Abstract
Background
The concomitant therapy of amiodarone with direct oral anticoagulants (DOACs) significantly increases the concentrations of DOACs and may increase the bleeding risks. Multiple real-world studies compared the concomitant therapy of amiodarone and DOACs versus the DOAC alone, but their main findings were contradictory.
Methods
A meta-analysis compared the concomitant therapy of amiodarone and DOACs versus DOACs monotherapy. Database searches through May 1, 2025, were performed. The primary outcome was major bleeding. The secondary outcomes included stroke or systemic embolism, any bleeding, all-cause mortality, gastrointestinal bleeding, and intracranial bleeding.
Results
This meta-analysis included a total of nine studies. There were no significant differences in major bleeding between the concomitant therapy of amiodarone and DOAC and DOAC monotherapy groups (OR 1.12; 95% CI 0.98, 1.27; P = .09; I2 = 64%). No significant differences in any bleeding (OR 1.18; 95% CI 0.88, 1.57; P = .27; I2 = 77%), gastrointestinal bleeding (OR 0.97; 95% CI 0.84, 1.11; P = .65; I2 = 56%), and intracranial bleeding (OR 1.14; 95% CI 1.00, 1.30; P = .05; I2 = 32%) were also found between the two groups. No significant differences in stroke or systemic embolism were found between the two groups (OR 0.86; 95% CI 0.74, 1.00; P = .05; I2 = 34%).
Conclusion and Relevance
The concomitant therapy of amiodarone and DOACs did not significantly increase the bleeding risks or decrease the risk of stroke or systemic embolism compared to the DOAC monotherapy. The drug-drug interactions between amiodarone and DOACs may not be clinically significant.
Background
The most common supraventricular tachyarrhythmia, atrial fibrillation (AF), can increase mortality and morbidity. 1 Especially, for patients with high stroke risks, long-term anticoagulation therapies are recommended to prevent stroke or systemic embolism due to AF. The American Heart Association/American College of Cardiology/American College of Clinical Pharmacy/Heart Rhythm Society guidelines for AF recommend direct oral anticoagulants (DOACs) over warfarin. 1 Amiodarone is one of the widely used antiarrhythmic agents for AF especially in patients with heart failure with reduced ejection fraction. Thus, the concomitant therapy of DOACs and amiodarone is commonly seen in clinical practice. All DOACs are substrates for p-glycoprotein or both p-glycoprotein/CYP3A4 enzymes, while amiodarone is a p-glycoprotein/moderate CYP 3A4 inhibitor. 2 A prospective pharmacokinetic study by Limcharoen et al 3 showed the apixaban trough level in the concomitant therapy of apixaban and amiodarone was increased by about 60% compared to the apixaban monotherapy. Also, a pharmacokinetic study by Ding et al 4 corroborated that the concomitant therapy of amiodarone and rivaroxaban increased the rivaroxaban level by about 50% to 100% compared to the monotherapy. Multiple observational studies evaluated this drug-drug interaction in practice, but the conclusions are conflicted among studies. The primary objective of our meta-analysis is to compile the existing studies and quantify the effects of the drug-drug interaction on bleeding and stroke outcomes in patients with AF.
Methods
Literature search words included (amiodarone) and (apixaban or rivaroxaban or dabigatran or edoxaban). Database searches were performed through May 1, 2025, using Cochrane Library, MEDLINE, Google Scholar, and Web of Science. Two independent scholars (KK and MH) searched the database and selected the articles. Studies were included if they had event rates of clinical outcomes in the concomitant therapy of amiodarone and DOACs and the control group. The control groups are DOACs only or concomitant therapy of DOACs and antiarrhythmic agents minimally interacting with DOACs. Studies were excluded if they were only available as conference abstracts, non-English articles, or event numbers were not available. The primary outcome was major bleeding. The secondary outcomes were stroke or systemic embolism, all-cause mortality, any bleeding, gastrointestinal bleeding, and intracranial bleeding. Newcastle-Ottawa Scale was selected to evaluate the qualities of observational studies. 5
A random-effects model was selected. I2 statistics was used for heterogeneity assessment. The Egger regression test was used for the publication bias. The analysis estimated an odds ratio (OR) and 95% confidence interval (CI), and a P-value < .05 was defined as a statistical significance. The Preferred Reporting Items for Systematic Reviews and Meta-analysis guidelines were adopted for the meta-analysis. 6 Our meta-analysis used RevMan 5.4.1 (Nordic Cochrane Centre, Cochrane Collaboration, Copenhagen).
Results
This meta-analysis for the primary outcome included a total of nine studies (Figure 1).4,7–14 All included studies were AF populations. Six were cohort studies and three were post-hoc analyses of the randomized controlled trials. Case-control studies by Gronich et al 15 and Shurrab et al 16 were excluded because the raw event data was not available for the analysis. Two studies by Gosselin et al 17 and Hanigan et al 18 were also excluded due to no access to raw event data for the included outcomes. The study by Zhang et al 19 was also excluded due to the lack of a control group. The study by Hill et al 20 was also excluded because only bleeding requiring hospitalization or emergency room visits was investigated as an outcome. The baseline characteristics of the included studies are described in Table 1. A total of 118,037 patients in the amiodarone and DOAC group and 224,539 patients in the DOAC group without amiodarone were included in the meta-analysis for the primary outcome.

Flow Diagram of the Literature Search.
Baseline Characteristics of Included Studies.

Abbreviations: AF, atrial fibrillation; CrCL, creatinine clearance; DOAC, direct oral anticoagulants; NR, not reported; Scr, serum creatinine.
Displayed as mean
There were no significant differences in major bleeding between the concomitant therapy of amiodarone and DOAC and DOAC only groups (OR 1.12; 95% CI 0.98, 1.27; P = .09; I2 = 64%) (Figure 2). No significant differences in any bleeding (OR 1.18; 95% CI 0.88, 1.57; P = .27; I2 = 77%) (Figure 2), gastrointestinal bleeding (OR 0.97; 95% CI 0.84, 1.11; P = .65; I2 = 56%) (Figure 3) and intracranial bleeding (OR 1.14; 95% CI 1.00, 1.30; P = 0.05; I2 = 32%) (Figure 3) were also found between the two groups. No significant differences in stroke or systemic embolism (OR 0.86; 95% CI 0.74, 1.00; P = .05; I2 = 34%) (Figure 4) or all-cause mortality (OR 1.58; 95% CI 0.72, 3.46; P = .26; I2 = 99%) (Figure 4) were found between two groups. The Egger regression test showed no significant funnel plot asymmetry (no significant publication bias).
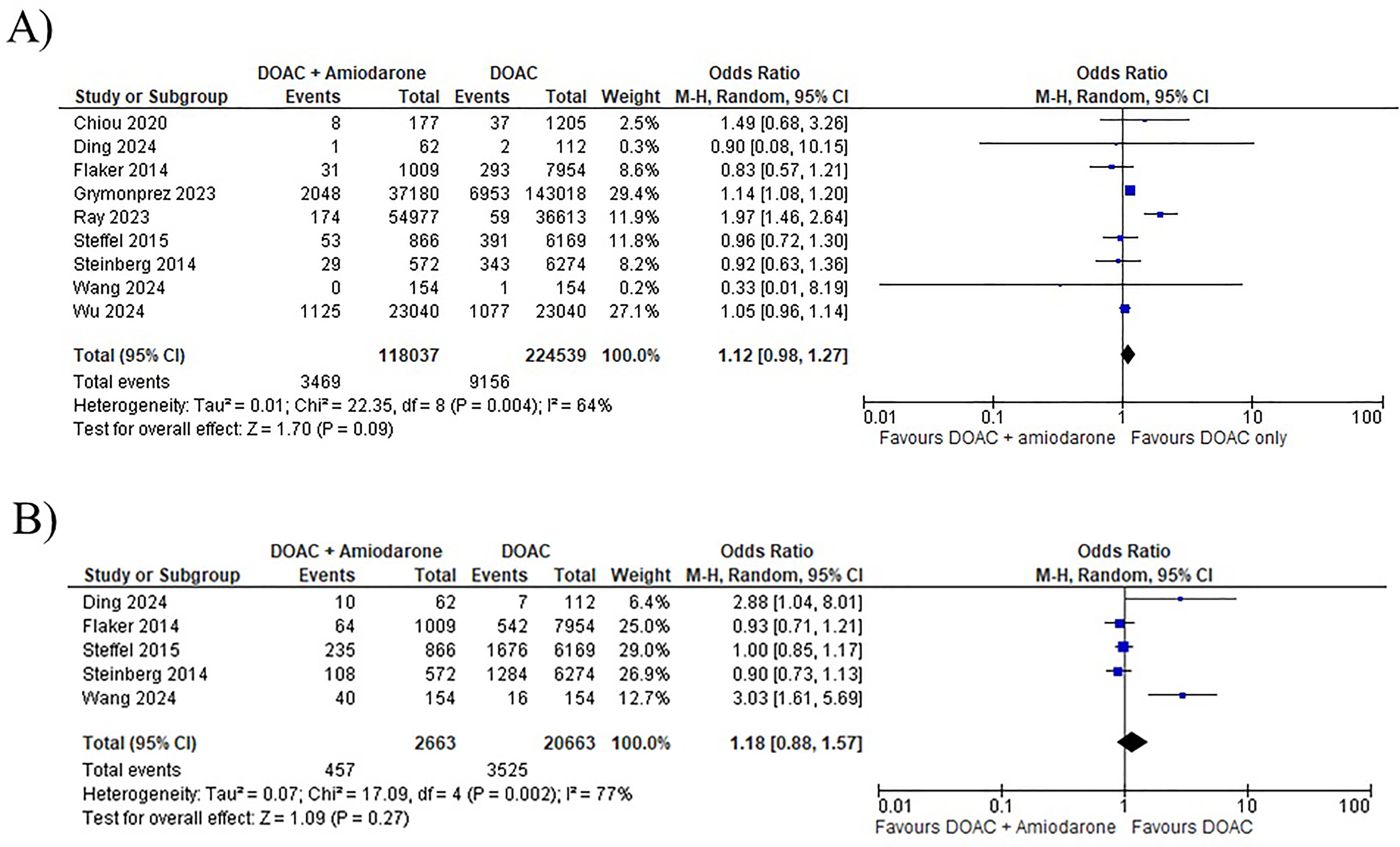
Forest Plots of the Estimated Effect of (A) Major Bleeding and (B) Any Bleeding in Patients Receiving the Concomitant Therapy of Amiodarone and Direct Oral Anticoagulants Versus Direct Oral Anticoagulant Only.

Forest Plots of the Estimated Effect of (A) Gastrointestinal Bleeding and (B) Intracranial Bleeding in Patients Receiving the Concomitant Therapy of Amiodarone and Direct Oral Anticoagulants Versus Direct Oral Anticoagulant Only.

Forest Plots of the Estimated Effect of (A) Stroke or Systemic Embolism and (B) All-Cause Mortality in Patients Receiving the Concomitant Therapy of Amiodarone and Direct Oral Anticoagulants Versus Direct Oral Anticoagulant Only.
Discussion
Our meta-analysis showed that the addition of amiodarone to DOACs did not significantly increase the risks of bleeding outcomes but also did not decrease the stroke or systemic embolism risk compared to the DOAC alone.
Amiodarone is a CYP 3A4 enzyme and p-glycoprotein inhibitor and DOACs are substrates of CYP3A4 and p-glycoprotein. 2 The concomitant therapy of amiodarone with DOACs is expected to increase the levels of DOACs due to the inhibition of CYP3A4 and p-glycoprotein. A prospective pharmacokinetic study by Ding et al 4 evaluated the effect of amiodarone in patients with AF receiving rivaroxaban. It was found that rivaroxaban levels significantly increased from 37.40 ± 60.70 to 77.65 ± 88.90 ng/mL. However, the study reports no significant differences between the two groups in prothrombin time and activated partial thromboplastin time. Another pharmacokinetic study showed that apixaban trough level in the concomitant therapy of apixaban and amiodarone group (n = 11) was significantly higher than the apixaban monotherapy group (n = 22) (162.05 [87.94-292.88] vs 108.49 [78.10-171.52] mcg/L). 3 However, bleeding events were numerically more frequent in the apixaban monotherapy (22.7% vs 18.2%).
The studies evaluating the concomitant therapy of amiodarone and DOACs revealed inconsistent findings in associations of bleeding and stroke risks. A single-center retrospective cohort study by Hanigan et al 18 showed a significantly increased risk of any bleeding in the concomitant therapy group receiving combined p-glycoprotein and moderate CYP 3A4 inhibitors and apixaban or rivaroxaban compared to the control group (only apixaban or rivaroxaban). However, most combined p-glycoprotein and moderate CYP 3A4 inhibitors were diltiazem at 65.6% to 75.0% and amiodarone at 26.1% to 28.6%. Thus, this study's main findings may not reflect the effect of amiodarone on the risks of bleeding in patients with AF. A large-scale retrospective cohort study by Chang et al 21 also found a significantly increased risk of major bleeding in the concomitant therapy group receiving DOACs and amiodarone compared to the DOAC alone group. Of note, the subgroup analysis showed that the significant associations were only shown in patients receiving dabigatran or rivaroxaban. Two posthoc analyses of DOAC randomized controlled trials reported the effects of amiodarone on clinical outcomes in patients receiving either DOACs (rivaroxaban in the ROCKET-AF and apixaban in the ARISTOLE trials) or warfarin.7,10 Although they did not specifically compare the concomitant therapy of DOACs and amiodarone with DOACs, the event rates of major bleeding and stroke or systemic embolism between the two groups were numerically comparable. Our meta-analysis also showed no significant differences in major bleeding and stroke or systemic embolism. These contradictory findings among studies may have been influenced by the differences in the DOACs, DOAC dosing, the concomitant antiplatelet therapy, age, and comorbidities.
Our meta-analysis has several limitations. First, some clinical outcomes were not evaluated because three or more studies were not available to report these outcomes. Second, all included studies were either observational studies or a posthoc analysis result of the randomized controlled trial. Third, this meta-analysis did not have enough power to analyze clinical outcomes based on the subgroup of an individual DOAC agent (dabigatran vs apixaban vs rivaroxaban vs edoxaban). Also, most included studies investigated the use of either rivaroxaban or apixaban. Thus, the main results may not apply to other DOACs. Fourth, the study did not have enough power to perform subgroup analyses based on renal or liver function. Finally, reported dosing regimens were not standardized among included studies, or details in dosing regimens were not reported in some studies. Future larger-scale study for subgroup analyses with various doses of DOACs is needed.
In conclusion, this meta-analysis revealed that the concomitant therapy of amiodarone and DOACs did not significantly increase bleeding risks or decrease the risk of stroke or systemic embolism, compared to the DOAC alone without amiodarone. The drug-drug interactions between amiodarone and DOACs may not be clinically significant.
Supplemental Material
sj-docx-1-cpt-10.1177_10742484251351148 - Supplemental material for The Concomitant Therapy of Direct Oral Anticoagulants with Amiodarone in Atrial Fibrillation: A Meta-analysis
Supplemental material, sj-docx-1-cpt-10.1177_10742484251351148 for The Concomitant Therapy of Direct Oral Anticoagulants with Amiodarone in Atrial Fibrillation: A Meta-analysis by Kazuhiko Kido, PharmD, PhD, Mikiko Shimizu, PhD, Tsuyoshi Shiga, MD, PhD, and Masayuki Hashiguchi, PhD in Journal of Cardiovascular Pharmacology and Therapeutics
Footnotes
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
Data sharing not applicable to this article as no datasets were generated or analyzed during the current study.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
