Abstract
We summarize the evidence for the safety and efficacy of catheter-directed thrombolysis (CDT) with and without ultrasound-assisted therapy for treating submassive and massive pulmonary embolism (PE) in a systematic review. The primary efficacy outcome was mortality. Outcomes were pooled across studies with the random-effects model. Twenty-four studies enrolled 700 patients in total; 653 received mechanical thromboembolectomy treatments for PE (mortality rate, 9% [95% confidence interval (CI), 6%-13%], P = .12; rate of minor complications, 6% [95% CI, 2%-13%]). In the ultrasound-accelerated thrombolysis (USAT) studies, the mortality rate was 4% (95% CI, 1%-11%) and in the non-USAT studies, it was 9% (95% CI, 6%-13%). Secondary safety outcomes were all bleeding events, which occurred in 12% (95% CI, 7%-20%) of the USAT studies and in 10% (95% CI, 5%-20%) of the non-USAT studies. Current clinical evidence does not prove USAT is superior over CDT methods.
Introduction
Venous thromboembolism, including pulmonary embolism (PE) and deep vein thrombosis, is a major contributor to the global disease burden and a leading cause of disability and lost income. 1,2 Although the 30-day survival for venous thromboembolism is 75%, sudden death is the clinical presentation of PE for almost a quarter of patients. 3,4 The primary treatment of PE is anticoagulation; however, thrombolysis may be used in high-risk patients. 5 –7 For these patients, current guidelines favor systemic use of thrombolytics over catheter-directed approaches. 5,6 In a recent meta-analysis 8 with over 2000 patients enrolled in randomized controlled trials, systemic thrombolysis was evaluated among patients with severe PE; the use of thrombolytics was associated with lower all-cause mortality compared to the use of anticoagulants alone, but this was in exchange for a higher risk of major bleeding (9.2%) and a 1.5% incidence of intracranial hemorrhage. Despite measurable reduction in PE-associated mortality, it is estimated that only 30% of hemodynamically unstable patients receive thrombolysis. 9
Catheter-directed thrombolysis (CDT) offers an alternative for patients who have a high risk of bleeding and contraindications for systemic thrombolysis. CDT may be administered conventionally through a catheter with multiple sideholes and direct thrombolytic infusion into the pulmonary arteries. 10 Delivery methods have evolved, however, plausibly improving the safety profile of the procedure. In addition, ultrasound-accelerated thrombolysis (USAT) is a novel pharmacomechanical thrombolysis method that combines CDT and ultrasound energy to improve the delivery of thrombolytic agents. 10 We sought to summarize the evidence for the safety and efficacy of conventional CDT versus USAT for the treatment of submassive and massive PE.
Methods
With the assistance of an expert research librarian, we performed a systematic review by searching several electronic databases, including Medline, Embase, Scopus, and Cochrane, for studies published from October 2009 through October 2014. We elected to exclude studies older than 2009 to focus on the more current outcomes and technology. The search terms were mechanical thrombectomy, mechanical thromboembolectomy, mechanical thrombolytic therapy, mechanical thrombolysis, and pulmonary embolism. We followed the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) Statement for reporting this systematic review. 11 The process of study selection is outlined in the Figure 1.
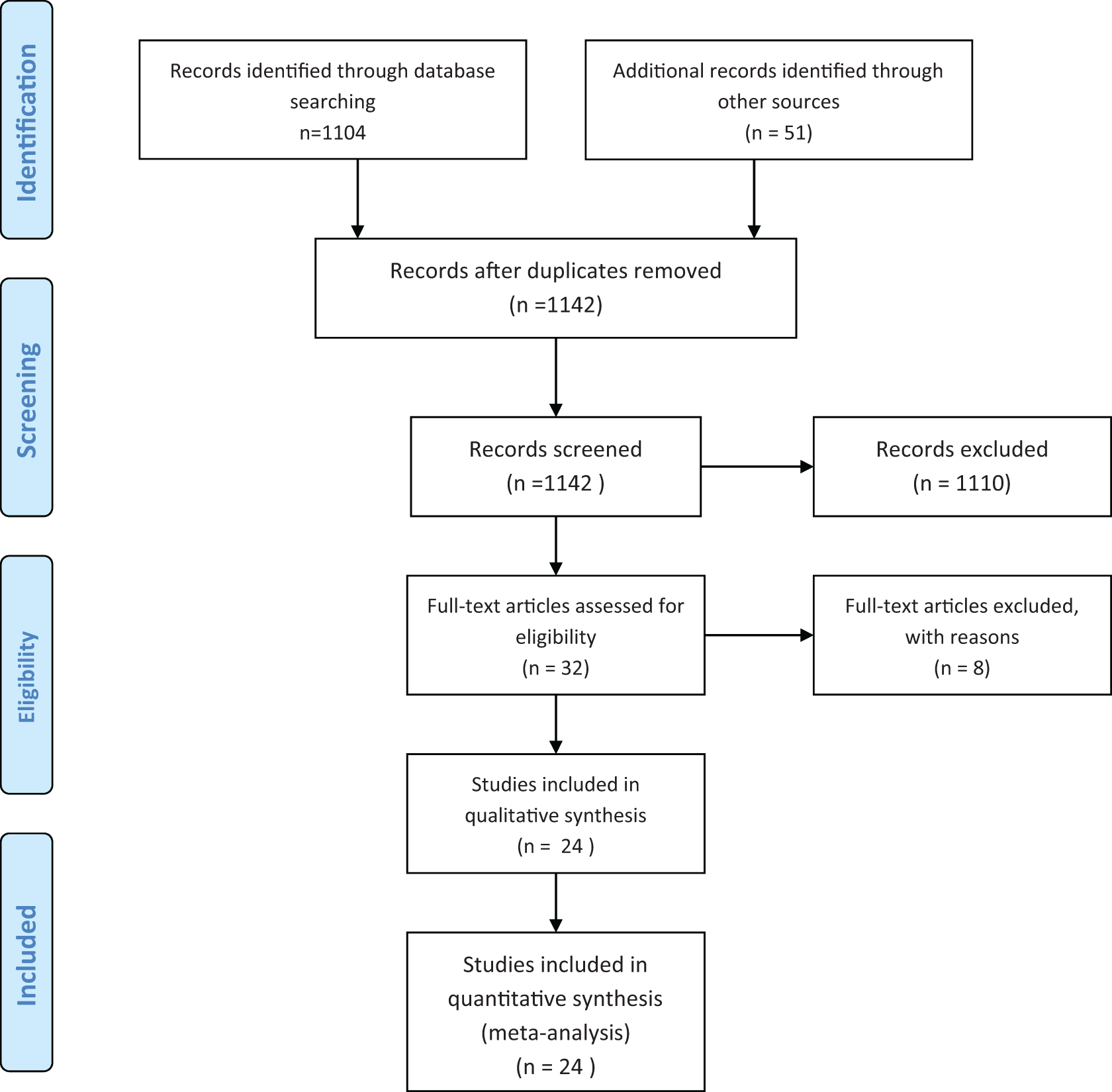
PRISMA flow diagram. (Adapted from Moher et al 11 and used with permission.).
There was no language restriction; we limited abstraction to case series with more than 5 patients. Unpublished abstracts were included in the search. All data were abstracted by at least 2 independent reviewers; if a discrepancy occurred, a third reviewer adjudicated the disagreement. Studies were included in the analysis if they enrolled patients with acute PE and provided outcome data on complications and mortality. Mortality was the main efficacy outcome. Complications were categorized as minor if no therapy or nominal therapy was required or as major if the complication required longer hospitalization or caused permanent adverse sequelae or death. 12 Bleeding complications were analyzed separately as a safety outcome and were defined as major bleeding if patients required a transfusion of blood products, if their hemoglobin level decreased by a least 2 g/dL, or if bleeding occurred in critical organs. We also extracted the method of diagnosis, severity of PE, clot burden as reported by the Miller index, 13 use of thrombolytic agents, pulmonary pressures, and hospital length of stay in days.
We evaluated the methodologic quality (risk of bias) of the included studies according to selected items relevant to uncontrolled studies, focusing on cohort selection, loss of follow-up, and ascertainment of the outcomes and exposure.
Statistical Analysis
The included studies were uncontrolled (ie, without a comparison group), so we could only estimate incidence rates (ie, the event rate at the end of study follow-up). Rates were pooled across studies with the random-effects model. 14 Heterogeneity was assessed with the I 2 statistic, with a value over 50% implying substantial heterogeneity. 15 We also conducted the Cochran Q test for heterogeneity, with a P value less than .05 being consistent with statistically significant heterogeneity. Data were insufficient to evaluate the presence and impact of publication bias.
Results
The electronic search identified 1104 references (Figure 1). A manual search identified 51 additional references. The meta-analysis included 24 studies with a total of 700 patients, of which 653 received endovascular treatments for PE. Although 2 studies by Engelhardt et al were selected for full review, only 1 study 16 was included because the second one contained duplicated information. The study by Lin et al 17 allowed for abstraction of USAT separately from conventional CDT; thus, the data were analyzed separately along with 5 more studies that were USAT-specific (Table 1). We included a total of 7 abstracts. Most of the studies were retrospective, and only 1 study was a randomized controlled trial. All studies allowed inclusion of both massive and submassive PE. The risk of bias was high because most of the studies included nonconsecutive patients and were uncontrolled (Table 2). The mean age of participants was 58 years (Table 1). Follow-up time ranged from 1 month to 5 years.
Demographics of Patients in 24 Studies of Catheter-Directed Thrombolysis.
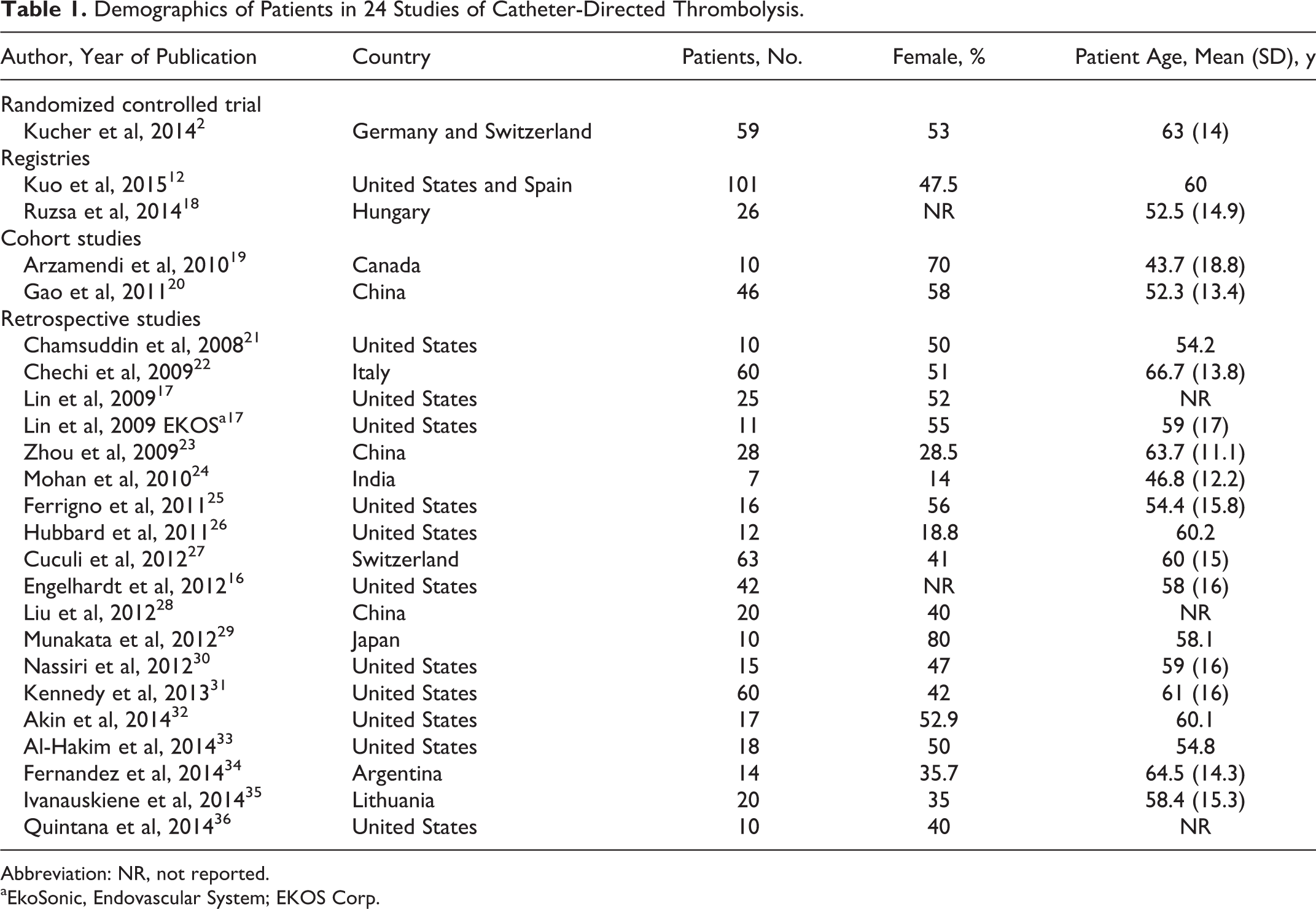
Abbreviation: NR, not reported.
aEkoSonic, Endovascular System; EKOS Corp.
Published Studies That Met Inclusion Criteria.
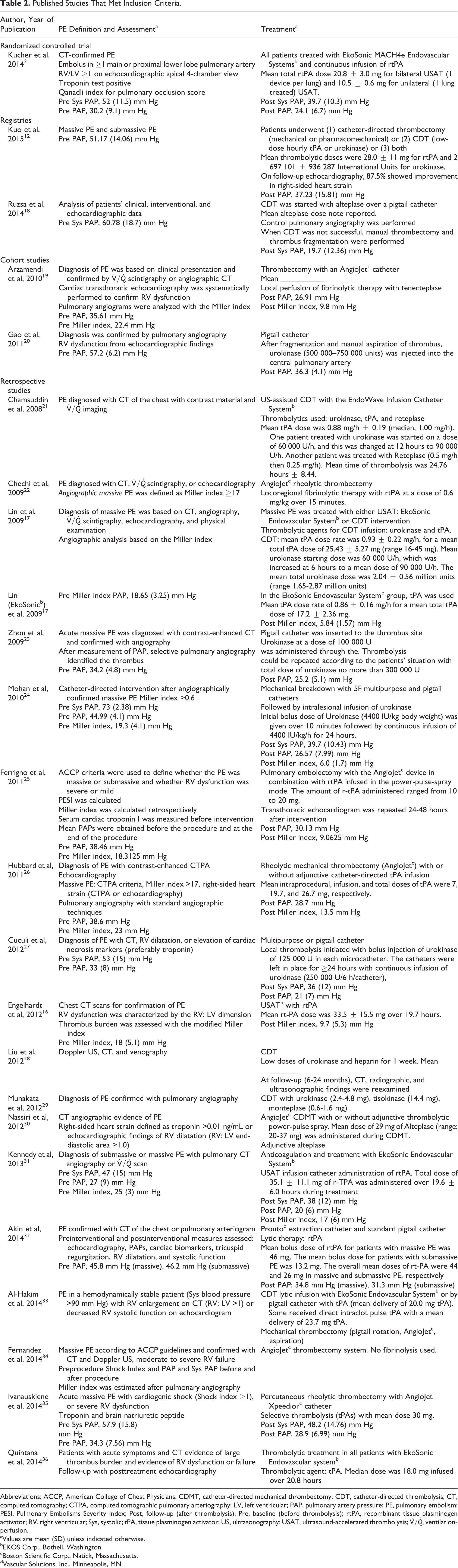
Abbreviations: ACCP, American College of Chest Physicians; CDMT, catheter-directed mechanical thrombectomy; CDT, catheter-directed thrombolysis; CT, computed tomography; CTPA, computed tomographic pulmonary arteriography; LV, left ventricular; PAP, pulmonary artery pressure; PE, pulmonary embolism; PESI, Pulmonary Embolisms Severity Index; Post, follow-up (after thrombolysis); Pre, baseline (before thrombolysis); rtPA, recombinant tissue plasminogen activator; RV, right ventricular; Sys, systolic; tPA, tissue plasminogen activator; US, ultrasonography; USAT, ultrasound-accelerated thrombolysis;
aValues are mean (SD) unless indicated otherwise.
bEKOS Corp., Bothell, Washington.
cBoston Scientific Corp., Natick, Massachusetts.
dVascular Solutions, Inc., Minneapolis, MN.
Table 3 presents the pooled estimates of the efficacy and safety incidence rates. The risk of death in the 18 non-USAT studies was 9% (95% confidence interval [CI], 6%-13%); there was no significant heterogeneity for this mortality estimate (P = .12). In the 6 USAT studies with reported mortality, 4% of the patients died (95% CI, 1%-11%). We could not discriminate USAT data from non-USAT data in the study by Al-Hakim et al 33 ; thus, these results were reported separately.
Meta-Analysis of Studies: Use of Catheter-Directed Thrombolysis With and Without Ultrasound-Assisted Thrombolysis.

Abbreviations: NA, not applicable; USAT, ultrasound-accelerated thrombolysis.
aOne study allowed separate data analysis for use of USAT versus no use of USAT.
Twelve studies used a formal PE stratification score. The most commonly used was the Miller index, reported in 10 studies. The average prethrombolysis Miller index was 19.4, and improvement was reported in all studies, with an average Miller index of 9.9 after the intervention. Pulmonary pressures were not consistently reported. Only 6 of 13 studies reported prethrombolysis mean pulmonary pressures; the average was 57 mm Hg. After the procedure, mean pulmonary pressure averaged 36 mm Hg.
In the 7 studies that reported a postintervention Miller index, there was improvement (Table 2); however, the data do not allow a pooled comparative analysis. Similarly, 6 of 13 studies reported systolic pulmonary artery pressures before and after the intervention. All reported improvement. None of the selected studies presented a quality-of-life evaluation. In addition, minor complications developed in 6% (95% CI, 2%-13%) of the patients in the non-USAT group and in 12% (95% CI, 7%-20%) in the USAT studies.
The main safety outcome was major bleeding. Among the 6 USAT studies, the incidence was 4% (95% CI, 1%-11%) compared to 10% (95% CI, 6%-14%) in studies that used alternative techniques. Secondary safety outcomes were all bleeding events, which occurred in 12% (95% CI, 7%-20%) of the USAT studies and in 10% (95% CI, 5%-20%) of the non-USAT studies.
Discussion
We report the best available evidence of the safety of modern endovascular treatment of PE. Although the risk of bleeding appears to be lower with USAT therapies, the heterogeneity between the trials and the paucity of adequate comparative data prevented us from reaching a definitive conclusion. The risk of death—as high as 9% despite advances in therapeutic techniques—stresses the need for further studies in this area.
In contrast to our results, Kuo et al 12 reported a meta-analysis with 594 patients in 35 studies before 2009. The pooled risk of major complications was 2.4% (95% CI, 1.9%-4.3%). None of the included studies evaluated USAT technology, and there were no comparative trials. We measured a similar rate of major complications for USAT compared to the 1 reported in the study by Kuo et al. 12 Engelberger and Kucher 10 summarized the data from 197 patients with high-risk PE abstracted from 7 studies. That systematic review was restricted to USAT, and the mortality at 3 months was 3.6% (95% CI, 1.4%-7.2%), with major bleeding complications developing in 3.6% of the patients. The complication and mortality rates that we estimated for USAT were similar. From the available information, it is not possible to define superiority of USAT for PE revascularization. The best estimates for USAT may be derived from the Ultrasound Accelerated Thrombolysis of Pulmonary Embolism (ULTIMA) trial by Kucher et al, 2 in which 59 patients who had acute PE and a ratio of right ventricular dimension to left ventricular dimension of 1.0 or more were randomly assigned to receive heparin or USAT (n = 30). Only the USAT group had significant improvement in the ratio, with no measurable increase in bleeding complications. There is, however, no prospective, randomized comparison between USAT and CDT. The study by Lin et al 17 retrospectively analyzed 25 patients with PE. The patients who received CDT and USAT had an improved Miller index, but no method was clearly superior.
Our study has potential limitations. We did not have a formal tool for measuring potential bias, this was a choice made given that the majority of the studies where noncomparative. Thus, the validated risk assessment tools were mostly inadequate for the type of publications. There was a great variation in the type and dose of thrombolytic. Although investigating this variable would have better explained our findings, this information was not sufficiently clarified in the manuscripts in order to create a pooled analysis.
Conclusions
We reviewed the best available estimate to date of using modern mechanisms of advanced PE therapy. Current clinical evidence does not prove USAT is superior over CDT methods.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
