Abstract
Rivaroxaban is a target-specific oral anticoagulant approved for the treatment of venous thromboembolism (VTE). On its major clinical trials, treatment was initiated directly with a 3-week dose of oral 15 mg twice daily followed by 20 mg every day for at least 3 months. We retrospectively evaluated an initial therapy for confirmed VTE with 1 to 18 days of enoxaparin (1 mg/kg twice daily parenteral) followed by oral rivaroxaban 20 mg every day. Of 49 patients, we found no symptomatic recurrence, no major bleeding, and only 1 clinically relevant nonmajor bleeding. We concluded in this pilot study that it is safe and effective to treat patients with enoxaparin course followed directly by a dose of 20 mg of rivaroxaban.
Introduction
Venous thromboembolism (VTE) is a prevalent and potentially fatal health problem affecting over 900 000 in the United States and 1 one million people in Europe annually. 1
Since the classic study by Barritt and Jordan, 2 anticoagulation has been the mainstay of therapy, initially with unfractionated heparin (UFH) being converted to vitamin K antagonists (VKAs).
This was the standard of care until low-molecular-weight heparin (LMWH) compounds were developed, offering incremental benefits over standard UFH and, in some instances, VKAs. 3 –6
Recently, evidence for target-specific oral anticoagulant (TSOAC) options for treatment of VTE has emerged. Rivaroxaban 7 and apixaban 8 do not require initial treatment with parenteral agents, whereas dabigatran 9 and edoxaban 10 does. However, even on the clinical trials, 90% of patients enrolled received at least 48 hours of parenteral therapy prior to TSOACs treatment. Until recently, of all TSOACs, only rivaroxaban had health authorities approval for the treatment of VTE in Brazil. Now, dabigatran is also an option, whereas apixaban has a pending approval. In our institution, physicians still do not feel confident in treating VTE without initial dose of parenteral anticoagulants, particularly to their familiarity and experience with LMWHs. The on-label instructions for treatment of VTE with rivaroxaban includes 15 mg twice daily for 3 weeks followed by 20 mg every day for 6 months. 11 This retrospective analysis aims to report the results on efficacy and safety of a parenteral anticoagulant therapy (1-18 days) followed by a TSOAC (rivaroxaban).
Materials and Methods
The review board of Hospital Israelita Albert Einstein (HIAE) approved this study. Our goal was to evaluate efficacy (recurrence of symptomatic VTE) and safety (major bleeding and clinically relevant nonmajor [CRNM] bleeding) in patients who received parenteral anticoagulant therapy (1-18 days) followed directly by 20 mg every day of rivaroxaban for as long as 3 years, in a retrospective fashion. The hypothesis for this pilot study was that the use of parenteral anticoagulation prior to TSOACs use did not affect efficacy and safety compared to results observed in major phase III clinical trials.
Medical charts were reviewed from 49 patients who were treated at HIAE for lower limb VTE with parenteral anticoagulant therapy followed by rivaroxaban for 6 months, from February 2011 to April 2015, searching for recurrence of symptomatic VTE and bleeding. Both outcomes were clinically evaluated by the same group that initially treated the patients during follow-up period, providing reliability on outcomes measurements. In addition, data on the curve of
The numerical variables were expressed using mean and standard deviation. The paired Wilcoxon test was used to compare initial and final concentrations of serum biomarkers. It is a cohort retrospective report of a series of cases, where sample size calculations or noninferiority margins were not required.
Results
The baseline characteristics of patients are summarized in Table 1. There was a predominance of male patients (n = 29; 59.2%). Age ranged from 20 to 91 years old (mean = 53.6; standard deviation [SD] =18.1). Body mass index ranged from 16.8 to 37.1 kg/m2 (mean = 27.5; SD = 4.2). The length of stay in hospital varied from 1 to 23 days, and the average was 5 days (SD = 3.4). The mean follow-up period was 219.9 days (SD = 256.7), with the range 7 to 1294 days.
Patient Demographics.
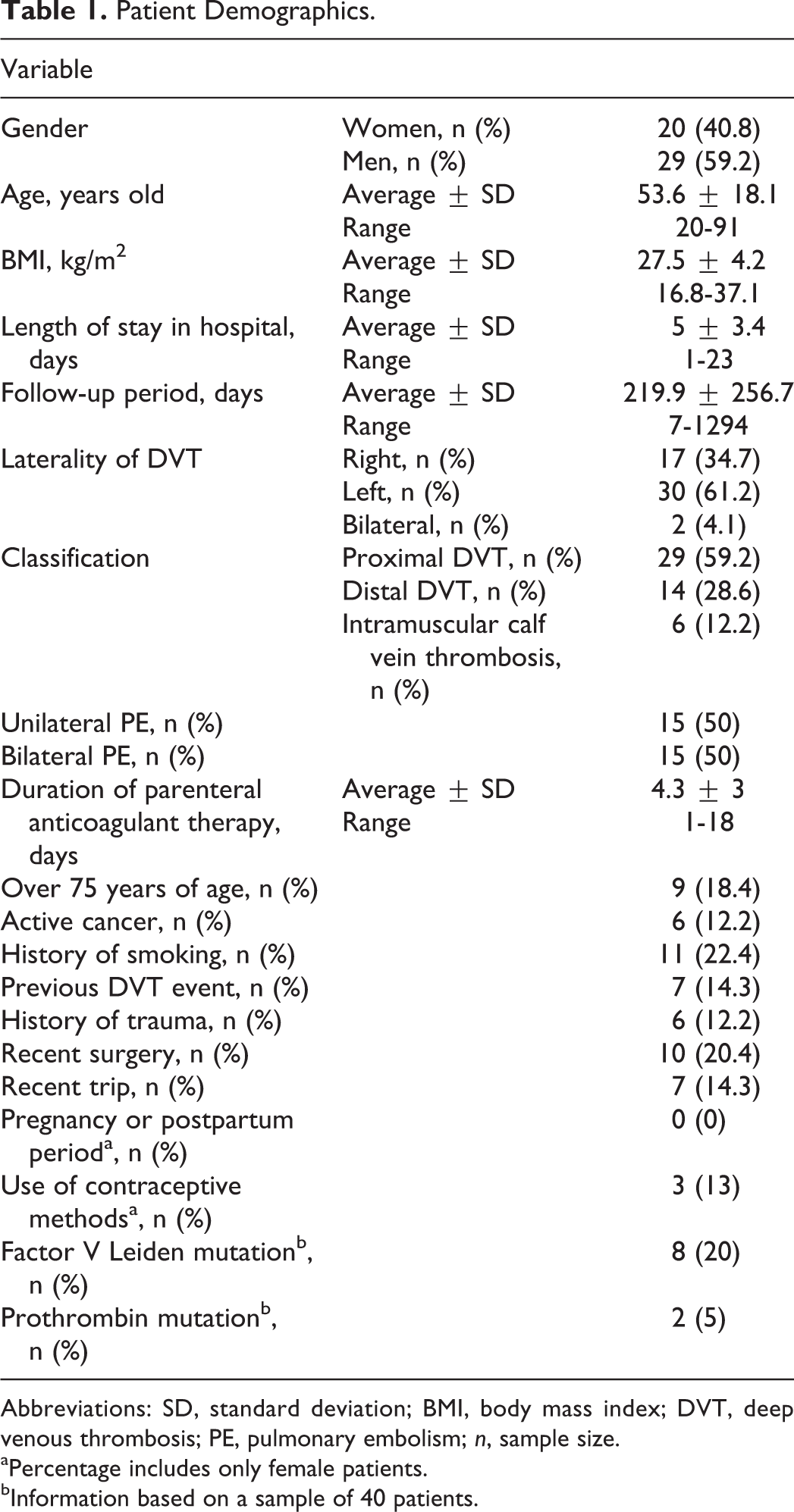
Abbreviations: SD, standard deviation; BMI, body mass index; DVT, deep venous thrombosis; PE, pulmonary embolism; n, sample size.
aPercentage includes only female patients.
bInformation based on a sample of 40 patients.
As we can see from the baseline data, 34.7% of the patients presented with deep venous thrombosis (DVT) of the right lower limb, 61.2% with left lower limb, and 4.1% with bilateral DVT. According to the site of the thrombosis, we observed that 59.2% had proximal (iliac, femoral, and popliteal veins) DVT, 28.6% had distal (tibial and peroneal veins) DVT, and 12.2% had intramuscular (gastrocnemius and soleal veins) calf vein thrombosis. The presence of pulmonary embolism (PE) was observed in 61.2% of the patients, and it was bilateral in 50% of these patients. The parenteral anticoagulant therapy was prescribed for an average of 4.2 days (SD = 3), with the range 1 to 18 days.
Among the risk factors analyzed, 18.4% of the patients were older than 75 years old by the time of the VTE diagnosis, 12.2% of the patients presented with active cancer, and 22.4% with a history of smoking. A previous DVT event was noted in 24.5% of the sample. It was observed that 12.2% of the patients had an antecedent history of trauma, 20.4% were recently submitted to surgery, and 14.3% had a recent trip (a trip in the last 30 days). None of the women was pregnant or in the postpartum period. The use of contraceptive methods was observed in 13% of female patients. Thrombophilia screening was available in medical records of 40 patients: Factor V Leiden mutation was positive in 20%, and prothrombin mutation was positive in 5% of this sample.
Data on both
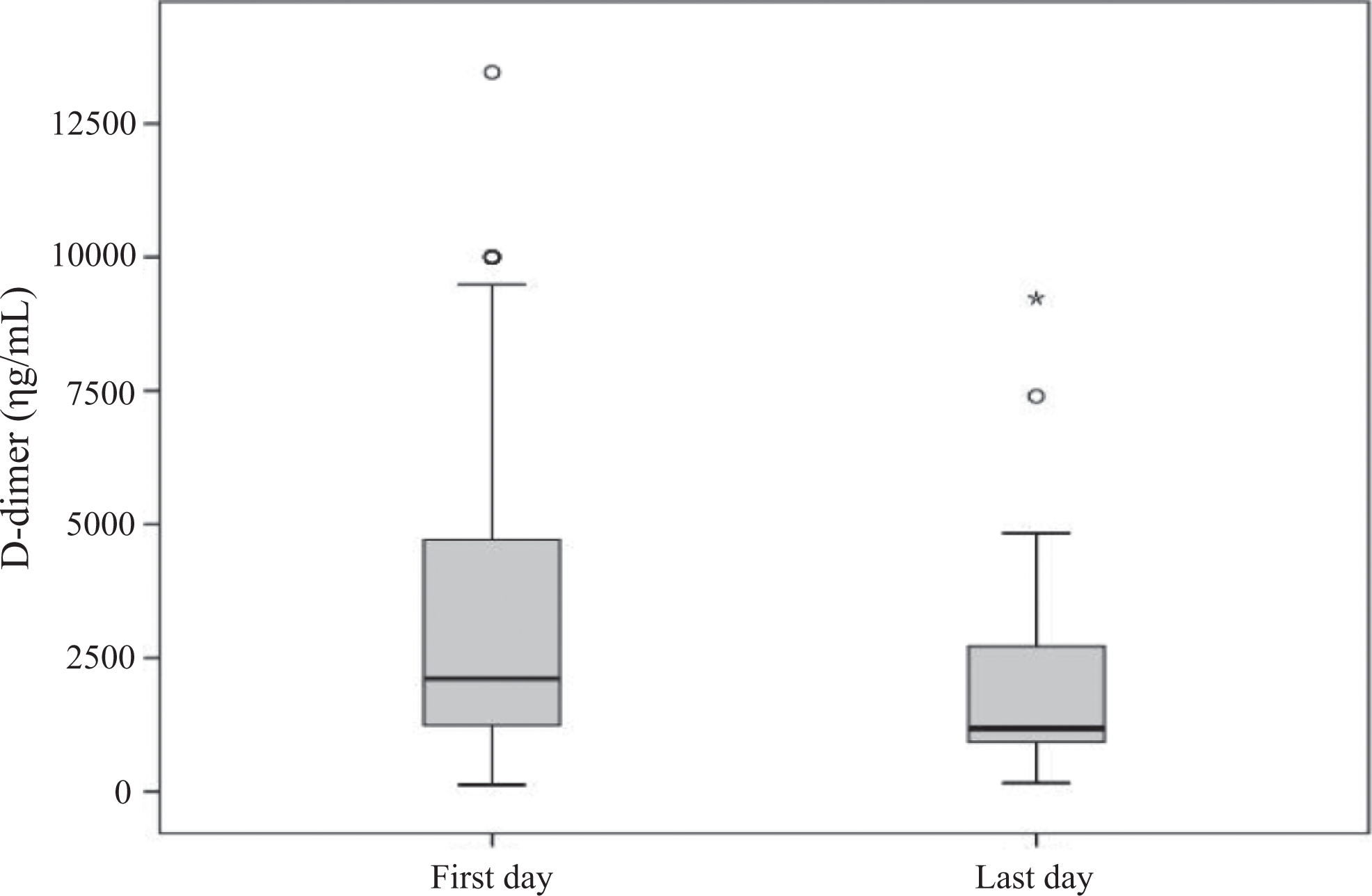
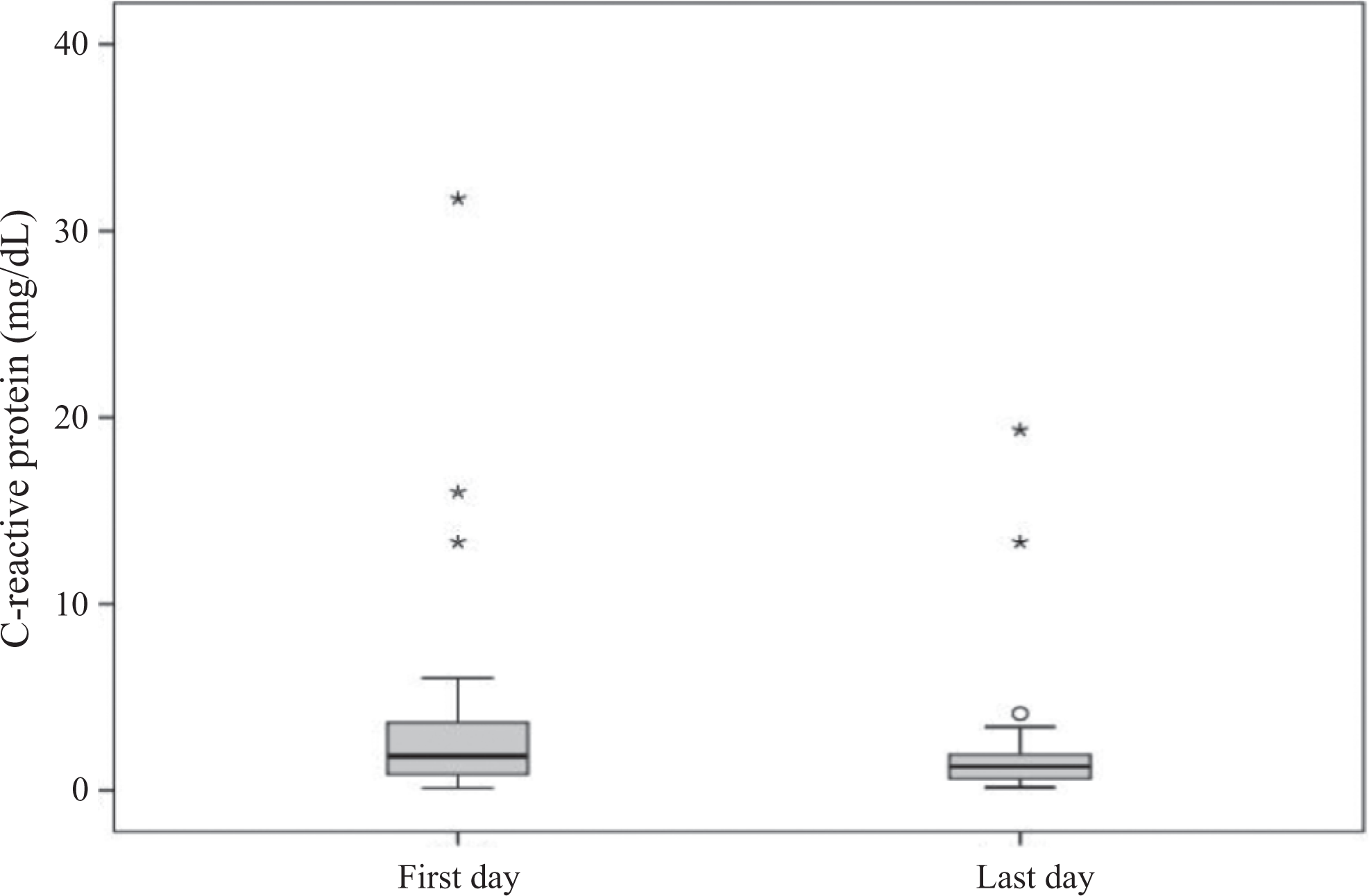
C-reactive protein (CRP) results (initial and final days of treatment) for patients with venous thromboembolism (VTE) submitted to enoxaparin followed by rivaroxaban 20 mg every day for 6 months.
Of the 49 patients, we found no symptomatic recurrence, no major bleeding, and only 1 clinically relevant nonmajor bleeding on the 6-month postindex event follow-up. It was a 41-year-old woman who presented with a painful swollen right leg 7 days before hospital admission as the index event (first episode of DVT). A duplex scan confirmed a popliteal and medial gastrocnemius vein thrombosis. The computed tomography scan was negative for PE. She has factor V Leiden mutation, a positive history of smoking, and had a calf muscle injury with medial gastrocnemius strain during running activity 30 days before hospital admission. The patient was treated with parenteral anticoagulant therapy for 1 day, was hospitalized for 2 days, was prescribed rivaroxaban 20 mg every day, and has a 322 days follow-up period. She presented with a single episode of hypermenorrhea after 3 months of the hospitalization, and the bleeding ceased after 1 day without the need to withdraw the oral anticoagulant.
Discussion
Until recently, VKAs have been the only oral anticoagulant available for the management of VTE. However, VKAs have a number of disadvantages that complicate patient care. The VKAs have unpredictable pharmacokinetics and pharmacodynamics, have food and drug interactions, and function within a narrow therapeutic window requiring frequent laboratory monitoring. 11,12 Therefore, the management of patients receiving VKAs is cumbersome.
The most recent advances in the management of patients with VTE are the TSOACs that directly inhibit factor Xa or factor IIa, which are strategically positioned in the coagulation cascade. Factor Xa is located at the intersection of the intrinsic and extrinsic coagulation pathways prior to its interaction with factor II (prothrombin). The TSOACs directly bind to activated factors X and II. These new drugs are fixed-dose oral agents that, unlike VKAs, do not require laboratory monitoring or dose adjustment and have a low potential for food and drug interactions.
The EINSTEIN investigators studied oral rivaroxaban for symptomatic VTE 7 and symptomatic PE. 13 The EINSTEIN investigators demonstrated that equal efficacy could be achieved in the treatment of acute VTE using rivaroxaban 15 mg orally twice daily for 3 weeks followed by 20 mg every day over the long term compared with conventional therapy using heparin followed by VKA. There were no differences in major and CRNM bleeding.
The
The major finding in our study is that the use of enoxaparin course did not increase neither recurrences nor bleeding events. We observed no recurrence, no major bleeding, and only 1 clinically relevant nonmajor bleeding. A limitation of this study is the lower number of events, attributable to the small sample size of this pilot evaluation. Since both drugs have short half-lives, we believe that they can be safely transitioned, with no overlapping. The advantage of been able to start therapy directly with an oral agent such as rivaroxaban is out-shadowed by the experience of clinicians by treating the initial phase of a VTE with enoxaparin. Due to its anti-inflammatory and other effects, physicians feel more confident in its initial effects, particularly in decreasing pain and protecting from PE extension. The drawbacks of this study are its limited sample size, not allowing us to build thrust worthy confidence intervals and its retrospective design. Another limitation was no standardization of duration of LMWH treatment. Nevertheless, it describes what we have been observing in our center: initial treatment with enoxaparin followed by a direct TSOAC transition. This study is a pilot one to a larger study using the same design, LMWH, or fondaparinux followed directly by 20 mg every day rivaroxaban, where parenteral period will be standardized for 5 to 11 days, following the design of major clinical trials in VTE treatment.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
