Abstract
Adults with hemophilia A (HA), hemophilia B (HB), and von Willebrand disease (VWD) frequently require surgery and invasive procedures. However, there is variability in perioperative management guidelines. We describe our periprocedural outcomes in this setting. A retrospective chart review from January 2006 to December 2012 of patients with HA, HB, and VWD undergoing surgery or invasive procedures was conducted. Type of procedures, management including the use of continuous factor infusion, and administration of antifibrinolytics were reviewed. Adverse outcomes were defined as acute bleeding (<48 hours), delayed bleeding (≥48 hours), transfusion, inhibitor development, and thrombosis. We identified 59 patients with HA and HB. In all, 24 patients had severe hemophilia and 12 had mild/moderate hemophilia. Twelve patients had inhibitors. There were also 5 female carriers of HA and 6 patients with VWD. There were 34 major surgeries (26 orthopedic, 8 nonorthopedic) and 129 minor surgeries. Continuous infusion was used in 55.9% of major surgeries versus 8.5% of minor surgeries. Antifibrinolytics were administered in 14.7% of major surgeries versus 23.2% of minor surgeries. In all, 4 patients developed acute bleeding and 10 patients developed delayed bleeding. Delayed bleeding occurred in 28.6% of genitourinary procedures and in 16.1% of dental procedures. Five patients acquired an inhibitor and 2 had thrombosis. In conclusion, patients with HA, HB, or VWD had similar rates of adverse outcomes when undergoing minor surgeries or major surgeries. This finding underscores the importance of an interdisciplinary management and procedure-specific guidelines for patients with hemophilia and VWD prior to even minor invasive procedures.
Introduction
Adults with hemophilia and von Willebrand disease (VWD) are living well into adulthood as a result of better access to treatments and safer products to prevent and control bleeding. Hemophilia A (HA) is an inherited deficiency of factor VIII (FVIII), and hemophilia B (HB) results from a deficiency in factor IX (FIX). The most frequent bleeding manifestation is hemarthrosis, which can lead to hemophilic arthropathy requiring joint replacement. von Willebrand disease is an autosomal-dominant (type 1 and type 2) or recessive (type 3) bleeding disorder with a mucocutaneous bleeding phenotype. Patients with hemophilia and VWD comprise the majority of bleeding disorder patients followed in the hemophilia treatment centers (HTCs) in the United States.
Patients with hemophilia and VWD require detailed coordinated care prior to surgery or invasive procedures because of an increased risk of hemorrhagic complications. In most cases, hemostasis can be attained by the administration of factor concentrates, desmopressin (1-desamino-8-
The objective of our study was to evaluate the outcomes of adult patients with hemophilia and VWD undergoing major surgeries or minor surgeries at our HTC.
Methods
The electronic medical records from surgeries and procedures in patients with hemophilia and VWD were reviewed from the Weill Cornell HTC from January 2006 to December 2012. Demographic data were collected including age, gender, classification of bleeding disorder, and type of procedure. Use of preoperative factor bolus, DDAVP, continuous factor infusion (CI), and antifibrinolytics was evaluated. Continuous infusion was administered as a bolus dose of FVIII followed by syringe pump infusion for 24 to 48 hours, with the goal FVIII or FIX activity of 80% to 110% by 1-stage clotting assay. Bolus dosing of FVIII or factor IX was targeted to an activity level of >80% with postoperative dosing goals based on the current WFH guidelines based on postoperative day. 1 In major surgeries, patients received factor dosing out to 7 to 10 days. In minor surgery, patients received factor for a shorter duration, usually 3 to 5 postoperative days. Inhibitor patients received bolus dosing with bypassing agents, either recombinant factor VIIa (rVIIa) or factor eight inhibitor bypassing activity (FEIBA). Outcomes included success of procedure, acute bleeding (<48 hours after procedure), delayed bleeding (≥48 hours after procedure), transfusion of blood products, inhibitor development, and thrombosis. Information on factor dosing, plasma levels attained, and length of factor exposure was collected. Antifibrinolytics were given either orally or intravenously (in the case of major surgery) and continued for 5 to 10 days postoperatively depending on the type of procedure. For example, dental procedures and those involving the oral mucosa were for 10 days.
Blood was collected in sodium citrate (3.2%) and then centrifuged at 4000 RPM to isolate plasma. The von Willebrand factor (VWF) activity was determined by platelet aggregation using the CHRONO-Log 700 (Havertown, Pennsylvania). A standard Bethesda-type inhibitor assay was used to assess inhibitors to coagulation factors and VWF. 7,8
Data were compared using Fisher exact test. All P values are 2-sided with statistical significance evaluated at the .05 alpha level. All analyses were performed in SPSS version 22.0 (SPSS Inc, Chicago, Illinois). This study was carried out after approval by the local institutional review board.
Results
The mean age of our patients was 58 years (range: 21-77 years). Six patients had VWD (3 type 1, 1 type 2A, and 2 type 3). Forty-eight patients had hemophilia (41 HA and 7 HB). Of the 48 patients with hemophilia, 24 were severe (factor activity <1%), 12 were mild or moderate (factor activity 1%-40%), and 12 had inhibitors. In addition, we included 5 female carriers of HA (Table 1). Patients with previously diagnosed high-titer inhibitors had inhibitor titers ranging from 7.1 to 2125 BU (median 24 BU). A total of 163 procedures were identified. There were 34 major surgeries (26 orthopedic, 8 nonorthopedic) and 129 minor surgeries (Tables 2 and 3). All patients received an initial bolus of factor replacement or DDAVP during surgical procedures. Administration of CI was performed in 55.9% of major surgeries and 8.5% of minor surgeries. Antifibrinolytics were used in 14.7% of major surgeries and in 23.2% of minor surgeries, excluding those involving the genitourinary (GU) tract.
Demographics of Study Population.a

Abbreviation: VWF, von Willebrand factor.
aN = 59 patients.
Complications With Major Surgeries.
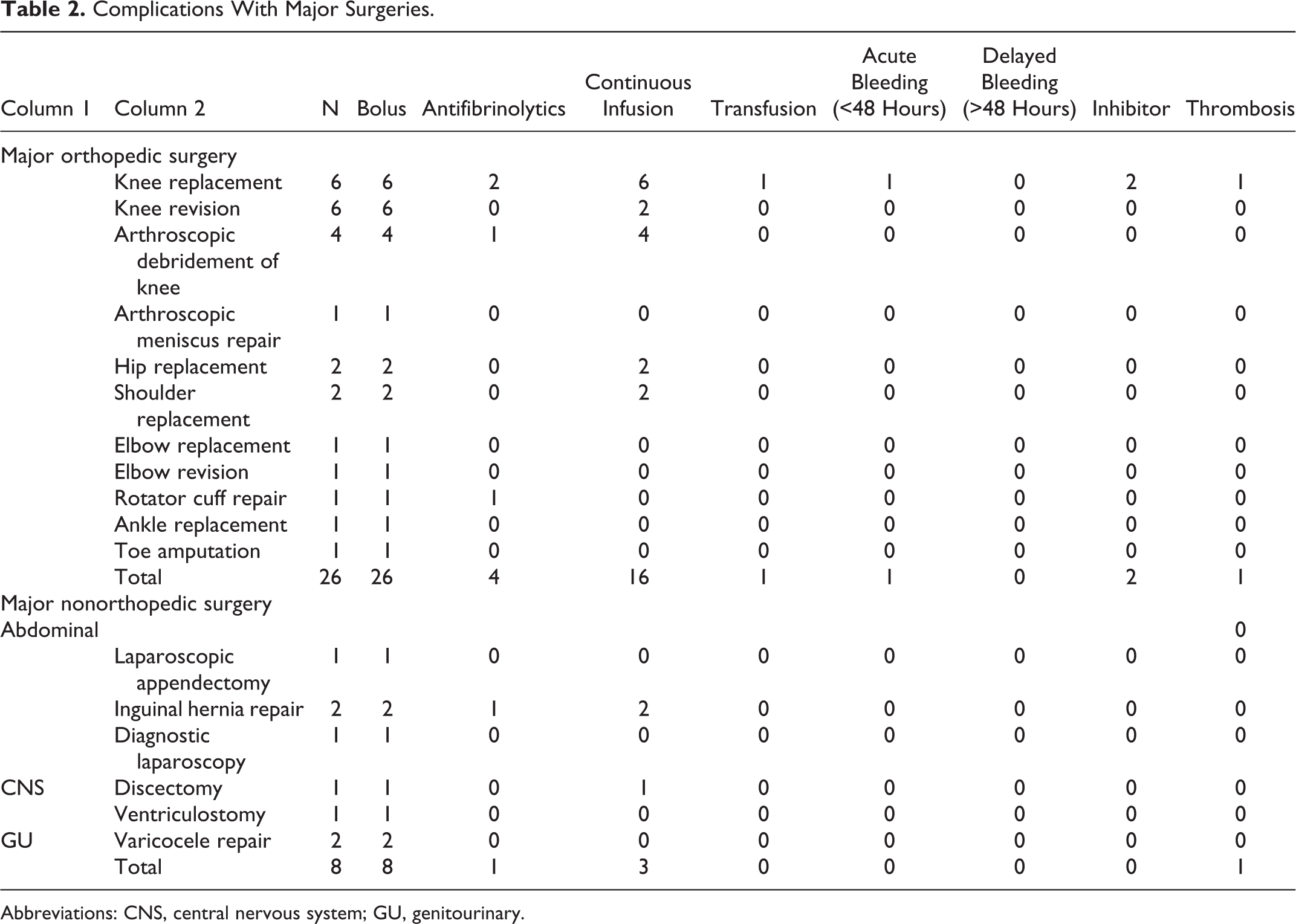
Abbreviations: CNS, central nervous system; GU, genitourinary.
Complications With Minor Surgeries.
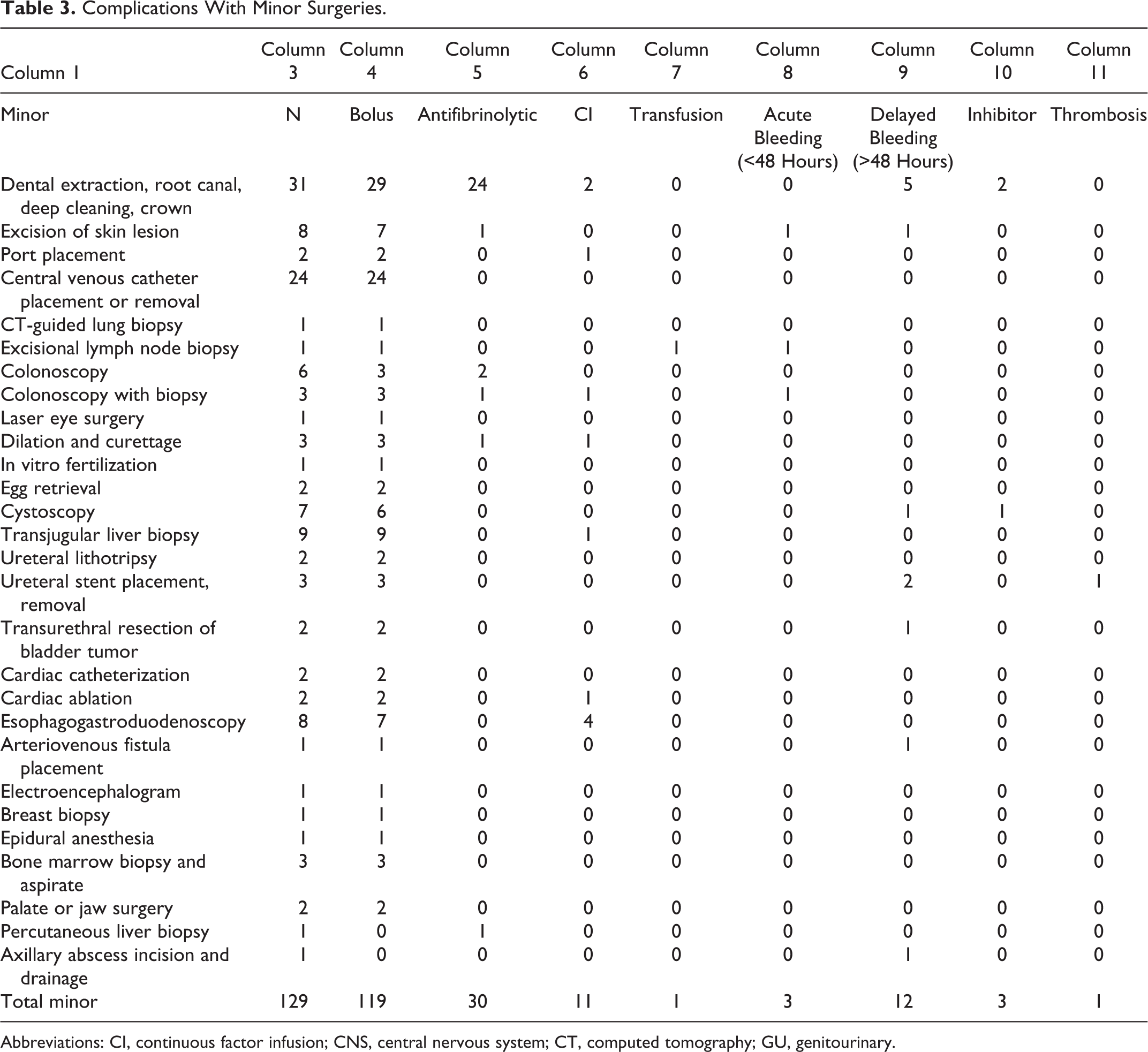
Abbreviations: CI, continuous factor infusion; CNS, central nervous system; CT, computed tomography; GU, genitourinary.
Acute bleeding was observed in only 6 procedures (3.6%): 1 after knee arthroplasty, 1 after colonoscopy with biopsy, 2 after liver biopsies, 1 after skin excision, and 1 post-lymph node biopsy. Delayed bleeding was noted in 4 GU procedures (1 cystoscopy with biopsy, 2 ureteroscopic stent placements, and 1 transurethral resection of bladder tumor), 5 dental procedures (tooth extractions), 1 excisional skin biopsy, 1 arteriovenous fistula placement, and 1 axillary incision and drainage (Tables 3 and 4).
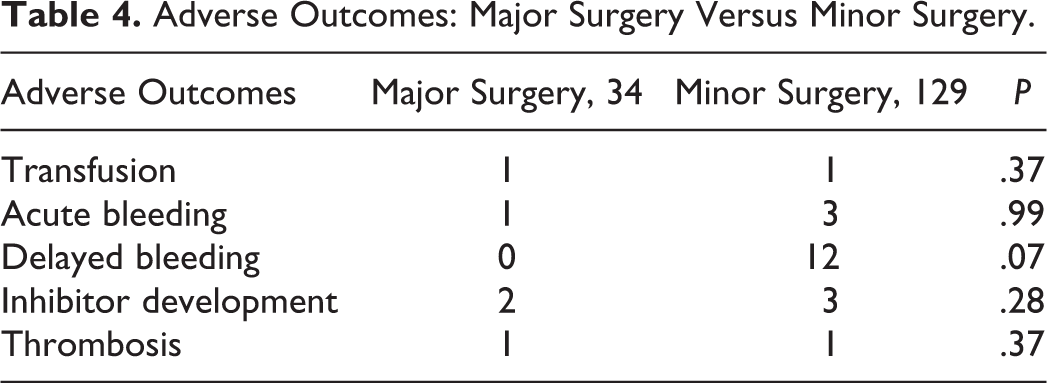
Adverse Outcomes: Major Surgery Versus Minor Surgery.
Inhibitor Development
Five patients (3 with hemophilia and 2 with VWD) developed an inhibitor during the study period. Inhibitor formation occurred in patients undergoing both minor and major surgeries (Table 4).
One patient with mild HA (FVIII 6%) developed a high-titer inhibitor (10.4 BU) after treatment with recombinant FVIII during right knee arthroplasty and prophylaxis during physical therapy. His mutation, Arg2169His, is associated with inhibitor development. 9 He had a family history of hemophilia without inhibitors. He is currently 5 years postsurgery with spontaneous recovery of FVIII activity to 8% and decrease in BU titer to 1.8. He had less than 150 exposure days (EDs) of factor prior to this procedure.
A second patient with severe HA developed an inhibitor 6 EDs after FVIII exposure during right knee arthroplasty. He had a personal history of high-titer inhibitor after laminectomy that resolved after rituximab treatment 7 years earlier. No mutation testing was available for this patient.
A third patient with mild HA (FVIII 8%) received recombinant FVIII prior to cystoscopy and developed an inhibitor (10 BU) after several infusions for ankle trauma 1 month after his procedure. His BU titer is now 0.7 with a detectable FVIII activity of 13%. His mutation, Asp2093His, has also been previously associated with inhibitor development. He also had received less than 150 EDs in his lifetime.
Two patients with type 3 VWD with over 150 EDs developed low-titer VWF inhibitors after dental procedures and plasma-derived VWF exposure. No mutation data are available for these patients. Both patients are currently using plasma-derived VWF with excellent hemostasis and partial resolution of VWF inhibitors.
Thrombosis
Symptomatic thrombosis occurred after 1 major surgery and 1 minor surgery (Tables 3 and 4). One occurred in a patient with severe HA admitted for right knee arthroplasty. He had a prolonged hospitalization for sepsis requiring a central venous catheter. The patient developed a symptomatic basilic vein thrombosis while receiving FVIII replacement. He was managed by observation and serial upper extremity compression ultrasounds. His thrombosis resolved without anticoagulation treatment.
A second patient with HA and an inhibitor was admitted for ureteral stent placement for nephrolithiasis. He received rVIIa for management of hematuria and traumatic elbow hemarthrosis. He subsequently developed asymptomatic right brachial vein thrombosis that was followed by serial ultrasounds and resolved spontaneously. He underwent stent removal without further complications. Systematic screening for asymptomatic thromboses was not performed during this study.
Discussion
In our adult patients with hemophilia and VWD, we demonstrated similar rates of adverse hematologic outcomes after both minor surgeries and major surgeries. Importantly, we also observed a trend toward more delayed bleeding after minor surgeries, especially in the oral and GU mucosa. This finding highlights the significant risks that affect patients with hemophilia and VWD from even minor invasive procedures. Aggressive replacement and treatment may be needed in some cases.
Although guidelines are available to provide adequate hemostasis for patients with hemophilia and VWD undergoing procedures, it is often difficult to take into account the broad range of procedures, potential complications, and concomitant medication use. 1,10 Specific treatment guidelines should be designed based on procedure type and anticipated outcomes. A recent Cochrane review emphasized the lack of adequate data on bleeding complications among patients with underlying bleeding disorders who are undergoing major and minor surgical interventions. 11
Institution-specific strategies are often used in the perioperative setting. Continuous infusion of factor provides several advantages including maintaining a constant factor level, minimizing time below trough factor levels where bleeding can occur, and decreasing overall factor use. 12 –14 Continuous infusion has been associated with inhibitor development in some reports 15 –17 but not others. 18 None of our patients exposed to CI developed inhibitors during the study period.
Unfortunately, CI is not easily used in patients with VWD or hemophilia with inhibitors because of dosing kinetics. All our known inhibitor patients were treated with pre- and postoperative bolus dosing of bypassing agents, however, several of these patients had bleeding complications. Although guidelines exist for surgical management on patients with inhibitors, hematologic and nonhematologic complications are still significant. 19,20 A recent study demonstrated that rotational thromboelastometry (ROTEM) and thrombin generation assays (TGAs) have a great potential to assess the effect of bypassing agents in patients with hemophilia with inhibitors. Thrombin generation assays seemed to be more sensitive than ROTEM in reflecting the difference in treatment response between activated prothrombin complex concentrates and rVIIa. Rotational thromboelastometry and TGA may be useful in the future for optimizing the perioperative care of these patients. 21
Inhibitors occurred in the setting of both minor and major surgeries. This finding is likely due to higher and more frequent dosing of factor during the perioperative period and the observation that non-severe hemophilia A patients remain at risk for inhibitor development. 48 Two of the 3 patients with hemophilia who developed inhibitors during exposure to FVIII had mild HA. Genotyping patients with hemophilia prior to a scheduled surgery or procedure may help in guiding the safety and intensity of factor replacement. 22,23 DDAVP may be used to limit factor exposure, and knowledge of the genotype will also be useful in predicting the response. 24
Patients with VWD types 1 and 2 and patients with mild or moderate HA can be managed with DDAVP for minor procedures. DDAVP binds to vasopressin receptors on the surface of endothelial cells, thereby increasing intracellular calcium, promoting trafficking of Weibel-Palade bodies to the EC plasma membrane. The result is an increase in circulating VWF and FVIII and improved systemic hemostasis. Response criteria derived from the MCMDM-1 VWD are an increase in VWF:RCo and FVIII above 50 IU/kg, whereas a partial response is defined as an increase in either level of less than 50 IU/kg but of at least 3-fold over baseline. 25 –27 The usual 3- to 5-fold increase in FVIII and VWF is associated with bleeding control in 70% of cases with VWD, albeit with variable responses. 25,26,28,29 Potential limitations for the use of DDAVP include its short duration of action and the need for fluid restriction. Perioperatively, fluid restriction is essential as patients may develop hyponatremia and are at risk of seizures. 30 Additionally, thrombosis has been reported in elderly patients using DDAVP. 31,32
We report a number of bleeds with dental procedures, despite the use of antifibrinolytics such as aminocaproic acid. Delayed bleeding is a recognized complication of dental surgeries and appears related to the severity of bleeding disorder and the intensity of the procedure. 33 Previous reports found that despite the use of factor replacement therapy prior to procedures and antifibrinolytic treatment for 5 to 7 days postprocedure, the bleeding rate remained was 1.9%. 3 Recent studies suggest that a postoperative factor trough level of 30% is desirable. However, duration of factor replacement and antifibrinolytic use is not clearly defined. 34 As our patients did not generally have long-term replacement therapy after dental procedures, the bleeding rate was higher than that cited in other studies.
We observed a high rate of cystoscopy bleeding approximately 7 days postprocedure in at least one of our patients even with postprocedure factor infusions. Urologic surgeries, such as transurethral resection of prostate, transurethral resection of the bladder, and prostate biopsies, can be performed with similar bleeding risk to patients without bleeding disorders as demonstrated in a recent study. 6 However, patients with bleeding disorders may experience increased need for irrigation and longer hospital admissions. 6 Hematuria was observed at similar rates both in patients with hemophilia and controls. In our study, there were 14 urologic procedures, 4 of which were complicated by delayed hematuria. It remains uncertain how aggressively postoperative hematuria should be treated in patients with hemophilia and VWD. The use of antifibrinolytics has been traditionally avoided by our center because of the GU obstruction risk, however, several studies suggest this may be a safe approach to treat prolonged hematuria in bleeding disorders. 35,36
We did not report a significant number of bleeding events in non-GU endoscopies, although delayed bleeding has been reported in colonoscopies with biopsies. 2 Antifibrinolytic therapy alone may be used as prophylaxis for patients with mild or moderate hemophilia undergoing gastrointestinal endoscopies without biopsy. 4 This strategy avoids excess factor exposure and the risk of inhibitor development.
Patients with bleeding disorders generally have a lower risk of postoperative thrombosis. The estimated incidence of thrombosis after a major orthopedic surgery is 0.5% in patients with hemophilia. 38 Some experts have recommended pharmacologic thromboprophylaxis after orthopedic surgery only if other risk factors are present, including advanced age, varicose veins, malignancy, inherited thrombophilias, and general anesthesia. 37, 38 We observed no lower extremity deep vein thrombosis or pulmonary embolism in our study group, although systematic screening for asymptomatic events was not performed. Both of our patients with thrombosis had symptomatic upper extremity thrombosis associated with indwelling catheters and inflammation. One was receiving rVIIa, which has been shown to increase thrombosis risk. 39,40 We do not routinely administer pharmacologic thromboprophylaxis to our patients with bleeding disorders regardless of severity, except in very specific cases. No patients in this study received pharmacologic thromboprophylaxis, and no clear consensus exists among treatment centers in the United States or Europe as to the role of this treatment in patients with hemophilia undergoing surgery. 10
Surgeries in patients with VWD have some treatment guidelines. 41,42 Dosing of replacement factor is generally based on levels modified from hemophilia factor goals or based on ristocetin cofactor activity. Some authors have advocated for ristocetin cofactor activity of 50% to 60% for minor and dental procedures and 80% to 100% for major surgeries, 43 although lower doses have been suggested by others for patients with type 2 VWD. 44 Orthopedic surgery can be performed safely, with overall improved joint function and minimal complications in VWD. 45 In the case of VWF-containing concentrates, thrombosis has been reported with persistent increased VWF activity levels (>150%), therefore, dose monitoring is essential. 46 Since most currently available VWF-containing concentrates have FVIII, it is also important to measure FVIII activity. 46 New guidelines for the use of recombinant VWF will also need to be established. 47
This study has several limitations including its retrospective design, small sample size, and a single-center experience that may not be reflective of other centers. Because of the retrospective design and variables in record availability, we were not able to systematically evaluate actual amounts of factor use and instead reported the treatments based on the desired factor activity. This approach is reasonable, given our adherence to World Federation of Hemophilia (WFH) guidelines in most settings related to dosing.
Our rate of postoperative bleeding in minor procedures was higher than that has been reported in other studies. We suggest several possible explanations. First, perioperative underestimation of bleeding risk may contribute to a false sense of safety by the patient, operator, and hemophilia staff, and treatment plans may not be implemented aggressively. We were not able to determine true adherence to our treatment plans in all settings. Secondly, although written treatment plans were provided, HTC personnel were not directly involved in patient care. Finally, insurance coverage issues and provider preferences in long-term use of antifibrinolytics are sources of variability that could directly contribute to delayed bleeding.
In conclusion, adult patients with hemophilia and VWD require a coordinated approach with a multidisciplinary team of hematologists, surgeons, endoscopists, and dentists trained in their care. Physicians should not underestimate the risks of bleeding, thrombosis, and inhibitor development, even in cases where the procedure may be minor. It is of utmost importance that clear treatment plans are outlined, factor replacement is maintained at optimal prespecified levels, and that the HTC remains involved throughout the entire duration of the procedure. Future efforts should adapt the current body of literature to create evidence-based procedure-specific guidelines for adults with these bleeding disorders.
Footnotes
Authors’ Note
JC and MD analyzed data and drafted the manuscript. PC performed data analysis and statistical support. FH and JB performed data collection and analysis. All authors contributed to manuscript preparation. These data were presented in part as a poster abstract at the 55th Annual Meeting of the American Society of Hematology; December 8, 2013; New Orleans, Louisiana.
Acknowledgments
The authors acknowledge Dr John Mitsios, who provided detailed information on the assay measurements for von Willebrand disease.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: JC has accepted consulting fees from Baxalta, CSL Behring, Novo Nordisk, and Bayer.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Dr Paul Christos was partially supported by the following grant: Clinical and Translational Science Center at Weill Cornell Medical College (UL1-TR000457-06).
