Abstract
March 2013 represented the 50th anniversary of the first license granted for a fibrinogen concentrate. In this review, we look at the history of bleeding management that led to the development of fibrinogen concentrate, discuss its current use, and consider future developments for this product.
Introduction
It is over 50 years since the first license for a fibrinogen concentrate (FIBRINOGENIO HUMANO LIO.) was granted in Brazil in 1963. Historically, fibrinogen came into focus because of reports suggesting it was a cardiovascular risk factor, and there is still no final answer as to whether it is a mediator, 1 or just a marker 2 or predictor, 3 of cardiovascular disease. It is only recently that research started focusing on the positive role of fibrinogen as a hemostatic agent, particularly in the management of perioperative bleeding.
The History of the Term “Fibrinogen”
Before we discuss the development of fibrinogen concentrate, we take a historical look at where the term “fibrinogen” first came from. As far back as the fifth- to fourth-century BC, Hippocratic doctors noted the presence of fibers in circulating blood. However, it was not until the end of the 17th century that Malpighi, using a single lens microscope, observed that whole blood clots have a fibrous and corpuscular component. 4 The term “fibrin” was introduced by Fourcroy in 1801. He demonstrated that plasma contained soluble substances and that the precursor of fibrin was present in plasma but not serum. A few decades later, in 1847, the term “fibrinogen” was invented by Virchow where he stated “should one want to give it a name, it could be called fibrinogen” (translated from German: “vielmehr, wollte man sie benennen, so könnte man sie höchstens Fibrinogen taufen.”). 5(p.581) Some consider that he created the term out of the words fibrin and oxygen, since he believed that “fibrin was a homogeneous mass formed from soluble fibrin after exposure of blood or exudates to oxygen,” 6(p.301) and others suggest that he composed the term by adding the syllable “ogen” for a precursor substance instead of the prefix “pro.” At the time, Virchow was experimenting with exudates, rather than with whole blood, and therefore mistakenly applied the term “fibrinogen” to decaying exudates rather than to the precursor of fibrin in whole blood as it was understood later. In fact, Virchow had not discovered fibrinogen, instead Denis (in his “Mémoire sur le sang” in 1859) was the first to recognize that plasma contained a clottable substance different from fibrin and went on to attempt to purify and characterize this protein. Denis proposed the name fibrinogen (independent of Virchow): “I believe that I should repeat once more that I do not mean to define a liquid fibrin, plasma fibrin, but rather a substance which is not at all fibrous and which is the origin of fibrin, clottable lymph, a substance which I would like to call ‘sérofibrine’ and which one might also designate by the name ‘fibrinogène’, if such a word would be admissible.” 7 Nevertheless, it took a further 20 years before fibrinogen was first isolated by Hammarsten (1879) who precipitated and purified it from horse plasma by salting-out with sodium chloride in what has become the classical procedure for fibrinogen preparation. 6
The History of Bleeding Management
Nowadays, bleeding management encompasses, among other things, the assessment of bleeding risk prior to surgery, and monitoring and managing blood loss during surgery, including appropriate management of bleeding. Historically, the development of therapeutic blood management can be traced back to the 17th century where the first animal-to-human blood transfusion was conducted in 1667. 8 However, it was not until 1818 that James Blundell, a British obstetrician, performed the first human-to-human transfusion—to save a patient from exsanguination. 9 The introduction of blood transfusion into the medical armamentarium was further advanced with the groundbreaking discovery of the ABO blood group by the Austrian–American immunologist and pathologist Karl Landsteiner in 1901, a feat for which he was later awarded the Nobel Prize. 10
Over the last 100 years, further developments have served to transform the practice of transfusion medicine. 11 One of the most relevant developments is the shift from using whole blood to using specific blood components (e.g., red blood cells, platelets, and fresh frozen plasma [FFP]) and further to the purified, virus-inactivated, plasma-derived products (such as factor concentrates). This latter development was made possible by the work of Edwin Joseph Cohn who discovered that the complex mixture of components in whole blood could be separated through a process of fractionation, commonly referred to as “Cohn fractionation.” 12 In 1947, he described 6 major plasma fractions, with fraction I containing most of the fibrinogen. Cohn emphasized the importance of separating different plasma components, as it was more efficient and less wasteful to use a particular component for a specific need rather than whole plasma. This approach proved of great value during World War II with the development of products such as fibrin foam helping to solve major problems connected with hemostasis. This change from the use of specific blood components to purified plasma-derived products for bleeding management represents a continuing paradigm shift.
Fibrinogen Concentrate: The Past and The Present
During major bleeding and replacement with red blood cell concentrates, fibrinogen is the first clotting factor to reach critically low levels. 13 Furthermore, low perioperative fibrinogen levels have been shown to be associated with increased bleeding. 14 Therefore, increasing the levels through fibrinogen supplementation appears essential to restore normal clotting function. 15 –17 The standard practice of replacing fibrinogen has focused on infusions of therapeutic plasma (eg, FFP) or cryoprecipitate; however, there are differences concerning the volume of each product. 18 Furthermore, there are various well-known disadvantages of using these products. 11,19 Some of the issues are related to the safety and purity of these products. Therefore, with the progression from using whole blood to allogeneic blood products (eg, plasma, red blood cells, platelet concentrates, and cryoprecipitate), the next consideration should be moving toward more purified, specific products such as factor concentrates (eg, fibrinogen concentrate). This represents the move from the use of a “liquid organ” to a pharmaceutical product.
Fibrinogen concentrate (FIBRINOGENIO HUMANO LIO.) was first approved for use in Brazil on March 4, 1963. This day is considered the international birth date of the product. Notably, fibrinogen concentrate and freeze-dried fraction I (prepared by Cohn fractionation of plasma) were already available before licensure of this product became obligatory. 20 –22 The European Union birth date of the product is January 4, 1966, 23 when fibrinogen concentrate (Human-Fibrinogen Behringwerke Konzentrat) was approved in Europe for the first time by the German Federal Ministry of Health. However, Behringwerke (a predecessor company of CSL Behring GmbH) had already commenced production of human fibrinogen concentrate in 1956, before registration of a product became mandatory with the Medicinal Products Act coming in to force in 1961 in Germany. In 1985, the production process was modified to include pasteurization as a virus inactivation step, and the product was renamed Haemocomplettan P (CSL Behring GmbH). Currently, all formulations of fibrinogen concentrate manufactured by CSL Behring are pasteurized, and over 3 million grams have been used since 1985. 24
At the time of writing, CSL Behring’s fibrinogen concentrate is approved for treatment and prophylaxis of acquired and congenital fibrinogen deficiency under the trade name Haemocomplettan P in several countries (Table 1). 25,26 In Israel, Haemocomplettan P is indicated for the treatment of acute bleeding episodes in patients with congenital fibrinogen deficiency, including afibrinogenemia and hypofibrinogenemia. The same product, licensed under the trade name RiaSTAP, is also available for the treatment of acute bleeding episodes in patients with congenital fibrinogen deficiency, including afibrinogenemia and hypofibrinogenemia, in Australia, Canada, Mexico, New Zealand, Puerto Rico, United States, and several European countries (Table 1). 25,26
Current Licensing Status of Fibrinogen Concentrate Manufactured by CSL Behring.
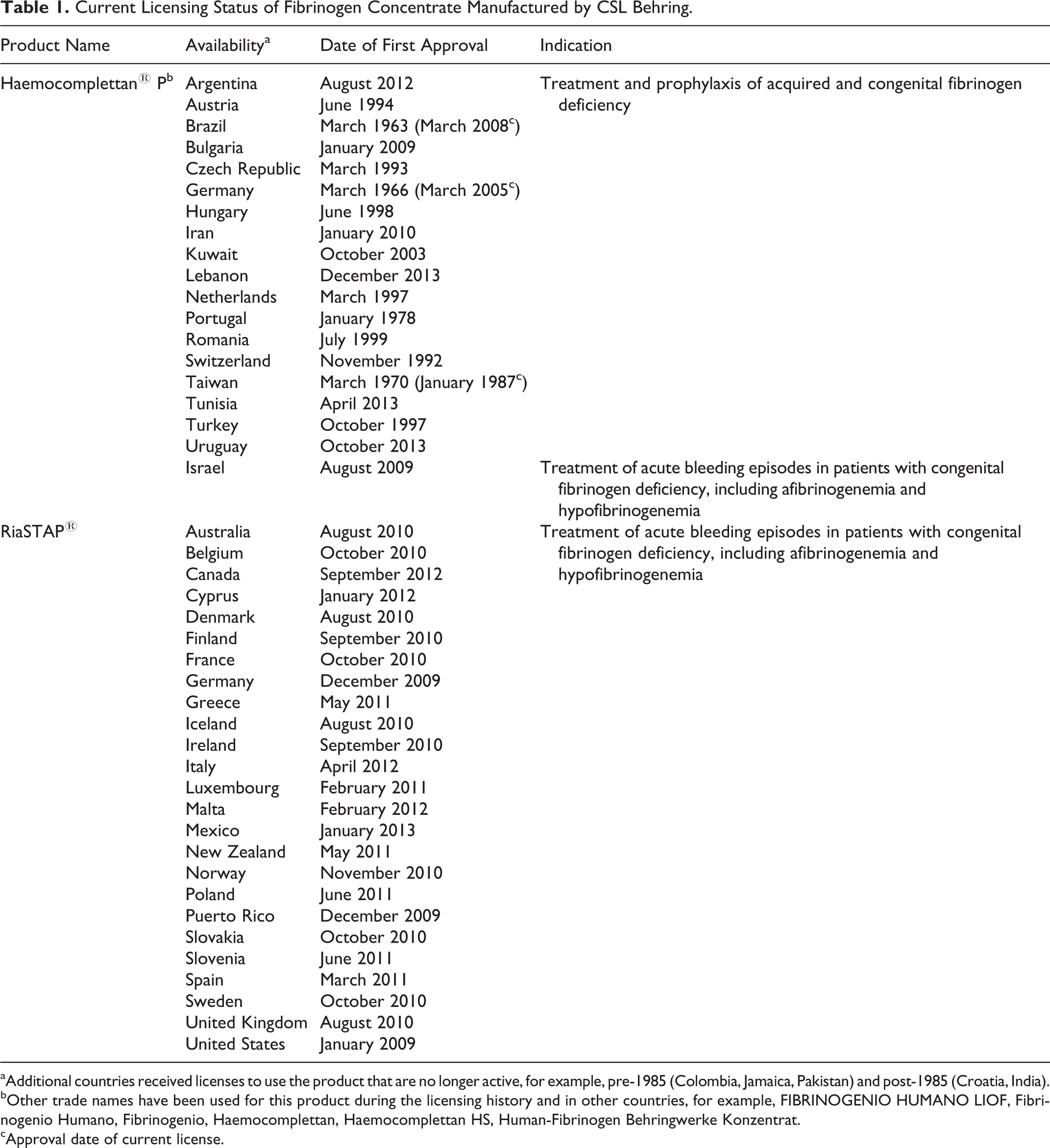
aAdditional countries received licenses to use the product that are no longer active, for example, pre-1985 (Colombia, Jamaica, Pakistan) and post-1985 (Croatia, India).
bOther trade names have been used for this product during the licensing history and in other countries, for example, FIBRINOGENIO HUMANO LIOF, Fibrinogenio Humano, Fibrinogenio, Haemocomplettan, Haemocomplettan HS, Human-Fibrinogen Behringwerke Konzentrat.
cApproval date of current license.
Current evidence suggests that fibrinogen concentrate has a good safety profile and delivers a standardized dose of fibrinogen. 27 Haemocomplettan P/RiaSTAP has been shown to be effective and well tolerated in a number of clinical studies of patients with congenital or acquired fibrinogen deficiency. 28 –35 A postmarketing surveillance study of Haemocomplettan P/RiaSTAP covering a 27-year period (1986-2013) found 28 spontaneous reports of possibly related thrombotic events for 2 611 294 g of distributed product. 27 This represents an incidence rate of 1 event per 23 300 doses.
The use of Haemocomplettan P/RiaSTAP to control bleeding and to decrease the use of allogeneic blood products during cases of acquired bleeding has been reported in a variety of clinical settings, including surgery, trauma, liver transplantation, and obstetrics. 28 –30,33,36 –43 Furthermore, recent systematic reviews confirm that the use of fibrinogen concentrate may reduce allogeneic blood product transfusion 44 –46 ; however, very often the trials are of low quality with a high risk of bias. Moreover, the measured outcomes, such as transfusion requirements, may be prone to performance bias. 47 Therefore, more randomized controlled trials are warranted before it can be confirmed whether fibrinogen concentrate has a definitive role in the management of bleeding. Additionally, some controversy still exists around the use of fibrinogen concentrate with regard to the appropriate level of fibrinogen to trigger treatment. 48,49 Ranucci and Solomon are right to note in their Editorial that there is currently a discrepancy among licences, guidelines, and clinical use and that there are open issues and gaps in knowledge that need to be addressed. 50
What Does the Future Hold for Fibrinogen Concentrate?
Four fibrinogen concentrate products are currently available in various regions 25 and according to public sources more are in development. 51 Other developments include the production of recombinant fibrinogen, first engineered by Lord et al in 1993. 52 The functional properties of a recombinant fibrinogen have been tested ex vivo in whole blood samples from a group of patients undergoing cardiac surgery. 53 According to the clinical trials registries (clinicaltrials.gov and clinicaltrialsregister.eu), there are currently 11 ongoing clinical studies investigating the use of fibrinogen concentrate in acquired bleeding. The results of these studies will potentially provide further evidence on the role of fibrinogen concentrate in the management of bleeding.
These ongoing investigations will provide additional evidence of fibrinogen concentrate in acquired bleeding, but fibrinogen may have a number of different applications beyond bleeding management. Thrombocytopenia is a common complication in hospitalized patients which affects blood clotting, and studies have shown that fibrinogen concentrate can increase the clot strength, independent of platelet count. 54,55 Further clinical investigations are necessary to confirm whether bleeding complications associated with thrombocytopenia could be managed through supplementation with fibrinogen concentrate. The role of fibrinogen and interference with the immune system is also worthy of further scrutiny, as a significant decrease in sepsis-induced mortality was observed in animals treated with fibrinogen concentrate in a rat model of sepsis-induced disseminated intravascular coagulation. 56
Advances in science and medicine have led to improvements in blood and blood products and in the way patients are treated. Fibrinogen concentrate seems to offer a specific option in the management of bleeding; however, further investigations are required to clearly define its role. Fibrinogen concentrate was described in 2009 as a “potential universal hemostatic agent” 57 ; the next 50 years will demonstrate whether this is the case.
Footnotes
Acknowledgments
Editorial assistance with manuscript preparation was provided by Meridian HealthComms, funded by CSL Behring.
Authors’ Note
Michael Wendt provided company and product information. Gerald Hochleitner developed the outline and researched the historical aspect of fibrinogen. All authors were involved in writing and reviewing the manuscript and gave final approval of the version to be published.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: R.C-F. has no conflict of interest to declare. G.H. and M.W. are employees of CSL Behring. A.T. received lecture fees and travel support from CSL Behring. D.R.S. was the chairman of the ABC Faculty and is the co-chairman of the ABC-Trauma Faculty, which both are managed by Physicians World Europe GmbH, Mannheim, Germany and sponsored by unrestricted educational grants from Novo Nordisk Health Care AG, Zurich, Switzerland, CSL Behring GmbH, Marburg, Germany and LFB Biomédicaments, Courtaboeuf Cedex, France. In the past 5 years, D.R.S. has received honoraria or travel support for consulting or lecturing from the following companies: Abbott AG, Baar, Switzerland; AMGEN GmbH, Munich, Germany; AstraZeneca AG, Zug, Switzerland; Bayer (Schweiz) AG, Zürich, Switzerland; Baxter AG, Volketswil, Switzerland; Baxter S.p.A., Roma, Italy; B. Braun Melsungen AG, Melsungen, Germany; Boehringer Ingelheim (Schweiz) GmbH, Basel, Switzerland; Bristol-Myers-Squibb, Rueil-Malmaison Cedex, France and Baar, Switzerland; CSL Behring GmbH, Hattersheim am Main, Germany and Berne, Switzerland; Curacyte AG, Munich, Germany; Daiichi Sankyo (Schweiz) AG, Thalwil, Switzerland; Ethicon Biosurgery, Sommerville, New Jersey, USA; Fresenius SE, Bad Homburg v.d.H., Germany; Galenica AG, Bern, Switzerland (including Vifor SA, Villars-sur-Glâne, Switzerland); GlaxoSmithKline GmbH & Co. KG, Hamburg, Germany; Janssen-Cilag AG, Baar, Switzerland; Janssen-Cilag EMEA, Beerse, Belgium; Merck Sharp & Dohme AG, Luzern, Switzerland; Novo Nordisk A/S, Bagsvärd, Denmark; Octapharma AG, Lachen, Switzerland; Organon AG, Pfäffikon/SZ, Switzerland; Oxygen Biotherapeutics, Costa Mesa, CA, USA; PAION Deutschland GmbH, Aachen, Germany; Photonics Healthcare B.V., Utrecht, Netherlands; ratiopharm Arzneimittel Vertriebs-GmbH, Vienna, Austria; Roche Diagnostics International Ltd, Reinach, Switzerland; Roche Pharma (Schweiz) AG, Reinach, Switzerland; Schering-Plough International, Inc., Kenilworth, New Jersey, USA; Tem International GmbH, Munich, Germany; Verum Diagnostica GmbH, Munich, Germany; Vifor Pharma Deutschland GmbH, Munich, Germany; Vifor Pharma Österreich GmbH, Vienna, Austria; Vifor (International) AG, St. Gallen, Switzerland.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: D.R.S’s academic department is receiving grant support from the Swiss National Science Foundation, Berne, Switzerland (grant numbers: 100 014_138545/1, 33CM30_124117, 33CM30_140339 and 406440-131268), the Ministry of Health (Gesundheitsdirektion) of the Canton of Zurich, Switzerland for Highly Specialized Medicine, the Swiss Society of Anesthesiology and Reanimation (SGAR), Berne, Switzerland (no grant numbers attributed), the Swiss Foundation for Anesthesia Research, Zurich, Switzerland (no grant numbers attributed), Bundesprogramm Chancengleichheit, Berne, Switzerland (no grant numbers attributed), CSL Behring, Berne, Switzerland (no grant numbers attributed), Vifor SA, Villars-sur-Glâne, Switzerland (no grant numbers attributed).
