Abstract
Unfractionated heparin (UFH) is a frequently utilized indirect anticoagulant that induces therapeutic effect by enhancing antithrombin (AT)-mediated procoagulant enzyme inhibition. In suspected heparin resistance (HR) during cardiopulmonary bypass, AT activity may be decreased and AT supplementation helps restore UFH responsiveness. The benefit of AT supplementation in HR over longer durations of UFH therapy is unclear. The objective of this study was to describe and evaluate the use of AT III concentrate in the intensive care units (ICUs) at our institution for improving UFH therapy response over 72 hours. A total of 44 critically ill patients were included in the analysis—22 patients received at least 1 dose of AT and 22 patients received no AT. Thirty (68.2%) of the 44 patients were receiving mechanical circulatory support. Baseline characteristics were similar between groups. The average AT activity prior to AT supplementation was 57.9% in the treatment group, and the median cumulative dose of AT was 786.5 U (9.26 U/kg) per patient. There were no significant differences observed in proportion of time spent in therapeutic range (31.9% vs 35.2%,
Introduction
Heparin therapies, specifically unfractionated heparin (UFH), are common agents used for therapeutic anticoagulation in both the operating room and the intensive care unit (ICU). Molecularly, heparinoids themselves do not contribute a direct anticoagulant effect. Heparin potentiates the activity of antithrombin (AT), an endogenous serine protease inhibitor that irreversibly binds various procoagulant enzymes, including thrombin and factor Xa. Heparin enhances AT-mediated effect more than 1000-fold by reducing the half-life of procoagulant enzymes and promoting their binding with AT. 1,2 Based upon the activity relationship between heparinoids and AT, supplementation of exogenous AT becomes a logical consideration when concerns over heparin resistance (HR) arise in a clinical setting.
Heparin resistance has various definitions but generally refers to a given patient requiring unusually large doses of intravenous heparin to achieve the desired therapeutic target. Heparin resistance is particularly common during cardiac operations with incidence rates reported as high as 20.8%. 3 Within the coronary artery bypass graft population, age >65 years, platelet count >300 000 cells/mm3, and preoperative heparin treatment and AT levels <60% have been identified as predictors for HR intraoperatively. 4 Cardiopulmonary bypass (CPB) causes substantial activation of the hemostatic system by creating an interface between blood and the extracorporeal circuitry as well as by the retransfusion of blood that has been exposed to thrombogenic surfaces. This profound hemostatic activation is thought to lead to a decrease in circulating protein buffers, including AT. 5,6
Antithrombin therapy has been assessed for restoring heparin responsiveness in HR patients undergoing cardiac surgery with positive results in multiple studies. 7 –9 Furthermore, AT concentrates are endorsed by evidenced-based guidelines to reduce plasma transfusions in patients with AT-mediated HR prior to CPB. 10 There is a lack of evidence on the direct comparison of AT to fresh frozen plasma (FFP) for HR treatment, and some investigators have proposed relative safety and efficacy advantages associated with AT supplementation in CPB. 11,12 Thus, in the ongoing and immediately perioperative periods of cardiac surgery, AT is a clinically useful option for restoring heparin responsiveness.
Heparin resistance is observed outside the operating room, but its management with AT supplementation in these settings is less studied. Heparin resistance is described in patients without a circulating AT deficiency, and within this subset, AT administration does not enhance heparin response. 3 Complicating the assessment of endogenous AT activity’s role in HR is the fact the heparin administration itself can decrease AT activity by as much as 30%. 13 Extracorporeal membrane oxygenation (ECMO) support reduces AT activity, which may produce a procoagulant state with decreased heparin response. 14 The increased utilization of ECMO in adult ICUs has led to many institutions monitoring and supplementing AT as a part of usual ECMO support. In a cross-sectional survey of 117 adult and pediatric ECMO centers, 82% reported performing AT testing and the majority of centers reported using FFP, recombinant, or pooled AT for supplementation. 15
Considering the paucity of evidence for AT supplementation outside the operating room despite a strong physiologic basis for the importance of AT activity in patients receiving heparin anticoagulation, further investigation into the use of AT supplementation during UFH administration in the ICU is warranted. The objective of this study was to describe and evaluate the use of AT III concentrate (Thrombate III; Bayer Pharmaceuticals, West Haven, Connecticut) in the ICUs at our institution for enhancing responsiveness in prolonged intravenous UFH therapy.
Methods
This retrospective cohort study was approved by the Colorado Multiple Institutional Review Board. Patients aged 18 to 89 years were eligible for inclusion into this study if at least 1 AT activity test was performed while receiving an intravenous infusion of UFH during an admission to an ICU at the University of Colorado Hospital from September 1, 2011, until July 31, 2015. Patients were excluded if they were known to have AT deficiency due to a genetic etiology.
Patients were placed into 1 of the 2 groups—a treatment group that received at least 1 dose of AT during their ICU stay or a control group that received no AT during their ICU admission. Patient matching was performed on the basis of the following characteristics: age, gender, baseline AT activity, baseline activated partial thromboplastin time (aPTT), heparin infusion rate at the time of AT test, and type of mechanical circulatory support (MCS) at the time of AT test. The type of MCS was divided into several categories including short-term ventricular assist device (VAD), durable VAD, ECMO, or no support. Intravenous UFH infusions were titrated to patient-specific goal coagulation parameters (aPTT, activated clotting time [ACT], or anti-factor Xa level) that were chosen by the providers. The normal range of aPTT at our institution is 24.2 to 33.9 seconds. The University of Colorado Hospital clinical laboratory performed all of the coagulation parameter testing per routine patient care using Stago (Diagnostica Stago, Parsippany, New Jersey) testing reagent for each respective test.
The outcomes of this study were evaluated between groups within the 72 hours following the initial AT activity test. This time period was chosen based upon the theoretical therapeutic duration of supplemental AT concentrate. 16 The time spent within the operating room was excluded from the analysis. If therapeutic anticoagulation was transitioned to a non-UFH agent, the data collected were censored at the time of initiation of the new agent. Outcomes included proportion of time within the therapeutic range (hours spent within the therapeutic range/duration of UFH anticoagulation up to 72 hours), duration of time until first therapeutic test, UFH infusion rate at goal, and clinically significant bleeding. A clinically significant bleeding event was defined as overt bleeding plus any of the following within 72 hours of AT administration: operating room intervention to correct bleeding not related to a technical issue within the MCS, a decrease in hemoglobin of greater than 2 g/dL in a 24-hour period, 2 or more transfusions of packed red blood cells in a 24-hour period, or death related to bleeding. Heparin responsiveness before and after AT activity assessment and AT supplementation (only in the AT treatment group) was calculated by determining the hourly UFH units required per second of additional aPTT prolongation, that is, change in hourly UFH dose/change in aPTT.
All statistical analyses for this study were performed using JMPPro v12.01 (SAS Institute Inc, Cary, North Carolina, 2012). Distribution of results was assessed for normality and analyzed via appropriate statistical tests. Nominal data including type of hemodynamic support, gender, and bleeding events were analyzed via Fisher exact test due to the small sample size. Wilcoxon rank sum test was utilized to assess nonparametric continuous data such as UFH infusion rates, baseline aPTT results, and therapeutic outcome parameters. Student
Results
Patient baseline characteristics are displayed in Table 1. Of the 44 patients included for analyses, 22 patients received at least 1 dose of AT while being admitted to an ICU and 22 patients received no AT during their entire hospital admission. Within the AT treatment group, the median cumulative dose was 786.5 U (9.26 U/kg) per patient. Antithrombin doses ranged from 500 to 2000 U, and the median number of AT administrations per patient was 1. Seven patients received multiple AT doses (range: 2-4 doses). There were no statistically significant differences in the baseline characteristics between the AT treatment and control groups. However, there was a strong trend toward a higher baseline UFH infusion rate in the AT treatment group. All of the patients who received AT supplementation were treated in the cardiothoracic ICU (CTICU). In the control group, 20 (91%) patients were admitted to the CTICU and 2 (9%) patients were admitted to the cardiac ICU. A total of 5 patients, 3 in the AT treatment group and 2 in the control group, were transitioned to alternative therapies (4 direct thrombin inhibitors and 1 low-molecular-weight heparin) for anticoagulation within 72 hours of the baseline AT activity test. Patients were most frequently titrated to goal aPTT (39 of 44, 89%), followed by ACT (3 of 44, 7%) and anti-factor Xa test results (2 of 44, 4%).
Baseline Characteristics.a
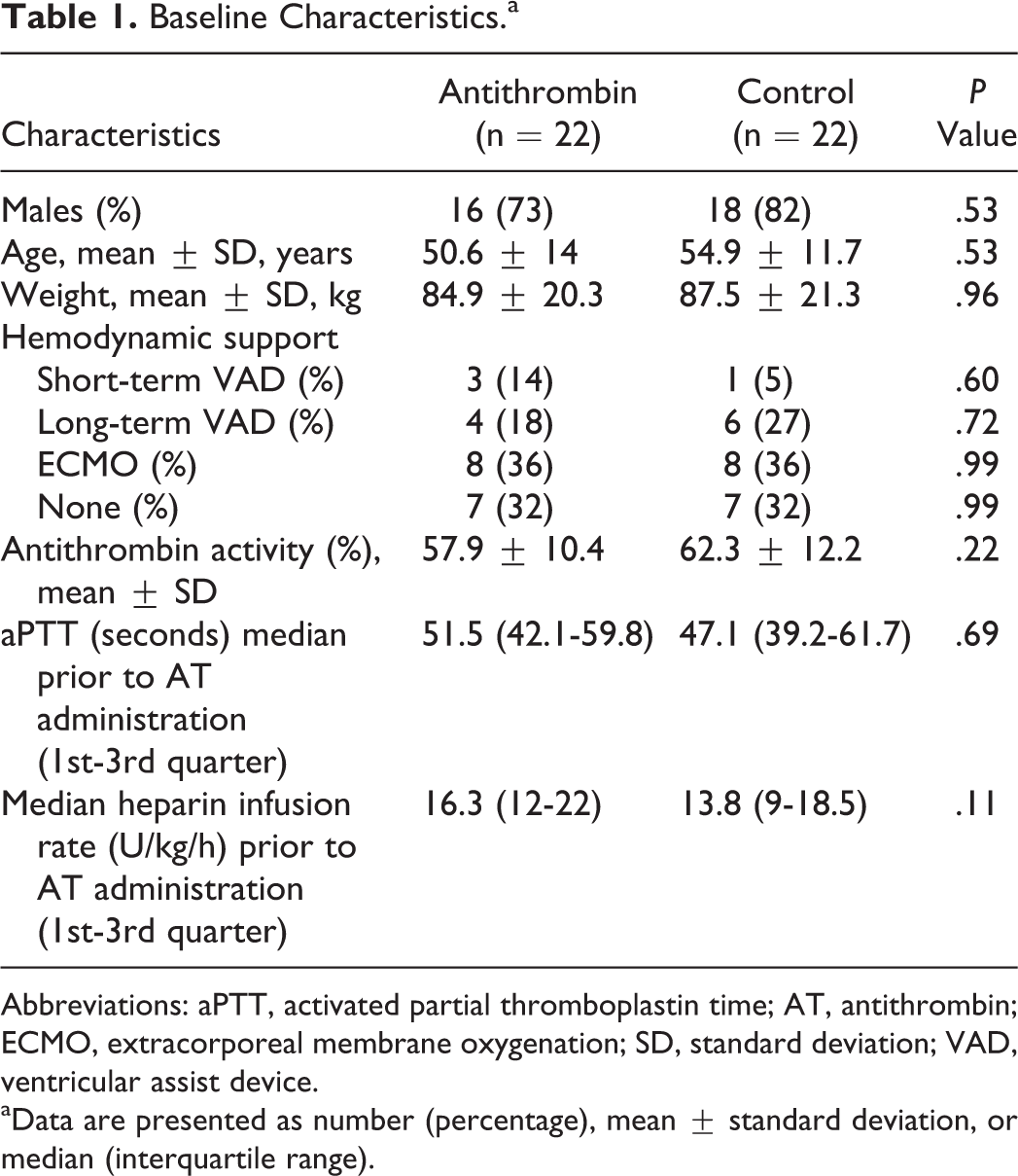
Abbreviations: aPTT, activated partial thromboplastin time; AT, antithrombin; ECMO, extracorporeal membrane oxygenation; SD, standard deviation; VAD, ventricular assist device.
aData are presented as number (percentage), mean ± standard deviation, or median (interquartile range).
Outcome results are displayed in Table 2. There were no significant differences observed in proportion of time spent in the therapeutic range (31.9% vs 35.2%,
Anticoagulation Outcomes.a
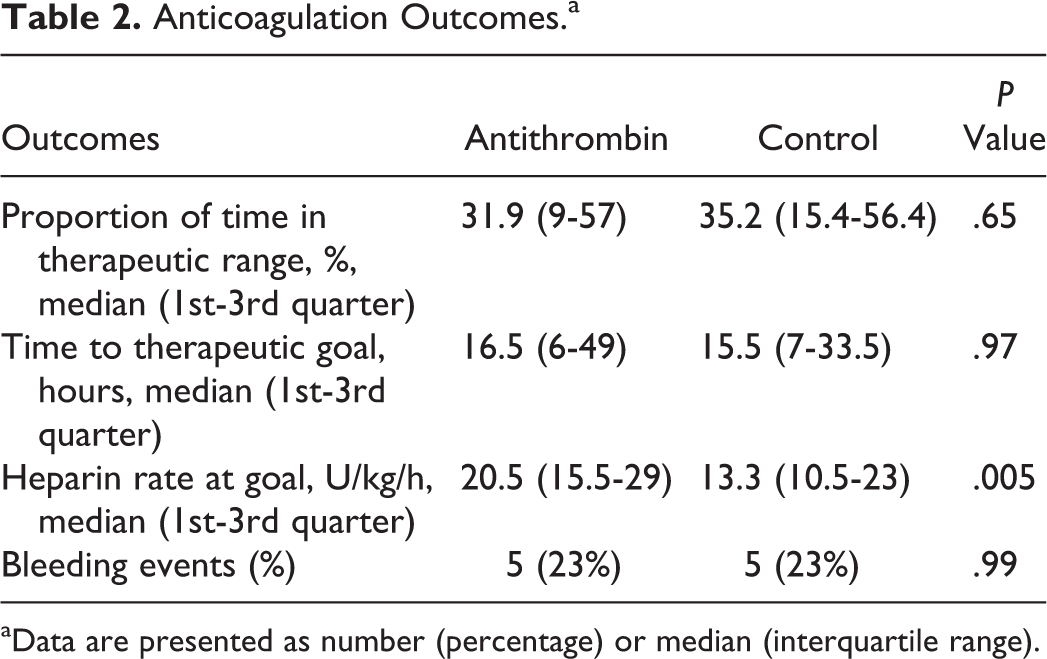
aData are presented as number (percentage) or median (interquartile range).
The resultant aPTT response to UFH therapy before and after AT activity assessment is displayed in Figure 1. In the approximately 4 to 6 hours between aPTT results, the median increase in UFH infusion rate in the AT treatment group was 1 (interquartile range [IQR]: 0-2) U/kg/h compared to 1 (IQR: 0-1.75) U/kg/h in the control group (
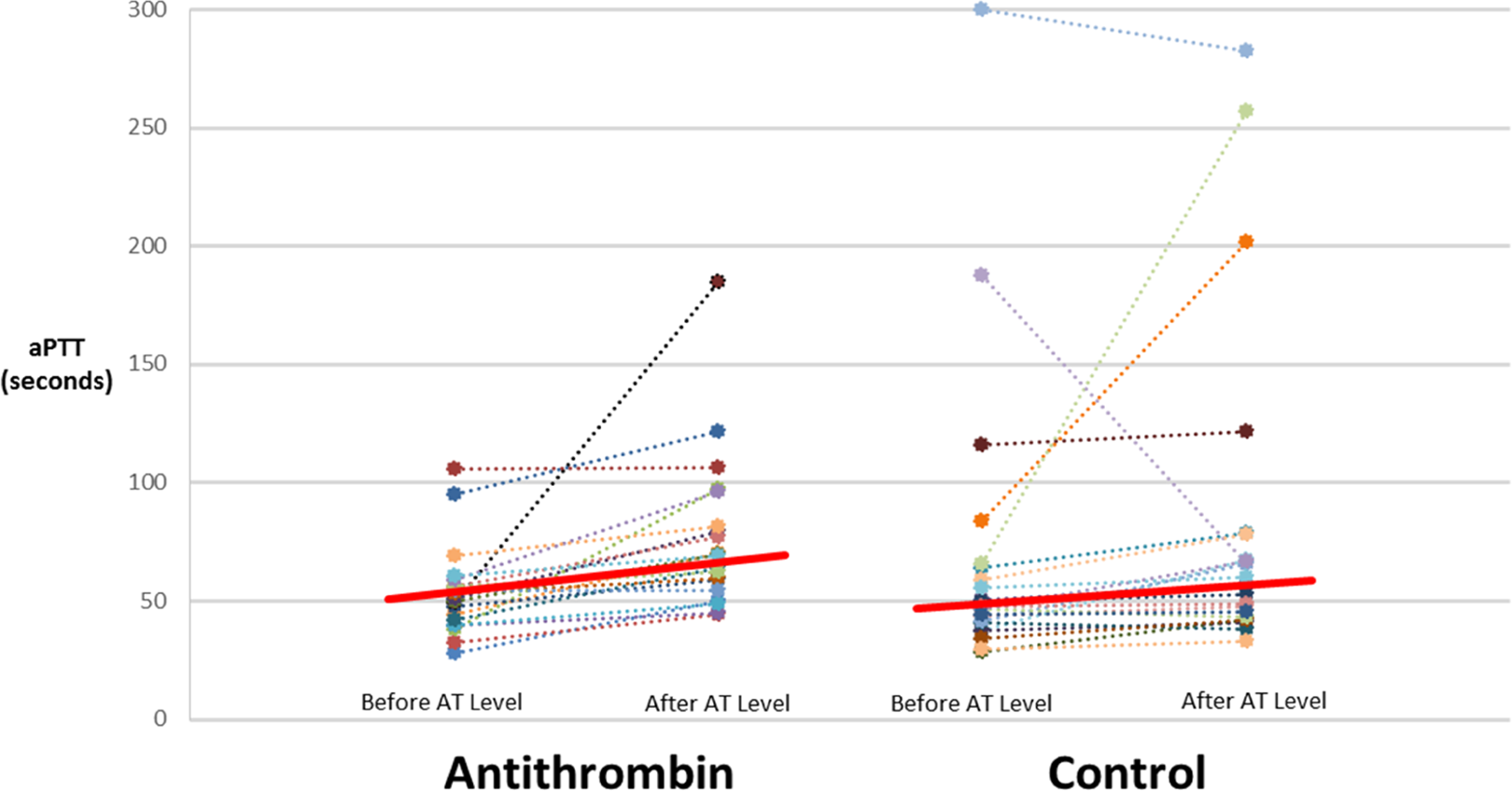
Activated partial thromboplastin time response before and after antithrombin activity assessment.
Discussion
This study describes the use of AT supplementation during prolonged UFH infusion within a subset of critically ill adult cardiothoracic patients at our center. When compared to a similar group of patients receiving UFH in the ICU without AT supplementation, there appeared to be no significant differences in anticoagulant response (eg, time to therapeutic goal or proportion of time in the therapeutic range) or frequency of clinically significant bleeding. The higher UFH infusion rate observed at goal in the AT supplementation group suggests that in cases of mild to moderate HR, a strategy of simply increasing the UFH dose to achieve the goal level of anticoagulation is at least as effective as combining UFH dosage increases with AT supplementation.
The lack of AT efficacy observed within our study cohort could be related to inadequate AT supplementation. Within the current literature, there is no clear target AT activity to ensure heparin responsiveness, and survey data indicate that AT targets during ECMO support vary significantly (30%-120%) from center to center. 15 Manufacturer-recommended dosing of AT concentrate is individualized for patients with congenital AT deficiency on the basis of baseline AT activity levels and the expected incremental increase of 1.4% per unit per kilogram of body weight. 16,17 Previous studies addressing AT supplementation in CPB for heparin sensitization have utilized smaller doses of AT concentrate (500-1000 U). 8,9 In these intraoperative studies, favorable anticoagulation response was documented despite observing increases in AT activity that did not approach normal values and a lack of correlation between post-heparin ACT and AT activity. 8,9 The biologic elimination half-life of AT concentrate is reported to be 2.5 days but is accelerated during UFH therapy, which may contribute to a lack of response observed over longer periods of heparin infusion with an intermittent daily dosing strategy of AT. 16
Studies within the pediatric population have investigated the effects of AT administration for patients receiving ECMO support with findings similar to what is reported in our work. Byrnes et al provided daily AT supplementation to 21 pediatric patients receiving venoarterial ECMO with AT activity levels <70% and found that despite increases in anti-Xa levels, neither heparin responsiveness nor circuit life was enhanced. 18 Niebler et al reviewed AT use in 28 pediatric patients receiving ECMO support with AT activity levels less than 80% and reported increases in AT activity but no difference in heparin infusion rate, ACTs, chest tube output, or red blood cell transfusion volume. 19 The results of our study add evidence that suggest intermittent daily doses of AT concentrate in critically ill adult patients does not reduce heparin infusion rates or affect clinically significant bleeding.
Multiple considerations related to UFH therapy may have contributed to the findings of this study. The baseline UFH rate in the AT treatment of 16 U/kg/h was not exceedingly high, which suggests that many patients within the AT treatment group were not HR by definition. Heparin resistance has been defined in a variety of ways by different investigators. 7 –9 One proposed definition is the requirement of an intravenous UFH dose greater than 35 000 U every 24 hours to prolong the aPTT 1.5 to 2 times the normal values. 20 Based on the mean values reported in Table 1, the average patient in the AT treatment arm would not have met this definition for HR and as such would be less likely to benefit from AT supplementation. Additionally, the majority of patients analyzed in this study were receiving some sort of MCS and recently underwent a surgical intervention. Considering bleeding risk within this patient population, anticoagulation practices were judicious. Patients were rarely given boluses of UFH for subtherapeutic tests and were started at low UFH infusion rates with upward titration. This approach could have contributed to delays in therapeutic target attainment, which brought the question of HR and AT activity into the clinical picture. Lastly, HR can occur due to a variety of mechanisms including increased heparin clearance, increased heparin binding to von Willebrand factor or factor VIII, inability to suppress platelet-related or clot-bound thrombin activity, and a multitude of other mechanisms. 21 Many of these mechanisms are not AT dependent, and supplementation would be less likely to restore heparin responsiveness.
One proposed benefit of AT supplementation during exposure to extracorporeal circuits is the theoretical restoration of heparin response without excess levels of UFH, thus mitigating the risk of hemorrhage due to heparin rebound. In a case series of 6 pediatric cardiac surgery patients, a continuous infusion of AT to a target activity of 100% was associated with a lower risk of bleeding. 22 In a randomized control trial in septic shock, high-dose AT was associated with increased hemorrhage rates in patients receiving heparin, and increased blood loss in CPB 12 hours postoperatively has been observed. 7,23 Our study reviewed a low-dose AT supplementation strategy during UFH infusion and found no apparent effect on bleed frequency. However, this analysis is likely underpowered to observe a potential difference in bleeding rates, and the retrospective assessment of clinically significant bleeding via the criteria used here is challenging in patients receiving ECMO who may be experiencing shear stress–related hemolysis.
The resource utilization associated with AT supplementation is another factor worth reviewing. The average wholesale price of AT III concentrate (Thrombate III; Bayer Pharmaceuticals) is US$4.66 per unit. 24 Based on this cost estimate, the acquisition cost associated with the average amount of AT supplementation provided in this study is US$3665 per patient. With the lack of clinical utility observed in this cohort, the cost-effectiveness of AT supplementation in patients with predominantly mild to moderate HR should be considered and weighed against the low acquisition cost associated with higher dosages of UFH.
There are limitations to our study. First, the sample size is small and the design was retrospective. Second, there was variation in the coagulation parameters, both in type and therapeutic range, selected by the providers to guide UFH therapy titration. This variation was not directly accounted for within the study design and may have been different between the treatment and control groups. We believe this difference to be minimal as the patients were similar in each group and were receiving treatment in predominantly 1 ICU under the care of the same providers within an identical time period. Third, the average patient included in this study was not definitively HR. Additional UFH titration, rather than AT supplementation, could have been utilized in the treatment arm, but the lack of a consensus definition for HR makes this a debatable point. Lastly, we did not perform any assessment of bleeding risk when matching patients and cannot be certain that bleeding risk was equal between the treatment and control groups.
In conclusion, our findings suggest that in ICU patients with mild to moderate HR receiving a prolonged UFH infusion, AT supplementation did not have a significant impact on anticoagulant response or frequency of clinically significant bleeding. Given the relatively high cost of AT supplementation and the lack of benefit observed in this work, a protocol-guided approach may help identify truly HR patients and limit AT supplementation to those more likely to benefit. Additional research is needed to define AT activity targets and to standardize AT supplementation practices in patients receiving prolonged UFH infusions.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
