Abstract
Aortic valve stenosis (AVS) is the most common valve disease in adults. Severe forms are associated with acquired von Willebrand syndrome (aVWS) with loss of the largest von Willebrand factor (VWF) multimers. Diagnostic gold standard is the VWF multimer analysis. Valve replacement rapidly restores the VWF structure. Uncertainty exists if this effect is permanent and how functional VWF assays perform compared with multimer analysis. We studied 21 consecutive patients with severe AVS before and 6 to 18 months after valve surgery and compared them with 14 controls without valve disease referred for coronary angiography. The VWF multimers, VWF antigen (VWF:Ag), VWF collagen binding capacity (VWF:CB), VWF:CB/VWF:Ag ratio, in vitro bleeding time (PFA-100), factor VIII coagulation activity (FVIII:C), and VWF ristocetin cofactor activity (VWF:RCo) were determined. In all patients with AVS, the large VWF multimers were strongly reduced (56 ± 13% of normal plasma); all controls had normal multimers. The PFA-100 collagen/ADP closure times (coll/ADP CT) were prolonged in patients with AVS compared with the controls (175 ± 56 seconds vs 86 ± 14 seconds, P < .001). The VWF:CB/VWF:Ag ratio was pathological in 20 of the 21 patients but normal in controls. After surgery, the multimers normalized in all patients and coll/ADP CT shortened (pre 184 ± 65 seconds vs post 102 ± 22 seconds; P < .001). The VWF:CB/VWF:Ag ratio strongly improved (P < .001) and normalized in 14 of 17 patients. In conclusion, all consecutive patients with severe AVS had an aVWS. The combination of coll/ADP CT and VWF:CB/VWF:Ag ratio detected the aVWS in all patients. More than 6 months after valve replacement, the VWF multimers were still normalized in all patients indicating a permanent cure of the aVWS.
Introduction
Aortic valve stenosis (AVS) is the most common valve disease in adults. Showing an increasing incidence with age and considering the demographic changes in the near future, it may become an epidemic disease. 1 The AVS progresses over several years until symptoms such as exertional dyspnea, angina, and syncopes develop. Severe stenosis may cause an acquired von Willebrand syndrome (aVWS) due to the loss of the largest multimers of the von Willebrand factor (VWF). This deficiency leads to a reduced hemostatic function of the VWF, which especially disturbs the primary hemostasis by affecting platelet adhesion under high-shear conditions. In a considerable number of patients, this causes an increased bleeding tendency, although in most patients it remains inapparent. 2 In 1958, Heyde, a general practitioner, first described the association of calcific AVS with gastrointestinal bleeding. 3 Laboratory diagnosis is demanding, since global coagulation assays are not affected. The diagnostic gold standard for aVWS type 2 is the VWF multimer analysis, which is a laborious and expensive method performed in specialized coagulation laboratories only.
Surgical valve replacement can rapidly resolve the aVWS. The concentration of the high-molecular-weight multimers of VWF normalizes within hours. 4 However, it is unclear if valve replacement completely and permanently corrects this coagulopathy. Most reports focus on short follow-up intervals from 2 to 4 weeks up to 6 months. 5 –7 Long-term outcomes are restricted to a case report. 8 In 2003, Vincentelli et al reported that 6 months postsurgery, the aVWS had recurred with prolonged collagen/ADP closure times (coll/ADP CT; PFA-100 in vitro bleeding time) in 66% and a pathologic VWF multimeric pattern in 74% of the initially restituted patients. 7
In our study, we evaluated patients with confirmed severe AVS scheduled for valve surgery preoperatively and at least 6 months postoperatively. The findings were compared with a control group of patients referred to coronary angiography without valve disease. We measured the VWF multimer pattern in conjunction with the markers of VWF function including collagen binding capacity (VWF:CB) and in vitro bleeding time (PFA-100). We hypothesized that severe AVS is accompanied by aVWS type 2, and aortic valve replacement would lead to a sustained cure of the aVWS. Furthermore, we evaluated the usefulness of the PFA-100 test system and the determination of the VWF:CB/VWF:Ag ratio, alone or in combination, to diagnose the aVWS.
Methods
The study was approved by the ethics committee of the University Hospital Aachen, Germany. Written informed consent was obtained from all participants.
Patients and Controls
We prospectively enrolled 21 consecutive adult patients (mean age 70 ± 9 years, 9 males, 12 females) with confirmed severe AVS accepted for surgical valve replacement. For the diagnosis, the following criteria according to the guideline of the European Society of Cardiology were used: (a) a mean transvalvular gradient measured echocardiographically or invasively during coronary angiography of at least 40 mm Hg and/or a calculated orifice area of <1.0 cm 2 , (b) a calcified valve with reduced mobility, and (c) clinical symptoms attributable to the valvular disease (eg, syncopal events, decompensated heart failure, or pulmonary edema).
The decision for surgical repair was based on a multidisciplinary conference judgment of functional status, complete cardiac evaluation, and clinical symptoms. Exclusion criteria were coronary artery disease with indication for more than 1 aortocoronary bypass, multivalvular disease, endocarditis, or denial of written consent.
From the initial patient group, 1 patient finally refused valve surgery and 3 patients were not available for the follow-up after at least 6 months (1 death, 2 not able to come), so that the comparison before and after surgery is based on 17 data pairs.
The control group consisted of 14 patients (mean age 67 ± 12 years, 8 males, 6 females, P = not significant vs patients) referred to coronary angiography during the same time period. These patients had no valvular or infectious disease, had no active neoplastic disease, and were all without bleeding history.
Blood Collection
After informed consent and in the patient group after acceptance for valve surgery, blood was carefully collected from an antecubital vein in citrated tubes (3.2%, 0.106 mol/L trisodium citrate; Sarstedt, Nümbrecht, Germany) and processed immediately. The first 5 mL of blood were used for routine blood chemistry. For the VWF analysis, citrated whole blood was sent to the cooperating external coagulation laboratory (Prof Budde) per mail overnight.
Coagulation Assays
The following coagulation parameters were analyzed in the central laboratory of the University Hospital Aachen using standard automated assay methods: prothrombin time (TPZ/Quick), activated partial thromboplastin time (aPTT), factor VIII coagulation activity (FVIII:C), VWF ristocetin cofactor activity (VWF:RCo), and in vitro bleeding time.
The in vitro bleeding time was measured using the collagen/ADP cartridge of the PFA-100 device (Dade Behring, Germany). The coll/ADP CT is not affected by concomitant therapy with acetylic salicylic acid. 9 The blood was analyzed after 1 hour of resting and within 4 hours after collection. The PFA-100 is a sensitive screening test for the primary hemostasis and determines the platelet function under high-shear conditions. Platelet defects, certain antiplatelet drugs, and VWF abnormalities lead to prolonged closure times indicating platelet malfunction. 10
The VWF analysis including multimeric pattern, VWF antigen (VWF:Ag enzyme-linked immunosorbent assay), and VWF:CB was performed in the Coagulation Laboratory Hamburg (Prof U. Budde, Hamburg, Germany). The VWF multimer pattern was analyzed using low-resolution agarose gel electrophoresis (1.2% agarose). The original method by Ruggeri and Zimmerman 11 was modified by Western blotting on nitrocellulose filters and use of the sensitive chemiluminescence technique. 12 Subsequently, the digital image was quantified by densitometry with the Fluorchem Instrument from Alpha Innotech (San Leandro, CA, USA). The multimers were categorized into large (>5500 kDa, oligomers 11 and larger), medium (oligomers 6-10), and small (oligomers 1-5). For quantitative analysis, the multimers were expressed as percentage of all multimers and as percentage of the normal plasma pool, which served as reference in each electrophoresis.
For the VWF:CB assay, 13 Nunc-Immuno plates (Thermo Scientific Nunc A/S, Roskilde, Denmark) were coated with 3 µg/mL type I/III collagen reagent Horm (95%/5%, Takeda, Linz, Austria) in 0.1 mol/L acetic acid overnight. After 4 washing steps, the standard curve and plasma samples were applied and incubated for 2 hours at 37°C. After washing, the horseradish peroxidase-labeled antibody (1.1 µg/mL, polyclonal rabbit antihuman VWF; DAKO, Glostrup, Denmark) was added. Thereafter, the plates were incubated for 2 hours at 37°C and washed 4 times. Absorbance was measured at 492 nm.
In order to characterize the functional integrity of VWF, we calculated the VWF:CB/VWF:Ag ratio. The ratio can become abnormal indicating VWF dysfunction even if the single parameters are within the normal range. In a subgroup of patients and controls, we also analyzed the multimer pattern and VWF:Ag level of the platelet VWF.
Statistics
All statistical calculations were performed using the software package SPSS 10.0 (SPSS Inc, Chicago, Illinois). Since according to the Kolmogorov-Smirnov test, the data were assumed to be normally distributed, and they are presented as mean values ± standard deviation. For analytical reasons, measured values of VWF:RCo, coll/ADP CT, and FVIII:C exceeding the upper limit of the assay method were set as upper limit plus 1 (VWF:RCo > 150%: 151%, coll/ADP CT > 300 seconds:301 seconds, FVIII:C > 250%:251%). Differences between groups were tested with the paired or unpaired t test where appropriate. For correlation analysis, the Pearson coefficient was used. A 2-tailed P value of <.05 was considered to indicate statistical significance.
Results
Clinical Data
In the patient group, the mean pressure gradient over the aortic valve was 63.5 ± 15.3 mm Hg (range 40-84 mm Hg). The calculated orifice area was 0.72 ± 0.27 cm2 (range 0.4-1.3 cm2). Valve surgery was uneventful without bleeding complications. None of the patients reported an increased bleeding tendency or bleeding complications in the preoperative course. Serum creatinine and platelet counts at baseline were similar in patients and controls (1.0 ± 0.26 mg/dL vs 1.0 ± 0.12 mg/dL; P = .881; 235 ± 80 g/L vs 267 ± 108 g/L, P = .365). Of the 20 implanted artificial aortic valves, 17 were mechanical (St. Jude Medical, Eschborn, Germany) and 3 biological prostheses (Baxter Perimount, Baxter Healthcare, Hechingen, Germany).
Standard Coagulation Assays
The routine coagulation assays prothrombin time (Quick) and aPTT were all in the normal range throughout the study. The FVIII:C levels were above the normal range (>140%) in all patients and in 3 of the 14 controls. The VWF:RCo was normal (reference range 50-150%) or variably increased (>150%) in all patients and controls without meaningful differences between groups (VWF:RCo 11 patients > 150%, 6 > 100%, 3 > 85%, 1 patient 61%; 12 controls > 150%, 2 > 100%). The VWF:RCo above the upper limit of the assay (>150%) was not further quantified. After valve replacement, all but 1 patient had a VWF:RCo > 150% (1 patient 123%).
von Willebrand Factor Analysis at Baseline
All patients with AVS revealed an abnormal multimer pattern with considerable reduction in the large VWF multimers resembling VWD type 2A (see Table 1, Figures 1A and 2A). Compared with a normal plasma pool, the large multimers were reduced to 56% with corresponding increase in the small multimers (∼130%). Although all VWF:Ag and VWF:CB levels were normal or elevated, the VWF:CB/VWF:Ag ratio was pathological (<0.8) in 20 (95%) of the 21 patients, revealing a significantly reduced ratio in the patients with AVS versus controls (0.65 ± 0.1 vs 1.0 ± 0.19, P < .001, reference range 0.8-2.0). Only 1 patient with AVS had a ratio of 0.85 at the lower limit of the normal range. The VWF:CB/VWF:Ag ratio did not correlate with the large multimers.
Baseline Values of Patients With AVS (n = 21) and Controls (n = 14).a
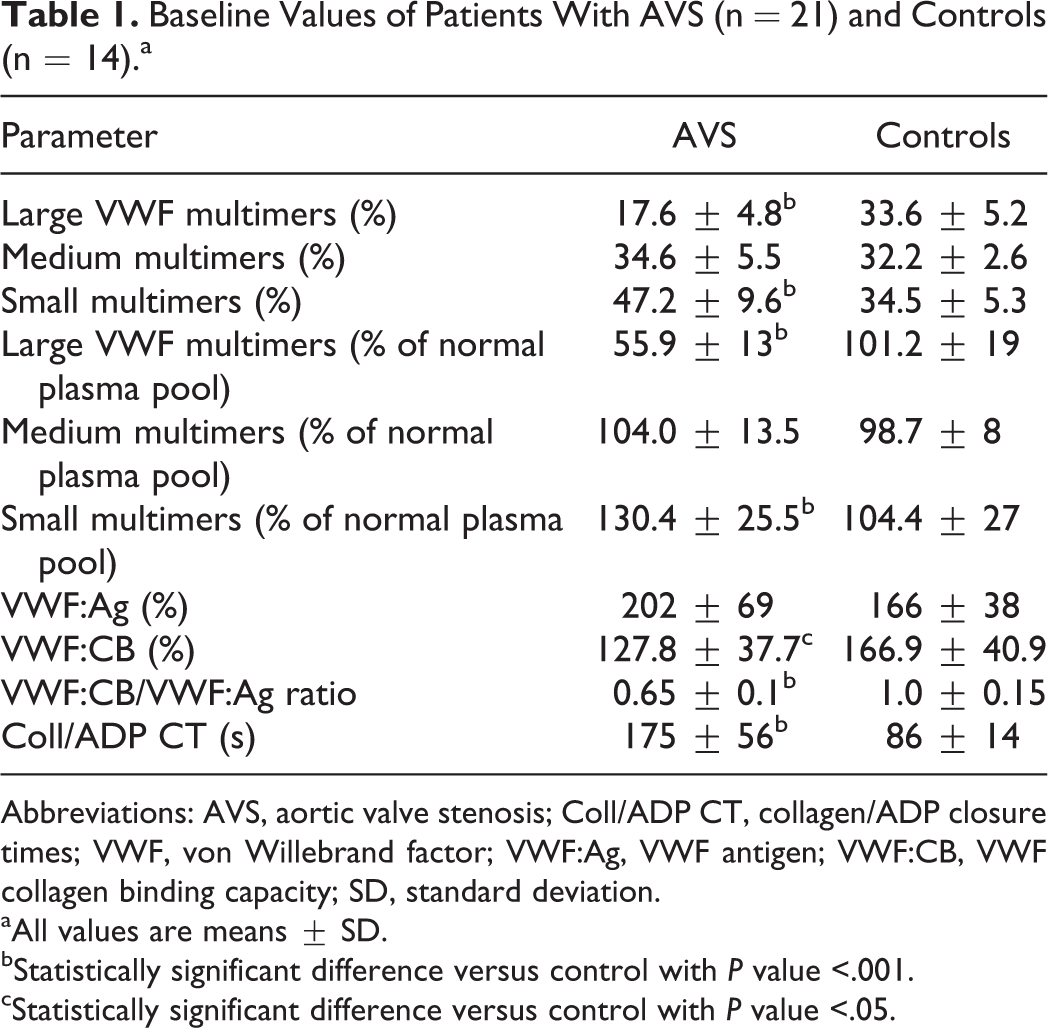
Abbreviations: AVS, aortic valve stenosis; Coll/ADP CT, collagen/ADP closure times; VWF, von Willebrand factor; VWF:Ag, VWF antigen; VWF:CB, VWF collagen binding capacity; SD, standard deviation.
aAll values are means ± SD.
bStatistically significant difference versus control with P value <.001.
cStatistically significant difference versus control with P value <.05.
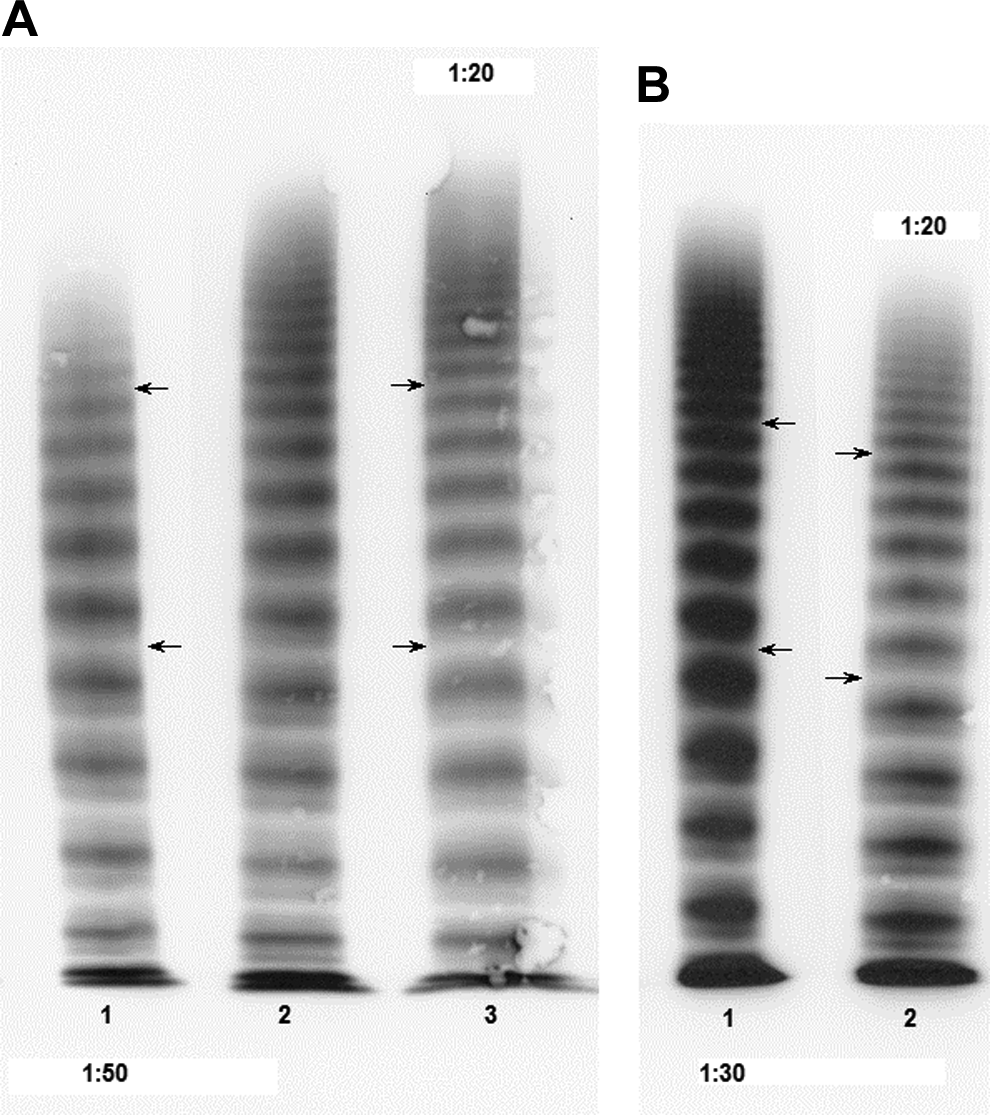
Representative von Willebrand factor (VWF) multimer electrophoresis in low-resolution gels: (A) before surgical correction of aortic valve stenosis (AVS): #1 patient with AVS, #2 control without AVS, #3 control (normal plasma pool [NPP]); (B) after surgical valve replacement: #1 patient, #2 control (NPP). The electrophoresis runs from top to bottom, therefore the large multimers are at the top of the figure. The arrows mark the limits between medium and large multimers (between oligomers 10 and 11) and small and medium multimers (between oligomers 5 and 6), respectively. Large multimers are reduced in the patient with AVS. After surgery, the multimer pattern has normalized.
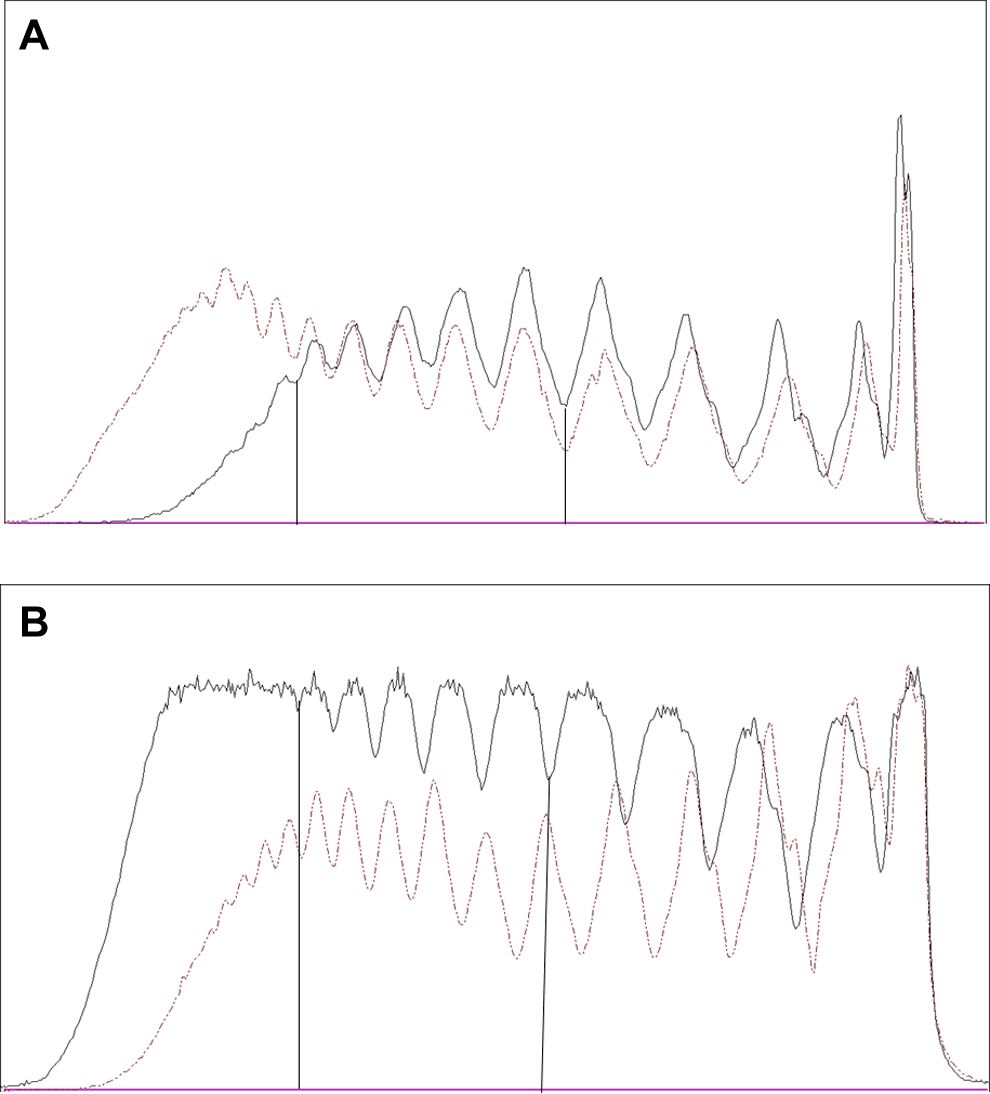
Representative densitometric tracings of the von Willebrand factor (VWF) multimers: (A) before surgical correction of aortic valve stenosis (AVS); (B) after surgical valve replacement; solid line = patient; broken line = control (normal plasma pool). The large multimers appear at the left side. The vertical lines mark the limits between medium and large multimers (between oligomers 10 and 11) and small and medium multimers (between oligomers 5 and 6), respectively.
All patients had a prolonged coll/ADP CT (range 106-300 seconds; reference range 62-104 seconds). There was no inverse correlation between the extent of the multimer reduction and the closure time prolongation.
All controls had a normal multimer pattern (Figure 1A) and a normal VWF:CB/VWF:Ag ratio (lowest value 0.89). The distribution of the VWF:CB/VWF:Ag ratios of patients and controls did not overlap. Two controls had a moderately prolonged coll/ADP CT (both 109 seconds), all others remained below 90 seconds.
The combination of the 2 parameters VWF:CB/VWF:Ag ratio and coll/ADP CT was able to correctly identify all patients with aVWS as confirmed by the multimer analysis (sensitivity 100%, VWF:CB/VWF ratio sensitivity 95%). The only patient with AVS with a low normal VWF:CB/VWF ratio (0.85) had a clearly abnormal coll/ADP CT (146 seconds). In the control group, the 2 patients with abnormal coll/ADP CT had a normal VWF:CB/VWF:Ag ratio and multimer analysis, suggesting that the pathological coll/ADP CT was not related to the VWF. Since all control patients had a normal VWF:CB/VWF:Ag ratio, the specificity of this parameter was 100%.
In 5 patients and 5 controls, a platelet VWF analysis was available. All samples showed a normal multimer pattern of the platelet VWF and normal VWF:Ag levels (10-48 U/10 11 platelets, reference range 10-50). The severity of the AVS measured as mean pressure gradient or orifice area did not correlate with VWF:CB/VWF:Ag ratio, large multimers, or coll/ADP CT.
Effect of Aortic Valve Replacement
At least 6 months postsurgery (range 6-18 months, mean 10.7 months), the VWF analysis was repeated. The results are shown in Table 2. We observed a complete normalization of the multimeric pattern (large multimers 101% of normal plasma pool; Figures 1B and 2B). Correspondingly, the VWF:CB/VWF:Ag ratio significantly improved in all patients and became normal in 14 (82%, range 0.9-1.4) of the 17 patients. In 3 patients, the ratio clearly improved from baseline 0.54 to 0.62 but remained slightly below the normal range (0.75-0.78). The coll/ADP CT significantly decreased in all patients (Table 2) but remained prolonged in 5 of the available 14 measurements (range 106-121 seconds), although shortened by 60 to 130 seconds compared with baseline. Interestingly, in these patients, the VWF:CB/VWF:Ag ratio had normalized (baseline 0.5-0.75 to 0.9-1.2), reflecting the restitution of the large VWF multimers. Obviously, the abnormal closure time was not explainable by a pathologic VWF.
Comparison of Coagulation Assay Results in Patients With AVS Before and At Least 6 Months After Valve Replacement Surgery (n = 17).a
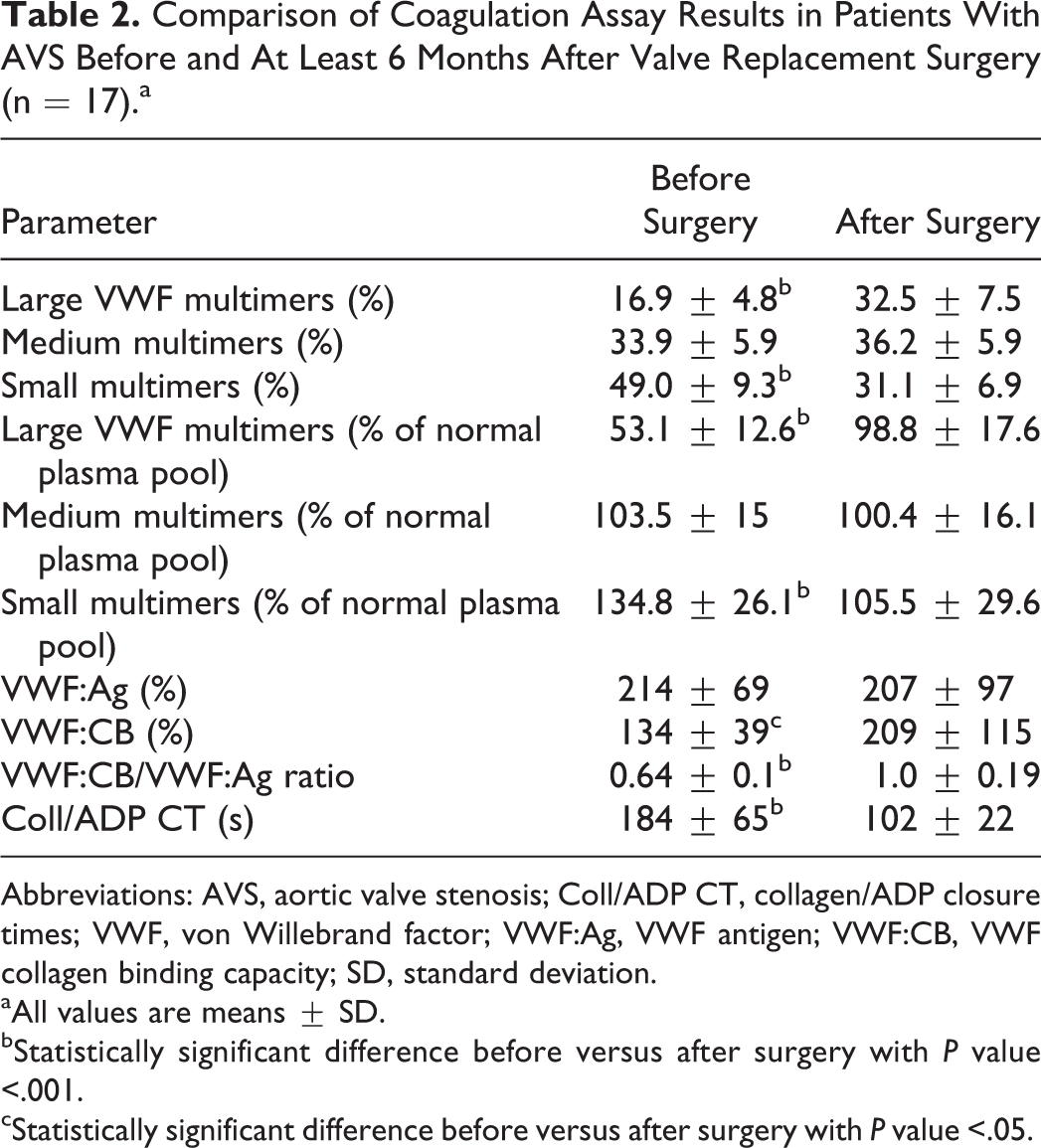
Abbreviations: AVS, aortic valve stenosis; Coll/ADP CT, collagen/ADP closure times; VWF, von Willebrand factor; VWF:Ag, VWF antigen; VWF:CB, VWF collagen binding capacity; SD, standard deviation.
aAll values are means ± SD.
bStatistically significant difference before versus after surgery with P value <.001.
cStatistically significant difference before versus after surgery with P value <.05.
Aspirin Use
Ten patients and 12 controls were treated with the nonsteroidal anti-inflammatory drug aspirin. The coll/ADP CT was not affected by this medication. The closure times were similar in patients and controls with or without aspirin medication.
Discussion
All 21 consecutive patients with severe AVS displayed an aVWS type 2 with a marked reduction in the large VWF multimers (∼55% of normal pool plasma). The reported prevalence of the aVWS in the literature is variable and ranges from 33% to over 80%. 14 –16 Some authors even did not find evidence for aVWS in patients with severe AVS at all or only in a minority and interpreted pathological platelet function indices as a primary platelet disorder. 17 –20 Most likely, these differences are due to the used methods and diagnostic criteria.
The valve replacement leads to a complete resolution of the aVWS evidenced by normalization of the VWF multimer pattern in all patients. In contrast to Vincentelli et al, 7 we found a permanent cure of the aVWS without evidence for recurrence. To our knowledge, we report for the first-time follow-up data of patients with AVS more than 6 months after surgery. Our data do not exclude that in the long run, the aVWS may recur due to the deterioration of the replaced valve.
Laboratory diagnosis of aVWS type 2 is hampered by the fact that routinely applied VWF parameters such as FVIII:C, VWF:Ag, and VWF:RCo are not affected. The VWF:RCo test measures the binding of VWF to the platelet glycoprotein 1b in the presence of ristocetin. The test is obviously more sensitive to intermediate large multimers, which are usually preserved in aortic stenosis. In contrast, the VWF:CB measures the physiological interaction of VWF with collagen, which mainly depends on the large multimers. Therefore, this parameter should be especially sensitive to aVWS type 2. We observed that VWF:CB was significantly different in patients and controls but remained in the normal range. Normalization of the VWF:CB to the VWF:Ag level by calculating the VWF:CB/VWF:Ag ratio unmasked the pathological VWF:CB and then clearly discriminated between patients with AVS and controls without overlap of the individual results. The sensitivity was 95%, with only 1 of the 21 patients displaying a low normal ratio. The specificity was 100%. Our results show that this parameter is able to reflect the loss of the largest VWF multimers and could serve as a valuable diagnostic test for aVWS type 2. This is in contrast to reports from other groups, which observed no difference or abnormal VWF:CB/VWF:Ag ratios in only few patients concluding that the parameter could not be recommended. 4,14,21 The most likely explanation for these discrepant findings is the type and source of the collagen used in the tests. 22,23 The collagen type I/III mixture used in our test system seems to have a high sensitivity, but most commercial assays use only 1 collagen type (I or III). This implies that the VWF:CB assays are not interchangeable. During the development and standardization of a VWF:CB assay, stability and reproducibility of the collagen source and coating as well as the sensitivity to the largest multimers have to be explicitly studied.
All patients had severely prolonged coll/ADP CT. This automated commercial assay is an established screening tool for platelet disorders and VWD. 10,24,25 In contrast to the collagen/epinephrine closure time, the coll/ADP CT is not affected by platelet aggregation inhibitors such as aspirin. 9 Since this parameter cannot discriminate between a platelet dysfunction and an abnormality of the VWF, a VWF specific and sensitive assay has to be combined in order to avoid a misclassification. After surgery, all patients showed a pronounced shortening of the coll/ADP CT associated with the normalization of the VWF multimers. In 3 patients, the closure time remained moderately prolonged pointing to a platelet dysfunction not related to VWF.
The clinical significance of the aVWS remains unclear. The loss of the large VWF multimers leads to aVWS, which may cause bleeding. Typically, AVS is associated with gastrointestinal bleeding, mostly from angiodysplasias, known as Heyde syndrome. 3,26 Bleeding resolves after valve replacement. 8,26 We did not find evidence for an increased bleeding tendency. A large retrospective cohort study in patients with AVS found a prevalence of gastrointestinal bleeding of only 1%.2 The major cardiac surgery is tolerated without increased blood loss or transfusion requirements. 15,16,27 This may be explained by the rapid restoration of the hemostatically most important large multimers during cardiopulmonary bypass as shown by Solomon et al. 4 In the rare cases of gastrointestinal bleeding associated with AVS, the patient should be screened for aVWS type 2 by measuring the coll/ADP CT and the VWF:CB/VWF:Ag ratio. Future studies should evaluate the correlation of progressing AVS with the development of the aVWS type 2. So far, it is not known at what time point or severity of the valve disease the aVWS becomes detectable. This requires a longitudinal observational study in a collective of well-characterized patients with AVS. Further studies are also needed to find out whether the diagnosis of aVWS should play a role in the decision process for valve replacement.
In conclusion, we found that all consecutive patients with severe AVS had an aVWS with loss of the large multimers. There was no evidence for an increased bleeding tendency. The combination of coll/ADP CT and VWF:CB/VWF:Ag ratio detected aVWS in all patients. The VWF multimer analysis could be reserved for indistinct cases. More than 6 months after valve replacement, the VWF multimers were still normalized in all patients indicating a permanent cure of the aVWS.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
