Abstract
Background:
In patients with venous thromboembolism (VTE), the study of the case fatality rate (CFR) of VTE recurrences and bleeding complications may be of help to balance the risks and benefits of anticoagulant therapy.
Objective:
To investigate the CFR with the direct oral anticoagulants (DOACs; dabigatran, rivaroxaban, apixaban, and edoxaban) in patients with VTE.
Methods:
We conducted a systematic review and meta-analysis of randomized clinical trials testing the DOACs versus standard initial treatment of VTE (parenteral anticoagulant for ≥5 days plus vitamin K antagonists [VKAs] for ≥3 months) and DOACs versus placebo or VKA for extended treatment. Two investigators independently extracted the data. A random effects meta-analysis was conducted using StatsDirect software.
Results:
Overall, 10 trials in 35 029 patients were included. During initial treatment, the rate of recurrent VTE per 100 patient-years (%/yr) and CFR (%) was similar in patients receiving DOACs or standard therapy (4.1%/yr vs 4.4%/yr;
Conclusion:
The use of DOACs was associated with fewer major and fatal bleedings and corresponding CFR than standard initial treatment of VTE, and fewer recurrent VTEs and mortality than placebo during extended therapy, although the CFR of recurrent VTE was not reduced.
Keywords
Introduction
Venous thromboembolism (VTE), including deep vein thrombosis (DVT) and pulmonary embolism (PE), is associated with significant morbidity and mortality. 1 Traditionally, guidelines of antithrombotic therapy have recommended to treat patients with acute VTE with a parenteral anticoagulant for approximately 1 week overlapping with and followed by an oral vitamin K antagonist (VKA; dose-adjusted to keep an international normalized ratio [INR] between 2.0 and 3.0) for at least 3 to 6 months (initial treatment). 2 At this point, the decision to continue with anticoagulant treatment (extended treatment) depends on the balance between the risk of VTE recurrences if anticoagulation is withdrawn and the risk of bleeding if it is continued. Patients with ongoing risk factors and patients with unprovoked VTE are often prescribed extended treatment provided that the bleeding risk is not excessive. 2
In recent years, several direct oral anticoagulants (DOACs) have been developed for the treatment of VTE, among other indications. The DOACs inhibit either thrombin 3 or activated factor Xa (FXa). 4 –6 They do not need parenteral administration, have less food or drug interactions than VKA, and can be given in fixed doses once or twice daily. 2
The case fatality rate (CFR) is defined as the proportion of patients who die as a consequence of a particular condition. This estimate, together with the absolute rates of recurrent VTE and bleeding, provides useful information to balance the risks and benefits of anticoagulant therapy. 7 The CFR reported during standard (3-6 months) anticoagulant treatment is approximately 11% for both recurrent VTE and major bleeding. 7 However, the CFR of recurrent VTE decreases over time during anticoagulation, while the CFR of major bleeding remains more stable. 8
As the DOACs and VKA have different mechanisms of action and the DOACs lack specific antidotes available, the CFR of recurrent VTE and bleeding may differ. Therefore, additional details above benefit/risk, including the estimation of the CFR of recurrent VTE and major bleeding by type of anticoagulant, could be helpful in refining therapeutic decision making with respect to the type and duration of anticoagulant therapy.
We conducted a systematic review and meta-analysis to assess the CFR of recurrent VTE and major bleeding events during anticoagulation with DOACs and compared them with the CFRs during initial standard treatment (parenteral anticoagulant plus VKA) and extended treatment (VKA or placebo) in patients with VTE.
Methods
We considered all randomized controlled trials comparing any DOAC (dabigatran, rivaroxaban, apixaban, or edoxaban) with standard treatment of VTE (eg, initial treatment with a parenteral anticoagulant plus an oral VKA [dose adjusted to maintain an INR between 2.0 and 3.0]) or comparing DOACs with VKA or placebo in the extended treatment of VTE in patients who had received anticoagulation therapy for at least 3 months.
Trial Identification and Data Collection
We searched MEDLINE and CENTRAL (up to February 1, 2014), clinical trial registries, relevant conference proceedings, and Web sites of regulatory agencies (see supplemental material for search strategy). No language restrictions were applied. Two investigators (AG-O and RL) independently and separately assessed trials for eligibility and extracted data. If a trial was covered in more than 1 report, we used a hierarchy of data sources, namely, public reports from regulatory authorities (US Food and Drug Administration, European Medicines Agency), peer-reviewed articles, reports from the Web-based repository for results of clinical studies, and other sources.
Study Characteristics and Quality
We collected data on patients’ characteristics (age and sex), percentage of patients evaluable for efficacy and safety, dosage used in the experimental and control groups, duration of treatment and follow-up, inclusion and exclusion criteria, and definitions of outcomes. We assessed study quality using the Cochrane Collaboration’s tool for assessing risk of bias in randomized studies. 9
Outcome Measures
We extracted data on the rate of symptomatic VTE recurrences (recurrent or new episode of symptomatic DVT or symptomatic PE, including fatal events) and major bleeding per study definition. The CFR of recurrent VTE was defined as the proportion of all recurrent VTE events resulting in death. 2 The CFR of a major bleeding event was defined as the proportion of all major bleeding events causing or contributing to death.
We also calculated (1) the case PE rate, defined as the proportion of all recurrent VTE events that corresponded to PE (nonfatal or fatal); (2) the case critical bleeding rate, defined as the proportion of all major bleeding events that were fatal or contributed to death (regardless localization) or were nonfatal but occurred in a critical site (eg, intracranial, intraspinal, intraocular, retroperitoneal, intraarticular, pericardial, or intramuscular with compartment syndrome); and (3) the rates of major bleeding depending on the sites of bleeding (intracranial vs extracranial), and corresponding CFR.
Statistical Analysis
We carried out direct comparisons between the effects of DOACs and standard treatment or placebo on an intention-to-treat basis, according to Preferred Reporting Items for Systematic reviews and Meta-Analyses (PRISMA) recommendations. 10 The analysis was made separately for studies focused on initial treatment and those on extended treatment of VTE.
The meta-analysis was conducted within treatment groups to obtain the average rate of events in each group and corresponding 95% confidence interval (CI; proportion meta-analysis) as well as between groups to obtain relative risks (RRs) and corresponding 95%CI (RR meta-analysis). To estimate the weighed rates and 95%CIs for the review’s primary outcomes, individual study estimates were converted to rates per patient-year of follow-up on the basis of the mean (or median) reported follow-up. Rates were transformed via the Freeman-Tukey double arcsine method
11
and were pooled performing an inverse variance-weighted random effects meta-analysis by conventional methods, as described by DerSimonian and Laird.
12
Heterogeneity was assessed using the Cochran Q test
13
and the Higgins I2 test.
14
A Cochran Q
Results
Search Results
The literature search identified 2987 articles, of which 300 were related to clinical trials or protocols using DOACs (Figure 1). Of these, 21 studies corresponded to treatment of VTE and were selected for further checking. We included 6 clinical trials on initial treatment of VTE with direct thrombin inhibitors (RE-COVER I and II) and FXa inhibitors (EINSTEIN-DVT, EINSTEIN-PE, AMPLIFY, and HOKUSAI-VTE) 16 –21 as well as 4 studies on extended treatment of VTE with direct thrombin inhibitors (RE-MEDY and RE-SONATE) 22 and FXa inhibitors (EINSTEIN-Extension and AMPLIFY-EXT). 18,23 Data from publications were complemented with 6 additional references to these studies from companies’ trial registries and public reports from regulatory authorities. 24 –32 We excluded from the main analysis 2 trials with ximelagatran (THRIVE and THRIVE III), 33,34 as the compound was withdrawn from the market on 2006, but we included them in one of the sensitivity analyses.

Study identification, selection, and exclusions.
Characteristics of Trials, Patients, Treatments, and Outcomes Measures
The 10 studies comprised 35 029 patients. Table 1 shows the characteristics of the trials, patients, and treatments. Of them, 6 studies focused on the initial treatment of VTE for at least 3 months (range: 3-12 months; Table 1). Mean patients’ age ranged between 55 and 58 years, with a predominance of male gender. Patients were excluded if they had contraindications to anticoagulation with heparin or VKA, if they had a PE with hemodynamic unstability, or if they received treatment with a thrombolytic agent, thrombectomy, or a vena cava filter for the index episode of VTE. All 4 studies focused on extended treatment enrolled patients who had completed at least 3 months of anticoagulation for VTE. Extended treatment of VTE was planned for 6 to 18 months (Table 1). Participants in the 3 placebo-controlled extension studies (RE-SONATE, EINSTEIN-Extension, and AMPLIFY-EXT) were included if they had not developed a symptomatic recurrence during prior anticoagulant therapy and if there was clinical equipoise about the continuation or cessation of anticoagulant therapy. On the contrary, participants in the single active-control study (RE-MEDY) were considered to be at increased risk of recurrent VTE on the basis of the site investigator’s assessment, thus comprising a high-risk population for VTE recurrence in which continued anticoagulant treatment was indicated.
Characteristics of Phase III VTE Treatment Studies With Direct Oral Anticoagulants (DOAC).
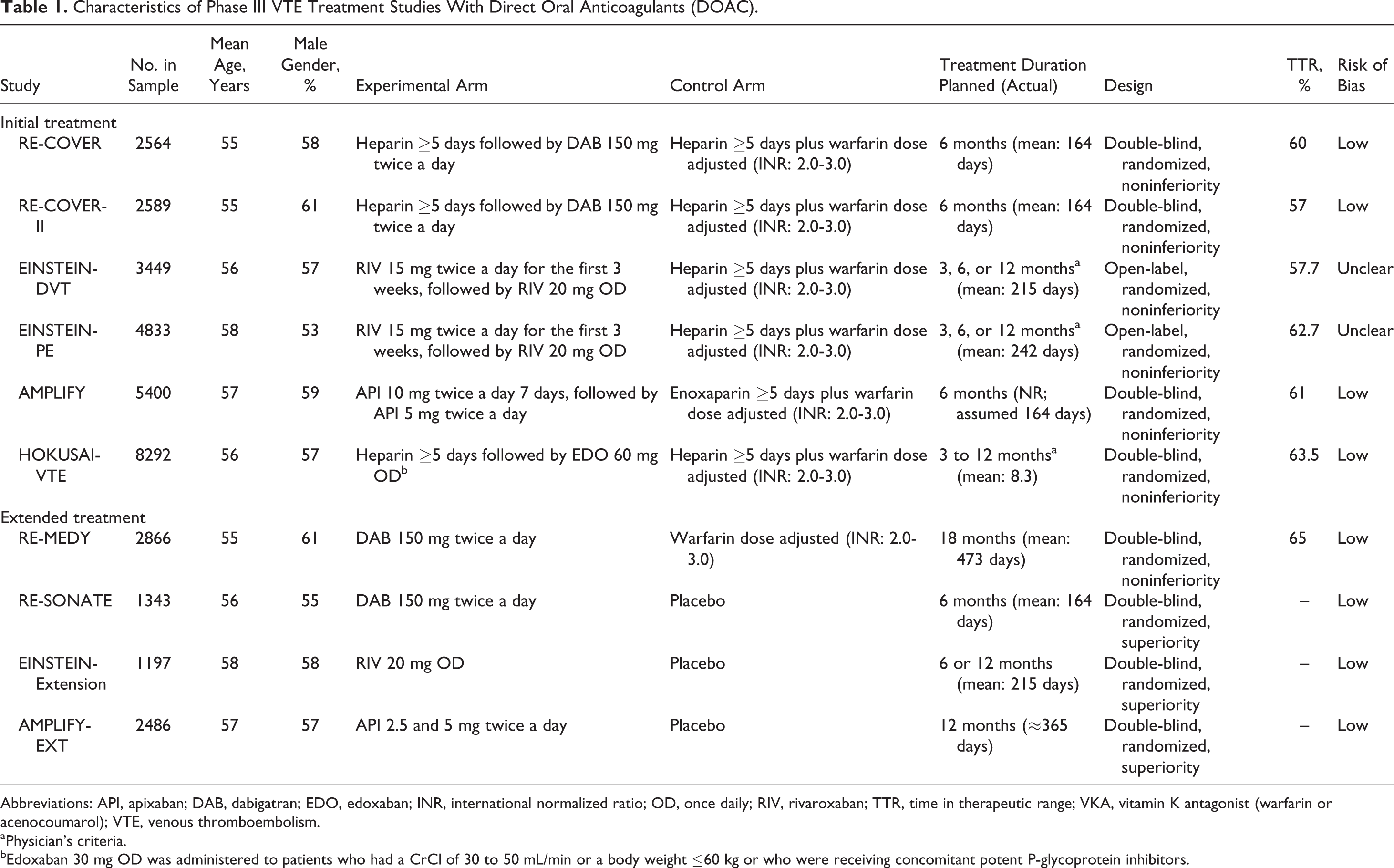
Abbreviations: API, apixaban; DAB, dabigatran; EDO, edoxaban; INR, international normalized ratio; OD, once daily; RIV, rivaroxaban; TTR, time in therapeutic range; VKA, vitamin K antagonist (warfarin or acenocoumarol); VTE, venous thromboembolism.
aPhysician’s criteria.
bEdoxaban 30 mg OD was administered to patients who had a CrCl of 30 to 50 mL/min or a body weight ≤60 kg or who were receiving concomitant potent P-glycoprotein inhibitors.
The experimental groups of studies with rivaroxaban and apixaban did not include initial therapy with a parenteral anticoagulant (single-drug approach). On the contrary, clinical trials with dabigatran and edoxaban required a lead-in treatment period with a parenteral anticoagulant (Table 1).
In all studies, DVT was diagnosed by compression ultrasonography or venography of leg veins, while PE was diagnosed with ventilation–perfusion lung scanning, angiography, or spiral computed tomography of pulmonary arteries. Fatal VTE was defined as death due to PE documented by objective diagnostic testing, autopsy, or death which could not be attributed to a documented cause and for which PE could not be ruled out (unexplained death). Major bleeding definitions used in all trials were consistent with the 2005 major bleeding definition of the International Society of Thrombosis and Hemostasis (ISTH; overt bleeding associated with a decrease in hemoglobin of 2 g/dL or more, or requiring a transfusion of 2 or more units of blood, or occurring in a critical site, or contributing to death). 35 Fatal bleeding was defined as major bleeding contributing to death. Suspected recurrent VTE events, major bleedings, and deaths were adjudicated centrally by independent committees in a blinded fashion.
The percentage of time that the INR was within therapeutic range (2.0-3.0; TTR) ranged between 57% and 65% across studies (Table 1).
The risk of bias was assessed as “low” in the 8 double-blind studies (Table 1). The risk was assessed as “unclear” in the EINSTEIN-DVT and PE studies 18,19 because they used an open-label design but the reports included appropriate information about random sequence generation, allocation concealment, handling of missing data, and blinded outcome assessment by a central adjudication committee. The full numbers of assessable patients, follow-up periods, and events in each trial are included in Supplementary Table S1.
Case Fatality Rate of Recurrent VTE Events
During initial treatment, the rate of VTE recurrences was similar in patients receiving DOACs or standard therapy (4.1%/yr vs 4.4%/yr; RR, 0.91; 95%CI, 0.79-1.05;
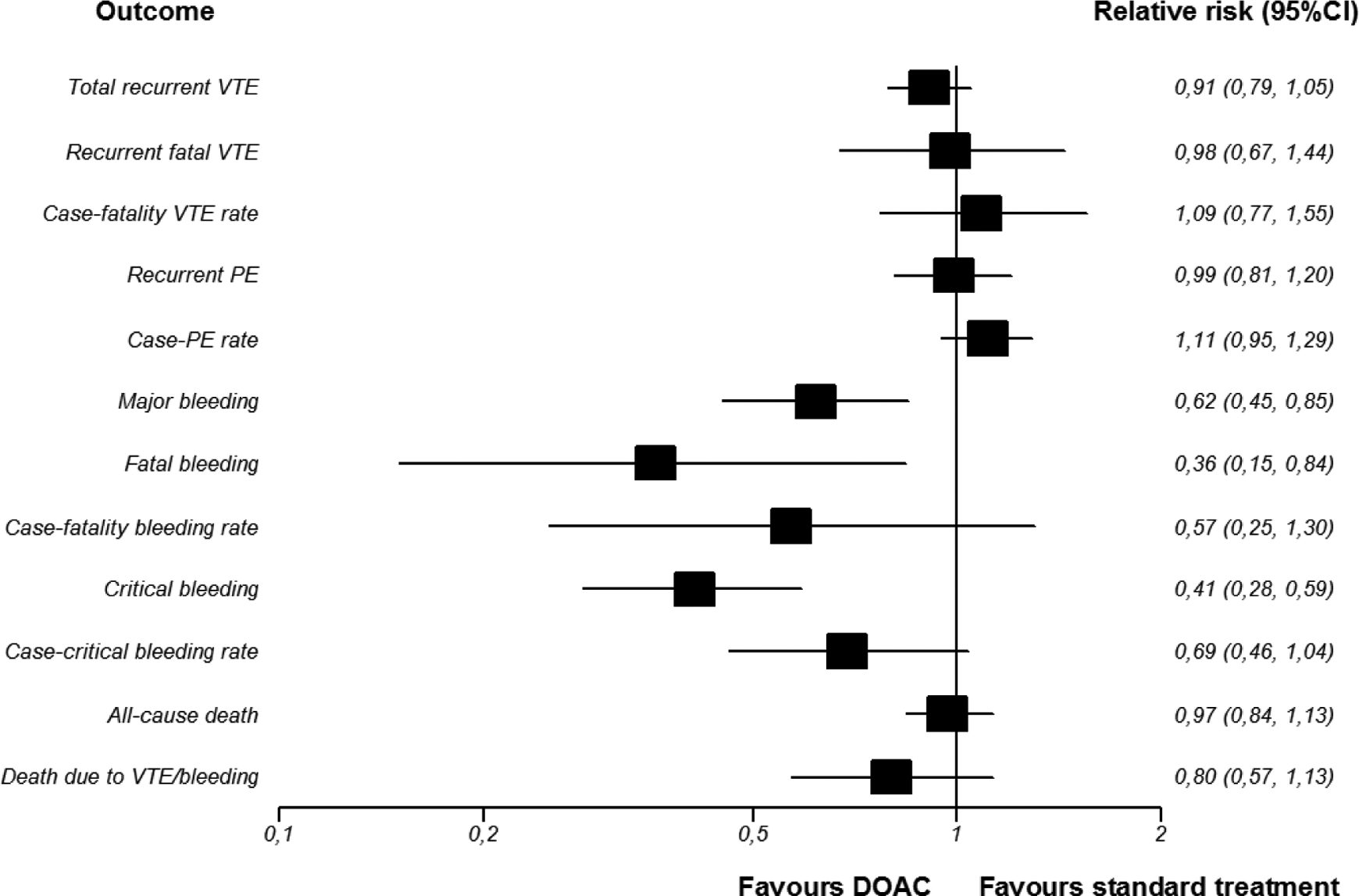
Forest plots of outcomes during initial treatment (DOAC vs standard treatment) (RRs, 95% confidence intervals). Random effects (DerSimonian and Laird). Heterogeneity VTE: total recurrent VTE I2 = 0%,
Recurrent VTE, Major Bleeding, and Death During Initial Treatment of VTE.
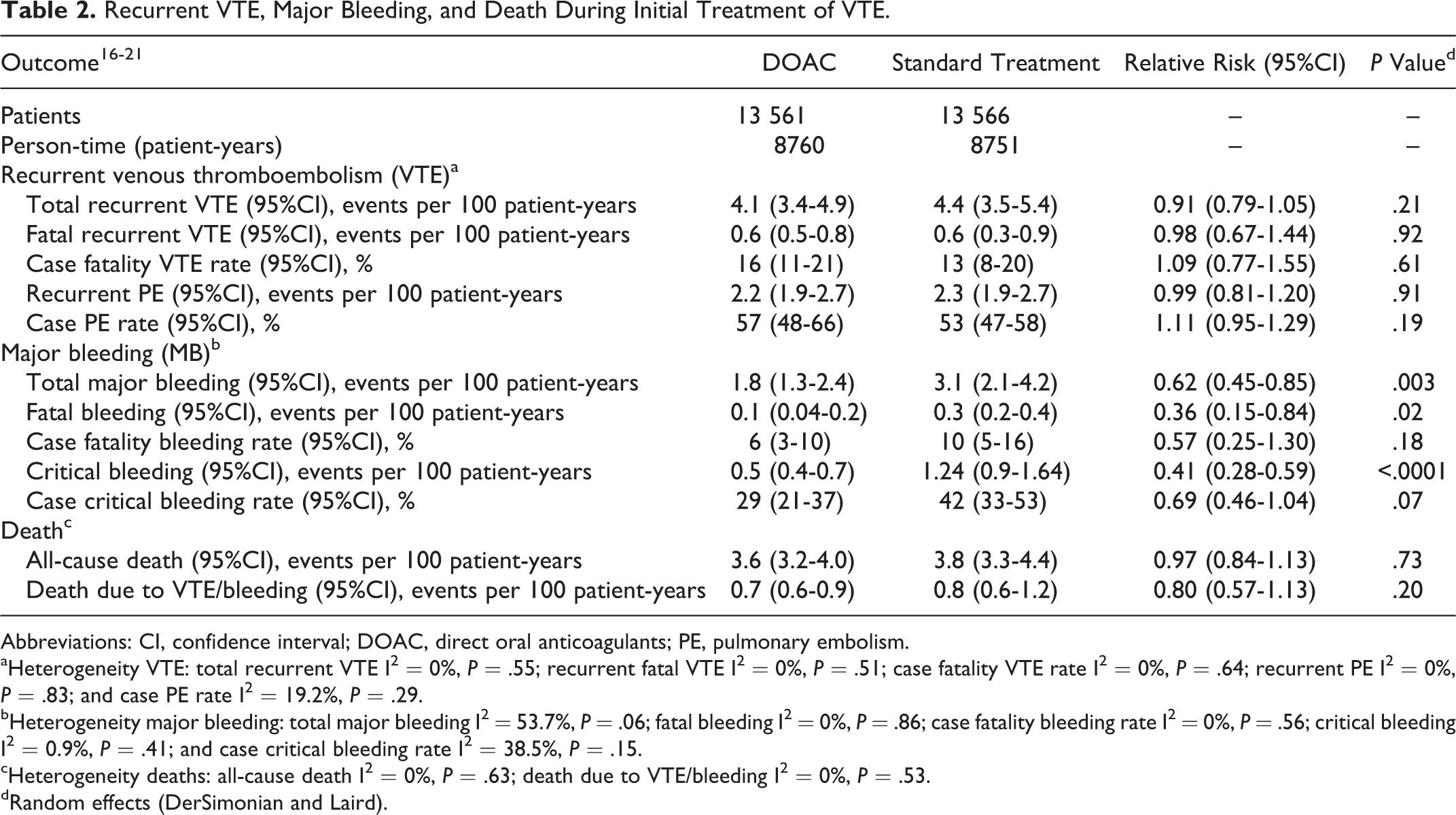
Abbreviations: CI, confidence interval; DOAC, direct oral anticoagulants; PE, pulmonary embolism.
aHeterogeneity VTE: total recurrent VTE I2 = 0%,
bHeterogeneity major bleeding: total major bleeding I2 = 53.7%,
cHeterogeneity deaths: all-cause death I2 = 0%,
dRandom effects (DerSimonian and Laird).
During extended treatment, the rate of total recurrent VTE events was significantly lower in patients receiving DOACs than in those with placebo (1.9%/yr vs 10.9%/yr; RR, 0.19; 95%CI, 0.13-0.27;

Forest plots of outcomes during extended therapy (DOAC vs placebo; RRs, 95% confidence intervals). Random effects (DerSimonian and Laird). No fatal bleedings were reported in extended therapy studies comparing the DOAC with placebo, and only 1 study reported at least 1 major or critical bleeding in the placebo group. Therefore, RRs of fatal and critical bleedings as well as case fatality and case critical bleeding rates could not be calculated. Heterogeneity VTE: total recurrent VTE I2 = 3.5%,
Recurrent VTE, Major Bleeding, and Death in the Extended Treatment of VTE.

Abbreviations: CI, confidence interval; DOAC, direct oral anticoagulants; PE, pulmonary embolism.
aHeterogeneity VTE: total recurrent VTE I2 = 3.5%,
bHeterogeneity major bleeding: total major bleeding I2 = 62.2%,
cHeterogeneity deaths: all-cause death I2 = 0%,
dRandom effects (DerSimonian and Laird).
eNo fatal bleedings were reported in extended treatment studies comparing the DOAC with placebo, and only 1 study reported at least 1 major or critical bleeding in the placebo group. Therefore, relative risks of fatal and critical bleedings as well as case fatality and case critical bleeding rates could not be calculated.
Only 1 study compared DOACs with warfarin for the extended treatment of VTE (RE-MEDY). The rate of total recurrent VTE events was similar (1.4%/yr vs 1.0%/yr; RR, 1.44; 95%CI, 0.76-2.80;
Case Fatality Rate of Major Bleeding Events
During the course of initial treatment for VTE, the rate of major bleeding events was lower with DOACs than with standard treatment (1.8%/yr vs 3.1%/yr; RR, 0.62; 95%CI, 0.45-0.85;
With respect to bleeding site, intracranial bleeding (ICB) accounted for 11% of all major bleedings occurring with the DOAC and 18% of all major bleedings associated with standard treatment. The remaining major bleedings were extracranial. Compared to standard treatment, the DOAC reduced both the risk of ICB (0.2%/yr vs 0.5%/yr; RR, 0.37; 95%CI, 0.21-0.68) and the risk of extracranial bleeding (1.6%/yr vs 2.7%/yr; RR, 0.66; 95%CI, 0.44-0.98). Adjusted CFRs of bleeding were significantly higher in those patients who had ICB than in those who had a major extracranial bleeding (19% vs 5%; RR, 4.17; 95%CI, 2.07-8.38), regardless of the type of anticoagulation.
During extended therapy, the rates of major bleeding events were low with both, DOACs and placebo (0.6%/yr vs 0.2%/yr), and there was heterogeneity across trials (I2 = 62.2%;
In the single comparison between dabigatran and warfarin for secondary prevention (RE-MEDY), there was no statistical difference between treatments in the rates of major bleeding events (0.7%/yr vs 1.3%/yr; RR, 0.52; 95%CI, 0.24-1.05;
All-Cause Death
During initial treatment, the all-cause mortality was similar in patients receiving DOACs or standard treatment (3.6%/yr vs 3.8%/yr; RR, 0.97; 95%CI, 0.84-1.13;
During extended therapy, DOACs were associated with a lower risk of death in comparison with placebo (0.6%/yr vs 1.1%/yr; RR, 0.39, 95%CI, 0.19-0.80;
No differences were found between dabigatran and warfarin in all-cause mortality in the RE-MEDY study (0.9%/yr vs 1.0%/yr;
Absolute Risk Differences and Number Needed to Treat to Benefit
During initial treatment, the use of DOACs significantly reduced the risk of major, critical, and fatal bleeding events in comparison with standard treatment, with corresponding number needed to treat to benefit (NNTB) of 85, 145, and 638 patients to avoid 1 additional event, respectively (Table 4).
Absolute Risk Difference in Events per 100 Patient-Years and Number Needed to Treat to Benefit (NNTB).

Abbreviations: CI, confidence interval; DOAC, direct oral anticoagulants; NNTB, number of patients needed to be treated for 1 additional patient to benefit; NNTH, number of patients needed to be treated for 1 additional patient to be harmed; PE, pulmonary embolism; VTE, venous thromboembolism.
aRandom effects (DerSimonian-Laird). A negative risk difference favors the DOAC versus standard treatment or placebo.
bNo fatal bleedings were reported in secondary prevention trials comparing the DOAC and placebo.
During extended treatment, the use of DOACs significantly reduced the risk of recurrent VTE, recurrent PE, and all-cause death in comparison with no anticoagulation, with corresponding NNTB of 11, 33, and 151 patients to avoid 1 additional event, respectively (Table 4).
Sensitivity Analyses
In the initial treatment of VTE, the RR point estimates of major bleeding and fatal bleeding consistently favored DOACs over standard treatment regardless of statistical model (fixed vs random effects model), type of DOAC (direct thrombin inhibitors or FXa inhibitors), inclusion/exclusion of studies at unclear risk of bias, and inclusion/exclusion of studies with parenteral anticoagulation lead-in (Table S2). Sensitivity analyses of recurrent and fatal VTE consistently showed no differences between DOACs and standard initial treatment of VTE.
In the extended treatment of VTE, the RR point estimates of recurrent VTE and all-cause death consistently favored DOACs over placebo. Confidence intervals were wide for infrequent events, including deaths and CFRs (Table S2).
Discussion
The assessment of the CFRs of recurrent VTE and of major bleeding adds useful information to the crude rates of both outcomes with competing anticoagulant treatment strategies and be helpful in refining therapeutic decision making with respect to the type and duration of anticoagulant therapy. In this systematic review of registration-track trials of the DOACs in the treatment of VTE, we found a potential benefit of the new compounds in terms of major bleeding rate and corresponding CFR in comparison with initial standard treatment of VTE and in terms of recurrent VTE and all-cause mortality versus placebo during extended treatment of VTE, although CFR of recurrent VTE was not reduced. In sensitivity analyses, the results were generally consistent under different assumptions, thus supporting the robustness of our study.
Traditional anticoagulation for initial and extended treatment of VTE is effective in reducing the risk of recurrent VTE but is associated with an increased bleeding risk. In addition, the development of major bleeding during anticoagulant therapy for VTE is a predictor of mortality. 36 Therefore, strategies that could minimize the risk of bleeding are needed. Our study provides some insights in this regard. The use of DOACs was associated with a lower rate of major bleeding, critical bleeding, including ICB, and a trend to lower CFR of bleeding in comparison with initial standard treatment of VTE, thus potentially improving current management of acute VTE. The CFR of major bleeding during acute treatment with DOACs observed in the present study (6%) was also much lower than that previously reported in a systematic review of clinical trials with standard therapy (11.3%). 7 Therefore, it is reassuring that, despite the DOACs currently lack specific antidotes, the clinical outcome of the DOAC-induced major bleedings seems no worse than those developed under standard treatment. The positive results of the DOAC with respect to CFR of major bleeding in comparison with previous reports with standard therapy seems driven by the reduction in the risk of ICB versus standard treatment, as patients who developed ICB had a 4-fold increased risk of death compared to patients who developed extracranial major bleedings in our review, regardless of the type of anticoagulation.
The CFR of recurrent VTE during the first 3 to 12 months of anticoagulation after an acute VTE in this study (between 13% and 16%) is consistent with the 11% to 16% CFR previously reported in the literature. 2,3 Beyond this period, the CFR of recurrent VTE in patients without treatment in our study fell to 4%, which is again comparable to the 3.6% previously reported. 7,8
The benefit of extending treatment of VTE could be considered positive when the expected rate of recurrent VTE with no treatment is at least 3-fold higher than the expected rate of major bleeding during anticoagulant therapy. 7 In this regard, the use of DOACs could help to widen the number of patients that might benefit from extended treatment of VTE, as the absolute benefit in recurrent VTE events avoided versus placebo (9%/yr) was much higher than the additional major bleedings incurred (0.4%/yr).
Despite the fact that DOACs were associated with a reduced risk of VTE and death than placebo during extended treatment, the CFR of recurrent VTE and the rate of VTE recurrences that were PE with DOACs (16% and 50%, respectively) were relatively high in comparison to those reported in the placebo group (4% and 34%, respectively). These data suggest a relatively more severe presentation of VTE in the few patients who developed recurrent VTE despite DOAC extended therapy than in the greater number of patients who had a recurrent VTE while receiving placebo. Therefore, it is arguable that patients who have a recurrent VTE despite extended anticoagulation therapy may represent a particularly high-risk population in which the clinical presentation and outcome may be more severe than in those patients who experience recurrent VTE after having stopped initial anticoagulant treatment. This issue is relevant in clinical practice and highlights the need for individualized therapeutic decision making. However, these data should be interpreted with caution, as the current study has some limitations to be acknowledged.
Study Limitations
First of all, fatal bleeding and fatal VTE events were infrequent. Consequently, absolute benefits of the DOACs versus standard treatment (regarding fatal bleeding) and versus placebo (regarding fatal VTE) were small and CIs for CFR were wide. Notwithstanding, fatal events only represent the “top of the iceberg” and the benefit of the DOACs was consistent for the more frequent outcomes of major bleeding when compared to standard initial treatment and total recurrent VTE in comparison with placebo. Second, the population included in these trials was heterogeneous with respect to baseline characteristics (eg, index event, risk factors of VTE). No disaggregated data on VTE-related and bleeding-related deaths were available depending on baseline characteristics and therefore we were unable to discern whether CFR might differ in relevant subgroups. Another limitation of our study is that we found heterogeneity in the risk of major bleeding across trials on extended treatment, thus leading to an imprecise estimate of major bleeding risk versus placebo. Indeed, the risk of major bleeding and critical bleeding was much higher during initial treatment than during extended therapy, which is consistent with previous findings showing that major bleeding rate is higher during the first 3 months of anticoagulation therapy (mainly during the first month) than afterward. 8 In addition, a selection bias cannot be ruled out as contributing factor, because extension studies included only selected patients that had completed at least 3 months of anticoagulation without having developed recurrent VTE or serious bleeding. These findings support our decision to meta-analyze the studies in the initial and extended treatment of VTE separately. The source results were obtained from registration-track trials, which means that the trials were conducted by selected sites, applying extensive exclusion criteria. Since the analysis of CFRs in our review is essentially an exploratory subgroup analysis in patients who had recurrent VTE or major bleeding during anticoagulant therapy or placebo, our analysis is subject to the general limitations applicable to subgroup analyses. 37 Finally, the dynamics of CFR of recurrent VTE and bleeding during anticoagulant therapy could not be estimated, as the time to fatal VTE or fatal bleeding events was not available. Although with limitations, our review is based on the best evidence available to date from randomized trials and provides an overall picture on the expected benefit-risk profile of the DOACs in the treatment of VTE that is enriched by the analysis of the CFRs.
Conclusions
The analysis of phase III trials on VTE shows that the use of DOACs is associated with fewer major and fatal bleedings and corresponding CFR than standard initial treatment of VTE, and fewer recurrent VTE and mortality than placebo during extended therapy, although the CFR of recurrent VTE was not reduced.
Footnotes
Author Contributions
A. Gómez-Outes contributed to conception and design, analysis, and interpretation, collected the data, drafted the article, critically revised the article, gave final approval, and agreed to be accountable for all aspects of work ensuring integrity and accuracy. R. Lecumberri contributed to conception and design, acquisition, analysis, and interpretation, collected the data, critically revised the article, and gave final approval. M. L. Suárez-Gea, A. I. Terleira-Fernández, and M. Monreal contributed to analysis and interpretation, critically revised the article, and gave final approval. E. Vargas-Castrillón contributed to analysis and interpretation, critically revised the article, supervised the study, gave final approval, and agreed to be accountable for all aspects of work ensuring integrity and accuracy.
Authors’ Note
The contents of this study are solely the responsibility of the authors and do not necessarily represent the official view of their institutions or any other party. Antonio Gómez-Outes and Emilio Vargas-Castrillón are the guarantors.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr Lecumberri declares personal fees from Bayer, Boehringer Ingelheim, Bristol-Myers Squibb, and Daiichi Sankyo and grants and personal fees from Rovi, outside the submitted work. Dr Monreal declares consultant fees from Sanofi, Bayer, and Daiichi Sankyo and grants from Bayer and Sanofi, outside the submitted work.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
