Abstract
Introduction
Plasma-derived von Willebrand factor containing FVIII concentrates (pdVWF/FVIII-C) are indicated as replacement therapy for patients with von Willebrand disease (VWD). This study assessed safety and efficacy associated with long-term real-world experience of the pdVWF/FVIII-C, Fanhdi®, in patients with VWD.
Methods
This observational, prospective, post-authorization cohort study was conducted at five centers in Spain. Patients with VWD were treated with the pdVWF/FVIII-C to achieve satisfactory hemostasis for on-demand (bleeding episodes and surgical/invasive procedures) and prophylaxis treatment. Clinical efficacy was evaluated as the response to treatment in both settings. Safety parameters were assessed.
Results
Fifteen VWD patients received at least one dose of the pdVWF/FVIII-C and were followed for 12 months. Forty-six bleeding episodes were reported for 9 (60.0%) patients, and 6 surgical/invasive procedures for 5 (33.3%) patients. Most frequently reported bleedings were gastrointestinal (3 [33.0%] patients) and gynecological (3 [33.0%] patients). No complications nor bleeding episodes related to surgical/invasive procedures were reported. Overall clinical efficacy of treatment (including on-demand and prophylaxis) achieved 100% excellent and/or good (n = 15 patients), being excellent for 7 (46.7%) patients. There were 27 treatment-emergent adverse events in 8 (53.3%) patients, 11 serious adverse events in 3 (20.0%) patients, but none of them were drug-related. No clinical signs and symptoms of immunogenicity or thromboembolic events were reported.
Conclusions
This real-world evidence study confirmed the efficacy of the pdVWF/FVIII-C as on-demand and/or prophylaxis treatment in patients with bleeding episodes or surgical procedures in VWD. Fanhdi® was well tolerated without any safety concerns.
Keywords
Introduction
Von Willebrand disease (VWD) is the most common autosomal hereditary bleeding disorder characterized by a dysfunction or a deficiency of von Willebrand factor (VWF). Under physiological conditions, VWF acts as a stabilizer and carrier of coagulation factor VIII (FVIII) and is involved in primary and secondary hemostasis. 1
VWD clinical manifestations include mucocutaneous bleeding such as epistaxis, menorrhagia, and oral bleeding, although patients with severe VWD tend to suffer hemarthrosis and gastrointestinal bleeding. 2 VWD is classified into three types, from Type 1 (less severe) to Type 3 (nearly total absence of VWF). 3
The aim of VWD treatment is to correct coagulation defects due to low circulating FVIII levels, and reverse abnormal platelet adhesion, resulting in prolonged bleeding time. There are three therapeutic strategies to control bleeding based on VWD type and severity, hemostatic challenge severity, and bleeding nature: non-replacement therapy, replacement therapy, and antifibrinolytic or topical agents.4,5 In non-replacement therapy, patients are treated with desmopressin. 6 In replacement therapy, patients are treated with cryoprecipitate or plasma-derived VWF/FVIII concentrates (pdVWF/FVIII-C) to re-establish platelet adhesion to the vascular sub-endothelium and stabilize and avoid rapid degradation of endogenous FVIII upon binding to it.7,8 Replacement therapy with VWF/FVIII-C is indicated in desmopressin non-responders patients or whom desmopressin is contraindicated.9,10 Lastly, antifibrinolytics promote hemostasis and wound healing without substantially altering VWF plasma concentration. 10
Fanhdi® (Grifols, Barcelona, Spain) is a highly purified, double-inactivated, human pdVWF/FVIII-C. It is presented in vials containing 250, 500, 1000, or 1500 IU of lyophilized FVIII, along with 300, 600, 1200, or 1800 IU of VWF, respectively. Fanhdi® was initially intended for used in patients with hemophilia A. 11 However, pdVWF/FVIII-C is also an effective and safe replacement therapy for patients with VWD, providing adequate hemostasis in surgical procedures, treatment of bleeding episodes, and for long-term prophylaxis.12–16 The development of FVIII inhibitors is a key parameter to be considered when a therapy with a pdVWF/FVIII-C is initiated, because patients with VWD may develop alloantibodies against VWF, presenting lack of hemostatic response. 17 In addition, rare thromboembolic events have been observed when FVIII coagulant activity (FVIII:C) are excessively high (>150 IU/dL), in patients with known thrombosis risk factors. 18
Real-world data are relevant to inform clinical decision-making. Despite the vast clinical experience during the clinical development of Fanhdi®, prospective real-world studies evaluating the product's long-term use are lacking. Therefore, this post-marketing study was designed to further supplement long-term data from clinical studies with that from real-life clinical practice. The aim was to evaluate safety, throughout the clinical assessment of immunogenicity and thrombogenicity, as well as overall clinical efficacy of long-term use of Fanhdi® in patients with VWD during routine clinical use.
Materials and Methods
Study Design
This phase 4, observational, multi-center, prospective, post-authorization cohort study was conducted between February 2019 and August 2022 at five sites in Spain. The study was performed at the request of the Spanish Agency of Medicines and Medical Devices (AEMPS) to examine the safety and clinical efficacy of long-term use of Fanhdi® in patients with VWD according to the current guidelines in routine clinical practice. 19 Data were collected from information captured during routine examinations and treatments performed by the investigator per standard of care, during a 12-month observational period starting with the first pdVWF/FVIII-C infusion. This 12-month follow-up was conducted to ensure a representative experience in the real-world clinical practice.
The study was conducted according to local laws, regulations, International Council for Harmonization, Good Clinical Practice, and the principles of The Declaration of Helsinki. Ethics committee of each participating site reviewed and approved the study protocol and the informed consent form (ICF). Patients signed the written ICF before any study-specific procedure took place.
Patients
The study included both male and female adult patients (≥18 year-old), diagnosed with hereditary VWD of any type and severity, who required replacement therapy with VWF/FVIII-C when desmopressin treatment was ineffective or contra-indicated, with a history of prior VWF concentrates treatment due to bleeding episodes and/or surgery/invasive procedures (on-demand or prophylaxis) and who were expected to experience bleeding episodes and/or surgeries/invasive procedures (including elective surgeries) requiring replacement therapy in the future or with active bleeding at the time of inclusion.
Key exclusion criteria were acquired VWD diagnosis, congenital or acquired platelet function disorder or other concomitant processes interfering with coagulation, positive anti-VWF or anti-FVIII antibodies (≥0.5 Bethesda units) or had been positive during their disease history, drug product intolerance, history of anaphylactic reactions to blood or blood components, participating in another clinical study or having compliance problems with the protocol.
Study Procedures
Patients were intravenously treated with pdVWF/FVIII-C according to the approved labeling for dose and frequency of administration as described in the Summary of Product Characteristics, 40-80 IU/kg of von Willebrand factor ristocetin cofactor (VWF:RCo) activity, and 20-40 IU/kg FVIII:C to achieve satisfactory hemostasis for prophylaxis and treatment of all bleeding episodes, surgical or invasive procedures, and prophylaxis. 11 Efficacy and safety profiles were monitored over a 12-month period post-initial pdVWF/FVIII-C infusion.
Efficacy Endpoints
The pdVWF/FVIII-C efficacy was assessed as on-demand (bleeding episodes and surgical procedures) and long-term (prophylaxis) treatment. For both, data regarding the use of pdVWF/FVIII-C or any blood derivative or antifibrinolytic agents, including the presence of complications during treatment, dose per infusion, number of infusions, and total pdVWF/FVIII-C dose were evaluated.
For bleeding episodes, data about reason (spontaneous or traumatic), and type of bleeding (major or clinically relevant non-major) were collected. According to ISTH guidelines, 20 major bleeding episodes were those with a fall in hemoglobin ≥2 g/dL, transfusion of ≥2 units of packed red blood cells or whole blood, bleeding episodes occurred in a critical location, or caused death. Clinically relevant non-major bleeds were those bleeds not meeting criteria for major bleeding but required any medical intervention. Likewise, data about severity (mild, moderate, or severe), location, and bleeding episode duration (time between first infusion and bleeding cessation) were recorded.
For surgical procedures, number of surgeries per patients, and type of procedure (minor surgery, major surgery, invasive procedures) were assessed. Major surgery was defined as surgical procedure that involved the use of regional or general anesthesia penetrated and exposed one of the major body cavities (eg, skull, chest, neck, abdomen) or one of the major joints (eg, shoulder, hip, spine), or stressed vital organs. Invasive procedures were those procedures that avoided the use of cutting open surgery in favor of closed surgery. Minor surgery was any procedure not considered major.
The hemostasis assessment for on-demand and long-term efficacy of prophylaxis treatment was rated based on a 4-point rating qualitative scale: (1) Excellent, when hemostasis was comparable with that expected for individuals without known bleeding disorders and no increase in FVIII/VWF dosing required; (2) Good, when hemostasis was slightly inferior to that expected for individuals without known bleeding disorders and minor increase in FVIII/VWF dosing required; (3) Poor, when reduced hemostasis was compared with that expected for individuals without known bleeding disorders, a significant increase in FVIII/VWF dosing was required, and there was no need for alternative therapy; (4) No response, when severe bleeding occurred despite FVIII/VWF therapy with a significant increase in FVIII/VWF dosing required and/or need for alternative therapy to control bleeding.
Safety Endpoints
Treatment-emergent adverse events (TEAEs), serious adverse events (SAEs), adverse drug reactions (ADR), clinical laboratory data (hematology, biochemistry), immunogenicity, thrombogenicity, vital signs (temperature, heart rate, respiratory rate, blood pressure) and physical assessments.
Immunogenicity studies aimed to detect the presence and titers of FVIII and VWF inhibitors. The presence and titers of these inhibitors were determined at the investigator's discretion per standard of care. Monitoring assessments of clinical signs and symptoms of thromboembolic events were conducted per standard of care in patients undergoing a surgical or invasive procedure.
Statistical Analysis
The sample size was based on clinical considerations and was not formally calculated. For continuous variables, descriptive statistics included the number of non-missing values, mean, standard deviation (SD), median, interquartile range, minimum and maximum. For categorical variables, descriptive statistics included counts and percentages per category. Both safety and efficacy analyses were performed in the safety population, defined as all patients enrolled in the clinical study who received at least one dose of pdVWF/FVIII-C and met all selection criteria. GraphPad Prism version 10 (GraphPad Software, Inc) was used for charting.
Results
Study Patients
A total of 18 patients were screened. Seventeen of them met all eligibility criteria and were enrolled in the study. Fifteen (88.2%) patients received at least one dose of pdVWF/FVIII-C, were included in the safety population, and completed the 12-month study period (Figure 1). The observed mean (SD) follow-up study period was 372.5 (182) days.

Flow diagram of patients through the study.
Demographics and clinical baseline characteristics are shown in Table 1. Overall, patients were women (60.0%) with a mean (SD) age of 44.7 (19.9) years. Type 2A VWD (46.7%) and type 1 VWD (33.0%) were the most frequently reported types of the disease. Thirteen (86.7%) patients had at least one medical condition. The four most frequently reported system organ classes were gastrointestinal disorders (n = 7, 46.7%), metabolism and nutrition disorders (n = 6, 40.0%), blood and lymphatic system disorders (n = 5, 33.3%), and vascular disorders (n = 4, 26.7%). The most frequently reported preferred terms for medical conditions were gastrointestinal angiodysplasia (n = 4, 26.7%) and hypertension (n = 3, 20.0%). In the 12-month period prior to baseline, seven (46.7%) patients were treated on-demand with FVIII/VWF-C for bleeding episodes, and ten (66.7%) patients were treated with VWF/FVIII-C for prophylaxis.
Demographic and Clinical Characteristics at Baseline in the Safety Population.

Data are expressed as n (%) unless stated otherwise.
Abbreviations: VWF:Ag, von Willebrand factor antigen; FVIII:C, factor VIII coagulant activity; VWF:RCo, von Willebrand factor ristocetin cofactor activity; SD, standard deviation.
Bleeding Episodes
A total of 46 bleeding episodes were reported in 9 (60.0%) patients. Six (40.0%) patients presented ≥4 bleeding episodes, three (20.0%) patients had one bleeding episode, and six (40.0%) patients did not have any bleeding episodes. The mean number of bleeding episodes per patient/month was 0.23.
All bleeding episodes were spontaneous, and most of them, 37 (80.4%), were clinically relevant non-major, and occurred in 7 (77.8%) patients. Regarding severity, 29 (63.0%) bleedings were moderate and occurred in 6 (66.7%) patients. Most frequently reported bleeding locations were gastrointestinal and gynecological, reported by 3 (33.3%) patients each.
Mean bleeding duration lasted 4.8 (4.3) days, and the mean number of pdVWF/FVIII-C infusions per bleeding episode was 2.6, with a mean pdVWF/FVIII-C total dose per bleeding episode of 76.0 IU/kg. There were four (44.4%) patients who received other hemoderivatives or antifibrinolytic agents (Table 2).
Description of Bleeding Episodes and Treatment with the pdFVIII/VWF Concentrate, Fanhdi®, in the Safety Population (n = 15).

Data are expressed as n (%) unless stated otherwise.
Abbreviations: SD, standard deviation; IU, international units.
Major bleed was defined as bleeding with a fall in hemoglobin of ≥2 g/dL, or with transfusion of ≥2 units of packed red blood cells or whole blood, or when occurred in a critical location, or if caused death. Clinically relevant non major was defined as bleeding that did not meet criteria for major bleeding or required any medical or surgical intervention to treat the bleeding.
Descriptive statistics of the average value per patient for all episodes reported (first column) and descriptive statistics per episode (second column).
Surgery or Invasive Procedures
Six surgeries/invasive procedures (3 [50.0%] major and 3 [50.0%] minor) were reported in 5 (33.3%) patients and were treated with the pdFVIII/VWF-C. No complications nor bleeding episodes related to surgical/invasive procedures were reported. None of the patients experienced a bleeding episode during the surgery/invasive procedure. On the patient level, the mean (SD) number of pdVWF/FVIII-C infusions per surgery/invasive procedure was 3.5 (1.7), the mean dose per infusion was 20.2 (8.1) IU/kg and the mean total dose per surgery/invasive procedure was 83.3 (48.4) IU/kg (Table 3).
Description of Bleeding Episodes Associated with Surgical or Invasive Procedures in the Safety Population (n = 15).

Data are expressed as n (%) unless stated otherwise.
Abbreviations: SD, standard deviation
Descriptive statistics of the average value per patient for all surgeries/invasive procedures (first column) and descriptive statistics per surgery/invasive procedure (second column).
Long-Term Overall Clinical Efficacy
A total of 476 pdVWF/FVIII-C infusions were administered during the study period; 358 (75.2%) were administered for prophylaxis, 96 (20.2%) for bleeding, and 22 (4.6%) for surgical/invasive procedures. The mean (SD) total dose per year was 1135.1 (1486.0) IU/kg, being 260.3 (308.4) IU/kg for on-demand, and 1749.6 (1405.3) IU/kg for prophylaxis. The mean number of infusions per patient varied between on-demand (8.4 infusions) and prophylactic treatment (51.1 infusions). Conversely, the pdVWF/FVIII-C dose per infusion for both treatment types were comparable: 28.5 (7.8) IU/kg for on-demand, and 28.5 (4.6) IU/kg for prophylactic treatment.
Global clinical efficacy for on-demand treatment achieved an excellent rating in 7 (50.0%) patients, and good in the other 7 patients (50.0%). For prophylaxis, global clinical efficacy was excellent in 2 patients (28.6%), and good in 5 patients (71.4%). Overall, treatment with pdVWF/FVIII-C had ratings of excellent in 7 (46.7%) patients and good in 8 (53.3%) patients. There were no episodes classified as poor or no response (Figure 2).

Global clinical efficacy of the use of pdVWF/FVIII concentrate, Fanhdi®, as treatment of all bleeding episodes and surgery or invasive procedures (on-demand), and for long-term prophylaxis.
Safety Assessment
A total of 9 (60.0%) patients reported 29 AEs (27 TEAEs and 2 non-TEAEs). In total, 12 SAEs were reported by 3 (20.0%) patients; 11 SAEs were TEAEs. Of the 11 serious TEAEs, nine were related to upper gastrointestinal bleeding (2 [13.3%] patients), one to colitis (1 [6.7%] patient), and one metastasis to liver (1 [6.7%] patient). All SAEs were unrelated to study drug (Table 4).
Overall Summary of Treatment-Emergent Adverse Events (TEAEs) and TEAEs Reported in the Safety Population by Preferred Term (PT) in Patients with at Least one TEAE.
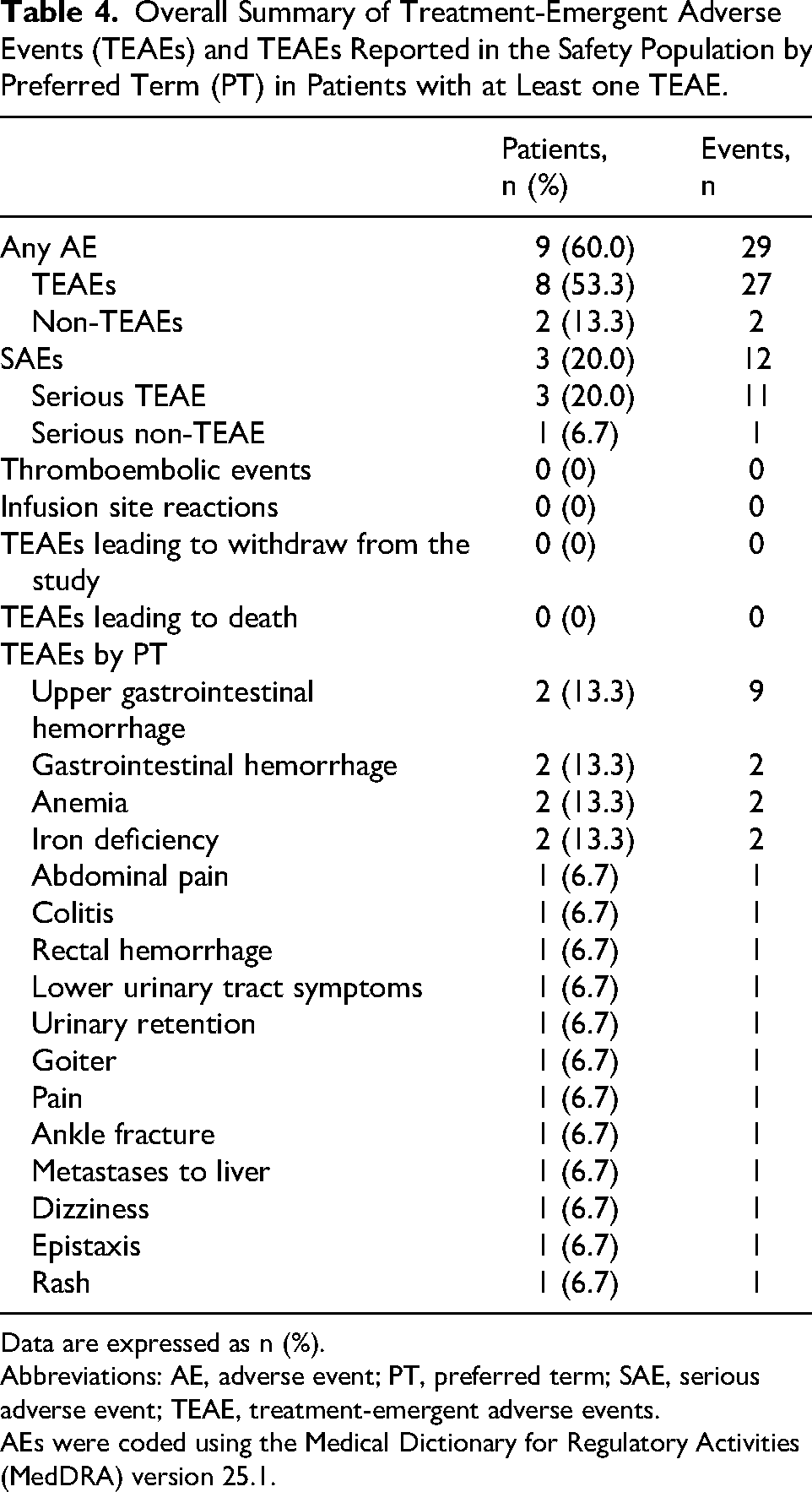
Data are expressed as n (%).
Abbreviations: AE, adverse event; PT, preferred term; SAE, serious adverse event; TEAE, treatment-emergent adverse events.
AEs were coded using the Medical Dictionary for Regulatory Activities (MedDRA) version 25.1.
Regarding intensity, most TEAEs were either mild or moderate. Eight severe TEAEs occurred in 3 (20.0%) patients: 7 events of severe upper gastrointestinal hemorrhage and 1 event of severe metastases to liver. No infusion site reactions, suspected treatment emergent ADRs, or confirmed ADRs were reported. There were 13 and 14 TEAEs temporally associated at 24 h and 72 h, respectively, in five (33.3%) patients. Likewise, there were no TEAEs leading to withdrawal from the study or fatal outcome.
The most frequently reported AEs were upper gastrointestinal hemorrhage (nine events in 2 [13.3%] patients), gastrointestinal hemorrhage, anemia, and iron deficiency (two events in 2 [13.3%] patients each).
Episodes of clinically significant decrease of hemoglobin, hematocrit, and erythrocytes levels were observed in 2 (13.3%) patients. These changes were associated with the reported bleeding episodes and SAEs. No other relevant changes in safety laboratory parameters were observed. No clinically meaningful abnormal vital signs and physical examination findings were reported.
Immunogenicity and Thrombogenicity
There were no clinical signs and symptoms of hypersensitivity or potential immunogenicity phenomena during the study. Immunogenicity tests were not part of the sites’ routine clinical practice and there were no clinical reasons justifying their indication. Patients undergoing surgical or invasive procedures were monitored for thromboembolic signs and symptoms as per the standard care protocol. No thromboembolic events were reported.
Discussion
This post authorization study in patients with VWD demonstrated the efficacy of pdVWF/FVIII-C treatment for prophylaxis and on-demand management of bleeding episodes and during surgeries or invasive procedures in the real-world setting. The favorable pdVWF/FVIII-C safety profile was confirmed with no safety concerns during the study up to the follow-up visit. Therefore, this real-world study contributed further data confirming previously published safety and efficacy results on the pdVWF/FVIII-C.12–16,21
Based on the data reported, pdVWF/FVIII-C was used according to its approved labeling and the target population was medically treated as expected in the VWD management. Although longer durations of follow-up are sometimes feasible, those studies may be retrospective, and as such may present higher variability in patient management due to a limited control over the data collection. 22 The prospective cohort evaluation allowed for robust safety evaluation of any potential clinical signs and symptoms of immunogenicity and thrombogenicity and other potential safety signals.
The efficacy assessment of any pdVWF/FVIII-C requires the enrollment of patients with severe VWD, defined as those with baseline VWF:RCo below the threshold of 15-20 IU/dL. 23 This study enrolled patients with any type and severity of VWD to provide representative data on the efficacy of pdVWF/FVIII-C in real-world clinical practice. Importantly, four patients had a mean VWF:RCo levels of 20.2 IU/dL at baseline, indicating that severe patients (more prone to suffer bleeding events) were also represented in this study. Although this representation may seem small, it aligns with previously reported prevalence rates,24,25 highlighting the rarity of severe VWD cases. Nine patients reported 46 bleeding episodes, including 2 (13.3%) patients with the severe form, type 3 VWD. Most of bleeding episodes were of moderate severity and few pdVWF/FVIII-C infusions (mean of 2.6 infusions/episode) were required to achieve an adequate hemostasis. Furthermore, 80.4% of the bleeding episodes did not require any other hemoderivative agent to control the bleeding. Notwithstanding, one patient reported a severe major upper gastrointestinal bleeding episode, which was reported as a SAE. Four patients who experienced an upper gastrointestinal bleeding episode required additional antifibrinolytic agents or hemoderivatives. These patients had angiodysplasia as coexisting medical condition, which might have affected their bleeding tendency. In fact, the management of patients with recurrent angiodysplasia is challenging in VWD. 26 Remarkably, none of these events were considered related to pdVWF/FVIII-C treatment.
The management of bleeding episodes during surgery depends on the VWD subtype, bleeding severity, procedure bleeding risk, and response to treatment. 27 This study covered patients with different severe VWD subtypes and bleeding severity, with an excellent and good hemostatic effect of the pdVWF/FVIII-C. In a total of six surgery procedures (3 major and 3 minor), the mean number of infusions per surgery (3.7) was lower than other VWF/FVIII-C.28,29 Overall, an adequate hemostasis was achieved in 80.0% of patients (83.3% of surgical/invasive procedures) without requiring any other hemoderivative and without complications related to the procedure. The pdVWF/FVIII-C has previously shown excellent bleeding control during surgical procedures rating excellent or good in 85.7% of major and minor surgeries. 30 Similar efficacy rates were observed with other pdVWF/FVIII-Cs.7,31
The clinical assessment of hemostasis and long-term overall clinical efficacy for on-demand treatment showed excellent (50.0%) or good (50.0%) responses for 100% of the patients. These results were aligned with a recent study with the same pdVWF/FVIII-C, which found a global clinical efficacy of 98.5% (52.1% excellent and 46.4% good) to treat bleeding episodes, 21 and with other VWF/FVIII-C. 32 Similarly, the investigators rated 100% of the outcomes as excellent (28.6%) or good (71.4%) for prophylaxis. Consistent with the literature, the same rating of excellent/good of the secondary long-term prophylaxis programs has been observed. 21
The pdVWF/FVIII-C has been used safely in multiple previous studies, supporting the favorable safety profile of the product.14–16,21,30 In the present study, there were no safety issues related to study drug infusion and no patients discontinued the study after receiving pdVWF/FVIII-C. The most frequent TEAEs were gastrointestinal hemorrhage, anemia, and iron deficiency, which were related to previous medical conditions. Importantly, none of the patients experienced suspected ADRs, and no deaths were observed during the study.
The development of alloantibodies to VWF represent a serious complication that can lead to loss of VWF/FVIII-C hemostatic response. 17 An interesting finding of the current study was that none of the patients developed clinical signs and/or symptoms of anti-VWF or anti-FVIII antibodies. Indeed, the antibodies tests were not performed because there were no clinical manifestations of hypersensitivity or potential signs/symptoms of immunogenicity to justify their indication.
Thromboembolic complications have been observed after repeated administration of VWF/FVIII-C which results in excessively high FVIII or VWF levels (>150 IU/dL), especially during treatment with low-purity VWF-C.7,18,33 Therefore, thrombogenicity needs to be assessed during the safety evaluation of VWF/FVIII-C. Remarkably, there were no thromboembolic events reported in this study among surgeries or invasive procedures, even during major surgeries. Altogether, the results indicated the pdVWF/FVIII-C safety profile was in line with the current product knowledge, 30 and with other pdVWF/FVIII-Cs.34,35
Our study does entail some limitations. For instance, the limited sample size of 15 patients, and especially the number of surgery or invasive procedures (6 procedures) may restrict the generalizability of the study's findings. A larger cohort would indeed yield more reliable data. In addition, plasma levels of VWF antigen, FVIII:C, and VWF:RCo were only collected at baseline and were not consistently reported during the study period, which constrains the understanding of treatment effects. Due to the observational nature of study, no interventions, such as obtaining specific laboratory data on immunogenicity to detect the presence and titers of FVIII and VWF inhibitors, could be performed. Therefore, data were obtained at the discretion of the investigator's standard of care according to their local clinical practice, in which if there was no medical indication to perform the mentioned tests, the assays were not conducted. This limitation restricts the in-depth assessment of potential immune responses in our study.
Conclusion
Overall, this study supports the safety and efficacy of pdVWF/FVIII-C (Fanhdi®) treatment in VWD for prophylaxis and/or on-demand treatment according to the standard clinical practice for bleeding episodes and/or surgery/invasive procedures. Fanhdi® was well tolerated without immunogenic or thromboembolic events reported during the 12 months follow-up, and without safety concerns.
Footnotes
Acknowledgements
Eugenio Rosado, PhD and Jordi Bozzo, PhD, CMPP (Grifols) are acknowledged for medical writing and editorial support in the preparation of this manuscript. The authors thank patients for their indispensable contribution.
Author Contribution
O. Benítez Hidalgo, C. Marzo Alonso, F.J. López Jaime, M. Carrasco, and M. M. Nieto Hernández: data collection, investigation, resources, and writing - review & editing.
N. Afonso, A. Lei, M. E. Aragonés, and M. Torres: data management and review, visualization, project administration, data interpretation and writing - review & editing; D. Whyms: formal analysis, visualization, validation, and writing - review & editing; K. Hanna, and J. Oliveras: conceptualization, methodology, supervision, and writing - review & editing. All authors critically revised, edited and approved the final manuscript.
Declaration of Conflicting of Interest
N. Afonso, A. Lei, D. Whyms, K. Hanna, M.E. Aragonés, M. Torres, J. Oliveras are full-time employes of Grifols and have no other competing interests to declare. The remaining authors stated that they had no interests which might be perceived as posing a conflict or bias.
Ethical Considerations
The study was conducted according in full conformance with appropriate local laws, regulations, International Conference on Harmonization, Good Clinical Practice and to the principles of The Declaration of Helsinki. Ethics committee of each participating research site reviewed and approved the study protocol and the informed consent form.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: OBH, CM, FJL, MC, MN were study investigators and received financial support for the data collection. This study was supported by Grifols, manufacturer of the pdVWF/FVIII concentrate, Fanhdi®.
Patient Consent Statement
Written informed consent form for the complete study was obtained from each patient at the screening visit before any study-specific procedure took place.
