Abstract
Objective:
Clopidogrel is a clinically important oral antiplatelet agent for the treatment or prevention of cerebrovascular disease. However, different individuals have different sensitivities to clopidogrel. This study assessed variants of different genes for association with response to clopidogrel, clinical outcome, and side effects in patients with ischemic stroke (IS).
Methods:
We consecutively enrolled 375 patients with IS after they received clopidogrel therapy, and venous blood samples were subjected to genotyping allelic variants of genes modulating clopidogrel absorption (ATP binding cassette subfamily B1,
Results:
Adverse events occurred in 37 patients (31 had IS recurrence, 4 died, and 2 had myocardial infarction) during the first 6 months of follow-up. Single locus analysis showed that only the
Conclusion:
Sensitivity of patients with IS to clopidogrel and clopidogrel-induced adverse clinical events may be multifactorial but is not determined by single gene polymorphisms.
Introduction
Stroke is a significant worldwide health problem and is one of the leading causes of death among the elderly individuals. 1 Patients with an ischemic stroke (IS) are also at a high risk of developing a recurrent stroke. To date, antiplatelet therapy using aspirin or clopidogrel is highly recommended for these patients. 2,3 A previous study demonstrated that clopidogrel was shown to be superior to aspirin to reduce the risk of IS. 4 However, a proportion of patients receiving clopidogrel treatment did experience recurrent ischemic stroke (RIS) or other vascular events, 5 referred to as clopidogrel resistance (CR), that is, poor or no response to clopidogrel treatment. 6 Several other studies also showed that reduced response to clopidogrel occurred in 4.8% to 51% of such patients. 7,8 This high variance can partially be attributed to the variability in study design and poor comparability among different measurement techniques, but genetic variants may also be responsible. However, the underlying mechanisms leading to a poor response to clopidogrel therapy have not yet been fully elucidated and are most likely multifactorial. For example, patient compliance, absorption or metabolic dysfunction, drug interactions, diabetes, or hypertension may influence the response to clopidogrel. 9 Thus, the genetic etiology of CR has also been proposed. 10
Clopidogrel is a prodrug requiring intestinal absorption and metabolic activation in the liver, then irreversibly blocking the binding of adenosine diphosphate (ADP) to the platelet
Materials and Methods
Ethical Statement
A protocol for this prospective study was reviewed and approved by the Ethics Committees of The People’s Hospital of Deyang City and The Third Affiliated Hospital of Wenzhou Medical College. Written informed consent was obtained from each participant before they were enrolled into this study.
Study Population
We consecutively enrolled 375 patients with IS with first-time stroke from these 2 hospitals between June 2014 and January 2015. These patients had IS within 72 hours and were treated with clopidogrel. The inclusion criteria were as follows: (1) Patients were ≥40 years of age; (2) patients had IS-related atherothrombotic or small artery disease according to the Trial of Org 10172 in Acute Stroke Treatment classification system 30 ; (3) patients had not taken clopidogrel for at least 7 days before admission; and (4) patients had a National Institutes of Health Stroke Scale (NIHSS) score <15.The exclusion criteria were (1) individuals who declined to participate in this study; (2)patients had an allergy to clopidogrel; (3) patients had cardiac cerebral embolism or any other determined or undetermined etiology of IS; (4) patients had undergone thrombolytic or anticoagulation therapy with warfarin or heparin within 7 days; (5) patients had received a proton pump inhibitor before or during hospital admission; (6) patients had hemorrhagic stroke, hematological diseases, autoimmune diseases, or other severe concomitant diseases; and (7) patients had a platelet count < 1 × 1011/L or > 4.5 × 1011/L.
All patients received standard therapies based on standard guidelines of treatment protocol, 2 that is, 75 mg clopidogrel (Sanofi Company Ltd., Beijing, China) once daily or clopidogrel (75 mg, once daily) plus aspirin (200 mg, once daily; Bayer Healthcare Company Ltd., Beijing, China), for the initial 2 weeks in patients with minor stroke (NIHSS score ≤ 3) or symptomatic carotid or intracranial artery stenosis, followed by treatment with clopidogrel alone (75 mg, once daily) for at least 6 months. Data on various risk factors, including body mass index, body weight, current smoking, diabetes mellitus, and hypertension were recorded; fasting blood samples were assessed for blood sugar, total plasma cholesterol, triglycerides, low-density lipoprotein cholesterol, high-density lipoprotein cholesterol, and homocysteine.
Adenosine Diphosphate-Induced Platelet Aggregation Assay
All patients were subjected to ADP-induced platelet aggregation assay before and after 7 to 10 days of clopidogrel administration. Platelet aggregation level was measured by light transmittance aggregometry. The procedure and the consistency were assessed according to our previous studies.
31,32
In brief, 5 mL of the fasting venous blood were collected in 660 μL of 3.8% sodium citrate and centrifuged at 200×
Selection and Genotyping of Single-Nucleotide Polymorphism
In this study, we selected a total of 12 single-nucleotide polymorphisms (SNPs) of relevant genes, including
The genotyping of these SNPs was performed in genomic DNA extracted from periphery blood samples using the matrix-assisted laser desorption/ionization time of flight mass spectrometry method according to our previous studies. 33,34 Each SNP was amplified using 2 specific polymerase chain reaction (PCR) primers and 1 extension primer (Table S1). Genotype call was performed in real time with Mass ARRAY RT software version 3.0.0.4 and analyzed using Mass ARRAY Typer software, version 3.4 (Sequenom Inc., San Diego, CA).
Each allele of these
Assessment of Clinical Outcome
Follow-up for each patient took place every month at our outpatient clinic for a period of 6 months. Drug compliance was monitored during outpatient visits or via telephone interview. Clinical data were collected from patients’ medical records or via phone interview by the referring physicians.
The primary adverse outcome of clopidogrel treatment was a composite of recurrent ischemic stroke (RIS), myocardial infarction (MI), and death during the first 6 months. Recurrent ischemic stroke was defined as a new focal neurologic deficit of vascular origin lasting for at least 24 hours, which was proved to be nonhemorrhagic by either computed tomography or magnetic resonance imaging scanning. Death was defined as vascular mortality due to MI, IS, and other vascular causes. Side effects included hemorrhagic episodes occurred within 6 months after treatment and the later was defined as the presence of any of the following: (1) symptomatic or asymptomatic hemorrhagic transformation (HT), symptomatic or asymptomatic intracerebral hemorrhage (ICH) and (2) extracranial hemorrhages (eg gastrointestinal bleeding, hematoma, hematuria, and skin or mucosal bleeding). Serious hemorrhage was considered as any symptomatic intracranial hemorrhage or any hemorrhage requiring blood transfusion or prolonged hospitalization.
Statistical Analysis
A χ2 test was performed to analyze the deviation of Hardy-Weinberg equilibrium for genotype frequencies and to compare genotype frequencies. Continuous variables were analyzed using Student
A significant independent predictor of CR was analyzed using a logistic regression analysis. The relative risk of a genotype with CR was expressed using odds ratio (OR) with a 95% confidence interval (CI). The Cox proportional hazards model was used to describe the risk of primary efficacy outcome and reported as values of the hazard ratio (HR) with 95% CI. All statistical analyses were performed using SPSS 16.0 (SPSS Inc, Chicago, Illinois), and all tests were 2 sided. A
Results
Characteristics of Patients
Among 375 patients with IS, 153 (40.8%) showed CR, whereas 222 (59.2%) showed CS. Clopidogrel resistance was significantly associated with greater age (
Baseline Characteristics of Patients With Ischemic Stroke.
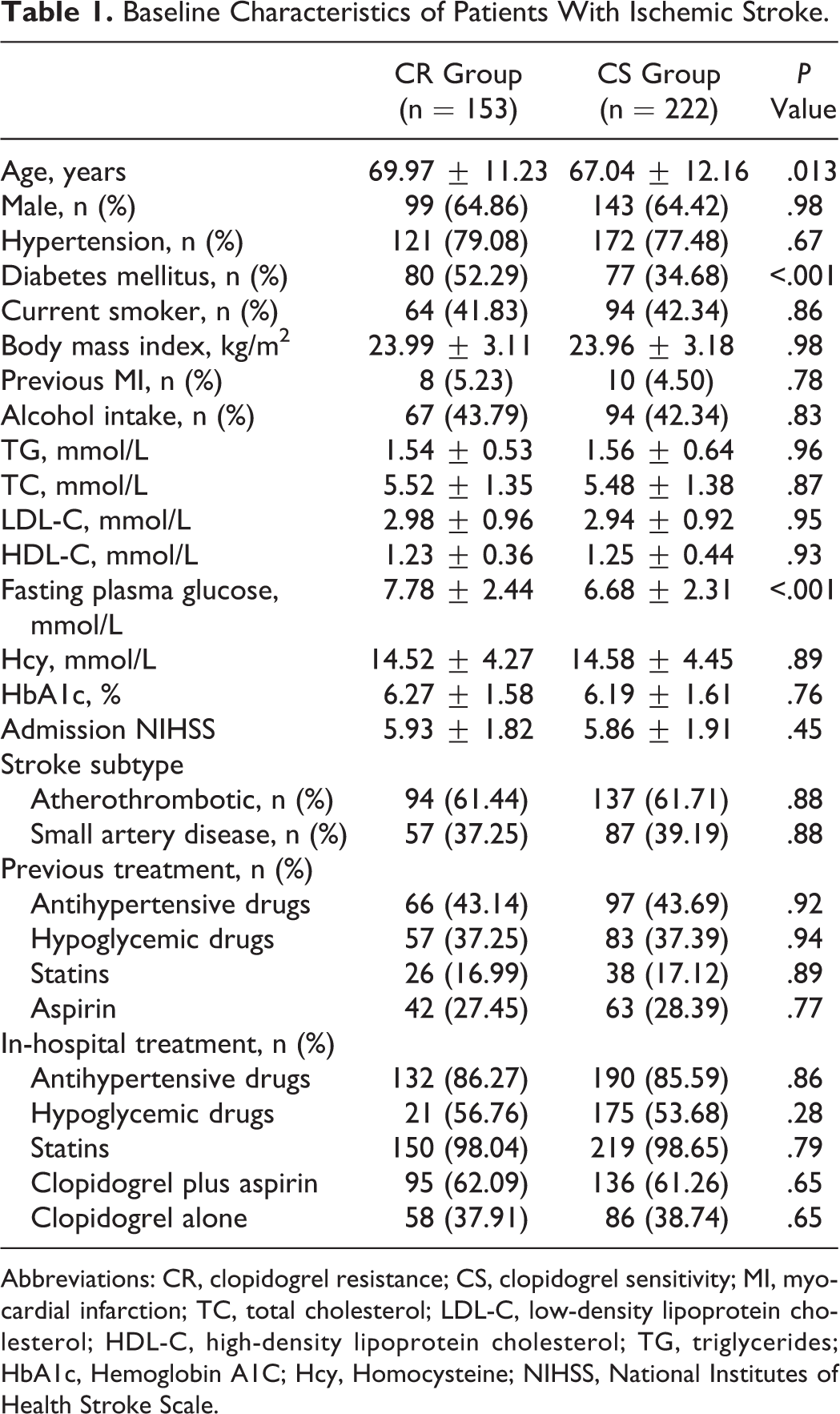
Abbreviations: CR, clopidogrel resistance; CS, clopidogrel sensitivity; MI, myocardial infarction; TC, total cholesterol; LDL-C, low-density lipoprotein cholesterol; HDL-C, high-density lipoprotein cholesterol; TG, triglycerides; HbA1c, Hemoglobin A1C; Hcy, Homocysteine; NIHSS, National Institutes of Health Stroke Scale.
Allelic Frequency, Gene–Gene Interaction, and Association With CR
The genotype distributions of these 12 genetic variants were in Hardy-Weinberg equilibrium. The allelic frequencies of rs701265, rs1439010, rs1371097, rs776746, rs4244285, rs16863323, rs2317676, and rs11871251 differed significantly between patients with and without CR (Table 2). However, only the
Association of SNPs With Responses of Clopidogrel Treatment in Patients With IS, n (%).
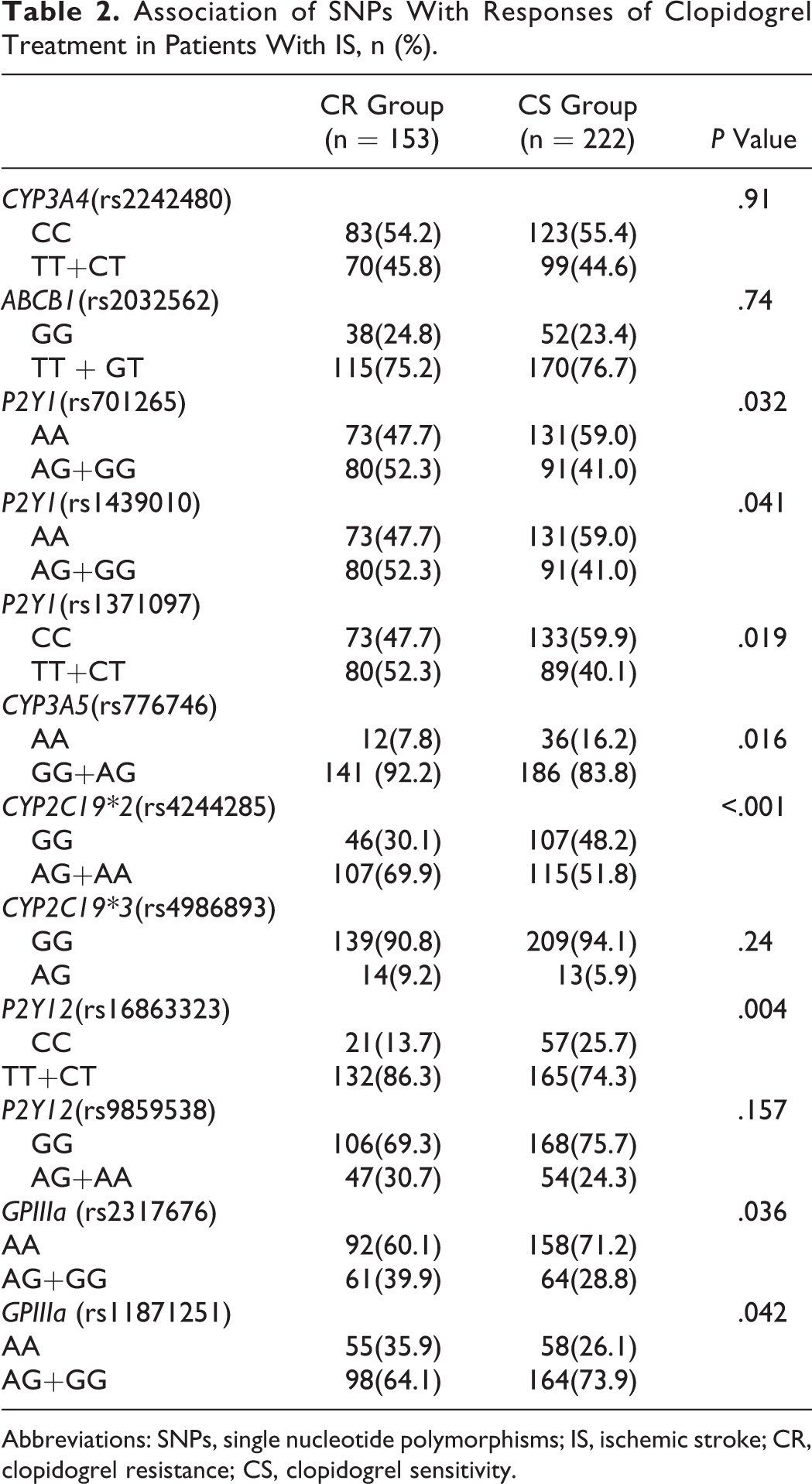
Abbreviations: SNPs, single nucleotide polymorphisms; IS, ischemic stroke; CR, clopidogrel resistance; CS, clopidogrel sensitivity.
We then assessed the association of the high-order interactions of SNPs with CR using the GMDR method. After being adjusted with covariates, the best model for CR included rs4244285, rs16863323, and rs2317676, which scored10 of 10 for cross-validation consistency and 9 of 10 for the sign test (
Associations Between Cerebral Infarction and Genotype Combinations.
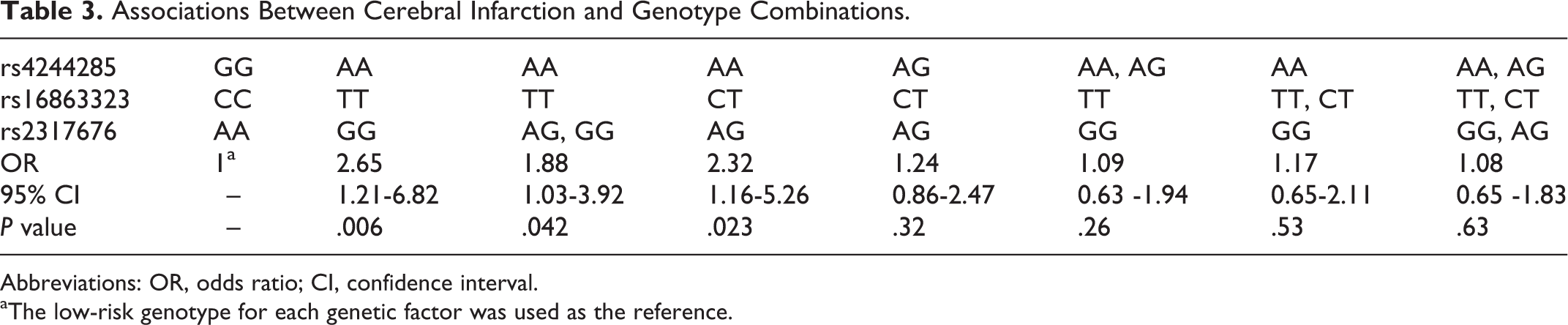
Abbreviations: OR, odds ratio; CI, confidence interval.
aThe low-risk genotype for each genetic factor was used as the reference.
Major Adverse Events in Patients With IS After Clopidogrel Treatment
Among these 375 patients treated with clopidogrel, 363 completed a 6-month follow-up; 149 (97.4%) of 153 patients showed CR, and 214 (96.4%) of 222 patients showed CS. Primary adverse events occurred in 37 patients (31 had RIS, 4 died, and 2 had MI) during the first 6-month follow-up. Clinically, patients with adverse events were older and had a higher prevalence of diabetes mellitus (Table 4). There was also a higher rate of adverse events in patients carrying the rs4244285 variant allele (genotype AA or AG) than in those with the wild-type allele (genotype GG). There was a higher rate of adverse events in patients carrying the rs776746 variant allele (genotype GG or AG) than in those with the wild-type allele (genotype AA). High-risk interactive genotypes (rs4244285, rs16863323, and rs2317676) were also associated with a higher rate of adverse events compared to genotypes with no such interaction (Table 4). The frequency of primary adverse effects and RIS was higher in patients after CR than those with CS (Table 5). There was no significant difference in the rate of extracranial hemorrhage, asymptomatic ICH, and asymptomatic HT between the CR and the CS groups (Table 5). There were no serious hemorrhage, symptomatic HT, or symptomatic ICH events observed in either of the 2 groups of patients.
Characteristics of Patients With or Without Adverse Events.a
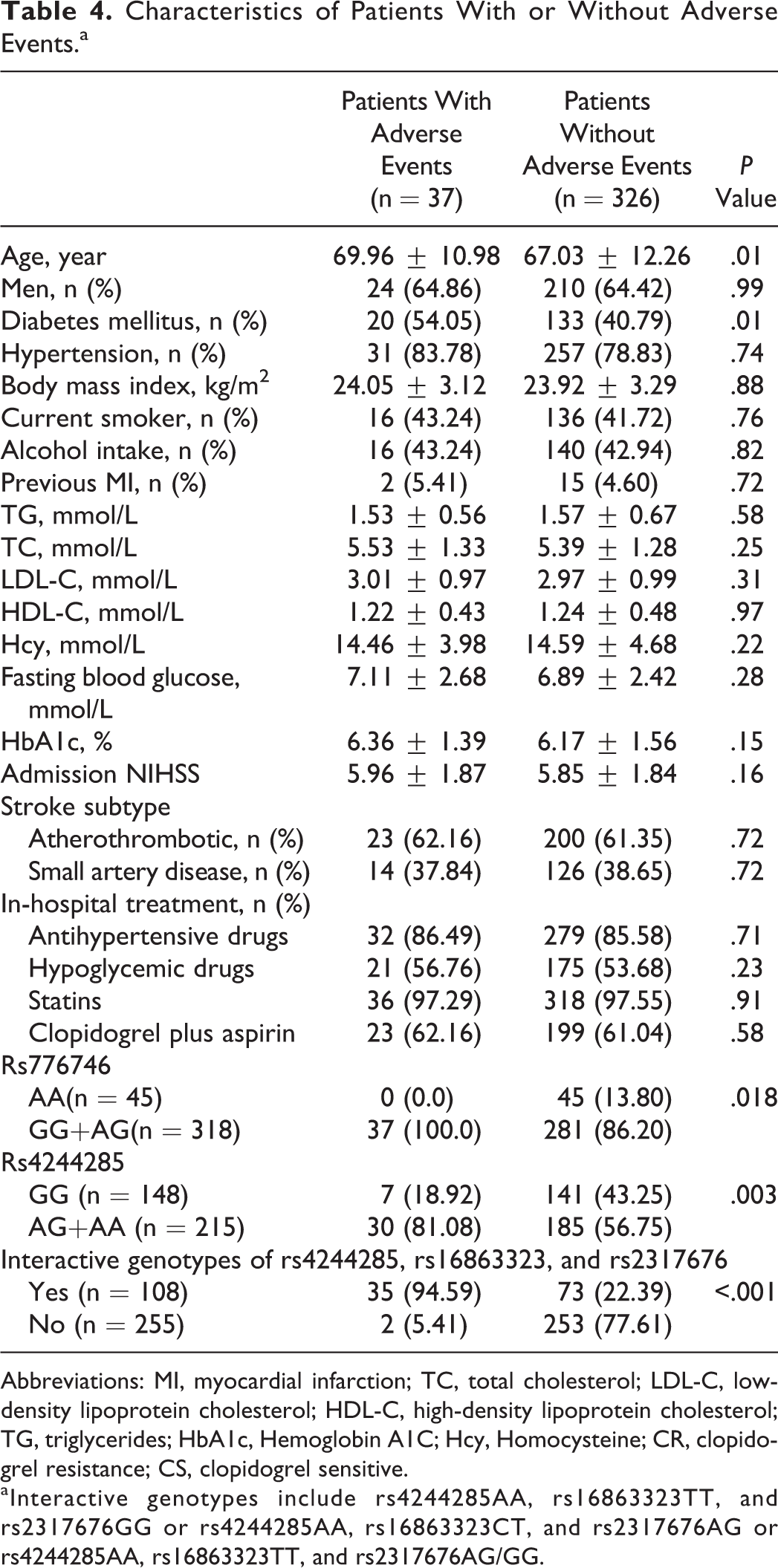
Abbreviations: MI, myocardial infarction; TC, total cholesterol; LDL-C, low-density lipoprotein cholesterol; HDL-C, high-density lipoprotein cholesterol; TG, triglycerides; HbA1c, Hemoglobin A1C; Hcy, Homocysteine; CR, clopidogrel resistance; CS, clopidogrel sensitive.
aInteractive genotypes include rs4244285AA, rs16863323TT, and rs2317676GG or rs4244285AA, rs16863323CT, and rs2317676AG or rs4244285AA, rs16863323TT, and rs2317676AG/GG.
Association of CR With its Adverse Clinical Outcomes.
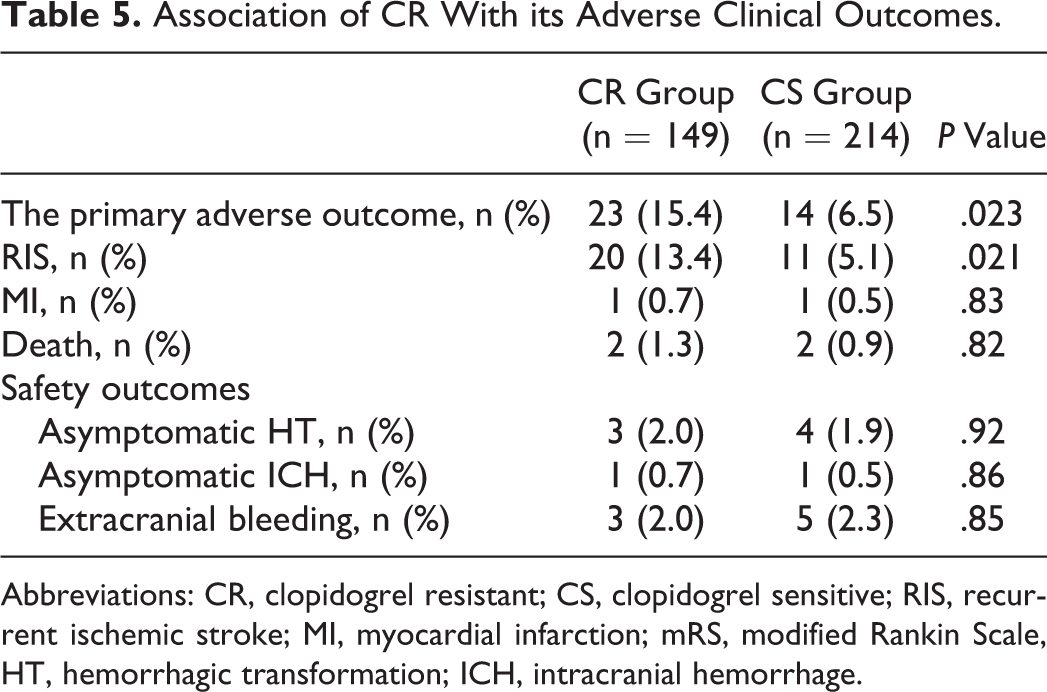
Abbreviations: CR, clopidogrel resistant; CS, clopidogrel sensitive; RIS, recurrent ischemic stroke; MI, myocardial infarction; mRS, modified Rankin Scale, HT, hemorrhagic transformation; ICH, intracranial hemorrhage.
Furthermore, Cox regression analysis revealed that diabetes mellitus (HR = 1.66, 95%CI: 1.12-3.84,
Cox Regression Analysis of Independent Predictors for Adverse Events.a
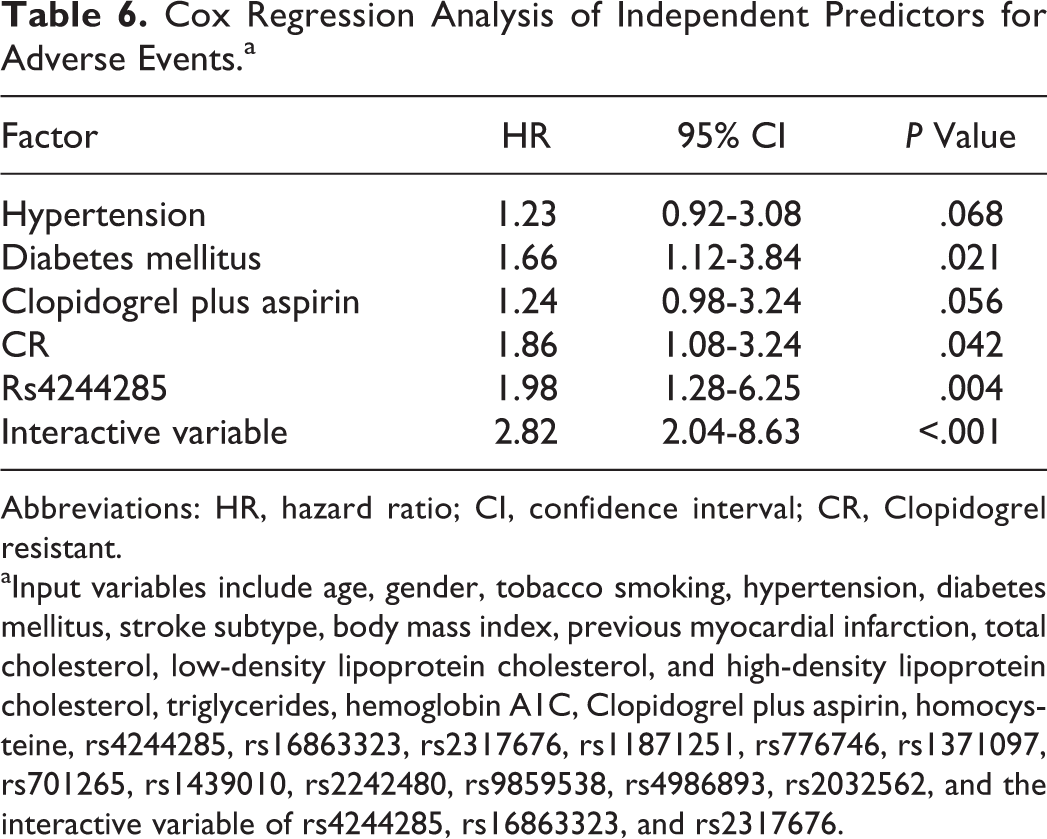
Abbreviations: HR, hazard ratio; CI, confidence interval; CR, Clopidogrel resistant.
aInput variables include age, gender, tobacco smoking, hypertension, diabetes mellitus, stroke subtype, body mass index, previous myocardial infarction, total cholesterol, low-density lipoprotein cholesterol, and high-density lipoprotein cholesterol, triglycerides, hemoglobin A1C, Clopidogrel plus aspirin, homocysteine, rs4244285, rs16863323, rs2317676, rs11871251, rs776746, rs1371097, rs701265, rs1439010, rs2242480, rs9859538, rs4986893, rs2032562, and the interactive variable of rs4244285, rs16863323, and rs2317676.
Discussion
In the current study, we analyzed different gene variants in blood samples from patients with IS having received clopidogrel therapy for their association with CS and adverse events, risk of IS recurrence, myocardial infarction, and death during 6 months of follow-up. Our data showed that there were 37 patients with adverse events: Single locus analysis showed that the variant
Indeed, clopidogrel is a prodrug, and in order to become active, it must be metabolized in the liver by several
Furthermore, the
Currently, there are no standardized treatment recommendations for CR. The dosing regime showed that the response to clopidogrel was beyond pharmacokinetics and
However, our current study does have several potential limitations. For example, it did not include data on the assessment of plasma clopidogrel levels and its active metabolite. The current study may also have a possible bias due to its relatively small sample size and short follow-up period. Moreover, although we genotyped multiple known functional variants in clopidogrel-relevant genes in this cohort of patients, some rare functional variants may have been left undetected in this population; thus, we were not able to exclude their role in the regulation of CR. In addition, the exact pathological mechanism of these gene–gene interactions or clopidogrel activity remains unclear, although we found that gene–gene interactions were involved in clopidogrel metabolism and biologic activity. Future studies with a larger set of genetic variants, a multicenter, large sample size, and a longer follow-up period should be conducted to elucidate the full extent of gene–gene interaction effects on CR.
In conclusion, our current study showed that gene–gene interaction among
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported in part by grants from the Deyang City Science and Technology Research Foundation (#2014SZ035) and the Scientific Research Foundation of Sichuan Provincial Health Department (#140025).
Supplemental Material
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
