Abstract
Background:
We assessed the effects of protamine overdosing on thrombelastometry, bleeding, and transfusions in patients after cardiopulmonary bypass (CPB).
Methods:
In group 1 (n = 15), representing the clinical standard, the protamine dose was based on the initial heparin dose, and group 2 (n = 15) received protamine based on the heparin concentration measured after CPB. Primary end points were thromboelastometric parameters. Secondary end points were perioperative blood loss and utilization of blood products.
Results:
During CPB, heparin concentrations decreased by 40%, resulting in overdosing of protamine in group 1. Thromboelastometry revealed longer clotting time (CT) in group 1 (P values < .05). Four patients in group 1 but none in group 2 had excessive prolonged CT values (>360 seconds) and concomitant microvascular bleeding, requiring substantial replacement of coagulation factors.
Conclusions:
Heparin dose-based protamine management leads to protamine overdosing with inhibition of the coagulation process. Protamine management guided by heparin concentration avoids these complications.
Introduction
Heparin reversal is a procoagulant effect of protamine. By itself, protamine also reveals anticoagulant effects that lead to dose-dependent prolongation of various coagulation parameters. 1 During cardiopulmonary bypass (CPB) surgery, a fixed weight-based heparin dose is commonly administered before commencing CPB. Usually, additional heparin boluses are given only if the activated clotting time (ACT) decreases below a defined target value. Since the ACT is affected by various factors such as temperature, hemodilution, status of the coagulation system, and platelet function, the ACT levels tend to remain in the target range during CPB despite decreasing heparin levels. 2,3 Therefore it is conceivable that protamine administration may be overdosed on the basis of the initial heparin dose and contribute to impairment of the coagulation system. This potential side effect of protamine has been studied in in vitro investigations and studies on platelet activation and smaller observational studies, but to date, only limited clinical data are available from controlled randomized studies in cardiac surgical population. 4 –6
We herein report the results of our clinical pilot investigation comparing a heparin dose-based protamine strategy with a heparin level-based protamine regimen in patients undergoing cardiac surgery with prolonged CPB. Our primary hypothesis was that particularly during the condition of a prolonged CPB, a protamine dose based on the initial heparin bolus would lead to an excess of protamine administration and impairment of the coagulation system. Our secondary hypothesis was that this condition would possibly translate into an increased need for the transfusion of coagulation factors and other allogeneic blood products. Therefore, we assessed the impact of the 2 strategies on the protamine dose, the different parameters of thromboelastometry, the utilization of allogeneic blood products, the perioperative blood loss, and the re-exploration rate.
Methods
The study was approved by the local branch of the ethics committee of the Ruhr University of Bochum in Bad Oeynhausen, Germany. The single-center study was performed at the Heart and Diabetes Centre NRW Bad Oeynhausen, Germany. The patients were enrolled between January 2011 and March 2012. All participants gave their written informed consent (see flowchart of CONSORT statement, Figure 1). Group 1 (n = 15) received a heparin dose-based protamine dose, and group 2 (n = 15) received a heparin level-based protamine dose. Only patients scheduled for mitral valve surgery and double to quadruple coronary artery bypass grafting, including a graft of the left thoracic internal artery, were included. A further inclusion criterion was a presumed CPB time of >90 minutes. Exclusion criteria are listed in Table 1. The study was registered at ClinicalTrials.gov as NCT01608971.
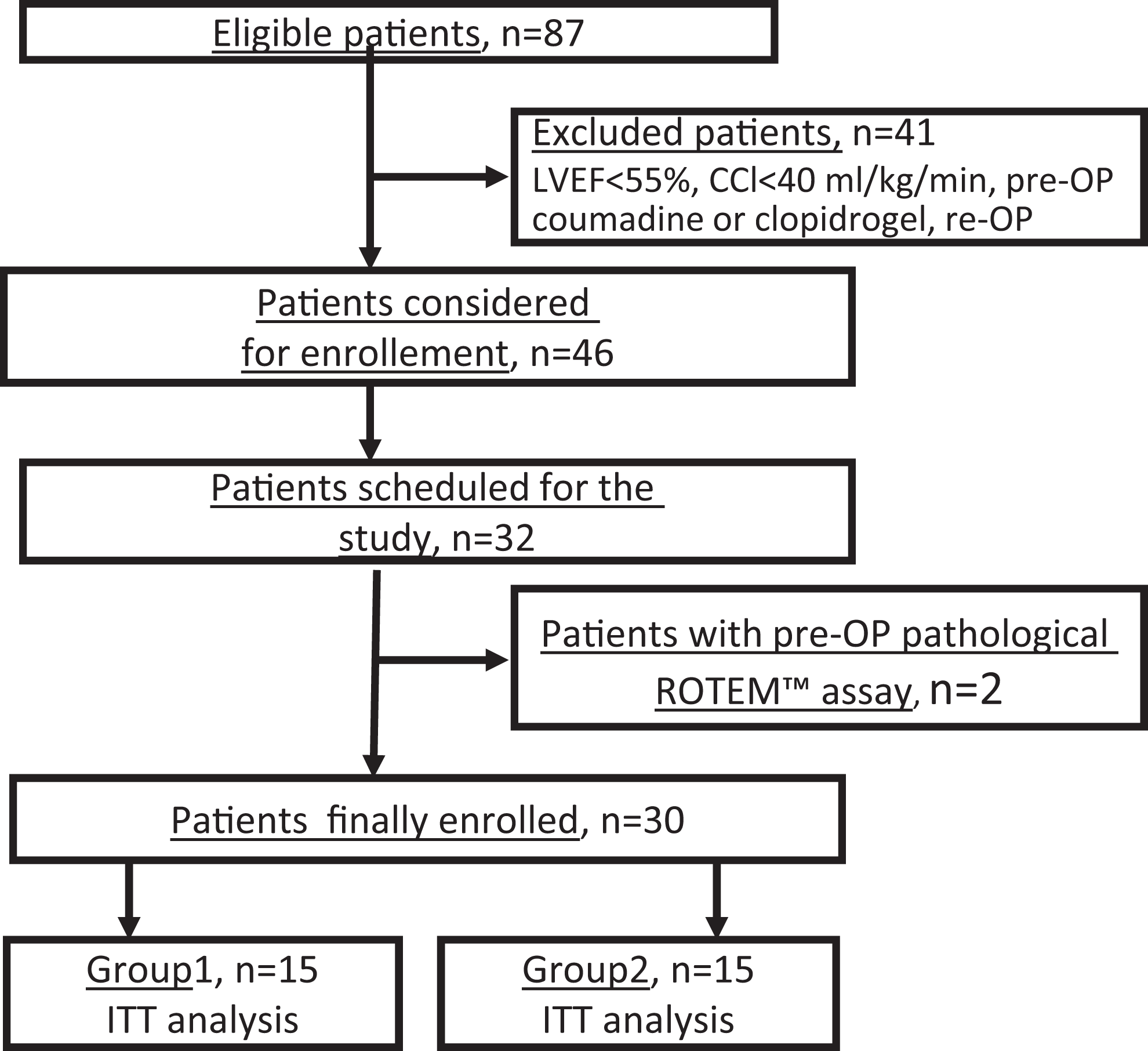
CONSORT flow chart. LVEF indicates left ventricular ejection fraction; CCL, creatinine clearance.
Exclusion Criteria.

Study Procedures
Before induction of anesthesia, the patients were allocated to 1 of the 2 different protamine dosing regimens by restricted randomization using 30 sequentially numbered opaque envelopes containing 1 of the 30 consecutive numbers. Even numbers were allocated to patients in group 1, while odd numbers were allocated to patients in group 2.
The solution containing the protamine dose was prepared by the perfusionist after commencing CPB (see the following paragraph about heparin and protamine management). Anesthesiologists, surgeons, and study nurses were blinded to the group assignment.
Anesthesia and CPB
A balanced type of anesthesia was used, which included boluses of fentanyl, etomidate, and pancuronium bromide, followed by a continuous infusion of remifentanil and vaporization of sevoflurane. Additionally, according to departmental standards, a continuous infusion of 0.25 µg/kg/min of milrinone was started in all patients after induction of anesthesia. Additional inotropic or vasoactive agents (dobutamine 3-5 µg/kg/min, adrenaline 0.05-0.2 µg/kg/min, and norepinephrine 0.05-0.2 µg/kg/min) were given if the cardiac index (CI) did not increase above 2.2 L/m2 body surface area (BSA) or if the mean arterial pressure remained at or below 70 mm Hg under milrinone alone.
In order to reduce the effect of body weight on hemodilution, a CPB system with a priming volume of 1100 mL was used in patients with a BSA of <1.8 m2, and a system with a priming volume of 1500 mL was used in patients with a BSA of >1.8m 2 . The CPB was conducted with open noncoated CPB circuits under mild hypothermia, with a core temperature of 32°C to 34°C. Cardioplegic arrest was achieved by infusing warm blood cardioplegic solution according to the Calafiore protocol.
All patients received tranexamic acid; a bolus of 1 g was administered to the patient, 0.5 g was added to the CPB priming volume, and a continuous infusion of 0.2 g/h was given throughout CPB.
Heparin and Protamine Management
Initially, a heparin bolus of 400 IU/kg was given. If the celite ACT (Actalyke ACT; Helena Lab, Beaumont, Texas) remained below the target value of >450 seconds, additional boluses of one-third of the initial dose were administered until the target value was reached. An additional dose of 10 000 units of heparin was added to the CPB priming volume.
Heparin concentrations were measured 5 minutes after commencing CPB using the white (range 3.4-6.8 IU heparin/mL) heparin protamine titration (HPT) cartridge of the Hepcon HMS Plus device (Medtronic Inc, Minneapolis, Minnesota) and 1 minute after termination of CPB using the gold-colored HPT cartridge (range 2.0-5.4 IU/mL heparin).
The total dose of protamine was always diluted to reach a volume of 100 mL, 80 mL of which was infused over a duration of 10 minutes directly after terminating CPB. The remaining 20 mL was given while the residual CPB blood was reinfused after arterial decannulation, which was usually performed 10 to 15 minutes after weaning from CPB.
In the heparin dose-based protamine group (group 1), an amount of protamine equivalent to the total initial heparin dose needed to achieve the target ACT of >450 seconds was given. In the heparin level-based group (group 2), the total protamine dose was calculated based on a 1:1 (10 mg protamine for reversal of 1000 IU heparin) reversal ratio of the actual heparin level determined by the Hepcon HMS Plus device after termination of CPB. The calculation of the total blood volume (BV; sum of patient and CPB priming volume) with the Hepcon HMS Plus device was performed as follows: first, the patient’s BSA was calculated according to the Mosteller formula (BSA, m2 = square root of height, cm × weight, kg/3600). Then, the BSA was used to calculate the patient’s BV (for male patients, BV = 2.8 × BSA m2; for female patients, BV = 2.4 × BSA m2). Finally, the priming volume of the CPB circuit was programmed.
Coagulation Tests
Fifteen minutes after the complete infusion of protamine, the INTEM, FIBTEM, and HEPTEM tests were performed on the ROTEM thromboelastometry system (TEM International GmbH, Munich, Germany). In all tests, the coagulation time (CT) is the period until clot formation starts; the clot formation time (CFT) is the period until the clot strength reaches 20 mm, and the maximal clot firmness (MCF) indicates the maximal clot strength (Figure 2). For the INTEM test, the intrinsic coagulation pathway is activated. In the HEPTEM test, heparinase is added to the INTEM reagent to detect the anticoagulant effects of any residual heparin. In the FIBTEM test, the extrinsic pathway of the coagulation system is activated, and a potent platelet inhibitor, cytochalasin, is added. Therefore, MCF reflects only the strength of the plasma (fibrinogen) clot. 7 –9 The reference values of the tests are given in Figure 2.
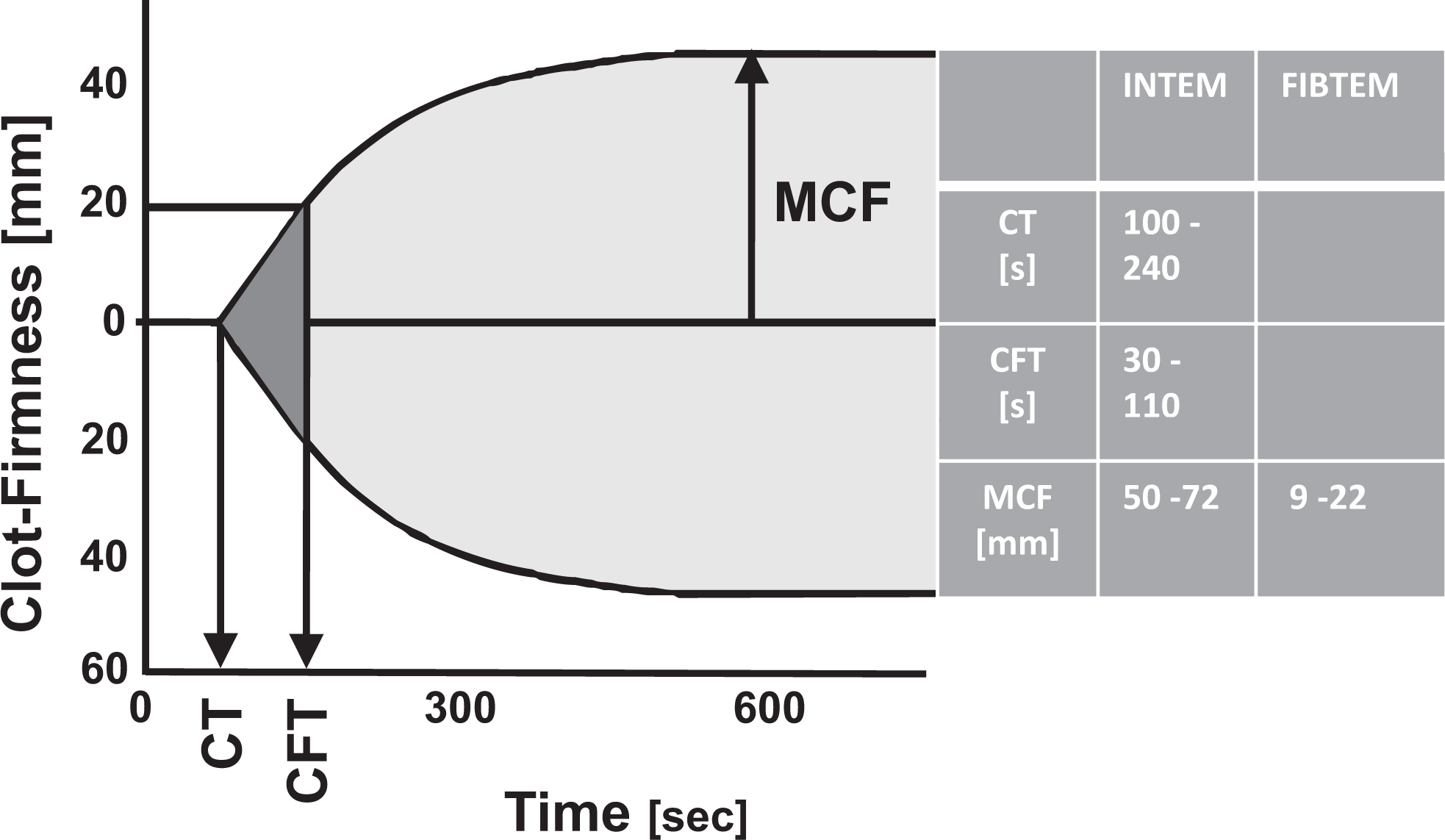
Parameters and reference ranges of ROTEM thromboelastometry. CT indicates coagulation time; CFT, clot formation time; MCF, maximum clot firmness.
Transfusion Triggers
During CPB, the hemoglobin (Hb) level triggering transfusion of packed red blood cells (PRBCs) was 8 g/dL. After CPB, in patients without need for inotropic support with dobutamine/adrenaline or noradrenaline and a CI >2.2 L/m2, the critical Hb level triggering transfusion of PRBC was defined as 8 to 9 g/dL. In patients requiring inotropic support with dobutamine/adrenaline or noradrenaline, the minimum target was 9 to 10 g/dL.
In patients with diffuse bleeding after protamine administration, management with fresh frozen plasma (FFP), prothrombin complex concentrate (Beriplex; CSL Behring, Marburg, Germany), fibrinogen concentrate (Haemocomplettan P; CSL Behring), and single donor apheresis platelet concentrates (PCs) was based on the results of the ROTEM analysis. However, these blood products were given only if the diffuse microvascular bleeding was observed in the surgical field.
We suggested the presence of residual heparin and gave additional protamine (0.5 mg/kg). When compared to the INTEM test, the CT was shortened by >10% in the HEPTEM test. In case of a moderate prolongation of the CT (240-360 seconds) in the INTEM test (and comparable result in the HEPTEM test), 3 to 4 units of FFP were transfused. If bleeding persisted, an additional 3 to 4 units of FFP or, in case of a prolongation of the CT > 360 seconds, 2000 to 4000 IU of prothrombin complex concentrate was given. Fibrinogen concentrate (2-4 g) was given when the MCF in the FIBTEM assay was <8 mm, and 2 apheresis PCs were transfused, if the MCF in the INTEM assay was <45 mm. The reference values of the ROTEM tests are listed in Figure 2.
Primary and Secondary End Points
The primary end point of the study was the impact of the 2 different protamine management strategies on the protamine dose and its effect on the hemostatic system as observed with various ROTEM assays. Secondary end points were utilization of allogeneic blood products, perioperative blood loss, and re-exploration rate.
Statistics
Due to the character of a “pilot investigation,” the sample size was not calculated. Categorical variables are presented as number or percentage of observations. Continuous variables are presented as median and interquartile range (IQR), since according to the Kolmogorov-Smirnov test, several variables were not normally distributed (P values below .05). For data comparison, we used the Mann-Whitney test, Fisher exact test, or chi-square test, as appropriate. P values <.05 were considered significant, and P values of .05 to .10 were considered borderline significant. Statistical analysis was performed with the software package PASW Statistics, version 18 (Predictive Analysis Software, Chicago, Illinois).
Results
All the 30 patients completed the study. Preoperative characteristics of the patients are presented in Table 2. There were no group-specific differences with regard to baseline characteristics of the patients and the preoperative ROTEM values. Surgical data are presented in Table 3. All patients underwent a procedure with a prolonged (>90 minutes) duration of CPB.
Preoperative Baseline Characteristics.a
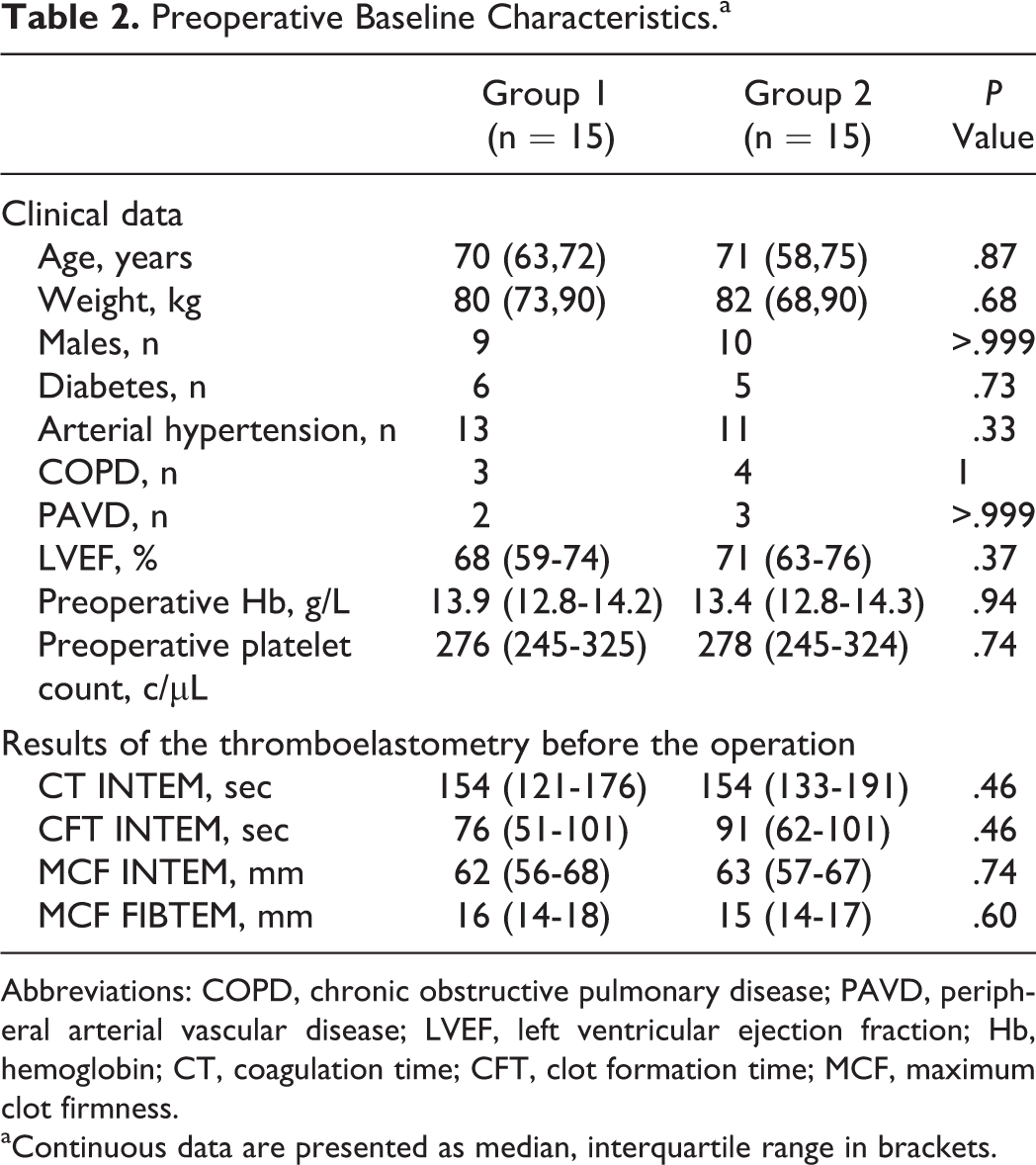
Abbreviations: COPD, chronic obstructive pulmonary disease; PAVD, peripheral arterial vascular disease; LVEF, left ventricular ejection fraction; Hb, hemoglobin; CT, coagulation time; CFT, clot formation time; MCF, maximum clot firmness.
aContinuous data are presented as median, interquartile range in brackets.
Intraoperative Data.a
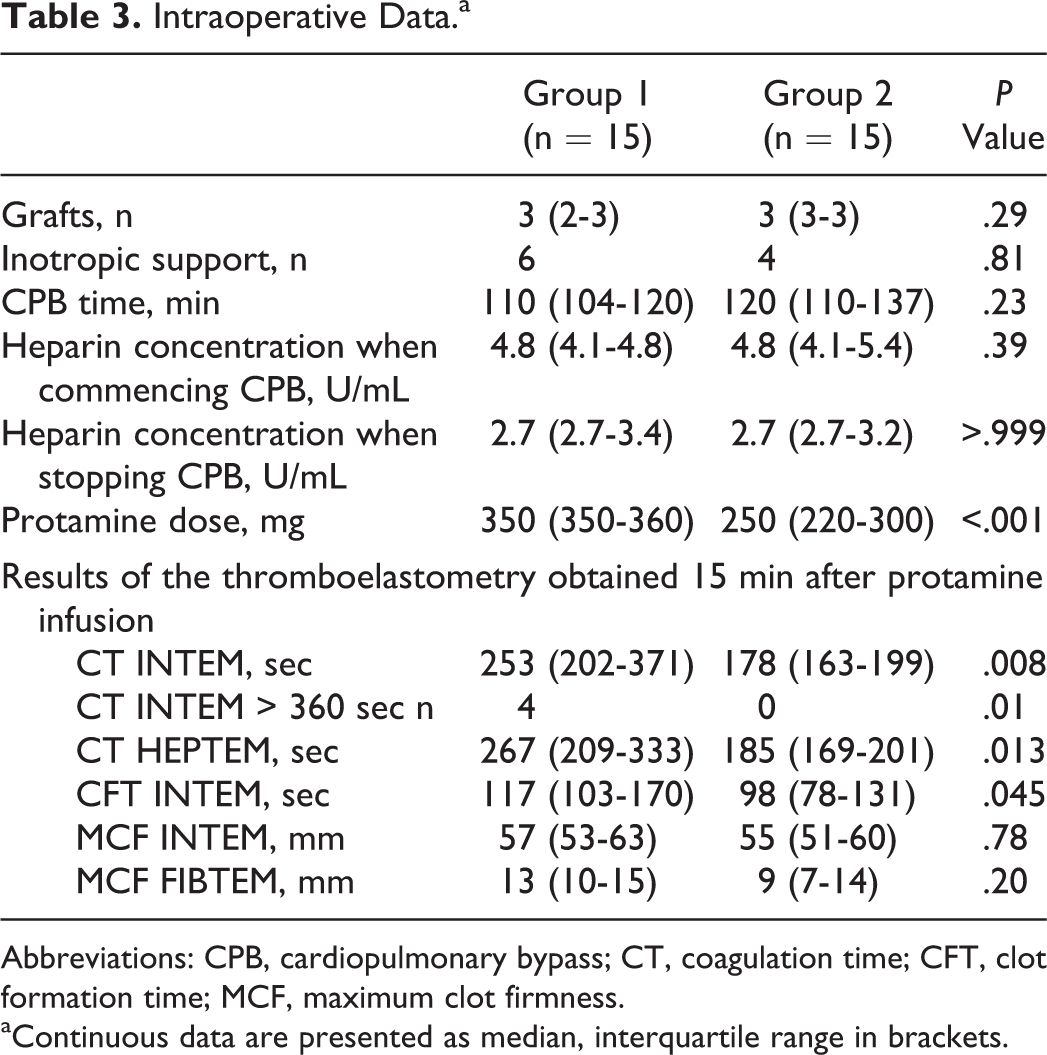
Abbreviations: CPB, cardiopulmonary bypass; CT, coagulation time; CFT, clot formation time; MCF, maximum clot firmness.
aContinuous data are presented as median, interquartile range in brackets.
In both the groups, heparin levels decreased significantly by approximately 40% from the levels measured 10 minutes after starting CPB to the time shortly after terminating CPB (Table 3). In group 1, this corresponded to an approximately 40% higher dose of protamine when compared to group 2. In group 1, the actually given protamine dose was 350.5 ± 50.7 mg, while the heparin concentration-based protamine dose would have been 210.18 ± 40.9 mg. In group 2, the heparin dose-based protamine dose would have been 370.2 ± 70.74 mg, while the given (heparin concentration based) protamine dose was 250.63 ± 40.7 mg.
Primary End Point
The INTEM test revealed significantly longer intraoperative CT and CFT values in group 1 when compared to group 2 and so did the CT HEPTEM test (Table 3). In group 1, 4 patients had excessive prolonged CT INTEM values (>360 seconds), whereas excessively prolonged CTs were not observed in group 2 (Table 3). In group 2, 1 patient needed an additional bolus of protamine due to residual heparin after protaminization. There were no differences in the CFT and MCF of the INTEM and FIBTEM test between the study groups.
Secondary End Points
Intraoperative blood loss and the need for perioperative transfusion of PRBC did not differ between the groups (Table 4). However, the amount of postoperative blood loss tended to be higher in group 1 when compared to group 2 (Table 4). One patient in group 2 required surgical re-exploration because of excessive postoperative blood loss due to bleeding from a side branch of the left internal thoracic artery. When this patient was excluded from data analysis, the difference in postoperative blood loss between the 2 groups reached statistical significance (median and IQR: 723 mL [600-850 mL] vs 610 mL [588-663 mL]; P = .033). There was a tendency toward more transfusions of FFPs and prothrombin complex concentrate in group 1 when compared to group 2 (Table 4). All patients had an uneventful subsequent postoperative course in the intensive care unit and were discharged from the hospital uneventfully.
Blood Loss and Transfusions.a
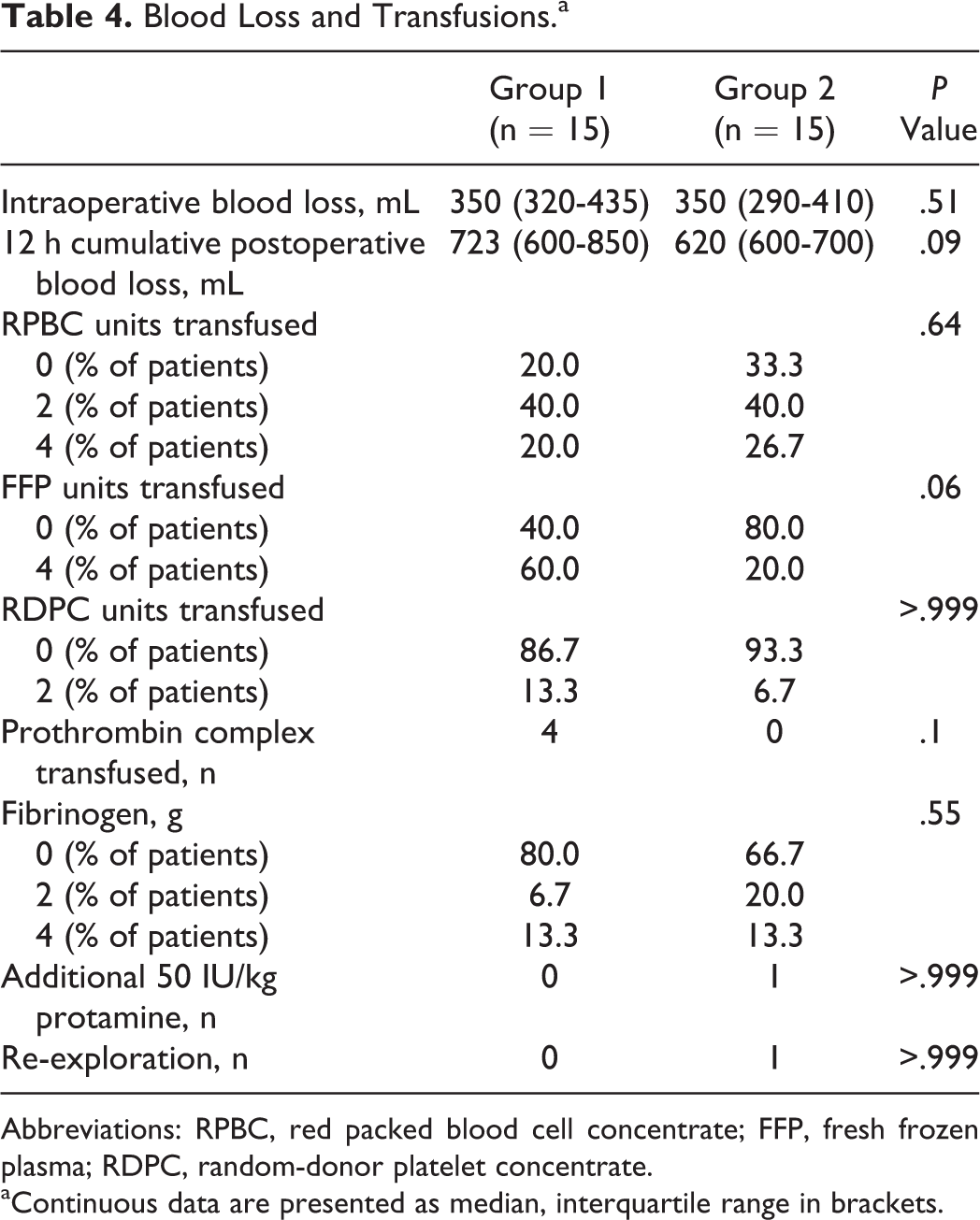
Abbreviations: RPBC, red packed blood cell concentrate; FFP, fresh frozen plasma; RDPC, random-donor platelet concentrate.
aContinuous data are presented as median, interquartile range in brackets.
Discussion
This study addressed the effects of 2 protamine dosing regimens on coagulation, bleeding, and transfusions in cardiac surgery with prolonged CPB. Our data show that heparin levels decreased significantly when using an ACT-based heparin management. Consequently, initial heparin dose-based protamine administration in group 1 resulted in an overdose. This caused disturbances of the hemostatic system as observed by a prolongation of the CT in the INTEM test of thromboelastometry. These disturbances in group 1 translated into a trend toward an increased need for transfusion of PRBCs, FFPs, and plasmatic coagulation factors. Increased postoperative hemorrhage and subsequent need for re-exploration, which occurred in 1 patient of group 2, was surgical and thus not related to the study intervention.
It is worth to note that in 1 patient in group 2, the Hepcon HMS Plus device underestimated the protamine demand. The Hepcon HMS Plus device determines heparin levels using a discontinuous method (gaps of 0.5-0.7 IU/mL heparin between the channels). Additionally, patient’s BV is calculated on the basis of the BSA. However, particularly in patients with heart disease and enlargement of the heart chambers, standard formulas might underestimate the patient’s BV. This system with immanent imprecision may serve as an explanation for this observation.
Coagulation is a complex process involving parallel positive and inhibitory feedback loops initiated by the interaction of the endothelium, plasma proteins, and platelets. Due to its complexity, it is nearly impossible to translate this process to in vitro test systems. Thromboelastometry does illuminate some phases of the coagulation process in vitro. Particularly, modified thromboelastometry with different activators and inhibitors of the hemostatic system is helpful for differentiating disturbances of the coagulation system and for guiding a differentiated therapy with protamine, coagulation factors, and/or PCs. 7 –9
In thromboelastometry, the CT represents the initial phase of thrombin generation/activation, until the beginning of the formation of plasma coagulate. The CFT represents the initial formation of a firm coagulate, while MCF represents the final strength of the thrombus achieved by the interaction of platelets with fibrinogen and fibrin. 7 –9
In our investigation, the protamine to heparin ratio in the patients in group 1 was approximately 1.4:1. In line with previous in vitro studies and a smaller clinical investigation, this protamine overdose prolonged the CT INTEM significantly. 4,5 Similar results, but however less pronounced, were observed regarding the CFT. As the CT represents the thrombin generation/activation phase, this observation is in line with recent data showing that protamine downregulates the thrombin generation by inhibition of the coagulation factor V and VII and by degradation of the coagulation factor Xa and factor Xa-antithrombin complex. 10 –12
Shigeta et al 6 investigated the effect of an approximately 2:1 protamine overdose on platelet activation. This clinical study revealed a reduced response of the platelets to activation by thrombin. Although thromboelastometry assesses some aspects of platelet function in the MCF, the method is not sensitive enough to detect more subtle changes in platelet function, such as those caused by antiplatelet drugs like aspirin or clopidogel. 13,14 This might explain why in contrast to the results of Khan et al our more moderate protamine overdose did not translate into the changes in the MCF. 4
The elimination half-life of protamine is approximately 5 minutes. 15 We infused protamine over a period of approxima-tely 20 minutes and obtained blood samples for thromboelastometry 15 minutes after completing the protamine infusion. Therefore, most of the protamine should have been cleared from the circulation when the blood samples were obtained. It is still conceivable that apart from its direct immediate anticoagulant effects, protamine initiates profound disturbances of the complex coagulation cascade with a persistent impairment of thrombin generation/activation. This would also serve as an explanation for the observation that in patients with a protamine overdose, the postoperative blood loss tended to be higher until 12 hours postoperatively despite transfusion of plasmatic coagulation factors.
The changes in the coagulation system after a protamine overdose were not consistent within the entire group of patients. Although some patient had normal coagulation tests and no signs of diffuse bleeding, 4 patients ended up with a severe disturbance of the coagulation system as shown by a CT INTEM test of >360 seconds and diffuse bleeding. Future studies should elucidate the potential mechanisms for the different interindividual responses.
Our study has some limitations; first, since this was a pilot study, we were unable to perform statistical power calculations before study begin. This may contribute to the fact that some results show only borderline significance. Second, the small number of included patients makes it necessary to confirm these initial clinical results in larger patient population and under more varied study conditions. Third, intraoperative blood loss was not quantified in defined time intervals throughout the procedure but only at the end of the operation. The occurrence of intraoperative diffuse bleeding was evaluated by clinical observation of the operation field, and therapy initiated immediately according to the results of the ROTEM. This might explain the fact that despite the marked disturbances in the hemostatic system seen in group 1, only a tendency toward group-specific differences in the perioperative blood loss and transfusion of RPBC was seen.
In line with preclinical, in vitro, and clinical studies, 4 –6,10 –12 our data indicate that heparin dose-based protamine administration leads to protamine overdosing and disturbs the coagulation system. Clinically, this results in diffuse bleeding and a tendency toward increased transfusion demands. Devices for calculating the individual protamine demand based on the measured heparin levels, such as the Hepcon HMS Plus used in our study, and devices for performing an in vitro dose response of protamine and calculating the individual protamine dose, such as the Hemochrone RxDx system, are available. 16 Such systems can reduce the risk of administering overdoses of protamine. Alternatively, if such systems cannot be used, careful in vivo titration of protamine may also be an option.
In conclusion, protamine management guided by heparin concentration avoids protamine overdosing and appears to be associated with less impairment of the coagulation system.
Footnotes
Authors’ Note
Institute for Anaesthesiology, Heart and Diabetes Centre NRW, Bad Oeynhausen, Ruhr-University Bochum, Germany.
Declaration of Conflict of interest
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article:Dr Koster has received honoraries from Medtronic Inc, Europe, for lectures and training session about the Hepcon HMS Plus. No other author has any potential conflict of interest to declare.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The work was supported by the Heart and Diabetes Centre North Rhine-Westphalia, Ruhr-University Bochum, Bad Oeynhausen, Germany. The Hepcon HMS Plus device and cartridges for the HMS were provided by Medtronic INC Germany, Dusseldorf, Germany.
