Abstract
Controversies exist over the currently recommended guidelines for the use of low-molecular-weight heparin (LMWH) in neonates. We retrospectively studied 30 neonates treated with LMWH and found a poor therapeutic response to recommended doses as measured by anti-Xa levels. Sixty percent of the study participants required their doses to be increased because of subtherapeutic anti-Xa levels during the initial course of their treatment. The mean starting enoxaparin dose was 1.53 ± 0.38 mg/kg. The mean enoxaparin dose, once therapeutic anti-Xa levels had been achieved, was 1.86 ± 0.50 mg/kg. Preterm and term infants required doses of 2.06 ± 0.61 mg/kg and 1.67 ± 0.26 mg/kg, respectively, to achieve therapeutic anti-Xa levels. In summary, our results suggest that higher initial doses are required to achieve therapeutic anticoagulation in neonates.
Keywords
Introduction
With medical and technological advances in maternal and newborn care, there has been a significant improvement in the survival and outcomes of newborns. 1 –3 However, increased interventions have, in part, led to an increase in the incidence of thromboembolic events in this population. 4 Registries from Canada, Germany, and the Netherlands have reported incidences of thrombotic complications in newborns of 2.4 per 1000 neonatal intensive care unit (NICU) admissions, 5.1 per 100 000 live births, and 0.7 per100 000 live births, respectively. 4 –7
Neonates admitted to the NICUs are particularly susceptible to thromboembolism (TE). These patients have higher rates of procedures performed such as the insertion of central venous lines (CVLs) and umbilical venous and peripheral arterial catheters. Other associated factors for TE in this group include the evolving maturity of the hemostatic system, the smaller caliber of blood vessels than adults, the need for long-term parenteral nutrition, and the presence of multiple comorbidities such as dehydration, polycythemia, sepsis, asphyxia, and various pulmonary and congenital cardiac diseases. 6 –9 The majority of neonatal thromboembolic events are usually asymptomatic and can lead to the development of acute life-threatening conditions such as cardiac, respiratory, or renal failure. Thus, the timely detection and treatment of TE in the neonatal population are ofparamount importance.
The use of anticoagulants in the pediatric population can prevent clot propagation and is associated with favorable outcomes. 10 –12 Low-molecular-weight heparin (LMWH) is being used more frequently in neonates. 13,14 However, the efficacy remains unproven in this age group. The potential advantages of LMWH are the reduced need for monitoring blood parameters, ease of subcutaneous administration, and a more predictable anticoagulant response. 15 Although guidelines exist for the use of LMWH in the pediatric population, these have been developed from small-scale retrospective or prospective cohort studies or extrapolated from the adult population and may not be appropriate during the neonatal period. 16 –18
Recent data suggest that newborns aged <2 months require a higher dose to achieve target anti-Xa levels. 14,18 –23 Additionally, it has been observed that preterm infants require higher doses than full-term infants. 14,20,24,25 In neonates, anti-Xa levels should be closely monitored during treatment with LMWH because immature renal and hepatic clearance mechanisms can predispose to wide fluctuations in anticoagulant responses. Limited information is available for documenting the fluctuations, which can be seen in anti-Xa levels in this age group. Current guidelines recommend a target anti-Xa level of 0.5 to 1 U/mL when measured 4 to 6 hours following a subcutaneous injection. 17 Thus, the use of LMWH in neonates has not been optimized, and the management of TE with LMWH also requires further exploration.
In view of the findings from previous studies and the controversies that exist over the doses of LMWH used in neonates, we performed a retrospective study with the primary aim to evaluate the use of LMWH in infants aged <28 days for the treatment of TE and the dosage required to maintain a therapeutic anti-Xa level. The secondary aim of the study was to evaluate the timing of LMWH doses.
Methods
Study Design and Patient Population
Following approval by the Hamilton Health Sciences/Faculty of Health Sciences Research Ethics Board (Ontario, Canada), all patients aged <28 days in the NICU at McMaster Children’s Hospital, with a documented TE who had received LMWH from June 2003 to July 2011, were identified through the hospital pharmacy records. A retrospective chart review of all the identified cases was then performed.
Data Collection
Data collection included birth weight, weight at diagnosis, gestational age, location of TE, diagnostic method to establish TE, date of diagnosis, date of treatment started, dates and results of all anti-Xa levels, all doses of LMWH, all dose adjustments based on anti-Xa levels, adverse events, duration of treatment, and outcome of TE.
Anti-Xa Assay
Anti-Xa activity was measured using a colorimetric assay from Stago Canada Ltd, Toronto, Ontario.
Statistical Analysis
Data were entered into an Excel spreadsheet and analyzed using standard descriptive methods and comparative statistics. All values were calculated as means ± standard deviations or medians when data were not normally distributed. Box plots were constructed using Sigma Plot 11.0, San Jose, California.
Results
Thirty-four neonates that had been prescribed LMWH (enoxaparin in all cases) within the first 2 months of life were identified. Among these 34 neonates, 4 were excluded from our analysis. In 1 case, the neonate had been prescribed enoxaparin prophylactically for a cardiac condition, and in 3 cases enoxaparin was started on a clinical presumption of thrombosis which was subsequently excluded by imaging, and therapy was discontinued prior to the administration of the third dose. Finally, there were 30 cases recruited for analysis; 5 were tested for a factor V Leiden or prothrombin G20210A mutation and none were found to be positive.
Baseline patient’s demographic data and details regarding the site of thrombosis are described in Table 1. The mean (range) gestational age of this cohort was 37 ± 4.34 weeks (29-43 weeks). The mean (range) weight at the time of thrombus detection was 2833 ± 1325 g (703-5353 g). In all, 16 patients were preterm (<37 completed weeks); 8 were low birth weight (LBW, <2500 g), 6 were very LBW (<1500 g), and 3 were extremely LBW (<1000 g). Forty-eight percent of the thrombi occurred in the thoracic or abdominal region and were related to the placement of CVLs. Thrombi in the limbs (21%) or central nervous system (14%) were the next largest groups with 10% of the cases having multifocal thrombi.
Characteristics of Neonates Receiving Low-Molecular-Weight Heparin (n = 29).

Abbreviation: SD, standard deviation.
Dose Titration Results of Low-Molecular-Weight Heparin in Neonates (n = 26).

Abbreviation: SD, standard deviation.
Enoxaparin Dose and Associated Anti-Xa Levels
We examined the mean starting dose of enoxaparin in this group, if a weight was recorded within 24 hours of the first dose. An accurate neonatal weight at the time the diagnosis of thrombosis was missing in 4 cases. In the 26 cases with accurate data, the mean (range) starting enoxaparin dose was 1.53 ± 0.38 mg/kg (0.95-2.26 mg/kg). The mean number of dose adjustments required prior to achieving a therapeutic anti-Xa level (defined as 0.5 U/mL or greater) was 1.7 ± 1.8, with a median of 1 dose adjustment due to the wide range encountered (0-8 adjustments). The median (range) time to achieve a therapeutic anti-Xa level was 3 days (0-16 days). The mean (range) dose of enoxaparin once therapeutic anti-Xa levels had been achieved was 1.86 ± 0.50 mg/kg (0.99-3.06 mg/kg). Subgroup analysis showed that a mean (range) dose of 2.06 ± 0.61 mg/kg (0.99-3.06 mg/kg) was required to achieve therapeutic anti-Xa levels in preterm infants and 1.67 mg/kg ± 0.26 mg/kg (1.21-2.13 mg/kg) in term infants, respectively.
The anti-Xa levels achieved after the initial dose of enoxaparin based on weight and gestational age are shown in Figure 1. Further analysis of the patient population revealed that 60% of the study participants required their doses to be increased because of subtherapeutic anti-Xa levels during the course of treatment. In all, 53% of the patients did not achieve therapeutic anti-Xa levels after the first dose, 10 of which were preterm. Cumulatively, 71% of the preterm or LBW neonates achieved subtherapeutic anti-Xa levels after initial doses. Anti-Xa levels were unavailable for one preterm infant. A total of 182 anti-Xa levels were performed, 40% of which were subtherapeutic, 50% were therapeutic, and 10% were supratherapeutic.
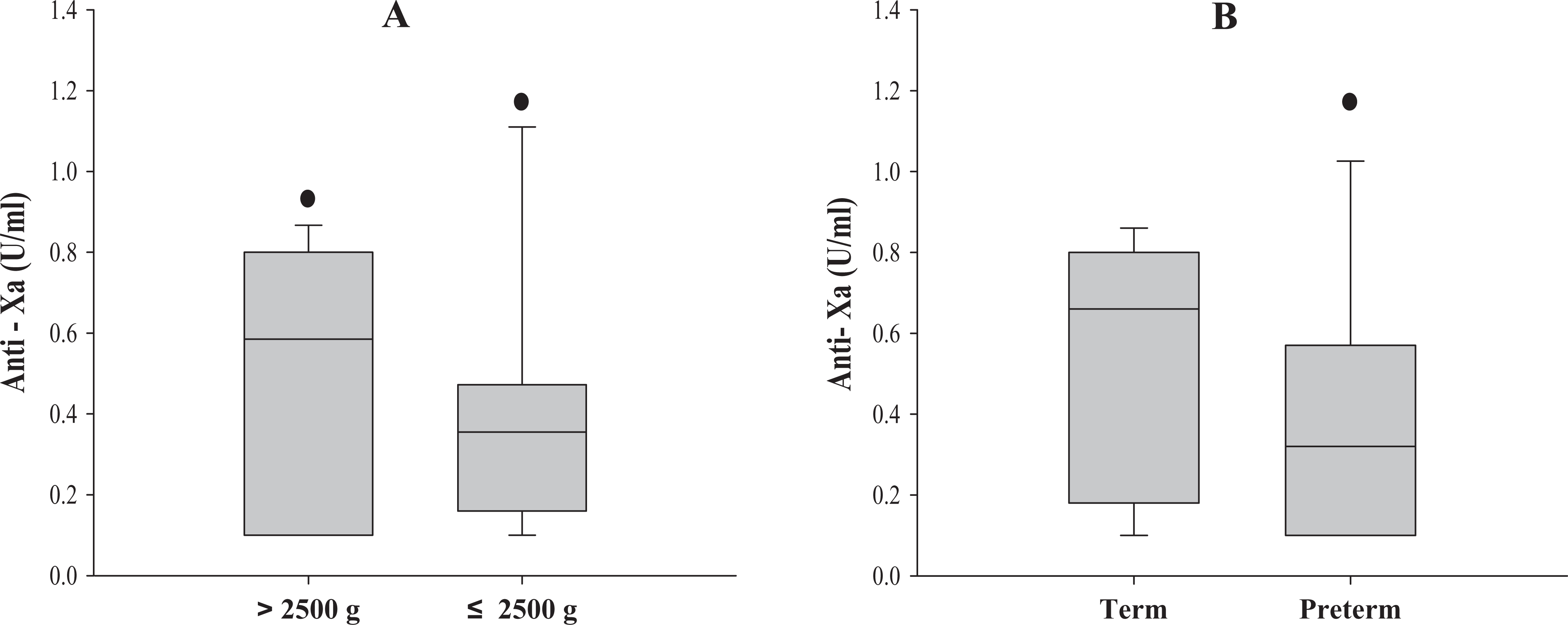
A, Anti-Xa levels achieved after initial dose of enoxaparin in neonates >2500 and ≤ 2500 g. Neonates ≤ 2500 g attained therapeutic anti-Xa levels less frequently than infants >2500 g. B, Anti-Xa levels achieved after the initial dose of enoxaparin in both term and preterm neonates. Preterm neonates achieved therapeutic anti-Xa levels less frequently than term neonates. The box plots represent anti-Xa levels achieved after the administration of the first dose of enoxaparin with median, 25th percentile (lower), and 75th percentile (upper) boundaries. Error bars indicate the 10th and 90th percentiles. Solid circles represent outliers.
Efficacy of LMWH
Documentation of the outcome of clot therapy (Table 3) was indeterminate in 10% of the cases. In the remaining 90% of the cases, 51% had complete resolution or decrease in the size of thrombus evidenced by imaging studies, and 35% had a stable thrombus that was nonsymptomatic.
Outcomes of the Treatment of Thrombi in 29 Neonates (n/%).

Adverse Effects
Overall, the therapy was well tolerated in this group, with 13% (n = 4) experiencing bleeding episodes; 2 infants had occult blood detected in the stool, in 1 case had bleeding documented at the injection site, and 1 neonate had a suspected adrenal hemorrhage on ultrasound examination. All of these cases had treatment withheld or the dose of enoxaparin reduced with subsequent successful dose reescalation. In one case, therapy was interrupted because of thrombocytopenia associated with sepsis. There was 1 death recorded, but the cause of death was unrelated to the presence of the thrombus or adverse effects of enoxaparin.
Discussion
The efficacy of LMWH for the treatment of thromboembolic diseases in the adult population has been established, 26,27 but similar evidence-based guidelines for neonates are still lacking. This is primarily due to the absence of robust studies confirming the safety and effectiveness of specific dosing regimens and monitoring parameters. 17 Current guidelines suggest an enoxaparin dosage of 1.5 mg/kg for every 12 hours for the treatment of thrombosis in infants less than 2 months old. 17 ,20
More recent studies 14 ,19,20 have shown that higher doses are required to achieve therapeutic anticoagulation. In a retrospective analysis, Lulic-Botica et al 28 reported that therapeutic anti-Xa levels were attained after 12.5 days of treatment starting with a mean dose of 1.4 ± 0.3 mg/kg and a mean cumulative dose of 2.1 ± 0.6 mg/kg. In their cohort of 26 infants, only 27% were within the recommended anti-Xa level therapeutic range, while 40% of the levels reached the desired target during maintenance therapy. Several other studies have highlighted the poor anti-Xa responses observed in neonates after the administration of currently recommended doses. Streif et al 14 conducted a prospective cohort study and noted that a dose of 1.7 ± 0.3 and 2.1 ± 0.6 mg/kg for every 12 hours was necessary to maintain therapeutic anti-Xa levels in term and preterm infants, respectively. Micheals et al 24 determined that the 10 preterm neonates in their study achieved therapeutic anti-Xa levels with a dose of 2.27 mg/kg given every 12 hours.
Data from our cohort similarly show that the administration of the initial recommended weight-adjusted doses were insufficient to achieve therapeutic anti-Xa levels. Final doses were found to be on average 1.4 times higher than the initial starting dose with a range increase of 1- to 3.04-fold. Previous studies have shown that an increase in the doses of 1.1- to 1.8-fold from the mean initial dose is required to achieve therapeutic anti-Xa levels. 14,19,24,28 Our results also suggest that preterm or LBW infants may require even higher initial doses than full-term infants. This may indicate that doses may be inversely proportional to age and birth weight of the neonate as has been documented in other studies. 18,29 Previous studies have also shown a stronger correlation between weight-adjusted doses and anti-Xa levels in term infants when compared to preterm infants. 20 There are various factors that may contribute to this finding. First, this may be due to the developing hemostatic system in preterm infants. 30 Second, preterm and LBW infants have immature renal and hepatic clearance mechanisms. Finally, it is well known that younger children have a higher volume of distribution that may affect the bioavailability of the drug in the circulation. It is also important to note that neonates with thrombotic events often are acutely ill with many associated comorbidities, which may further impact the degree of anticoagulation achieved.
Malowany et al 19 extensively reviewed the data available on the use of LMWH from 1996 to September 2007. The authors recommend that for the treatment of thromboembolic disease, an initial starting dose of 1.7 mg/kg every 12 hours should be used for term neonates and 2.0 mg/kg every 12 hours for preterm infants. The findings of our study support the dosage recommendations by Malowany et al 19 Furthermore, whole milligram doses were used more frequently in our study which suggests that the use of whole milligram doses may be appropriate in the neonatal population as opposed to precise doses. 18
Substantial fluctuation in anti-Xa levels was observed in our study population. This trend was also evident even after the attainment of initial therapeutic anti-Xa levels subsequent to doses being increased. Although a large proportion of neonates in our study had transiently supratherapeutic anti-Xa levels, there were no reports of major bleeding events in our cohort, consistent with previous studies that do not show an association between high anti-Xa levels and bleeding events. 14,31 Furthermore, majority of the neonates in our study improved after treatment, suggesting that LMWH is effective for the management of TE in this age group.
A substantial delay in the achievement of therapeutic anti-Xa levels was observed in our cohort due to the requirements for dose adjustments. In settings of acute or life-threatening TE, this delay could mean extensive clot propagation and progression of ischemia. Newborn infants grow relatively rapidly and experience alterations in their body composition during the first few months of life. This unique attribute may lead to unpredictable levels of anticoagulation in these patients, which may in part explain the fluctuations seen in anti-Xa levels. This highlights the need for further evaluation of dosage regimens in this age group in order to achieve appropriate therapeutic anticoagulation and improve clinical outcomes.
Our study is limited because of its retrospective design. However, it provides a view of real-world practices regarding the use of LMWH in the NICU and underscores the necessity for further studies to establish optimal dosing regimens as well as the safety and efficacy of LMWH in newborns. Our study is also consistent with the trends observed in many other centers and further demonstrates that current guidelines for the treatment of neonatal TE should be reevaluated.
Conclusion
In conclusion, this study supports the recent recommendations for the use of higher LMWH doses in neonates for the treatment of TE and further strengthens the notion that the treatment of a thromboembolic event in this population is a unique and complex entity, distinct from adults and older children. Further, well-designed clinical trials are necessary to assess the efficacy of LMWH in order to establish therapeutic dosage parameters for neonates. A prospective study using weight-adjusted fixed doses evaluated in large sample sizes is timely and merits consideration.
Footnotes
Authors’ Note
Dr Anthony K. Chan holds a McMaster Children’s Hospital/Hamilton Health Sciences Foundation Chair in Pediatric Thrombosis and Hemostasis.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
