Abstract
Background:
Recurrent miscarriage (RM) is one of the most common clinical problems in reproduction with no definite cause in about 50% of the cases. The study aims to evaluate the effect of low-molecular-weight heparin (LMWH) in the treatment of women with RM negatively tested for antiphospholipid antibodies (APAs).
Methods:
An open-labeled registered randomized controlled study (NCT 01608347) included women who attended the outpatient clinic in Assiut Women Health Hospital and Nag-Hamady Central Hospital, Egypt, with 3 or more unexplained RM. Eligible participants were randomly assigned into 2 groups. The study group included 150 patients receiving LMWH (Tinzaparin sodium 4500 IU) subcutaneous daily injection with 500 µg folic acid once daily orally started once positive pregnancy test till the 20th week of gestation. The control group included 150 patients receiving the same dose of folic acid alone. The primary outcome of the study was the rate of continuation of a viable pregnancy after 20 weeks of gestation.
Results:
There was no significant difference between both groups as regards age, parity, or number of previous miscarriages. There was a significant increase in women who continued their pregnancy beyond 20 weeks in the study group compared to the control group (73.3% vs 48%, respectively; P = .002). The take-home baby rate was also significantly higher in the LMWH group compared to the control group (P = .001).
Conclusion:
Early start of LMWH decreases the incidence of miscarriage in the first 20 weeks of pregnancy in women with unexplained RM negative for APAs.
Introduction
Recurrent miscarriage (RM), also referred to as recurrent pregnancy loss (RPL), is traditionally defined as 3 or more consecutive miscarriages occurring before 20 weeks of gestation. 1 It is one of the most common clinical problems in reproduction, yet a definite cause can be established in only 50% of cases. 2 Many etiological factors have been proposed, but none of them has been fully substantiated. Recurrent miscarriage has been directly associated with maternal thrombophilic disorders, 3 parental chromosomal anomalies, 4 and structural uterine anomalies 5 and indirectly with maternal immune dysfunction and endocrine abnormalities. 6
The association between miscarriage and antiphospholipid antibodies (APAs) was first noticed in the latter third of the last century. The antiphospholipid syndrome (APS) is characterized by the presence of APAs, associated with venous and/or arterial thrombosis and/or pregnancy loss. 7 The adverse pregnancy outcomes associated with the presence of APAs include recurrent fetal loss, intrauterine growth restriction (IUGR), and severe preeclampsia. 8
Testing the effect of heparin in the treatment of cases with RPL but negative for APA has been done in few animal and clinical studies. Animal studies showed that cases with disorders suspicious for APS but who had negative test results for lupus anticoagulant (LAC) and anticardiolipin (aCL) antibodies are carrying antibodies pathogenic to murine pregnancy. 9 So, this study was focused on the probability of the presence of untested LAC and aCL antibodies or very low levels of APAs by commonly used methods that may explain RM in women with negative APAs.
Randomized prospective study was done to assess the efficacy of early thromboprophylaxis of low-molecular-weight heparin (LMWH) in women with RM without identifiable causes. The results showed that there is a significant reduction in the incidence of both early and late miscarriages. 10
Cochrane Database Systemic Review (2014) shows randomized comparative studies for treating RM in women without APS. Although the result of meta-analysis shows no significant differences between LMWH and other antithrombotic drugs as low-dose aspirin (LDA), they expressed the need for large randomized controlled trials (RCTs) to solve this problem. 11
The current study is an RCT evaluating the effect of LMWH versus no treatment in the treatment of women with RM who is negative for APAs testing.
Methods
The current study was an open-labeled registered RCT at www.ClinicalTrials.gov (NCT01608347). The ethical review board of Assiut Faculty of Medicine approved the study. The study participants were recruited from the outpatient obstetrics clinics of Women Health Hospital, Assiut University and Nag-Hamady Central Hospital during the period between the January 01, 2011, and the end of December 2014.
Eligible Participants
All pregnant women presented to the above hospitals with RM, defined as 3 or more spontaneous consecutive miscarriages before 20 weeks’ gestation, were included in the study. They were clinically examined and investigated for different known causes of RM including APS and endocrinal or uterine anatomical disorders. Informed written consent was obtained for participation after reading the patient information sheet or having it discussed with them if cannot read.
We included in our study pregnant women between 20 and 35 years of age with a regular marital life with the same partner, regularly menstruating before current pregnancy, and got spontaneous conception.
We excluded women with polycystic ovarian syndrome, any endocrinal abnormalities such as diabetes mellitus, thyroid disorders, or history of abnormal uterine cavity proved by sonohysterography or hysteroscopy before pregnancy. Also women with positive consanguinity and those who refused to participate in the study were excluded. All included women were tested for APA by measurement of aCL-immunoglobulin (Ig) G and IgM antibodies by an enzyme-linked immunosorbent assay (Selisa; Cambridge Life Sciences, England). The results considered positive when the aCL-IgG >8.4 IU/mL and aCL-IgM >7 IU/mL.
Intervention
All eligible participants were recruited to receive one of the following treatments once pregnancy test was positive until the end of the 20th week of pregnancy. Group 1 (study [LMWH] group) included 150 patients who had received 500 µg folic acid (Mepaco-Medifood, Egypt) tablet daily together with tinzaparin sodium 0.4 mg/kg (Innohep 4500 IU; LEO Pharma A/S, Denmark) subcutaneous daily injections. Group 2 (control group) included 150 patients and received folic acid only in the same dose without LMWH.
Study Outcomes
The primary outcome of this study was the continuation of viable pregnancy beyond 20 weeks of gestation. The secondary outcomes were the take-home baby rate, miscarriage rate, occurrence of pregnancy complications such as IUGR or preeclampsia, and any side effects of the drug used, both to the mothers or their babies (maternal bleeding, heparin-induced thrombocytopenia, pain and bruising at injection sites, and teratogenicity).
Follow-Up Schedule
All study participants were followed up until the 20 weeks of gestation using standard antenatal care frequency usually done to pregnant women in obstetrics clinic. Additionally, each participant was subjected to: First ultrasound scan: Transvaginal scan was done to evaluate fetal viability at 6 weeks from the last menstrual period. Two other scans were done transabdominal around the 10th and the 20th weeks of pregnancy. In each scan, confirmation of fetal viability was done in addition to searching for any congenital malformations.
The antenatal records of the patients including fetal viability and any other complications were recorded in the data collection sheet up to the delivery. Side effects of drug treatments were also recorded.
Randomization
Randomization was done using a computer-generated random table. Eligible patients were randomly assigned to 1 of the 2 study groups. Allocation concealment was ascertained using serially numbered closed opaque envelopes. Each envelope was labeled with a serial number and had a card noting the intervention type. Once allocation has been obtained, it could not be changed.
Sample Size
The sample size calculation was based on the primary outcome (viability of the fetus at 20 weeks of gestation). Previous study by Badawy et al showed that RM rate was 8.8% without treatment and 4.4% with LMWH treatment.10 That means LMWH can reduce early pregnancy loss rate by 50%. Using Epi-info computer software, with an α of 10% and power 80%, with group ratio 1:1, the number of patients needed was 228 (114 patients in each group) taking in consideration a dropout rate of <10%.
Statistical Analysis
The data were collected and analyzed using the Statistical Package for Social Science (version 21; SPSS Inc, Chicago, Illinois). The demographic data were compared between the treatment groups. A Student t test was used to compare the continuous outcome measures. For dichotomous variables, χ2 was used to estimate the significance value. For analysis, P < .05 was considered to be significant.
Results
Of 691 pregnant women with a history of RM examined for participation in the study, 300 (150 in each arm) of them were enrolled in the study. The causes of exclusion and the study flowchart are shown in Figure 1. There were no significant differences between the 2 groups in their age, parity, number of previous miscarriages, body mass index (BMI), and history of preeclampsia or IUGR at the time of enrollment (Table 1).
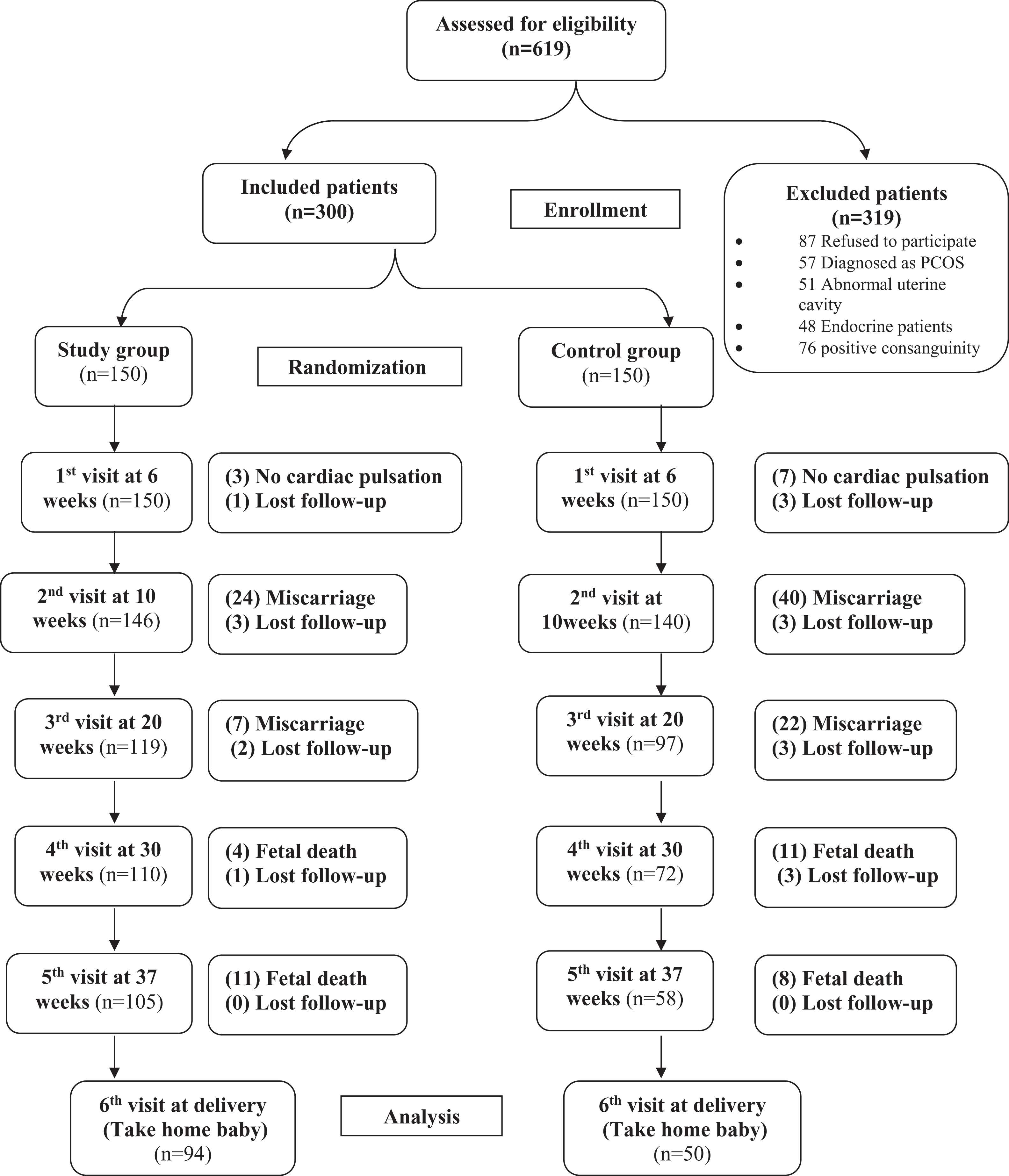
Flowchart showing the results of all visits during the study in both groups.
Baseline Characteristics of the Study Participants.
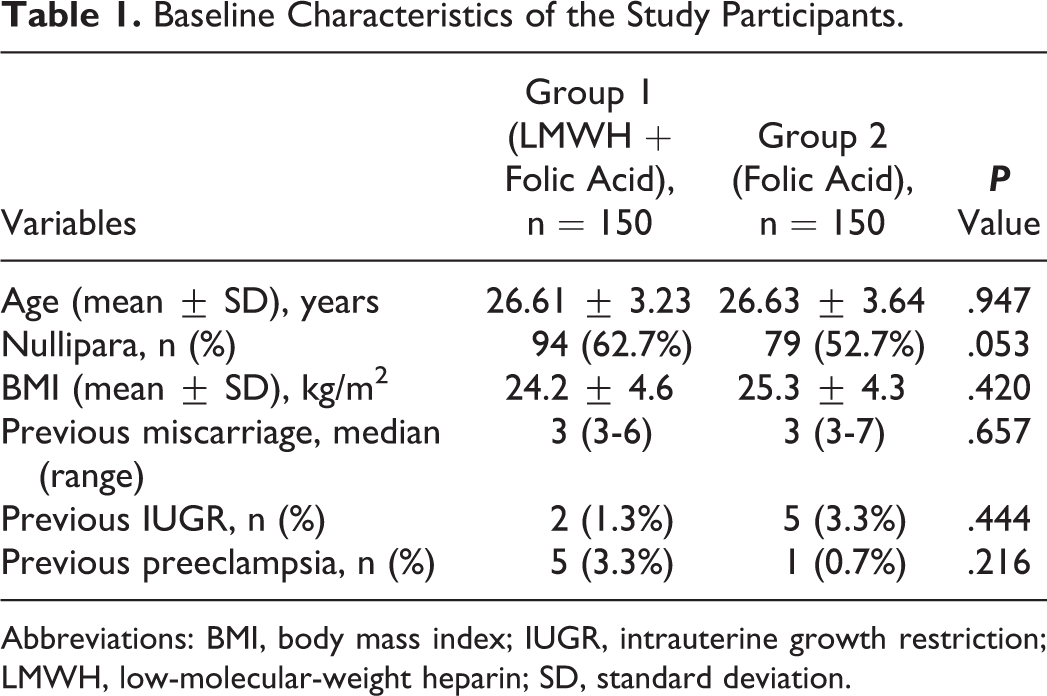
Abbreviations: BMI, body mass index; IUGR, intrauterine growth restriction; LMWH, low-molecular-weight heparin; SD, standard deviation.
The number of women who continued with viable pregnancy beyond 20 weeks was 110 (73.3%) in the LMWH group (group 1) versus 72 (48%) in the control group (group 2) with statistically significant difference (P = .002). The LMWH group had a significantly lower number of miscarriages before 20 weeks (34 [23.6%] vs 69 [48.9%], P = .002) and a significantly higher number of take-home babies (94 [65.7%] vs 50 [36.2%], P = .001) compared to the control group. There was also a significant difference in the number of fetal deaths after 20 weeks (15 [13.7%] vs 19 [27.5%], P = .031; Table 2).
The Study Outcomes Among the 2 Groups.a
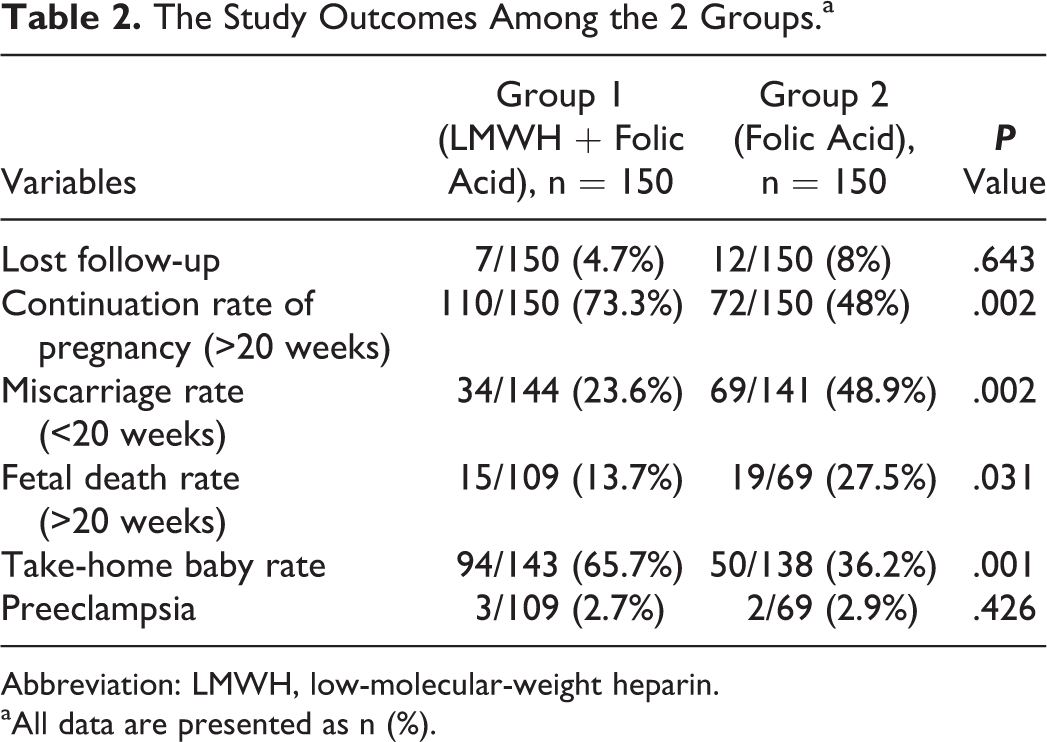
Abbreviation: LMWH, low-molecular-weight heparin.
aAll data are presented as n (%).
As regards pregnancy complications, the number of women who developed late-onset preeclampsia was not significantly different between the 2 study groups, 3 (2.7%) of the 109 in the LMWH group versus 2 (2.9%) of the 69 in the control group. Only 2 cases delivered prematurely (<37 weeks) in the combination group versus no cases in the control group. None of the women in either group developed an early-onset preeclampsia, IUGR, or thromboembolic complication during pregnancy.
Maternal adverse events were the development of thrombocytopenia in 2 women in the LMWH group versus none in the control group. Thirty-two (22.3%) women in the LMWH group displayed ecchymosis and indurations at the injection sites, but they continued treatment until the 20th week. No cases of maternal bleeding and no fetal congenital anomalies were detected in both groups.
Discussion
In this reported registered RCT, LMWH (tinzaparin) given at daily dose of 4500 IU leads to a significantly lower rate of miscarriages and higher rate of live births in pregnant women with a history of unexplained RMs negatively tested for APAs.
Low-molecular-weight heparins are important antithrombotic agents, and they enhance the inhibitory effect of antithrombin on activated factor X (FXa), thrombin (FIIa), activated FIX, and activated FXII. 12 Tinzaparin sodium has a more balanced ratio of anti-Xa and anti-IIa activities than other LMWHs. Tinzaparin sodium also has a greater effect on the release of tissue factor pathway inhibitor, a physiological inhibitor of thrombosis, than other LMWHs. 13 In addition to its anticoagulant action, LMWH works by reduction in fetal loss by binding to phospholipids, thereby protecting trophoblast phospholipids from attack and promoting successful implantation in early pregnancy. 14 To be valuable, LMWH may need to be given at the time of implantation.
A Cochrane Systemic Review (2005) on the treatment of RM in women without APS concluded that there was no significant difference between women treated by LDA as compared to those treated by the addition of LMWH. The study concludes the great need of large RCTs to further clarify this issue. 15 Since the initiation of our study in 2010, the results of several randomized trials were reported in unexplained RM.
An early RCT by Badawy and colleagues showed that early thromboprophylaxis by LMWH (enoxaparin sodium 0.2 mL, 20 mg, once daily subcutaneously) in women with a history of RM without identifiable causes was superior to no treatment. 10 Other investigators, however, suggested that no need to treat unexplained RM without APS or inherited thrombophilia with heparin or aspirin because of lack of evidence of any benefit and potential risks of therapy. 16
In our study, we administered LMWH to compare it against no treatment. We used a fixed dose of LMWH (tinzaparin 4500 IU/24 h) regardless of the BMI or the number of previous miscarriages. The above dose considered a prophylactic dose against thromboembolic complications according to Philippe et al who defined prophylactic dose as 2500 to 4500 UI/24h for tinzaparin. 17
The pregnancy outcome by the end of the first 20 weeks of pregnancy was our primary outcome. The miscarriage rate was 23.6% in the LMWH versus 48.9% in the control group, a difference that reached a statistically significant value. These results were keeping on track with the study of Badawy et al. 10 Also, our results keeps with Fawzy et al who achieved a live birth rate of 81% using enoxaparin 20 mg/d in women with unexplained RM when compared to the control group with a live birth rate of 48%; all had a negative hereditary thrombophilia work-up. 18 Moreover, the Enoxaparin for habitual abortion (HABENOX) trial reported that the live birth rate was higher with LMWH (71% vs 61% for enoxaparin and aspirin, P = .45). 19
Also our results agreed with the results of Zolghadri et al in which the live birth rate was 83.7% after thromboprophylaxis (LDA + LMWH) in the study group and was significantly higher than that in the control group (54%). 20 The higher live birth rate in their study than that achieved in our cohort may be due to giving LMWH up to 36 weeks of pregnancy. Additionally, our bigger sample size may demonstrate amelioration of the effect of early boosting of pregnancy. Further trial head-to-head between restricted first-trimester administrations versus continuous administration of LMWH in such patients is needed to reach a conclusion.
After registering our RCT, a Cochrane Review published by de Jong et al included 9 studies that were heterogeneous with regard to study design and treatment regimen. 11 The reviewers excluded 3 studies because of a high risk of bias. They concluded from the results of other 6 RCTs to not support the use of anticoagulants in women with unexplained RM. The results of our registered study might be included in their next reanalysis.
In 2015, a multicenter study was conducted by Schleussner et al to determine whether LMWH increases ongoing pregnancy and live birth rates in women with unexplained RM. 21 They concluded that daily LMWH does not increase ongoing pregnancy or live birth rates in women with unexplained RM. These results were not in accordance with our results giving attention that the LMWH used was dalteparin sodium and for up to 24 weeks’ gestation. The difference in the type of heparin and the nature of study participants may be behind the difference in our results.
In a study conducted by Pasquier et al, they reported in a recent randomized double-blind, placebo-controlled study that enoxaparin given at the daily dose of 40 mg did not improve the chance of a live birth in nonthrombophilic women with a history of unexplained RM. They detected a minimal clinically important difference of 10% in live birth rates. 22
There were no cases having congenital anomalies in both study groups. The LMWH does not cross the blood–placental barrier, given the safe use of drug by pregnant women. 23 Maternal side effects to LMWH use were limited as out of 150 participants, 78.7% had no side effects and 21.3% developed ecchymosis at the site of injection. These results coincided with de Jong et al who found local skin reactions (pain, itching, and swelling) to the injection site of LMWH were reported in almost 40% of patients. 11 This also agreed with Monien et al as they had no severe side effects of LMWH seen in their study. 24
The present study showed no significant difference in other pregnancy complications rather than miscarriage between both groups; this is in agreement with Zolghadri et al. 20 Similarly, Yuksel et al reported no difference in the maternal and neonatal side effects among their study participants. 25 These results may reassure us about the safety of using LMWH during pregnancy without special laboratory follow-up. 26 Most of the recent RCTs confirmed the safety of LMWH in pregnancy. 22
The study had its limitations including not having paternal genetic testing for chromosomal abnormalities, which is defined as one of the causes that should be excluded before diagnosing a recurrent unexplained miscarriage.
Conclusion
The use of LMWH (tinzaparin) in a fixed dose of 4500 IU starting from positive pregnancy test expresses 20th week of gestation decreased both early and late miscarriage rate and increase live birth rate in patients with RM negatively tested for APA. Further RCTs with extension of the period of treatment with LMWH may increase the percentage of take-home babies.
Our initial results were presented as a poster presentation in the American Society of Reproductive Medicine (ASRM) 67th Annual Meeting, October 20-24, 2012.
Footnotes
Acknowledgments
The authors acknowledge LEO Pharma Company, Egypt, for their support of providing the drug to the participants.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
