188-PRPP systems. Armstrong et al also observed the inhibitory effect of MST-188 on ADP-induced aggregation when MST-188 was directly added to WB. 19 This inhibitory effect was not observed when MST-188 was directly added to the PRP. According to Armstrong, 19 the reason for inhibition of ADP-induced aggregation in WB-MST-188 was the effect of MST-188 on RBC membranes. Edwards et al 20 showed that decreased aggregation with ADP in -MST-188-WBPis due to the enhancement of the disaggregation following the initial platelet aggregation, and not the RBC-related effect.
The limitation of this study is that the effect of MST-188 on platelet aggregation function was studied with only 1 method. Other methods such as flow cytometry may provide more explanation. To demonstrate an interaction between MST-188 and platelet receptors, its effect on platelet signaling has to be studied. The importance of phospholipid membrane of platelet and MST-188 interaction in the role of TxA2 synthesis has to be investigated since MST-188 is hydrophobic and can affect the phospholipid cell membrane in which the TxA2 is synthesized. Another limitation of this study is that the platelet counts were not determined before and during the experiment which could have explained whether or not this increased functionality is as a result of high platelet count in MST-188 systems or high receptor function despite low platelet count.
In conclusion, this study suggests that MST-188 modulates specific receptors on platelet surface such as ADP and collagen receptors. Since these receptors have a dominant role in aggregation, the protective effects of MST-188 on functionality of these receptors may contribute to the restoration of platelet function upon storage. Thus MST-188 supplementation to platelets stored in the blood bank may be useful in prolonging the functionality of platelets in patients requiring platelet transfusion.
Footnotes
Authors’ Note
The primary author is a visiting professor from the Department of Hematology, Ondokuz Mayis University, Turkey. One of the authors (Marty Emanuele) is an employee of Mast Therapeutics and has contributed to the development of the protocol and its execution.
Acknowledgments
The authors are thankful to Dr Eva Wojcik of the Department of Pathology for her support for the completion of this study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by internal research funding from the Department of Pathology, Hemostasis and Thrombosis Research Laboratories.
