Abstract
Background:
In randomized controlled trials, non-vitamin K antagonist oral anticoagulants (NOACs) demonstrated noninferiority to vitamin K antagonists (VKAs) in patients who spent limited time in therapeutic range (TTR). In real-life patients, TTR is known to vary significantly across countries and health-care settings.
Objective:
We aim to evaluate the quality of VKA treatment in Swiss primary care (PC) by comparing patients’ median TTR to levels achieved in the phase III NOAC trials RE-LY, ROCKET-AF, ARISTOTLE, and ENGAGE-AF-TIMI 48. Patient characteristics affecting TTR control shall be estimated.
Methods:
This is a retrospective longitudinal study in Swiss PC patients receiving VKA for ≥6 months. We identified patients from the PC research database FIRE (Family medicine International Classification of Primary Care Research using Electronic medical records) and calculated TTR according to Rosendaal formula. Comparative data from NOAC trials were retrieved from medical literature. Linear regression models were used to assess predictors of TTR.
Results:
Primary care encounters of 215 patients were analyzed. Like in the NOAC trials, median observation period was 2.2 years, but patients were older (67.9% vs 38% ≥75 years) and differed in terms of concomitant diseases and drugs. Median TTR was 75% (65% in the NOAC trials). Female sex was independently associated with a lower TTR and significantly modified by increasing age.
Conclusion:
Practitioners should consider that patients in NOAC trials are only partly representative of PC patients, particularly in terms of TTR control. Only a minority of the observed patients would require a therapy switch to NOACs due to inadequate TTR. Further research is needed in order to identify specific features of care management that are associated with these outcomes.
Introduction
Optimal management of long-term anticoagulant treatment for the prevention and treatment of thrombosis, cardiovascular, and cerebrovascular events is subject of increasing interest, mainly due to the availability of non-vitamin K antagonist oral anticoagulants (NOACs). The introduction of NOACs questioned the usefulness of the vitamin K antagonists (VKAs), whose efficacy and safety are known to depend largely on the quality of anticoagulation control. Reports on patients’ time spent in therapeutic range (TTR) of international normalized ratio (INR) level in daily practice of various countries suggest that the quality of anticoagulation control with VKA is poor, especially in primary care settings. 1 Reported TTR commonly ranges between 52% and 64%, but also lower values (29%) have been observed. 2 –24
Specialized anticoagulation clinics (ACCs) and patients’ self-monitoring tend to show better outcomes. However, in light of the high prevalence of patients in need of anticoagulant treatment, it does not seem feasible that solely ACCs could take care of these patients, and self-monitoring may not be feasible for everyone. 15 –22,25 –29
NOACs could represent a solution to this dilemma, since their pharmacologic properties simplify the administration and do not require monitoring. 30 However, the body of evidence in favor of NOACs is of questionable significance for the primary care setting, since it is predominantly based on phase III randomized controlled trials (RCTs) with noninferiority design and carefully selected patient populations treated in specialized centers for atrial fibrillation. 31 –35 These approval studies show noninferiority of NOACs when compared to the VKA agent warfarin, whereby patients under warfarin treatment spent on average (median) 65% of the follow-up time within the therapeutic range of INR 2 to 3. 35 Owing to important differences in study design and a lack of head-to-head trials, whether or not the different NOAC agents are equivalent with regard to efficacy and safety has not yet been verified. 36 –40 Data on the effectiveness of the agents (ie, the extent to which the agents achieve their intended effect in the usual clinical setting) are derived from observational studies, not from RCTs, and often lack information on the TTR of patients under VKA treatment. 41 –47 So it remains unclear whether these studies compared NOAC performance to high- or low-quality VKA treatments. The validity of these comparisons is therefore limited. As far as the NOAC agent dabigatran is concerned, advantages over warfarin were only seen in study centers where TTR was less than 72%. 48
By contrast, there are also examples of high-quality TTR control in daily practice. Previous reports from Sweden and the Netherlands documented TTR benchmarks as high as 76% and 81%, respectively. 49 –51 Moreover, a subanalysis of the ROCKET AF trial revealed global geographic region as being the strongest predictor of patients’ individual TTR independent of patients’ characteristics such as age and comorbidities. 52 This finding is likely to reflect different structures of care and support systems to manage warfarin and different regional barriers to frequent INR testing and warfarin dose changes. Under daily practice conditions—outside clinical trials—local differences also include the use of VKA agents other than warfarin (eg, phenprocoumon, fluindione, and acenocoumarol) that differ considerably regarding their half-lives, further affecting the comparability of local settings. 18,53,54
Therefore, it seems unjustified to extrapolate findings made under study conditions or in foreign health-care settings and to define a one-size-fits-all decision on first-line anticoagulant agents and ideal provision of care services without discussing the current standard of TTR control in specific care settings.
It is the aim of our study to contribute data on TTR control achieved in Swiss primary care practices to the discussion. As in many other countries, long-term anticoagulant therapy in Switzerland is mainly a task of general practitioners (GPs).
55
Available VKA agents are phenprocoumon and acenocoumarol. Most frequently, phenprocoumon is used. The INR monitoring is usually performed using a point-of-care device in order to adjust VKA dosage prescription during the same consultation if necessary. To date, the average TTR achieved in this setting is unknown, and it is unclear whether the patient populations of the RCTs are representative of Swiss primary care patients. We aim: to evaluate the stability of anticoagulant treatment in Swiss primary care expressed as patients’ average TTR of INR 2 to 3 when compared to the TTR levels achieved in the warfarin arms of NOAC approval studies, RE-LY (Randomized Evaluation of Long-Term Anticoagulation Therapy), ROCKET-AF (Rivaroxaban Once Daily Oral Direct Factor Xa Inhibition Compared with Vitamin K Antagonism for Prevention of Stroke and Embolism Trial in Atrial Fibrillation), ARISTOTLE (Apixaban for Reduction in Stroke and Other Thromboembolic Events in Atrial Fibrillation), ENGAGE AF-TIMI 48 (Effective Anticoagulation with Factor Xa Next Generation in Atrial Fibrillation -Thrombolysis in Myocardial Infarction 48)
31
–34
; to evaluate the characteristics of patients who receive long-term anticoagulant treatment with VKAs for ≥6 months in Swiss primary care; to compare these characteristics to baseline characteristics of intention-to-treat (ITT) populations included in the above-mentioned RCTs; and to assess the impact of patient characteristics known to affect anticoagulation control with VKA measured by TTR.
Materials and Methods
In this retrospective longitudinal study, we calculated the TTR of Swiss primary care patients receiving long-term anticoagulant treatment with VKAs for ≥6 months without interruption based on recorded results of INR tests according to Rosendaal’s formula. 56 The combined ITT population included in the NOAC approval studies RE-LY, ROCKET-AF, ARISTOTLE, and ENGAGE-AF-TIMI 48 served as a comparator to our study population. We will subsequently refer to this population as the NOAC RCT population.
FIRE Database
Eligible patients were identified from the primary care research database FIRE (Family medicine International Classification of Primary Care [ICPC] Research using Electronic medical records) receiving long-term anticoagulant treatment with VKAs for ≥6 months. The FIRE database is the core of an ongoing health services research project at the Institute of Primary Care at the University and University Hospital of Zurich, Switzerland. Details about the project, the data structure, and the validation of the database were reported previously. 57,58 In short, the database established in 2009 contains information on all physician–patient contacts in participating primary care practices around Switzerland. Data are extracted from routinely used electronic medical records, fully anonymized and aggregated by individual consultation dates and randomly generated, deidentifiable but unique patient ID numbers. The data set covers patients’ age, sex, reasons for encounter according to the ICPC-2 classification, 59 vital signs, laboratory test results as well as type and dosage of prescribed medication according to Anatomical Therapeutic Chemical/Defined Daily Dose Classification (ATC/DDD) coding established by the World Health Organization. 60 According to current Swiss law on human research, retrospective analysis of anonymously collected medical routine data requires no approval by regional ethics committees. 61
Patient Population
All patients with INR tests recorded between May 2009 and January 2015 were eligible and included based on the following criteria: prescribed VKA treatment for at least 6 months (this criterion would exclude patients with VKA treatment due to a first event of deep vein thrombosis and time-limited risk of thromboembolic disease) and no prescription of heparin agents during VKA treatment period (this criterion would exclude patients with periprocedural interruption of VKA treatment and heparin bridging).
Patients’ baseline characteristics were directly obtained from the FIRE database or derived from the available data set as follows: Indication for anticoagulant treatment was derived from the ICPC-2 codes assigned to the patient during VKA treatment; presence of concomitant chronic conditions was derived both from ICPC-2 codes and pharmaceutical cost groups based on ATC/DDD coding according to the established algorithms of O’Halloran et al and Lamers et al, respectively. 62,63 This approach has previously been validated for the FIRE database 58,64 ; all drugs prescribed for at least 6 months were considered to be long-term medication and counted as concomitant drugs; CHADS2 score of stroke risk in patients with atrial fibrillation according to ICPC-2 coding was calculated according to Gage et al. 65 We used the CHADS2 score instead of the CHA2DS2-VASc score for comparison reasons because the NOAC approval studies exclusively reported the CHADS2 score. Creatinine clearance (mL/min) was calculated based on serum creatinine values according to the Cockcroft-Gault equation. 66 Bleeding events were not registered in the database.
Comparative Studies
Baseline characteristics of the NOAC RCT population were extracted from the data sets of the RE-LY, ROCKET-AF, ARISTOTLE, and ENGAGE-AF-TIMI 48 studies published in medical literature. 31 –35
Outcome Definitions
We defined good anticoagulation control as TTR >70%, moderate control as TTR 61% to 70%, poor control as TTR 40% to 60% and ineffective control as TTR <40% based on guideline recommendations and current literature. 38,67 –74
Statistics
Data are presented as frequencies and percentages, mean and standard deviation (SD), or median and interquartile range (IQR), where appropriate. If not indicated otherwise, all percentages refer to the total number of included patients. First, correlation of TTR and age, sex, overall number of chronic conditions and drugs taken, hypertension, diabetes, congestive heart failure, creatinine clearance, CHADS2 scoring, length of observation (years), number of physician–patient contacts, and number of INR tests was assessed using univariable linear regression models. Results are presented as coefficients, 95% confidence intervals, and P values. A P value < .1 was considered as statistically significant. In a second step, all variables showing a significant impact on TTR in univariable analysis were included in a multivariable linear regression model. Subsequently, robustness of the results was tested by adding GPs as random variable to the model (mixed model) in order to adjust for clustering effects. Then, a P value <.05 was considered as statistically significant. All statistical analyses were done in R (version 3.0.2; The R Foundation for Statistical Computing, Vienna, Austria).
Results
Consultation data of 670 patients with INR tests recorded between May 2009 and January 2015 were eligible. Of these, 256 patients received VKA treatment for less than 6 months and were thus excluded. Another 199 patients were excluded because of periprocedural interruption of VKA treatment and heparin bridging (flowchart in Figure 1). Two hundred and fifteen patients treated by 12 physicians fulfilled the inclusion criteria and were included in the analysis. Patient characteristics are summarized in Table 1. The NOAC RCT population included 71 683 patients. Of these, 29 272 were allocated to warfarin treatment.
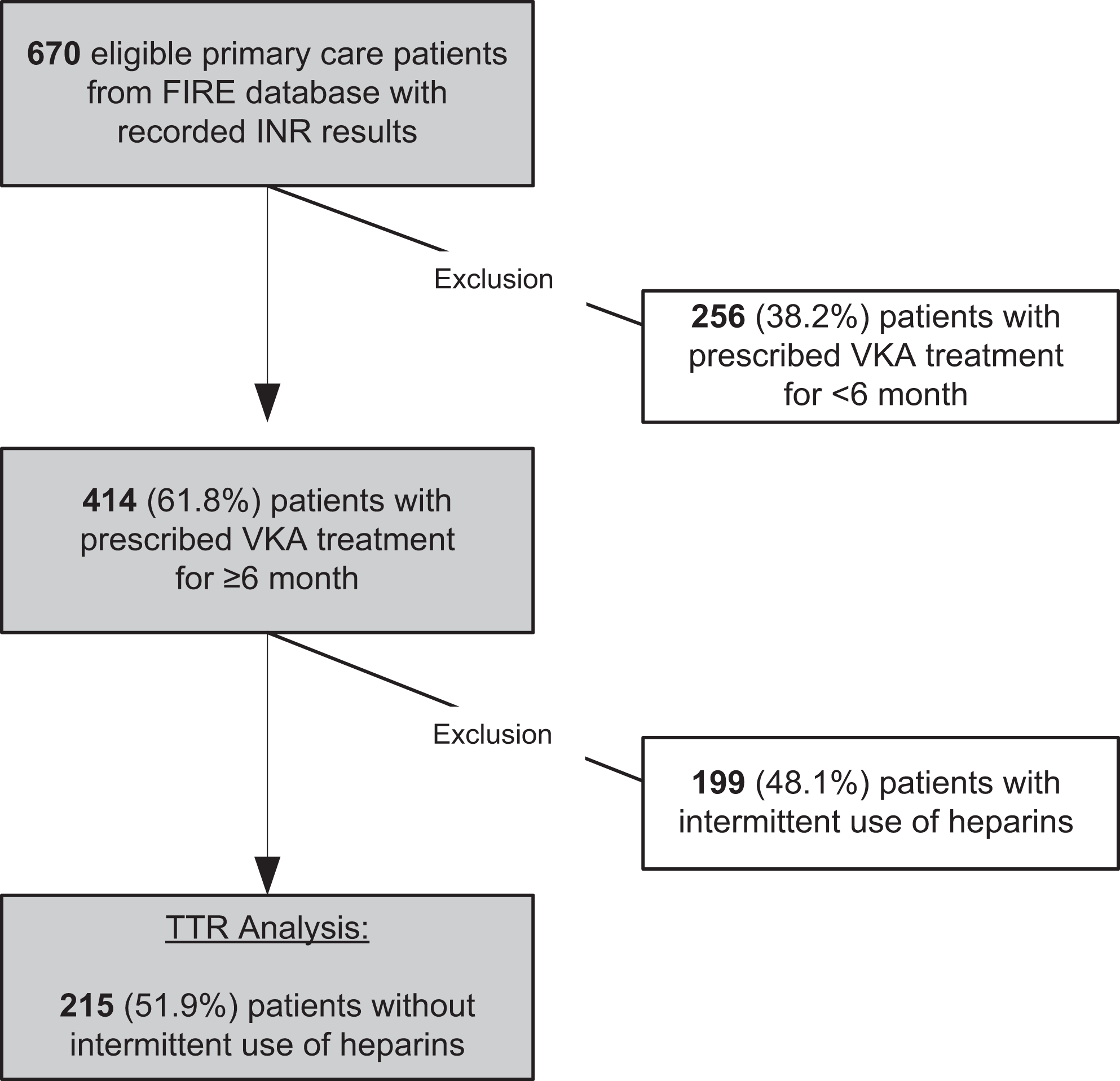
Flowchart of study sample selection—patients with vitamin K antagonist (VKA) therapy.
Baseline Characteristics of Included Patients in Comparison to the Intention-to-Treat Population of the NOAC Approval Studies.
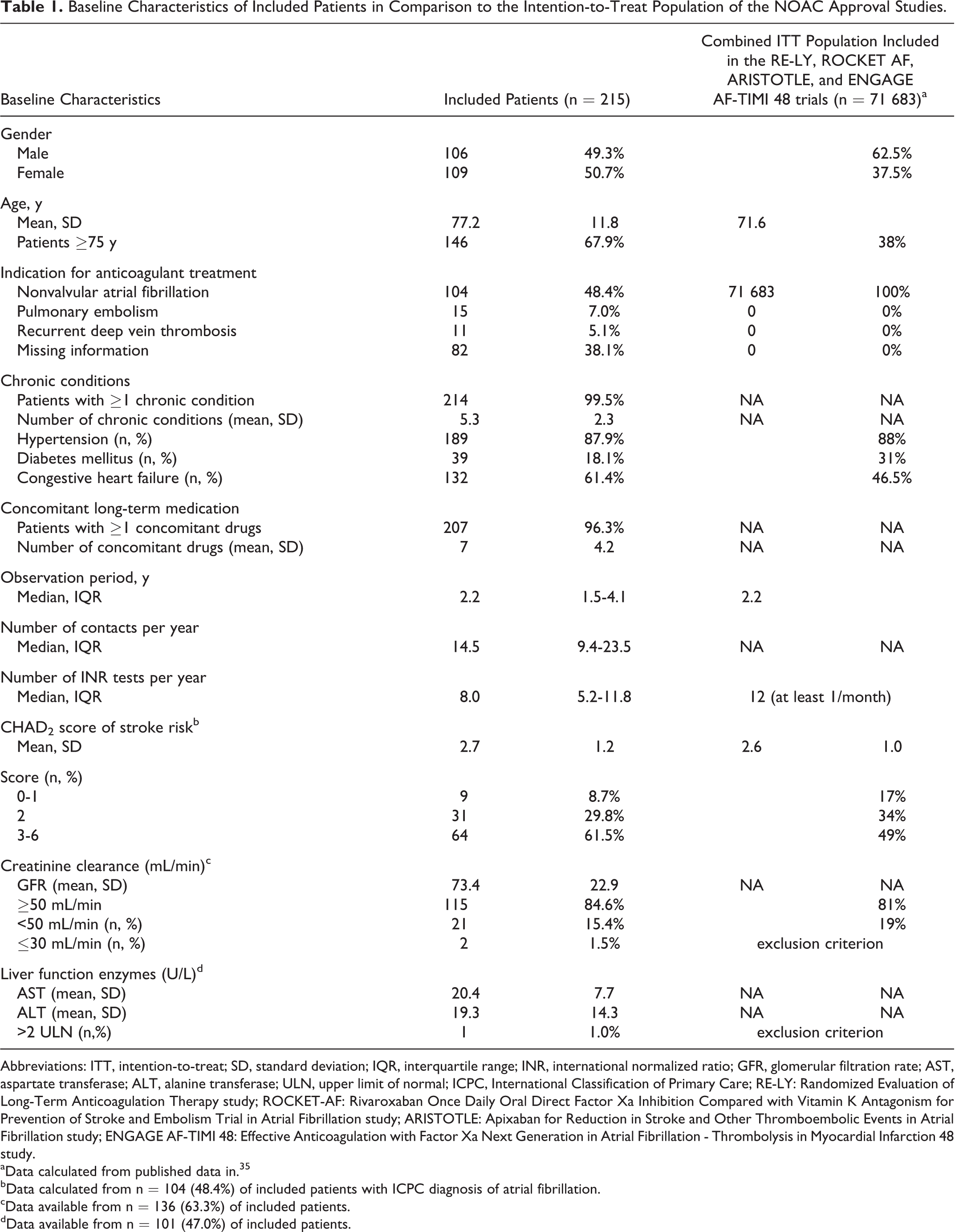
Abbreviations: ITT, intention-to-treat; SD, standard deviation; IQR, interquartile range; INR, international normalized ratio; GFR, glomerular filtration rate; AST, aspartate transferase; ALT, alanine transferase; ULN, upper limit of normal; ICPC, International Classification of Primary Care; RE-LY: Randomized Evaluation of Long-Term Anticoagulation Therapy study; ROCKET-AF: Rivaroxaban Once Daily Oral Direct Factor Xa Inhibition Compared with Vitamin K Antagonism for Prevention of Stroke and Embolism Trial in Atrial Fibrillation study; ARISTOTLE: Apixaban for Reduction in Stroke and Other Thromboembolic Events in Atrial Fibrillation study; ENGAGE AF-TIMI 48: Effective Anticoagulation with Factor Xa Next Generation in Atrial Fibrillation - Thrombolysis in Myocardial Infarction 48 study.
aData calculated from published data in. 35
bData calculated from n = 104 (48.4%) of included patients with ICPC diagnosis of atrial fibrillation.
cData available from n = 136 (63.3%) of included patients.
dData available from n = 101 (47.0%) of included patients.
Our patients were similar to the ITT population included in the NOAC RCTs in terms of length of observation (both median 2.2 years), rate of concomitant hypertension (87.9% vs 88%), and CHADS2 scoring (median 2.7 vs 2.6). Differences were found in terms of sex, age, rate of concomitant diabetes mellitus and congestive heart failure, creatinine clearance, and number of INR tests per year and patient (Table 1). In our study population, more patients were female (50.7% vs 37.5%) and aged 75 years or older (67.9% vs 38%). The proportion of patients with concomitant diagnosis of diabetes mellitus was lower than in the NOAC approval studies (18.1% vs 31%), whereas the proportion of patients with congestive heart failure was higher (61.4% vs 46.5%). In sum, more patients in our study showed a creatinine clearance of ≥50 mL/min (84.5% [n = 115/136] vs 81.5%), but unlike in the NOAC approval studies, also 2 (1.5%) patients with creatinine clearance ≤30 mL/min were included. The median number of GP encounters within our study population was 14.5 times per year, and INR was tested every 6 weeks (median 8 tests per year). The NOAC approval studies requested at least monthly follow-up contacts and INR tests in patients receiving warfarin—that is, a minimum of 12 tests per year.
Number of chronic conditions other than those leading to an indication for anticoagulant treatment, number of additional drugs taken, and liver function parameters (aspartate transferase, AST; alanine transferase, ALT) of the NOAC RCT population have not been publicly published. In our study population, the mean number of concomitant chronic conditions was 5.3 (SD 2.3). Apart from VKA, patients took 7 (SD 4.2) concomitant drugs. Mean values of both AST and ALT were within the reference ranges as specified by the International Federation of Clinical Chemistry and Laboratory Medicine, 75 but 1 patient (n = 1/101, 1%) showed values >2 times the upper limit of normal (ULN). Such values constituted an exclusion criterion in the RELY, ARISTOTLE, and ENGAGE-AF-TIMI 48 trials. 76 –78 No patient showed values >3 times ULN (exclusion criterion in the ROCKET-AF trial 34 ).
Time in Therapeutic Range
Individual median INR in our study population was 2.4. Patients spent median 75% of treatment time within the therapeutic range of INR 2 to 3. Median TTR in the warfarin arm of the NOAC RCTs was 65% (Table 2). In our study, 60.9% of patients reached or exceeded this threshold (TTR ≥ 65%). The majority of our patients showed good (55.8% with TTR > 70%) or moderate (9.8% with TTR 61%-70%) anticoagulation control. Poor and ineffective control was found in 20.9% and 13.5%, respectively. The median treatment time in a subtherapeutic INR range (INR < 2) was 10.1% (IQR 1.8%-29.6%), and the median treatment time above the TTR (INR > 3) was 5.5% (IQR 0%-17.2%).
International Normalized Ratio and Time in Therapeutic Range (TTR) Achieved in the Study Population in Comparison to the Intention-to-Treat Population of the NOAC Approval Studies.
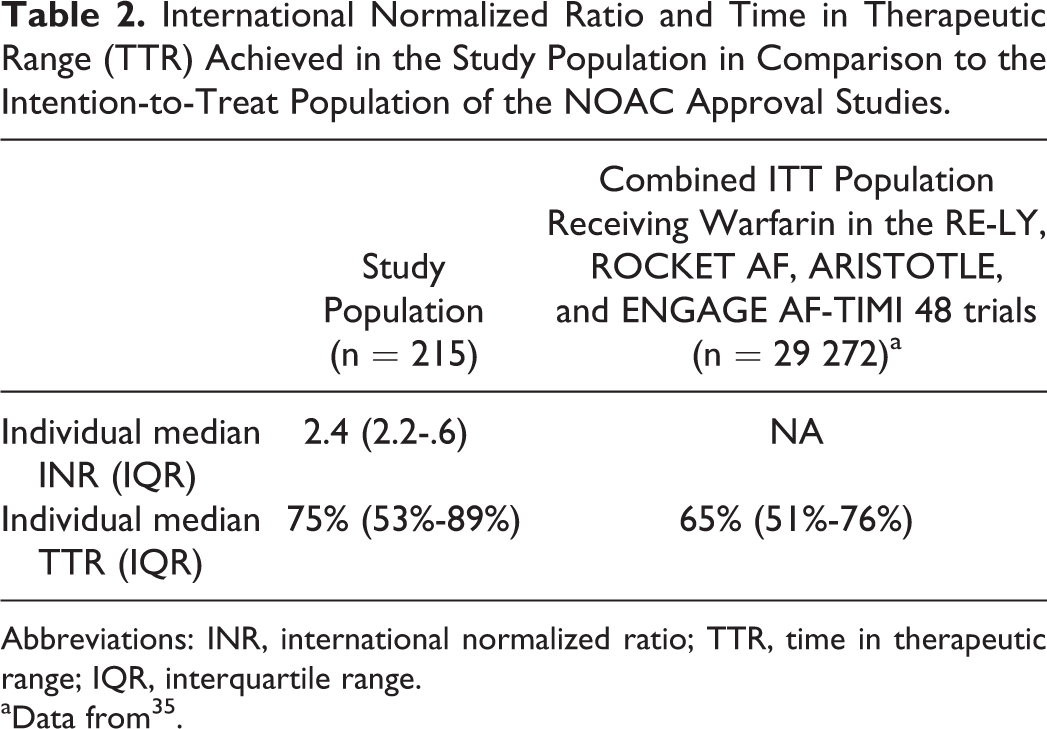
Abbreviations: INR, international normalized ratio; TTR, time in therapeutic range; IQR, interquartile range.
aData from 35 .
Univariable linear regression analysis demonstrated significant associations between lower TTR and patients’ age, sex, number of concomitant chronic conditions and diagnosis of congestive heart failure, and significant associations between higher TTR and length of observation and number of INR tests (Table 3). In the subsequently performed multivariable regression analysis, sex remained a significant predictor of TTR and was significantly modified by age (P = .0059 for the interaction term sex × age). This result remained unchanged when controlled for a cluster effect originating from GPs. Age was not an independent predictor of TTR, but a significant interaction between age and sex was found. A cluster effect was not present (Table 4).
Coefficients and 95% CIs From Univariable Regression Models.
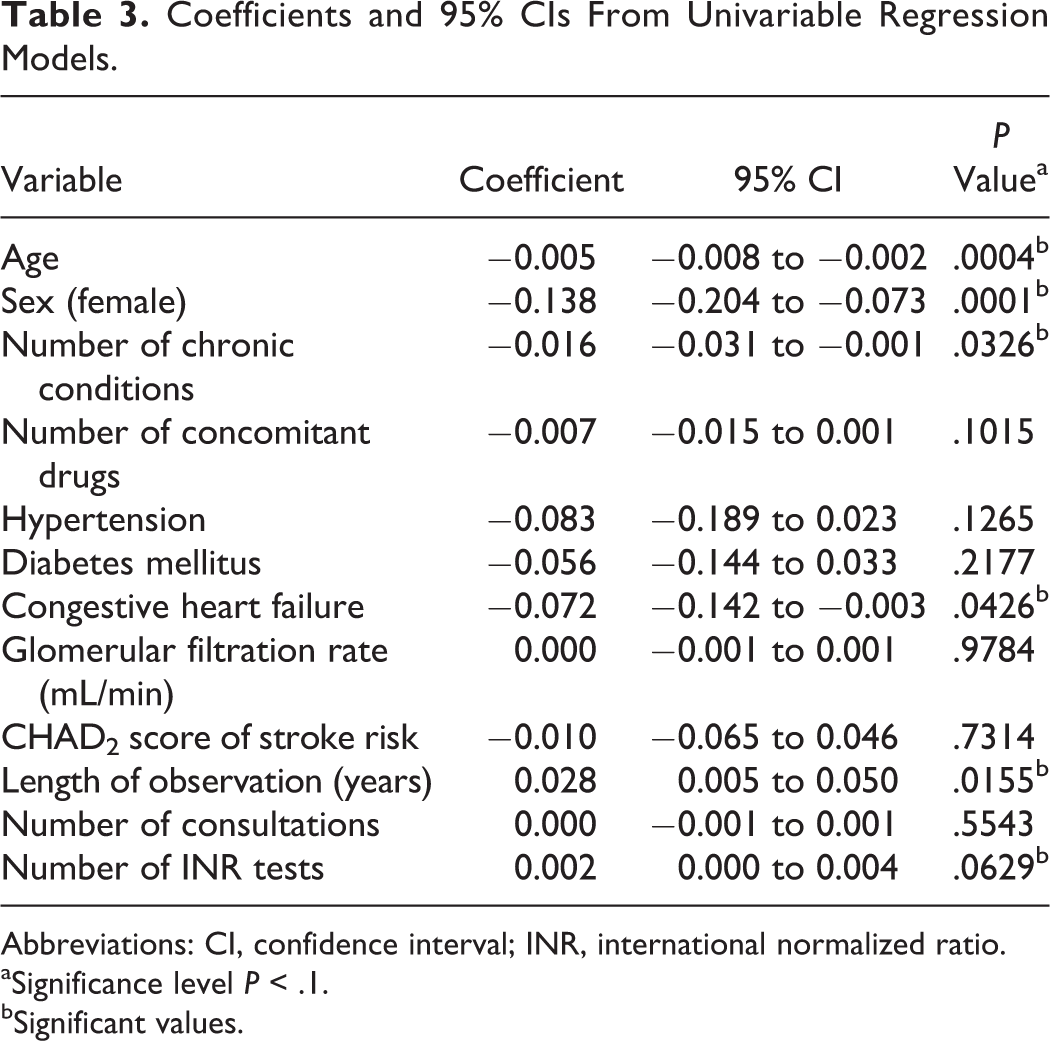
Abbreviations: CI, confidence interval; INR, international normalized ratio.
aSignificance level P < .1.
bSignificant values.
Coefficients and 95% CIs From a Multivariable Regression Model (Mixed Model): TTR = Age × Sex + Number of Chronic Conditions + Congestive Heart Failure + Length of Observation + Number of INR Tests With GP as Random Variable.
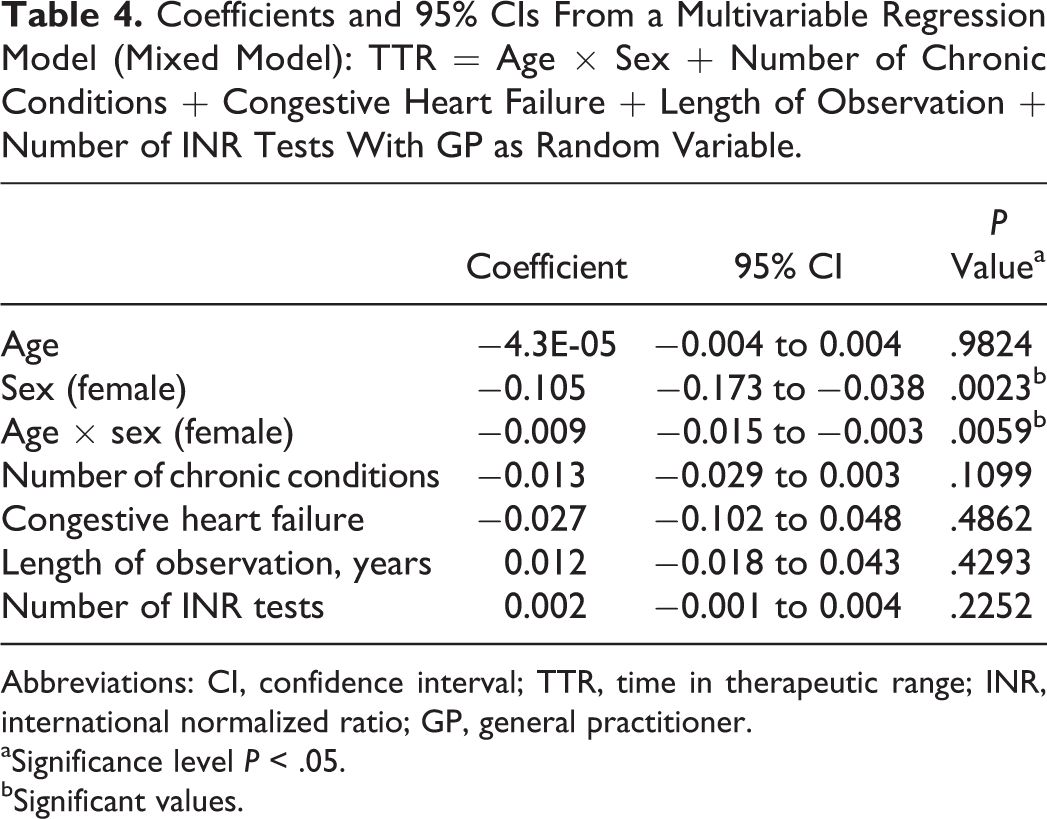
Abbreviations: CI, confidence interval; TTR, time in therapeutic range; INR, international normalized ratio; GP, general practitioner.
aSignificance level P < .05.
bSignificant values.
Discussion
In this study, we examined the quality of anticoagulant treatment with VKA in Swiss primary care patients, measured by individual TTR. It is the first study of this kind in Switzerland and showed a remarkably high level of TTR control. Median TTR was 75% exceeding not only the median TTR level achieved in the NOAC RCTs but also TTR levels reported from daily practice settings in other Western countries and is comparable to the data on anticoagulation self-management in Switzerland. 79 This finding ranks Switzerland with the best performing countries, Sweden and the Netherlands, which had previously reported TTR benchmarks of 76% and 81%. 49 –51
Previous estimations of the quality of VKA treatment control in Switzerland did not forecast our finding. In the absence of real-life data, these estimations were made based on regional analyses of NOAC RCTs. They suggested that TTR levels achieved in Swiss patients would be of minor quality. In the RE-LY trial, for instance, Swiss study centers showed a mean center TTR of 68%. 48 In the ROCKET-AF trial, mean individual TTR of Swiss patients was below the average baseline TTR of Western Europe (63.2%). 52 The impact of such estimations must not be underestimated. According to current European guidelines, patients with a TTR below 70% should receive NOAC instead of VKA. 38 Based on the above-mentioned estimations of TTR levels achieved in the Swiss health-care setting, one could conclude that the majority of Swiss patients under VKA treatment had to be switched to NOAC, and patients should generally be started on NOAC in order to avoid a few promising but strenuous VKA therapy initiation. 71 Not only would large quantities of therapy changes challenge patients and clinicians, but the mass of new NOAC prescription would also have relevant economic influence on the country’s obligatory health insurance system covering anticoagulant treatment. It has been shown that NOACs can be cost effective in specific settings, despite higher pharmaceutical costs because overall costs for anticoagulation-related outpatient visits and hospitalizations were reduced compared to warfarin treatment. 80 –84 However, just like clinical outcomes, cost-efficiency estimations have also been demonstrated to depend on TTR levels achieved in the VKA group. 85,86
Our findings, now, reduce concerns about the magnitude of the patient population requiring therapy changes by suggesting that the majority of Swiss patients maintain stable TTR levels and would not require therapy changes. Moreover, regression analysis indicated that TTR was independent of a wide range of patient characteristics. We conclude that all GPs were apparently able to control TTR irrespective of confounding influences.
Strengths and Limitations
Of course, it needs to be acknowledged that our study sample was small compared to the overall number of patients included in the NOAC RCTs or in foreign observation registries. 5,8,24 One might further argue that there are a number of potential confounders impacting on TTR that we did not include in our regression models. In response to these claims, it is to note that the strength of our study is not only the use of local data but also setting-specific data. Two aspects are to note.
All figures must be seen in proportion to the limited availability of regional data. Numbers of Swiss patients recruited in the RE-LY and ARISTOTLE trials are not publicly published, but it is known that TTR information on Swiss patients in the ROCKET-AF study were derived from only 4 Swiss patients. 87 In the ENGAGE-AF-TIMI 48 trial, zero Swiss patients received warfarin. 88 In comparison, we present results from 215 patients.
It was not the aim of this study to newly identify all potentially imaginable determinants of TTR control. We assessed the impact of those baseline characteristics in our data set that are known to affect anticoagulation control in order to control for potential confounders.
Thus, our results should not be generalized to other health-care settings nor be understood in a way that TTR control is always independent of patient characteristics. It is widely verified that patient characteristics such as concomitant diseases and female sex—also significant in our study—can act as significant predictors of TTR. 2,8,12,51,71 Explanations for these observations are, however, scarce. It is likely that some of the previously reported associations of TTR and patient characteristics are random effects due to multiple testing in very large data samples. On the other hand, it may also be well assumed that the previously observed effects actually reflect underlying differences of patients’ health literacy, self-management resources, adherence to medical advice, or access to medical care, in brief sociodemographic and infrastructural features of the health-care setting. 89
The results of our mixed models regression analysis indicate that features of the Swiss primary care setting, other than number of consultations and INR tests, might overrule the effect of patient characteristics on TTR control and ensure stable TTR maintenance. For instance, the point-of-care testing in GP practices is an important feature that is known to influence TTR. 90,91 It will be a task of future research to examine this hypothesis with adequate methods.
It was the aim of our study to evaluate TTR control in the circumscribed setting of Swiss primary care and call one-size-fits-all recommendations of first-line anticoagulant treatment into question. Indeed, our results highlight the importance of local and setting-specific TTR assessments by demonstrating that real-life TTR outcomes in individual health-care settings may deviate from expected values derived from RCTs or observation studies in foreign health care systems. It is important to note that our patients achieved better TTR results than the NOAC control group population, even though our patients were remarkably older than the NOAC ITT population and presented a lot of concomitant chronic diseases and polypharmacy. This raises concerns about the gold standard of VKA treatment that served as a reference in the NOAC RCTs and should be taken into account when deriving practice guidelines from RCT results. Initial observations indicating that patients enrolled in NOAC RCTs are only partly representative of patients with atrial fibrillation in clinical practice were made in Korea. 92 Our study confirms these findings now within a European context, but unlike the Korean study, we found that when TTR is considered, fewer patients than expected would require NOAC therapy instead of VKA.
Of course, TTR should not be the only basis of decision making. Bleeding events are another reason for switching to NOACs but were out of scope of this study. None of the observed patients stopped VKA treatment because of bleeding events, but information on patients was limited to GPs medical record entries. Thus, occurrence of bleeding events leading to consultation of other health-care providers cannot be ruled out entirely. Bleeding prevalence in Swiss VKA patients should be examined in a next step.
The burden of treatment should also be considered when selecting anticoagulant agents, and in many situations, the ease of use will speak in favor of NOACs. Nevertheless, it must be acknowledged that periodical INR monitoring required under VKA treatment imposes not necessarily a burden for patients and primary physicians. A recent study suggested that it could also enhance patient–physician relation and therapy adherence and offer opportunities for the physician to provide additional care to patients, such as adjusting the dosage of antihypertensive drugs, giving smoking cessation advice, or treating high cholesterol. 93 Our findings now show that concomitant comprehensive care for other conditions does not impair physicians’ resources in controlling INR and ensuring high-quality TTR control. This is of particular relevance for daily practice in primary care worldwide, given the high prevalence of multimorbidity in primary care patients. 94
As long as published data on NOAC use in elderly, multimorbid patient populations is scarce, general recommendations of NOACs as first-line treatment in such patients should be taken with caution. In the meantime, physicians should instead tailor anticoagulation management according to the individual needs of their patients with regard to the heterogeneity, age, multimorbidity, and risk profile of patients in primary care settings. Our findings show that high-quality TTR can be achieved even in such vulnerable patients.
Conclusion
Choice of anticoagulant agents is a complex medical decision and should be taken in comprehensive view of patients’ individual risk–benefit profile. Practitioners should consider that the noninferiority of NOACs has been proven in RCTs which enrolled patients who are only partly representative of patients in primary care, particularly in terms of TTR maintenance when receiving VKA treatment. The observed majority of our patients would not require an immediate switch to NOACs due to low-quality TTR. Further research is needed in order to identify specific features of care management that are associated with these outcomes, since most patients’ characteristics were insignificant predictors of TTR.
Footnotes
Acknowledgments
We would like to thank all participating GPs in the FIRE project.
Authors’ Note
SD, OS, DM, and BG developed the study protocol. SD and FV extracted and analyzed data. SD drafted the manuscript. All authors read and approved the final manuscript. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research received funding from the Swiss Academy of Medical Science (SAMS), Gottfried and Julia-Bangerter-Rhyner-Foundation.
