Abstract
Background:
The outcome of patients with upper extremity deep vein thrombosis (UEDVT) has not been consistently compared with that in patients with lower extremity deep vein thrombosis (LEDVT).
Methods:
We used the Registro Informatizado de Enfermedad Trombo Embólica (RIETE) registry to compare the outcomes during the course of anticoagulant therapy in patients with UEDVT versus outcomes in patients with LEDVT.
Results:
As of August 2015, 37,366 patients with acute DVT had been enrolled in RIETE: 35094 (94%) had LEDVT, 1334 (3.6%) non-catheter related UEDVT (672 unprovoked and 662 provoked) and 938 (2.5%) had catheter-related UEDVT. During the course of anticoagulation, patients with unprovoked UEDVT had a higher rate of DVT recurrences (hazard ratio [HR]: 2.22; 95% CI: 1.37-3.43) and a similar rate of PE recurrences or major bleeding than those with unprovoked LEDVT. Patients with non-catheter-related provoked UEDVT had a similar outcome than those with provoked LEDVT. Among patients with UEDVT, those with non-catheter related unprovoked UEDVT had a lower rate of PE recurrences (HR: 0.06; 95% CI: 0-0.35) and major bleeding (HR: 0.20; 95% CI: 0.08-0.46) than those with catheter-related UEDVT or those with non-catheter related provoked UEDVT (HR: 0.10; 95% CI: 0.004-0.60; and 0.22; 95% CI: 0.08-0.52, respectively). On multivariable analysis, any difference had disappeared.
Conclusion:
During the course of anticoagulation, patients with UEDVT had a similar outcome than those with LEDVT. Among UEDVT patients, there were some differences according to the presence of catheter or additional risk factors for DVT. These differences disappeared after adjusting for potentially confounding variables.
Keywords
Introduction
Venous thromboembolism (VTE) represents a major cause of morbidity and mortality in the United States. 1 The Joint Commission and National Quality Forum monitors VTEs as one of its core performance measures, specifically VTE prophylaxis. 2 The occurrence of lower extremity deep vein thrombosis (LEDVT), averaging approximately 187 000 cases per year in the United States, is considered the third most frequent vascular disease after myocardial infarction and stroke. 3,4 Patients with acute LEDVT may have risk factors such as surgery, prolonged length of procedures, weakness or paresis, malignancy, and advanced age. Additionally, upper extremity deep vein thrombosis (UEDVT) is diagnosed in approximately 4% to 10% of all cases with deep vein thrombosis (DVT) and is categorized into noncatheter-related and catheter-related UEDVT. 5 –15 Noncatheter-related UEDVT may be idiopathic or associated with cancer, estrogen use, or other risk factors. Catheter-related UEDVT may be secondary to a catheter, a pacemaker, or a port system. Few studies have compared the natural history of patients with UEDVT (in terms of symptomatic VTE recurrences or major bleeding) during the course of anticoagulant therapy with those with LEDVT.
Registro Informatizado de Enfermedad TromboEmbólica (RIETE or in English: Computerized Registry of Patients with Venous Thromboembolism) is a multicenter, ongoing, international (Spain, Belgium, Czech Republic, France, Greece, Israel, Italy, Latvia, Republic of Macedonia, Switzerland, United States, Canada, Ecuador, and Venezuela) observational registry of consecutive patients with symptomatic, objectively confirmed, acute VTE. 16 –20 Since its inception in 2001, the design of the RIETE is to record data including the clinical characteristics, treatment patterns, and outcomes in patients diagnosed with VTE. Using the RIETE cohort, the present study compared the clinical characteristics, treatments, and outcome during the course of anticoagulation of patients with UEDVT versus those with LEDVT.
Patients and Methods
Inclusion Criteria
Consecutive patients with symptomatic, acute DVT or pulmonary embolism (PE) confirmed by objective tests (compression ultrasonography or contrast venography for DVT; helical computed tomography [CT] scan, ventilation–perfusion lung scintigraphy, or angiography for PE) were enrolled in RIETE. Patients were excluded if they were currently participating in a therapeutic clinical trial with a blinded therapy. All patients (or their legal power of attorney) provided written or oral consent for participation in the registry, in accordance with local ethics committee requirements. This analysis was approved by the ethics committee of the Hospital Universitari Germans Trias i Pujol (Badalona, Spain) and by the institutional review board of NorthShore University Health System (Evanston, Illinois).
Physicians participating in the RIETE registry made all efforts to enroll consecutive patients. Data were recorded onto a computer-based case report form at each participating hospital and submitted to a centralized coordinating center through a secure website. To ensure the validity of the information entered into the database, one of the specially trained monitors visited each participating hospital and compared information in 25 to 50 randomly chosen patient records with the information entered into the RIETE database. For data quality assessment, monitors assessed 4100 random records from all participating hospitals that included 1 230 000 measurements. These data showed a 95% overall agreement between the registered information and the patient records. The RIETE also used electronic data monitoring to detect inconsistencies or errors and attempted to resolve any discrepancies by contacting the local coordinators.
Study Design
We conducted a retrospective cohort study that used prospectively collected data from patients enrolled in the RIETE registry. All patients diagnosed with upper or lower DVT in RIETE were evaluated. Patients with UEDVT were then categorized by noncatheter-related unprovoked DVT, noncatheter-related provoked DVT, and catheter-related UEDVT. The major outcome was the rate of symptomatic VTE recurrences and major bleeding events appearing during the course of anticoagulant therapy. Bleeding events were classified as “major” if they were overt and required a transfusion of 2 U or more of blood, or were retroperitoneal, spinal, or intracranial, or when they were fatal. Fatal PE, in the absence of autopsy, was defined as any death appearing within the first 10 days after PE diagnosis, in the absence of any alternative cause of death. Fatal bleeding was defined as any death occurring within 10 days of a major bleeding episode, in the absence of an alternative cause of death.
Deep vein thrombosis was considered to be unprovoked in the absence of recent immobility, surgery, bone fracture, active cancer, estrogen use, pregnancy, puerperium, or long-term travel. Immobilized patients were defined as nonsurgical patients who had been immobilized (ie, total bed rest with bathroom privileges) for ≥4 days in the 2-month period prior to VTE. Surgical patients were defined as those who underwent a surgical intervention in the 2 months prior to VTE. Recent bleeding was defined as a major bleeding episode <30 days prior to VTE. Active cancer was defined as newly diagnosed cancer, metastatic cancer, or cancer that was being treated (ie, surgery, chemotherapy, radiotherapy, support therapy, or combined treatments).
Treatment and Follow-Up
Patients were managed according to the clinical practice of each participating hospital (ie, there was no standardization of treatment). Patients were followed up in the outpatient clinic or physician’s office. During each visit, any signs or symptoms suggesting VTE recurrences or bleeding complications were noted. Each episode of clinically suspected recurrent DVT or PE was investigated by repeat compression ultrasound, lung scanning, helical CT scan, or pulmonary angiography as appropriate. Most outcomes were classified as reported by the clinical centers. However, if staff at the coordinating center were uncertain how to classify a reported outcome, that event was reviewed by a central adjudicating committee (<10% of events).
Statistical Analysis
Categorical variables were compared using χ2 test (2-sided) and Fisher exact test (2-sided). Odds ratios (ORs) and corresponding 95% confidence intervals (CIs) were calculated, and a P value <.05 was considered to be statistically significant. The association between the type of DVT and outcome was assessed using the Cox proportional hazards regression model, estimated by a forward step method. All variables achieving a significance level of ≤.1 on univariable analysis were considered for inclusion in the logistic regression model. Statistical analyses were conducted with SPSS for Windows Release 17.0 (SPSS, Inc, Chicago, Illinois, USA).
Role of the Funding Source
The funders of the study had no role in study design, data collection, data analysis, data interpretation, or writing of the report. The corresponding author had full access to all the data in the study and had final responsibility for the decision to submit for publication.
Results
As of August 2015, 37 366 patients with acute DVT had been enrolled in RIETE. Of these, 35 094 (94%) had LEDVT, 1334 (3.6%) had noncatheter-related UEDVT, and 938 (2.5%) had catheter-related UEDVT. Among patients with noncatheter-related UEDVT, 672 (50%) had unprovoked and 662 had provoked UEDVT.
Unprovoked UEDVT Versus Unprovoked LEDVT
Patients with unprovoked UEDVT were 19 years younger than those with unprovoked LEDVT and less likely to have chronic lung disease, anemia, renal insufficiency, or prior VTE (Table 1). At baseline, they less likely presented with symptomatic PE concomitantly (OR: 0.37; 95% CI: 0.29-0.47). Most patients in both subgroups (85% and 91%, respectively) received initial therapy with low-molecular-weight heparin (LMWH), at similar doses. Then, most (74% and 81%) switched to vitamin K antagonists (VKAs) for long-term therapy. During the course of anticoagulant therapy, patients with unprovoked UEDVT had a higher rate of DVT recurrences than those with unprovoked LEDVT (hazard ratio [HR]: 2.22; 95% CI: 1.37-3.43) and a similar rate of PE recurrences or major bleeding.
Clinical Characteristics, Treatment, and Outcome in Patients With Unprovoked UEDVT Versus in Those With Unprovoked LEDVT.
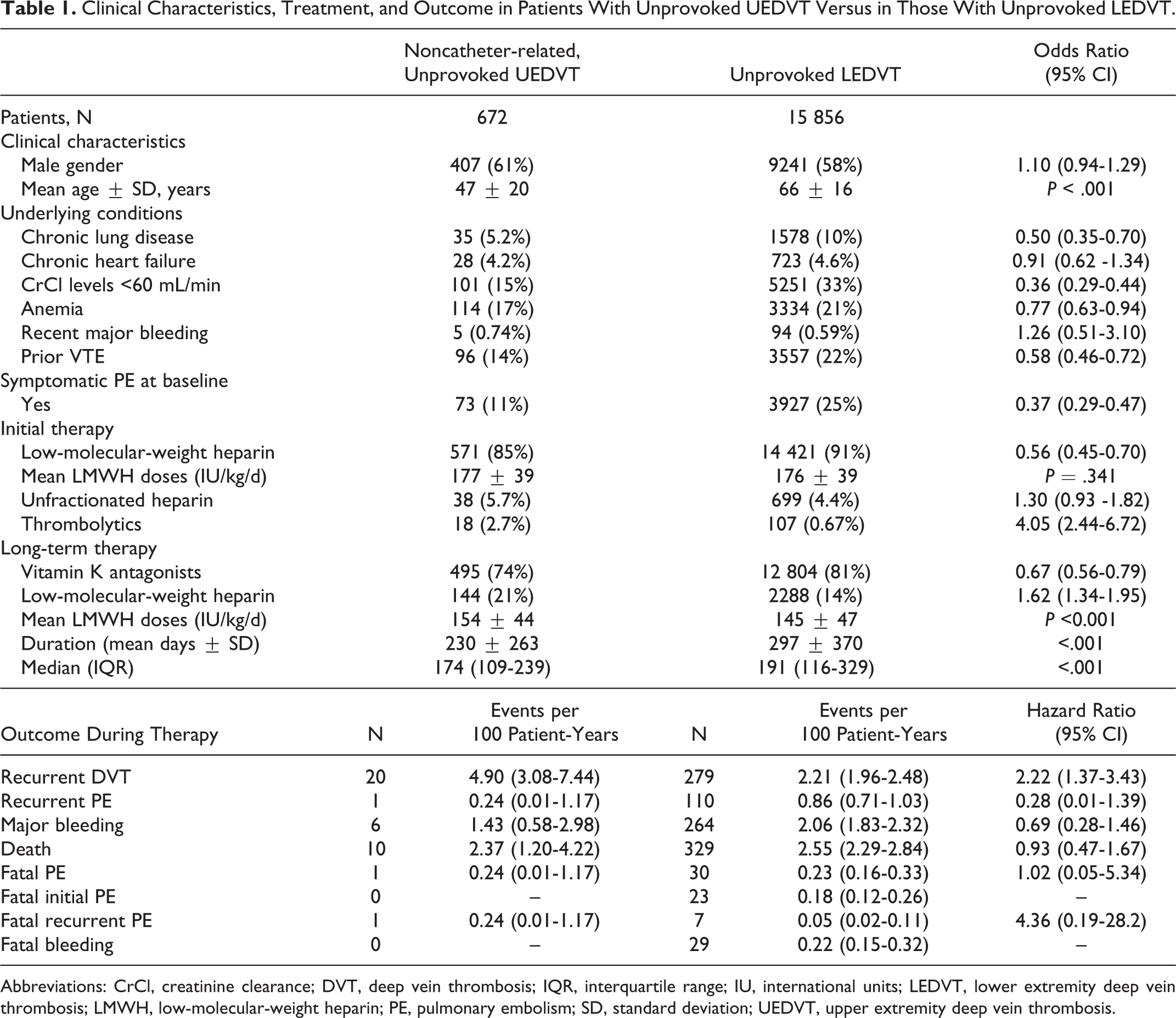
Abbreviations: CrCl, creatinine clearance; DVT, deep vein thrombosis; IQR, interquartile range; IU, international units; LEDVT, lower extremity deep vein thrombosis; LMWH, low-molecular-weight heparin; PE, pulmonary embolism; SD, standard deviation; UEDVT, upper extremity deep vein thrombosis.
Noncatheter-Related, Provoked UEDVT Versus Provoked LEDVT
Compared with patients with provoked LEDVT, those with noncatheter-related provoked UEDVT were 8 years younger and less likely to have renal insufficiency, recent surgery, or recent immobility but more likely to have active cancer or use of estrogens (Table 2). At baseline, patients with noncatheter-related provoked UEDVT less likely presented with symptomatic PE than those with provoked LEDVT (HR: 0.47; 95% CI: 0.38-0.59). Most patients in both subgroups (89% and 91%, respectively) treated with LMWH, but only 38% of patients with UEDVT and 55% of those with LEDVT switched to VKA for long-term therapy. During the course of anticoagulant therapy, patients with noncatheter-related provoked UEDVT had a similar rate of PE recurrences, DVT recurrences, or major bleeding than those with provoked LEDVT.
Clinical Characteristics, Treatment, and Outcome in Patients With Noncatheter-Related, Provoked UEDVT Versus Those With Provoked LEDVT.
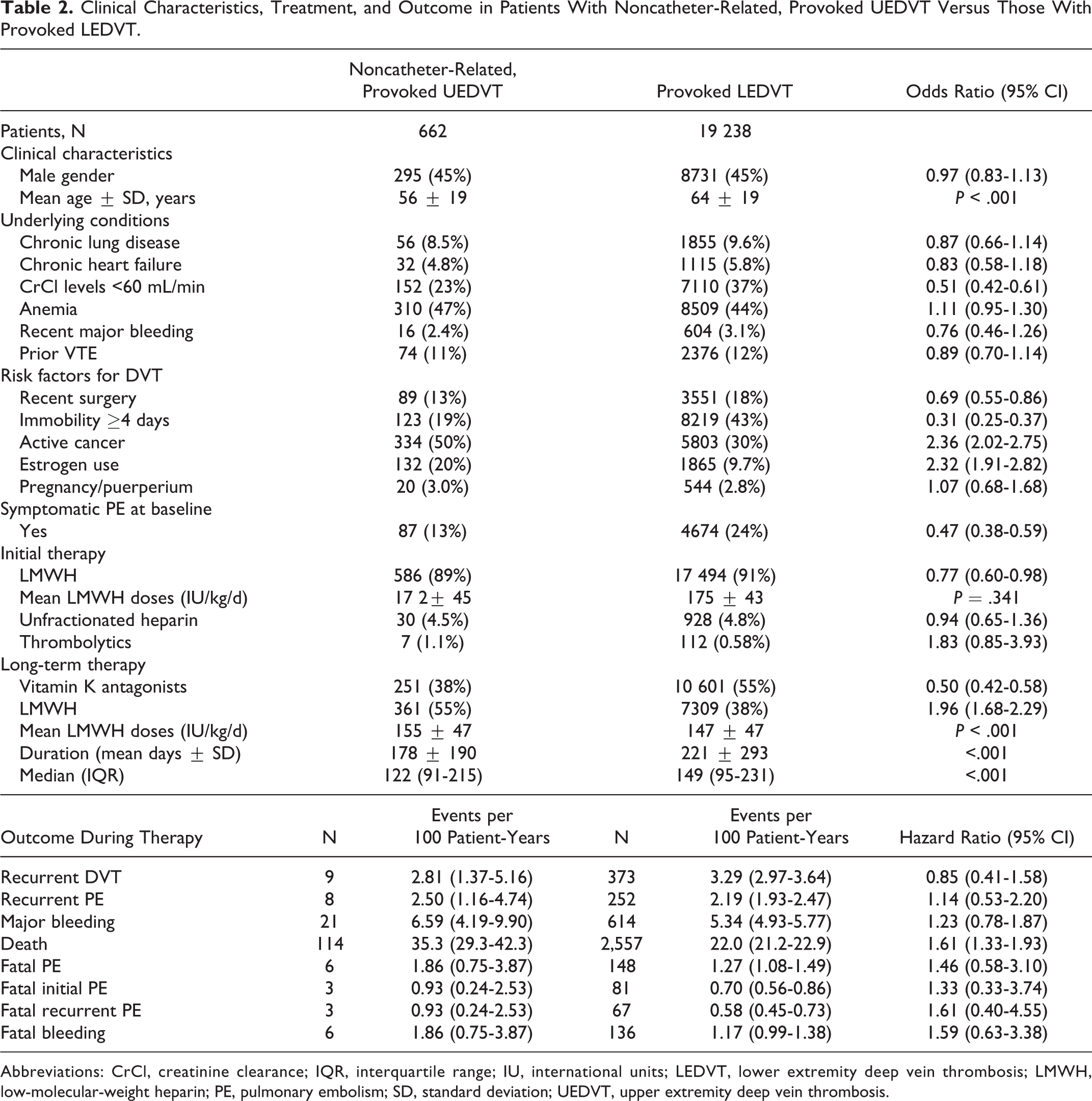
Abbreviations: CrCl, creatinine clearance; IQR, interquartile range; IU, international units; LEDVT, lower extremity deep vein thrombosis; LMWH, low-molecular-weight heparin; PE, pulmonary embolism; SD, standard deviation; UEDVT, upper extremity deep vein thrombosis.
Differences Between Patients With UEDVT
Patients with catheter-related UEDVT (central line 386, peripheral line 93, port system 243, pacemaker 91, other 10) were significantly older than those with unprovoked UEDVT and younger than those with noncatheter-related, provoked UEDVT (Table 3). They were most likely to have underlying diseases such as chronic lung disease or heart failure, renal insufficiency, anemia, or recent major bleeding than those with noncatheter-related UEDVT. At baseline, patients with catheter-related UEDVT were less likely to present with concomitant PE than the other 2 subgroups (Table 3). During the course of anticoagulant therapy, patients with noncatheter-related unprovoked UEDVT had a lower rate of PE recurrences (HR: 0.06; 95% CI: 0-0.35) and major bleeding (HR: 0.20; 95% CI: 0.08-0.46) than those with catheter-related UEDVT or those with noncatheter-related provoked UEDVT (HR: 0.10, 95% CI: 0.004-0.60 and 0.22, 95% CI: 0.08-0.52, respectively). On multivariable analysis, any difference in outcome between subgroups had disappeared and could be explained by differences in the proportion of patients with cancer, concomitant diseases, or risk factors.
Clinical Characteristics, Treatment, and Outcome in Patients With Catheter-Related Versus in Those With Noncatheter-Related Upper Extremity DVT.a
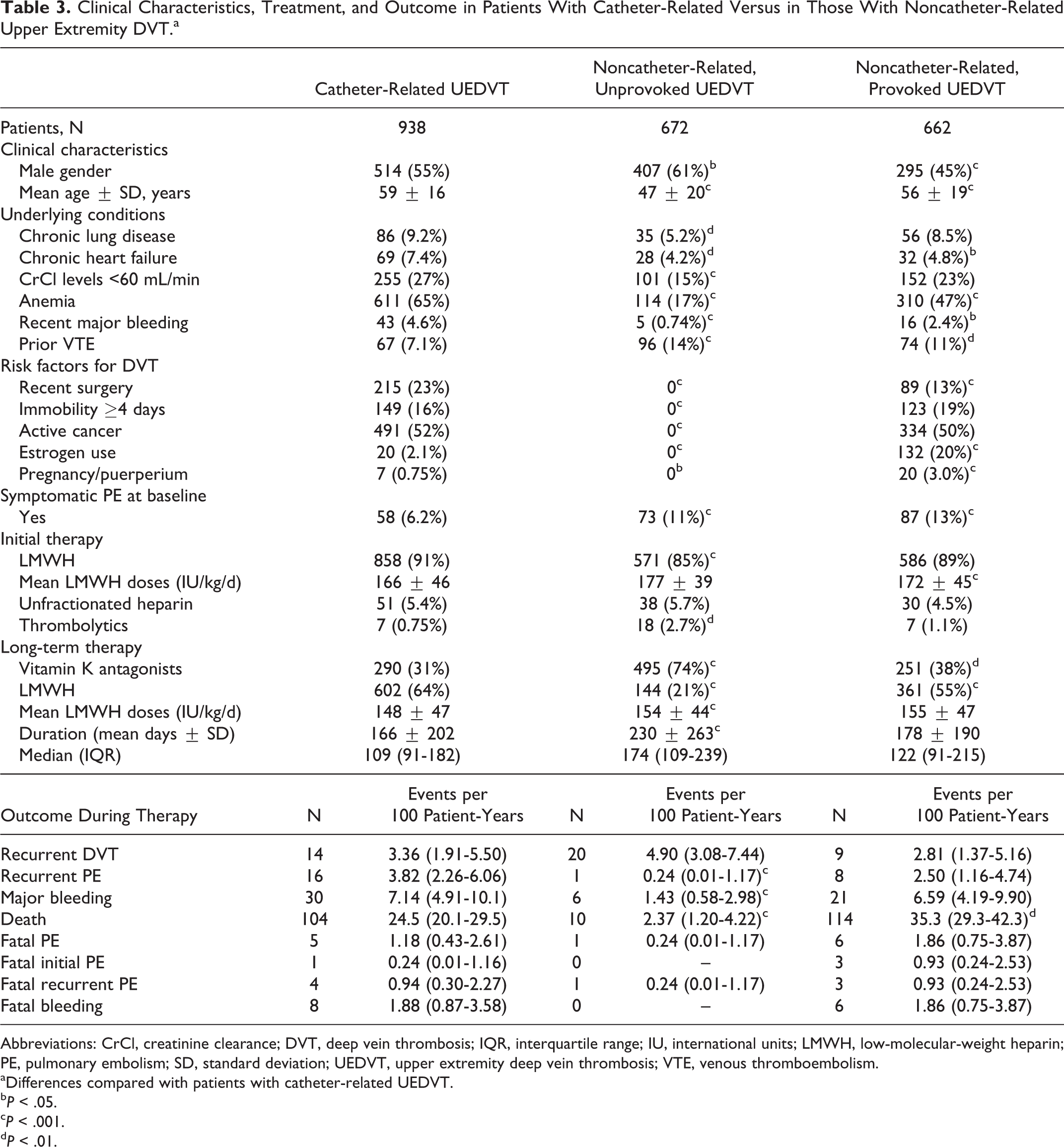
Abbreviations: CrCl, creatinine clearance; DVT, deep vein thrombosis; IQR, interquartile range; IU, international units; LMWH, low-molecular-weight heparin; PE, pulmonary embolism; SD, standard deviation; UEDVT, upper extremity deep vein thrombosis; VTE, venous thromboembolism.
aDifferences compared with patients with catheter-related UEDVT.
b P < .05.
c P < .001.
d P < .01.
Discussion
Traditionally, patients with UEDVT have been considered at low risk to develop PE. However, no studies have consistently compared the outcome during the course of anticoagulation in patients with UEDVT versus LEDVT. Our data, obtained from a large series of consecutive patients with acute DVT, confirm that patients with UEDVT less likely presented with PE at baseline than those with LEDVT (9.8% vs 25%, respectively), as already reported. Interestingly, however, the rate of PE recurrences appearing during the course of therapy in patients with UEDVT was not inferior to the rate in those with LEDVT. In fact, among patients with catheter-related UEDVT, the rate was higher, although according to the results of the multivariable analysis, it may not be attributed to the DVT itself but to potentially confounding variables, particularly cancer. In our series, 1 in every 3 patients with UEDVT developing PE recurrences (8 of 25, 32%) died of recurrent PE. Thus, its clinical relevance should not be underestimated.
Current guidelines of anticoagulant therapy recommend the same initial and long-term therapy for patients with LEDVT or UEDVT. 21,22 In our experience, patients with noncatheter-related UEDVT were much younger than those with LEDVT, less likely to have chronic lung disease or renal insufficiency, and less often presented with PE at baseline. On the contrary, over half (58%) of the patients with catheter-related UEDVT had cancer, and 65% had anemia. We might have expected that their outcome during therapy would have been different. If proven, we might have suggested that they would probably benefit from different anticoagulant strategies (in intensity or duration). However, we failed to find any difference in outcome after adjusting for potentially confounding variables. Thus, our data confirm that there seem to be no reason for treating patients with UEDVT differently.
There are several limitations to the present study. First, it was a retrospective analysis of patients who were recruited consecutively, thereby subject to possible selection bias. Second, there was not a standardized type of anticoagulation treatment in our cohort. Therefore, it is unknown as to which type and dose of various treatments is the most efficacious in the reduction or causation of events such as recurrent VTE or bleeding. Third, patients in the RIETE database were selected in several different countries. The variability of practices in different countries could potentially affect the study’s outcomes. For instance, the dosing and timing of pharmacologic treatment can vary according to each individual country’s pattern of practice, underlying disease process, and/or presence or absence of a diagnosed malignancy. In addition, there may have been variables that were unaccounted for in our multivariate analysis. Finally, a variety of practitioners entered data into the registry, which may lend itself to potential inaccuracies in the data being reported.
In summary, after comparing LEDVT patients to those with UEDVT, we found a number of differences in clinical characteristics, underlying diseases, initial presentation, and therapeutic strategies. However, their outcome during the course of anticoagulant therapy was similar. We also found significant differences in clinical characteristics between the 3 subgroups of patients with UEDVT, but any difference in outcome had disappeared on multivariable analysis. This study suggests that practitioners may be able to treat those patients with UEDVT in a similar manner to those with LEDVT. Further prospective trials, however, are needed to clarify this notion.
Footnotes
Appendix A
Coordinator of the RIETE Registry: Manuel Monreal. RIETE Steering Committee Members: Hervé Decousus, Paolo Prandoni, and Benjamin Brenner. RIETE National Coordinators: Raquel Barba (Spain), Pierpaolo Di Micco (Italy), Laurent Bertoletti (France), Inna Tzoran (Israel), Abilio Reis (Portugal), Marijan Bosevski (R. Macedonia), Henri Bounameaux (Switzerland), Radovan Malý (Czech Republic), Philip Wells (Canada), and Manolis Papadakis (Greece). RIETE Registry Coordinating Center: S & H Medical Science Service.
Members of the RIETE Group: Spain: M. A. Aibar, V. Andújar, J. I. Arcelus, M. Arroyo-Cozar, A. Ballaz, R. Barba, M. Barrón, B. Barrón-Andrés, J. Bascuñana, A. Blanco-Molina, I. Casado, A. Culla, J. de Miguel, J. del Toro, J. A. Díaz-Peromingo, C. Falgá, C. Fernández-Capitán, C. Font, L. Font, P. Gallego, F. García-Bragado, P. Garcia-Brotons, V. Gómez, J. González, E. Grau, M. Guil, L. Guirado, J. Gutiérrez, L. Hernández-Blasco, L. Jara-Palomares, M. J. Jaras, D. Jiménez, R. Jiménez, B. Lacruz, R. Lecumberri, J. L. Lobo, L. López-Jiménez, R. López-Reyes, J. B. López-Sáez, M. A. Lorente, A. Lorenzo, O. Madridano, P. J. Marchena, J. M. Martín-Antorán, M. Martín, F. Martín-Martos, M. Monreal, M. V. Morales, F. J. Muñoz, D. Nauffal, J. A. Nieto, M. J. Núñez, M. Odriozola, S. Otalora, R. Otero, B. Pagán, J. M. Pedrajas, M. L. Peris, J. A. Porras, A. Rivas, L. Ramírez, M. A. Rodríguez-Dávila, V. Rosa, P. Ruiz-Artacho, N. Ruiz-Giménez, R. Ruiz-Ferreras, A. Sampériz, R. Sánchez, T. Sancho, O. Sanz, S. Soler, J. M. Suriñach, G. Tiberio, C. Tolosa, J. Trujillo-Santos, F. Uresandi, B. Valero, R. Valle, J. Vela, G. Vidal, C. Vilar, and J. Villalta; Belgium: T. Vanassche and P. Verhamme; Canada: P. Wells; Czech Republic: J. Hirmerova, R. Malý, Ecuador: E. Salgado, France: L. Bertoletti, A. Bura-Riviere, D. Farge-Bancel, A. Hij, I. Mahé, A. Merah, I. Quere; Greece: D. Babalis, M. Papadakis, I. Tzinieris; Israel: A. Braester, B. Brenner, I. Tzoran; Italy: A. Apollonio, G. Barillari, A. Bertone, F. Biolora, M. Ciammaichella, D. De Blasis, P. Di Micco, P. Ferrazzi, G. Lessiani, C. Lodigiani, D. Mastroiacovo, F. Pace, R. Pesavento, M. Pinelli, P. Prandoni, L. Rota, D. Tonello, A. Tufano, A. Visonà, and B. Zalunardo; Latvia: E. Drucka, D. Kigitovica, and A. Skride; Republic of Macedonia: M. Bosevski and M. Zdraveska; Switzerland: A. Alatri, H. Bounameaux, L. Calanca, L. Mazzolai; United States: J. A. Caprini; Venezuela: J. C. Serrano.
Acknowledgments
The authors express gratitude to Sanofi Spain for supporting this Registry with an unrestricted educational grant. The authors also express gratitude to Bayer Pharma AG for supporting this Registry. Bayer Pharma AG’s support was limited to the part of RIETE outside Spain, which accounts for a 22.4% of the total patients included in the RIETE Registry. The authors also thank the RIETE Registry Coordinating Center and S & H Medical Science Service, for their quality control data, logistic, and administrative support.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
