Abstract
Introduction:
The prompt assessment and the reversal of direct oral anticoagulants (DOACs) are urgent matters in the emergency care setting. Thus, we planned to elucidate the adequate prothrombin time (PT) test for the evaluation of the anticoagulant effects of various DOACs.
Methods:
The anticoagulant effects of rivaroxaban, apixaban, and edoxaban were measured with 3 PT tests (Triniclot PT Excel S, Neoplastin R, and Thromborel S). Human plasma was spiked with each DOAC at a range of 0 to 1000 ng/mL, and the PT was measured using each PT test. In another series, the reversal effect of either 4-factor prothrombin complex concentrate (PCC) or activated PCC (aPCC) was evaluated with each PT test.
Results:
All PT reagents correlated with the concentrations of each DOAC, however, the reactivity was considerably different between the DOACs and the PT tests. A prolonged PT with DOACs was reversed both by PCC and aPCC in a dose-dependent manner; however, Triniclot PT Excel S showed reprolongation of the PT with a higher dose of PCC.
Conclusion:
The proper choice of PT test is necessary for the assessments of the anticoagulant activity of DOACs. It is also important to understand the different characteristics of each PT test for the assessment of the reversal effects of PCC.
Introduction
Life-threatening bleeding such as massive and intracranial hemorrhage (ICH) during anticoagulant therapy with direct oral anticoagulants (DOACs) is a present danger. 1,2 It would be undoubtedly favorable if real-time monitoring for the anticoagulant activity of these agents was available.
The measurement of antifactor Xa activity is a possible candidate, 3,4 however, this procedure is not consistently applicable in most of local laboratories. Instead, the prothrombin time (PT) test is more suitable if it is appropriately sensitive for practical use. The usefulness of PT tests for the monitoring of certain DOACs has been revealed in previous reports, 5,6 and thus, we planned the comprehensive comparison of various PT tests for all available DOACs in this study.
In the “Guidelines for the Management of Spontaneous Intracerebral Hemorrhage,” 7 the American Heart Association Stroke Council recommended treatment with prothrombin complex concentrates (PCCs) or activated PCC (aPCC) for the patients with ICH under the treatment of DOACs. However, there have been no randomized trials of reversing agents for DOACs among patients with ICH or other major bleeding complications. Since the reactivity of the PT test is reported to be different depending on the DOACs, we need to examine which PT test for each DOAC is most suitable as far as the reversal effect is concerned. We further tried to elucidate the characteristic difference in various PT tests and their potentials as monitoring tests for the reversal effects of PCC and aPCC.
Materials and Methods
Spike Test
Pooled citrated normal human plasma was spiked with rivaroxaban, apixaban, or edoxaban at increasing concentrations to assess the impact in the different kinds of PT tests. Rivaroxaban and edoxaban were kindly donated by Bayer Healthcare (Wuppertal, Germany) and Daiichi Sankyo (Tokyo, Japan), respectively. Apixaban was purchased from Selleckchem (Houston, Texas). Working solutions of 100, 300, 500, 700, and 1000 ng/mL of each DOAC were obtained from the stock solution at a dose of 1.0 mg/dimethyl sulfoxide 1.0 mL by mixing with pooled citrated normal human platelet-poor plasma.
Prothrombin Time Test
The PT was measured with the following method: 50 μL of plasma samples were incubated at 37°C up to 60 seconds and mixed with 100 μL of calcium and thromboplastin. The different thromboplastin reagents used were Triniclot PT Excel S (trade name in Japan: Shinplastin Excel S; Kyowa Medex, Tokyo, Japan), Thromborel S (Dade Behring, Newark, New Jersey), and Neoplastin R (Diagnostica Stago, Asnieres, France). Clotting time was measured on COAGTRON 350 (Kyowa Medex) for all PT tests.
Reversal Effect of PCC
After each DOAC was incubated at final concentrations of 0, 100, 300, 500, 700, or 1000 ng/mL with the pooled plasma for 15 minutes, 4-factor PCC (PPSB-HT; Nihon Pharmaceutical, Tokyo, Japan) was added at concentrations of 0, 0.63, 1.25, 2.5, or 5.0 U/mL, and these aliquots were incubated at 37°C. After 15 minutes of incubation, the PT was measured with 3 PT tests. For the comparison of the magnitude of reduction, reduction rate (RR) was calculated: RR = PT (PCC 0 − PCC 1.25)/PT (without DOACs and PCC).
Reversal Effect of aPCC
Similar to PCC, after each DOAC was incubated at final concentrations of 0, 100, 300, 500, 700, or 1000 ng/mL with the pooled plasma for 15 minutes, aPCC (FEIBA; Baxter, Vienna, Austria) was added at concentrations of 0, 0.63, 1.25, 2.5, or 5.0 U/mL, and these aliquots were incubated at 37°C. After 15 minutes of incubation, the PT was measured with 3 PT tests. For the comparison of the magnitude of reduction, RR was calculated: RR = PT (aPCC 0 − aPCC 1.25)/PT (without DOACs and aPCC).
Statistical Analysis
Statistical analysis was performed using the StatView program (Abacus Concepts, Berkeley, California). The Pearson product-moment correlation coefficient was used to correlate the distribution of PT values between the PT reagents. Overall statistical significance among serial measurements was analyzed with a repeated-measures analysis of variance, followed by an individual comparison with the paired t test as appropriate. P < .05 was considered statistically significant.
Results
Spike Test
The spike tests were repeated 3 times, and the same results were obtained. The summarized results of 3 independent experiments for each DOAC are depicted in Figure 1. A concentration-dependent prolongation of the PT was found in all DOACs at a concentration from 0 to 1000 ng/mL. The relationship between the PT and each DOAC concentration was almost linear for each PT test at the range of examined concentrations, and thus, slopes (slope of the linear regression line) and R 2 (coefficient of determination) were compared between the reagents. The slopes of correlations between the concentrations of rivaroxaban and the PT were greatest in Neoplastin and Triniclot followed by Thromborel (P < .01). The order was Triniclot followed by Neoplastin and Thromborel in apixaban and Triniclot followed by Neoplastin and then by Thromborel in edoxaban (Table 1).
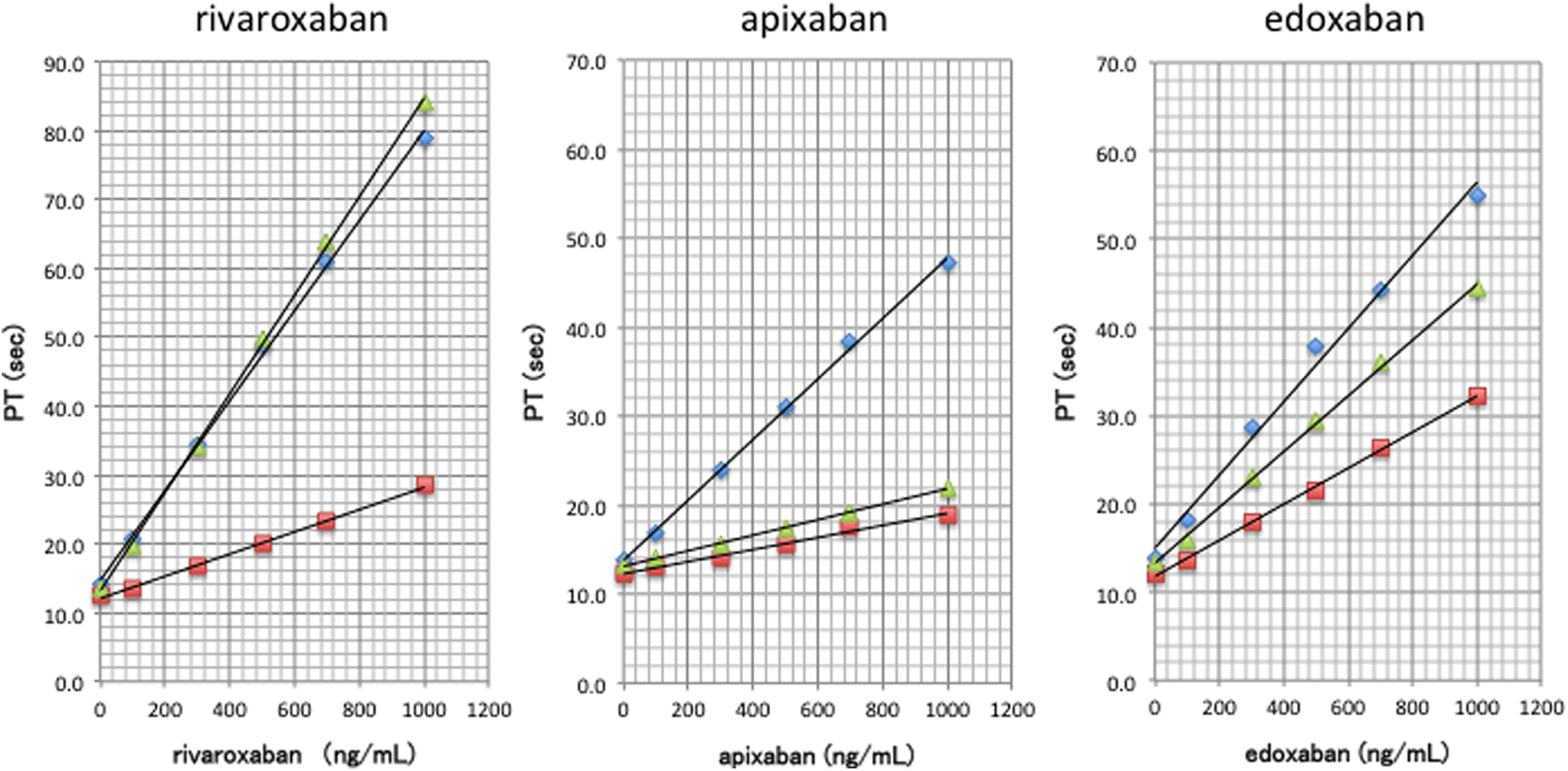
Impact of direct oral anticoagulants (DOACs) on the prothrombin time (PT). Direct oral anticoagulants prolonged the PT concentration dependently. The relationship was almost linear in all PT tests, and the magnitude of responses to DOAC depended on the PT reagent. The response ranged from 0 to 1000 ng/mL. ⋄Triniclot Excel S (blue), ▴Neoplastin R (green); ▪Thromborel S (red).
Prothrombin Time Reagents Evaluated.a
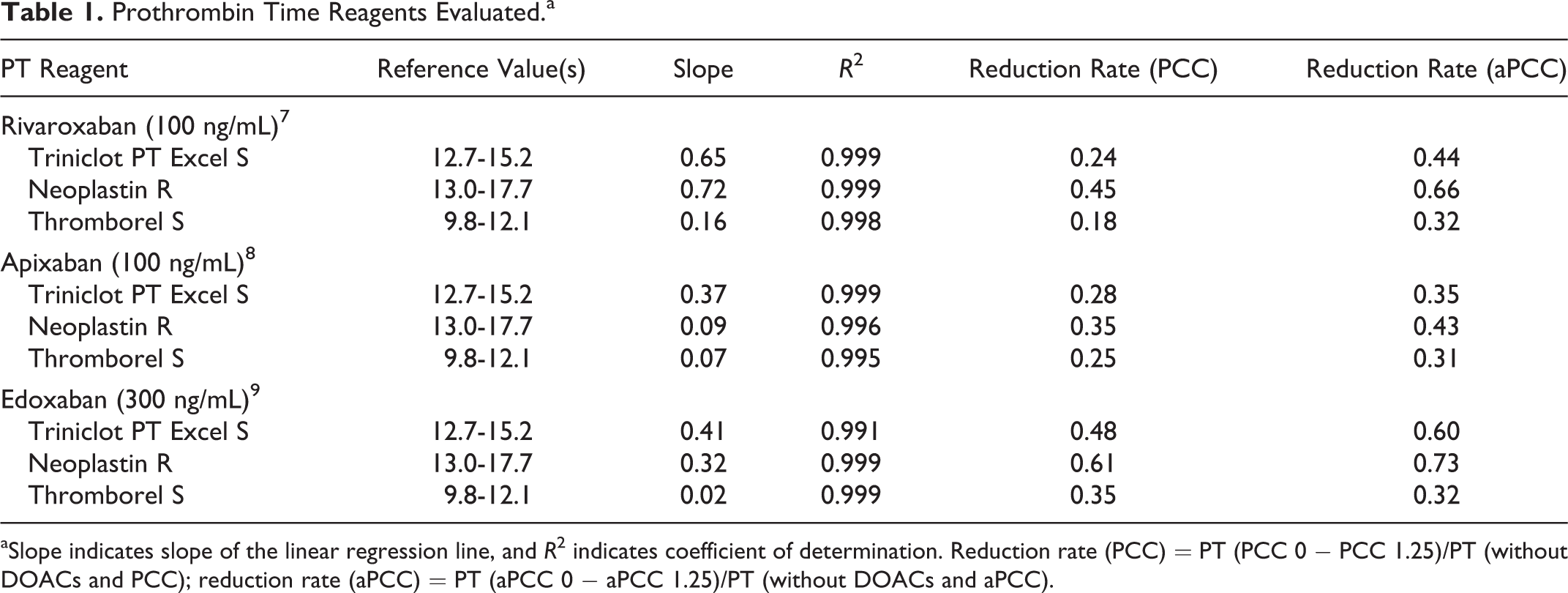
aSlope indicates slope of the linear regression line, and R 2 indicates coefficient of determination. Reduction rate (PCC) = PT (PCC 0 − PCC 1.25)/PT (without DOACs and PCC); reduction rate (aPCC) = PT (aPCC 0 − aPCC 1.25)/PT (without DOACs and aPCC).
Reversal Effect by PCC
The reversal effect of PCC was examined 3 times for each DOAC and each PT test, and the representative results are shown in Figures 2 to 4. A significant prolongation of the PT was observed in the samples preincubated with each DOAC from 0 to 1000 ng/mL without PCC. When PCC 1.25 U/mL was added to the peak therapeutic concentration of rivaroxaban-spiked samples (100 ng/mL), 10 the PT decreased most prominently with Neoplastin and was reduced from 19.7 to 13.6 seconds (RR, 0.45; Table 1). In the case of Triniclot, the PT decreased at PCC concentrations between 0.63 and 1.25 U/mL (RR, 0.24) and then the PT started to prolong until PCC dose of 5.0 U/mL. The PT at 5.0 U/mL of PCC was 26.4 seconds and longer than the original PT (20.4 seconds [PCC 0 U/mL]). Thromborel and Neoplastin did not show such reprolongation, and the PT reached a plateau when the PCC dose was 2.5 U/mL. In case of the therapeutic concentration of apixaban (approximately 100 ng/mL), 11 the PT measured with Triniclot decreased from 17.6 seconds (PCC 0 U/mL) to 13.6 seconds (1.25 U/mL), and the RR was 0.28. Similarly, 1.25 U/mL of PCC reduced the PT by 4.7 seconds (RR, 0.35) and 3.1 seconds (RR, 0.25) in Neoplastin and Thromborel, respectively. In case of the therapeutic concentration of edoxaban (approximately 300 ng/mL), 12 the PT reduced by 6.0 seconds (RR, 0.48), 8.2 seconds (RR, 0.61), and 4.2 seconds (RR, 0.35) in Triniclot, Neoplastin, and Thromborel, respectively.
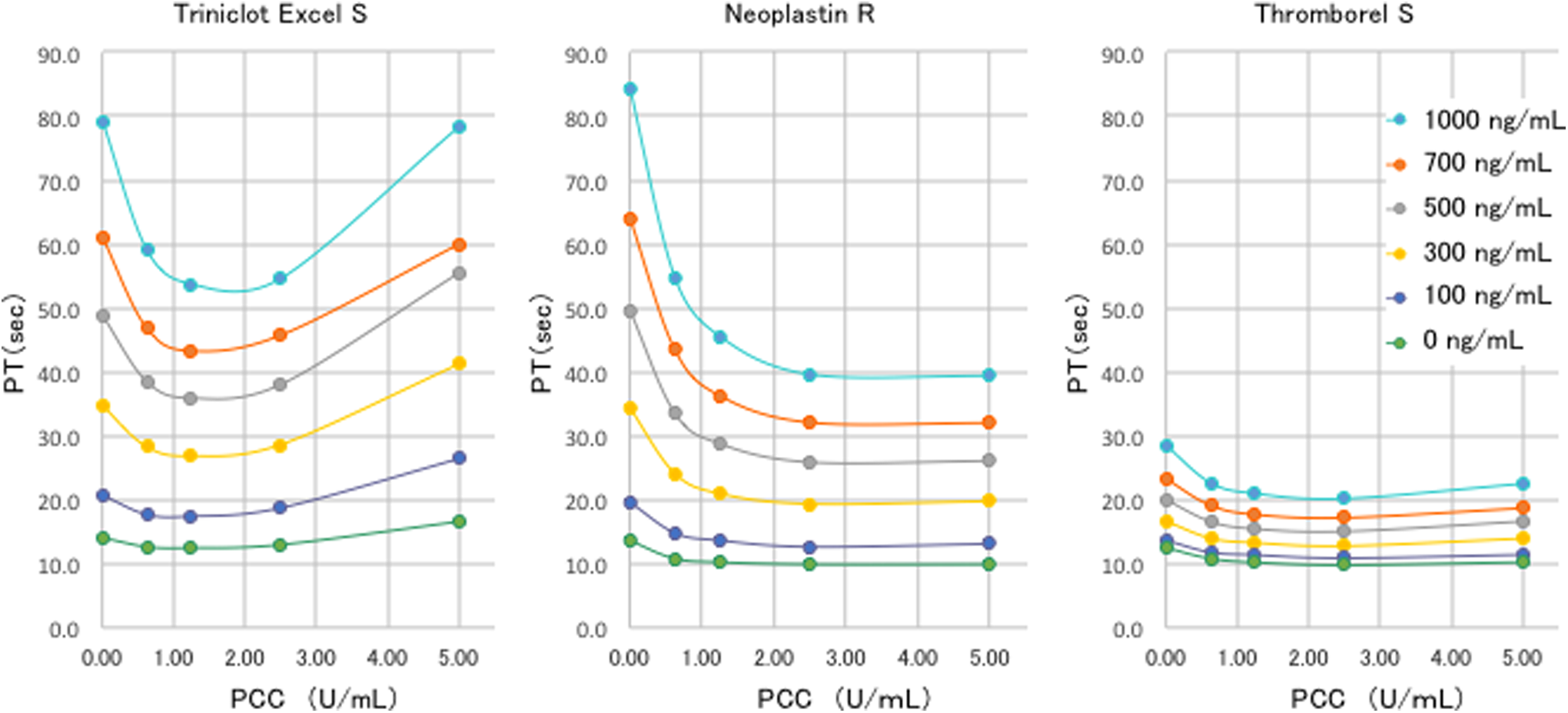
Impact of prothrombin complex concentrate (PCC) on the prothrombin time (PT) under the use of rivaroxaban. A concentration-dependent decrease in the PT was recognized after PCC administration. In case of Triniclot Excel S, the PT decreased in a dose-dependent manner at a PCC dose between 0.63 and 1.25 U/mL. The PT started to prolong again when the PCC dose was over 2.5 U/mL. Such reprolongation was not observed in Neoplastin R and Thromborel S, where the PT reached a plateau at the PCC concentration of 2.5 U/mL.
Impact of prothrombin complex concentrate (PCC) on the prothrombin time (PT) under the use of apixaban. A concentration-dependent decrease in PT was recognized after PCC administration. In case of Triniclot Excel S, the PT decreased in a dose-dependent manner at a PCC dose between 0.63 and 1.25 U/mL. The PT started to prolong again when PCC dose was over 2.5 U/mL. Such reprolongation was not observed in Neoplastin R and Thromborel S, where the PT reached a plateau at the PCC concentration of 2.5 U/mL.
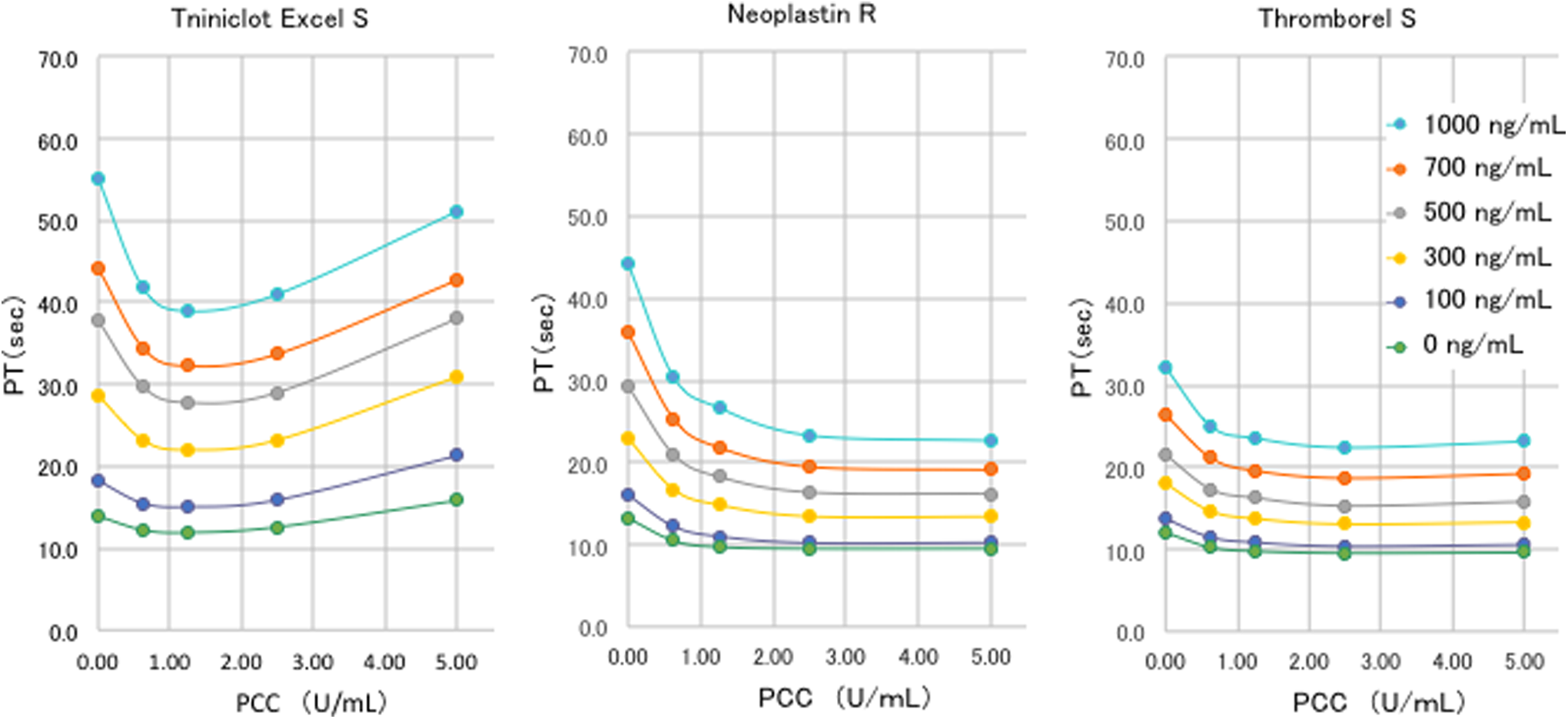
Impact of prothrombin complex concentrate (PCC) on the prothrombin time (PT) under the use of edoxaban. A concentration-dependent decrease in the PT was recognized after PCC administration. In the case of Triniclot Excel S, the PT decreased in a dose-dependent manner at a PCC dose between 0.63 and 1.25 U/mL. The PT started to prolong again when the PCC dose was over 2.5 U/mL. Such reprolongation was not observed in Neoplastin R and Thromborel S, where the PT reached a plateau at the PCC concentration of 2.5 U/mL.
Reversal Effect by aPCC
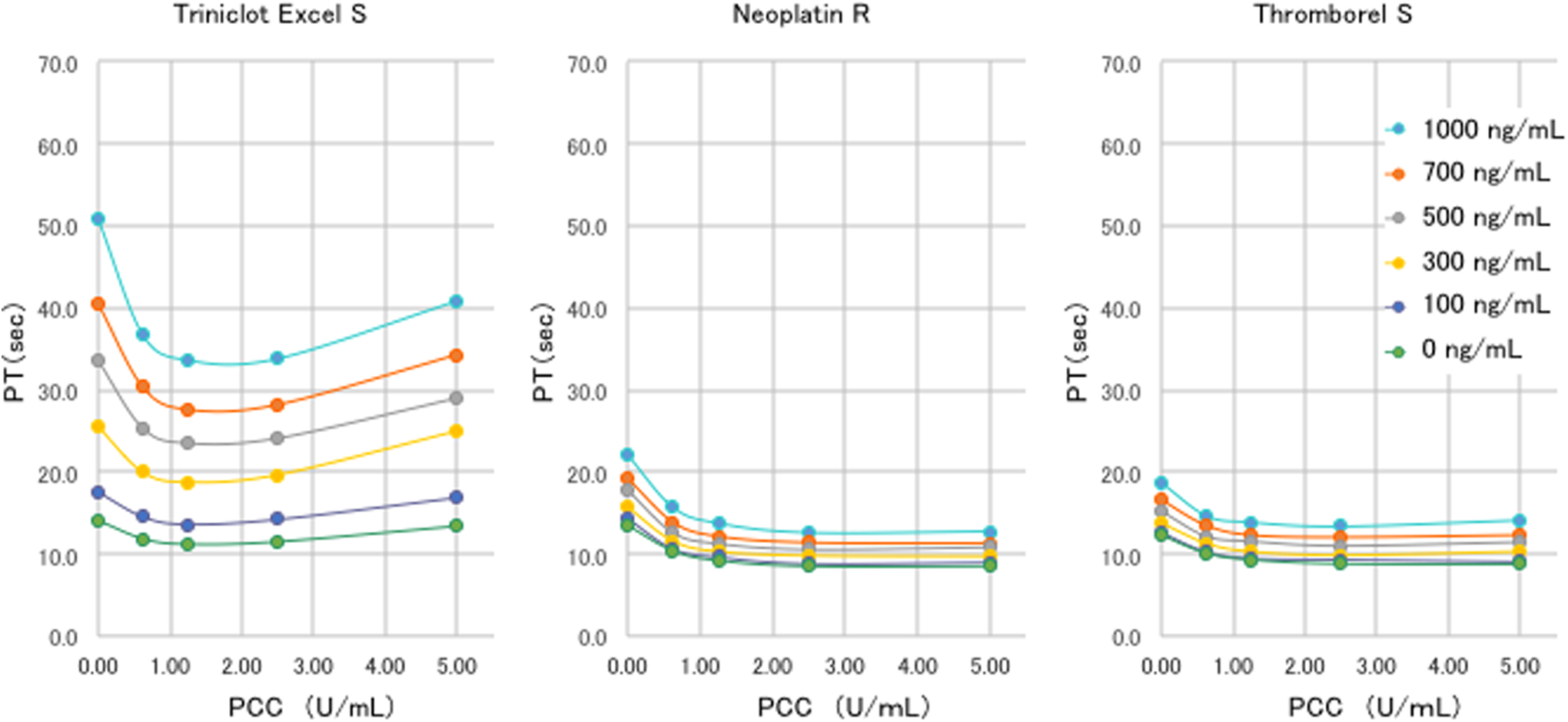
The examination for the reversal effect of aPCC was also repeated 3 times for each novel oral anticoagulant drugs (NOAC) and each PT test, and the representative results are shown in Figures 5 to 7. In a similar manner to PCC, the PT prolonged most with 100 ng/mL of rivaroxaban in Neoplastin and Triniclot, and 1.25 U/mL of aPCC reduced the PT measured by Neoplastin from 19.7 to 10.8 seconds with an RR of 0.66. The RR was 0.44 in Triniclot and 0.32 in Thromborel. Regarding the reprolongation of the PT recognized in PCC, such a phenomenon was not seen in any of the PT tests. The PT reached a plateau at an aPCC dose of 2.5 U/mL, and all 3 PT tests demonstrated similar trends. The changes in the PT were smaller in apixaban in all PT tests, and when the apixaban concentration was 100 ng/mL, the RR was 0.35 in Triniclot, 0.43 in Neoplastin, and 0.31 in Thromborel. In case of an edoxaban concentration of 100 ng/mL, 1.25 U/mL of aPCC reduced the PT by 0.60, 0.73, and 0.32 in Triniclot, Thromborel, and Neoplastin, respectively.
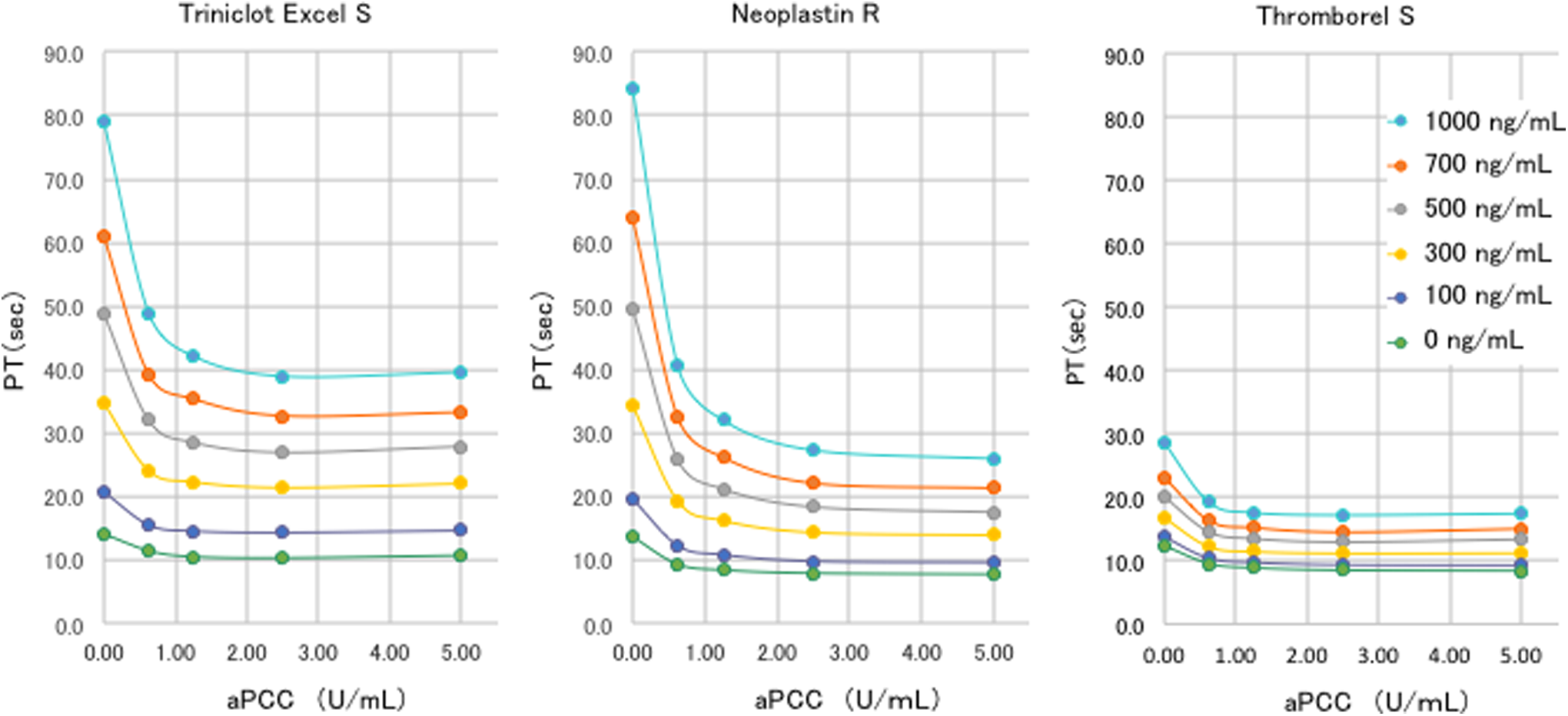
Impact of activated prothrombin complex concentrates (aPCC) on the prothrombin time (PT) under the use of rivaroxaban. A concentration-dependent decrease in PT was recognized after aPCC administration. When 0.63, 1.25, or 2.5 U/mL of aPCC was added to the plasma samples that had been spiked with rivaroxaban, each PT test showed the decrease in the PT in a dose-dependent manner. However, a further 5.0 U/mL of aPCC did not add any further effect.
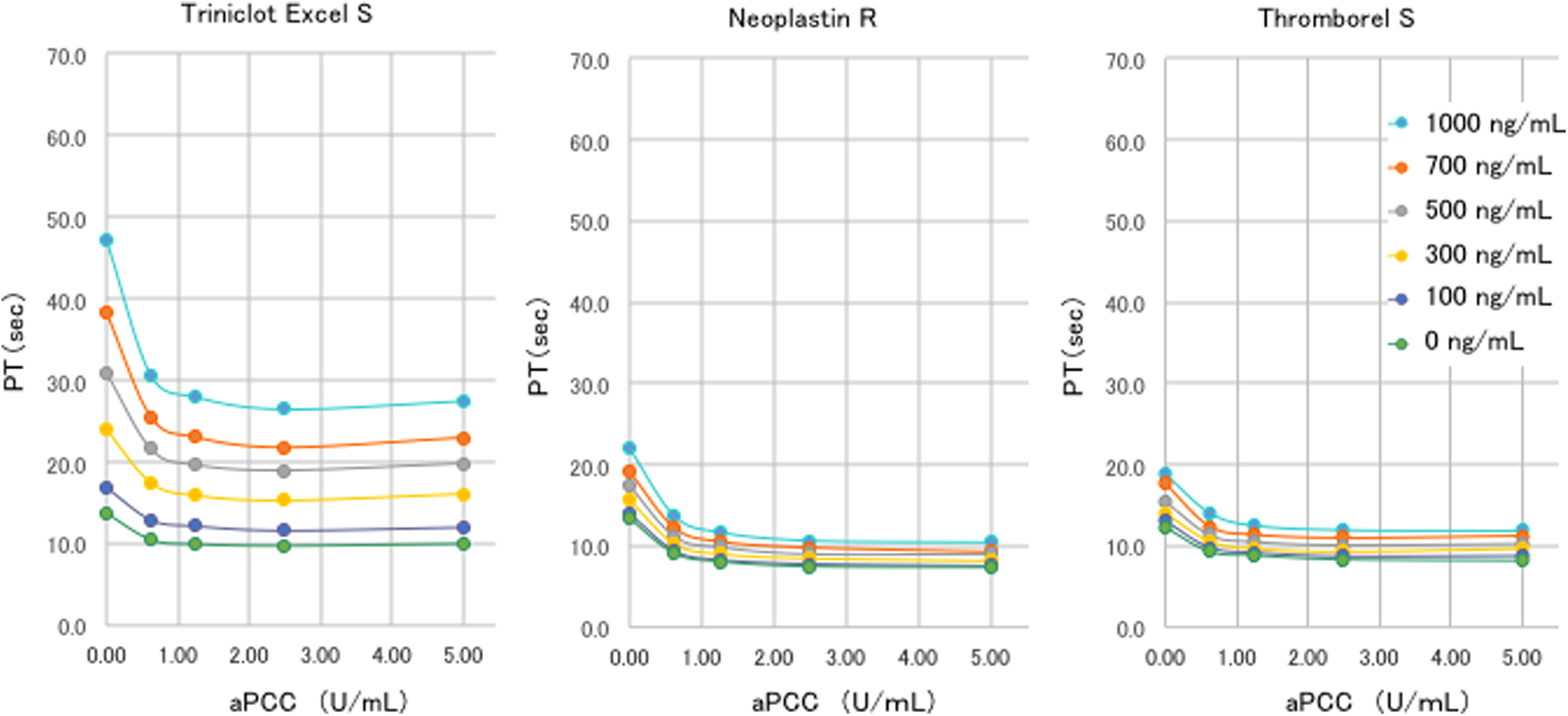
Impact of activated prothrombin complex concentrates (aPCC) on the prothrombin time (PT) under the use of apixaban. A concentration-dependent decrease in PT was recognized after aPCC administration. When 0.63, 1.25, or 2.5 U/mL of aPCC was added to the plasma samples that had been spiked with rivaroxaban, each PT test showed the decrease in the PT in a dose-dependent manner. However, a further 5.0 U/mL of aPCC did not add any further effect.
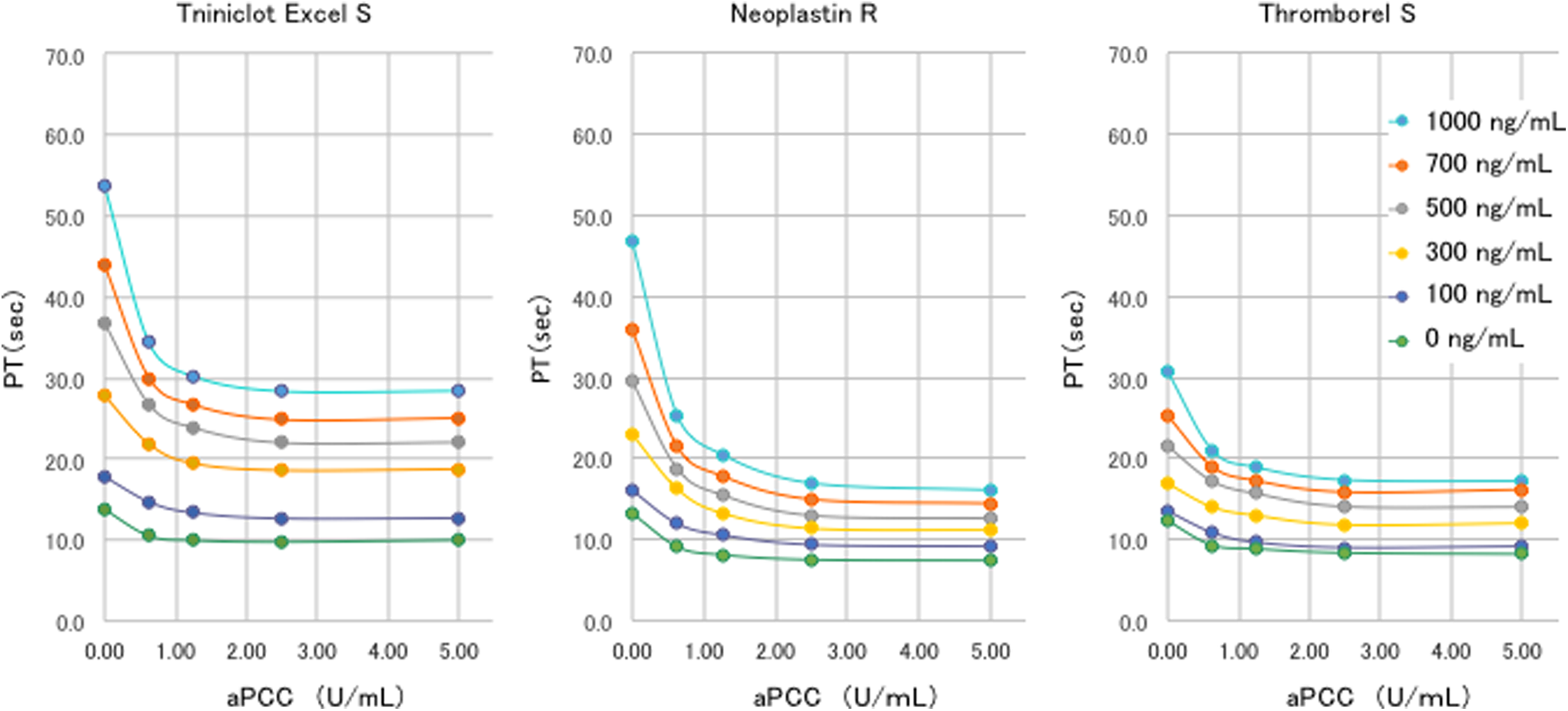
Impact of activated prothrombin complex concentrates (aPCC) on the prothrombin time (PT) under the use of edoxaban. A concentration-dependent decrease in the PT was recognized after aPCC administration. When 0.63, 1.25, or 2.5 U/mL of aPCC was added to the plasma samples that had been spiked with rivaroxaban, each PT test showed the decrease in PT in a dose-dependent manner. However, a further 5.0 U/mL of aPCC did not add any further effect.
Discussion
The present study showed that there was a good linear correlation between spiked DOAC concentration and the PT value of the 3 PT reagents. This study also revealed that there were considerable differences among the PT reagents in the magnitude of responses to DOACs. The reactivity for rivaroxaban would be sufficient in Neoplastin and Triniclot but not in Thromborel. In case of edoxaban, the reactivity would be acceptable in Triniclot but would not be sufficient in Thromborel. In the steady state of rivaroxaban therapy for the stroke prevention in nonvalvular atrial fibrillation (20 mg once daily), the peak plasma concentration at 3 hours after intake of rivaroxaban was reported to be ranging from 159.6 to 359.8 ng/mL. 11 Although significant prolongation of the PT was recognized with this range, considerable variability among the PT reagents existed. Thus, we have to consider which PT tests are most suitable for the detection of activity. We examined 3 popular PT tests; Neoplastin and Triniclot would be clinically useful for the monitoring of rivaroxaban, and Triniclot will be suitable for edoxaban. As for apixaban, Dale et al 6 reported that Triniclot showed moderate reactivity (slope = 0.24, R 2 = 0.993) to apixaban. Our measurement also resulted in a similar result (slope = 0.37, R 2 = 0.999), and we speculated the Triniclot might be useful to detect the apixaban dose. On the other hand, Douxfils et al 13 reported that PT tests were not sensitive enough to evaluate the activity of apixaban. Kanemoto et al 14 reported that PT values measured by Triniclot after 5-mg intake were 15.4 ± 1.7 seconds at the peak. Since the reference value of Triniclot is 12.7 to 15.2 seconds and the mean peak PT is just above the reference value, it might be difficult to detect the anticoagulant activity as a prolonged PT. This issue should further be examined in the clinical practice.
According to the sensitivity, the peak DOAC level after the standard dose (rivaroxaban, 20 mg once daily; apixaban, 5 mg twice daily; edoxaban, 60 mg once daily) intake can be measurable by adequate PT tests in cases of rivaroxaban and edoxaban. However, since the reported trough level of rivaroxaban was 22.3 ng/mL, 15 all PT tests may not be sensitive enough to detect this low range.
Although not all PT tests are sufficiently sensitive, we think that monitoring with the PT is still valuable. The incidence of serious bleeding is reportedly 2.12% per year when rivaroxaban was administered at 20 mg/d, 16 1.27% when 5 mg of apixaban was administered twice daily, 17 and 2.75% per year when edoxaban was administered at 60 mg/d, 18 and it was also reported that proper dosing decreases the fatal events. 19 Since the activity of DOACs peaks at 1 to 3 hours, 20 we recommend that the peak PT value should be checked with a proper test for the effective and safe control. In addition, the measurement of the PT will be extremely useful in the emergency setting. Except for the developing of specific antidotes, 21 –23 it has been widely accepted that PCC or aPCC is efficacious for the reversal of DOACs, 24 –27 and administration of PCC 25 to 50 U/kg or aPCC 30 to 50 U/kg has been generally recommended if an overdose was suggested. 27 Thus, it will be favorable if the PT could be used to the necessity of the treatment and could reflect the reversal effects of prohemostatic agents. Several reports have reported the usefulness of PT tests for those purposes, 28 however, only a few studies have compared the performance of different PT tests, and the results are inconsistent. 29,30
Prothrombin complex concentrate contains inactive factors II, IX, X, and VIIa, and aPCC contains activated factor VII and factors II, IX, and X. 8 The American Heart Association guidelines recommended the use of PCC or aPCC as a potential reversal agent. 7 In the present study, the PT measured with Triniclot decreased dose dependently from a PCC dose of 0.63 to 1.25 U/mL, and then the PT started to prolong when the PCC dose was over 2.5 U/mL. This phenomenon was estimated to reflect the effects of heparin added to PCC. Prothrombin complex concentrate and aPCC were originally developed for the replacement of vitamin K-dependent clotting factors. Since the thrombotic adverse events have been reported as a major adverse event, 9 heparin was added to the second-generation PCCs. For example, PPSB-HT contains 0.25 U of heparin sodium in 1 U of PCC. Then, does this heparin interfere with hemostasis? We think the phenomenon was observed only in an ex vivo study. The anticoagulant effect of heparin may not be of concern in practical use because the recommended PCC dose is 50 U/kg maximal (approximately 1 U/mL).
Interestingly, the reprolongation effect of PCC was observed only in the Triniclot. This is because other PT tests contain protamine to reverse heparins. Thus, we think Triniclot is more suitable for evaluating the net reversal effect of PCC.
In summary, a linear and dose-dependent prolongation of PT was recognized in cases of rivaroxaban and edoxaban at the therapeutic range. Triniclot will be the most appropriate PT test for the monitoring of DOACs. For the evaluation of the reversal effects of heparin-containing PCC, we think Triniclot is more suitable than the others. After all, it is important to keep it in mind that there are considerable differences between the PT tests.
Finally, there are some limitations in this study. First, we examined the PT in relatively high-concentration DOACs to examine the reversal effects in overdose cases. However, the PT in lower concentrations of DOACs needs to be examined to evaluate the monitoring effect in daily practice. Second, we examined only a limited number of PT tests in the present study. Since considerable differences were revealed in this study, physicians need to recognize the reactivity and the characteristic of PT tests used in their own facilities. Finally, since each PT test showed linear correlation between the concentration and PT, it would be more favorable if the data could be comparable by simple equation.
Conclusion
Prothrombin time tests will not be suitable for the routine monitoring of DOACs, however, they could be useful in certain occasions such as overdose, complicating the critical bleeding and considering the treatment with PCC or aPCC. However, since significant variability exists, physicians should be aware of the sensitivity of their own assays to each DOACs. Prothrombin time tests such as Triniclot and Neoplastin will be the useful assays to measure pharmacodynamic effects of rivaroxaban and edoxaban in the emergency care setting. The reversal effects of PCC and aPCC could also be evaluated by proper PT tests. It is therefore suggested that PCC and aPCC could better be used for emergency reversal of DOACs under monitoring by these PT tests.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: M. Emmi works for Kyowa Medex Co Ltd.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
