Abstract
Traditional anticoagulants, such as low-molecular-weight heparin and vitamin K antagonists, have been the mainstay for the treatment of venous thromboembolism (VTE) in the hospital setting and after discharge. These anticoagulants are effective but are associated with some limitations that may lead to their underuse. Based on the results of the EINSTEIN clinical trial program, the oral, direct factor Xa inhibitor rivaroxaban is approved for the treatment of acute deep vein thrombosis (DVT) and pulmonary embolism (PE) and for the prevention of recurrent VTE. The single-drug approach with rivaroxaban is now available in both the hospital and the outpatient settings and may overcome some of the limitations of traditional agents. This review provides hospital physicians with an overview of the practical management of rivaroxaban and a critical evaluation of its use for the treatment of DVT and PE, including in specific clinical settings and special patient populations.
Introduction
Venous thromboembolism (VTE) occurs at a rate of 1 to 2 cases per 1000 persons per year. 1,2 Hospitalized patients in particular are at increased risk of VTE and, therefore, require adequate anticoagulation. 3 When an acute VTE occurs, initial anticoagulation is essential to prevent thrombus extension and the occurrence of fatal or nonfatal pulmonary embolism (PE); treatment beyond the initial phase aims to prevent recurrence and, ultimately, to dissolve the thrombus by preventing extension. 4
Evidence-based guidelines for the treatment of acute deep vein thrombosis (DVT) and PE recommend the use of low-molecular-weight heparin (LMWH), unfractionated heparin (UFH), fondaparinux, and vitamin K antagonists (VKAs; after initial overlapping anticoagulation until the international normalized ratio [INR] is 2.0-3.0); anticoagulation of these patients is recommended for at least 3 months. 4 The LMWHs, UFH, and fondaparinux are fast-acting parenteral anticoagulants that have a good safety profile in terms of bleeding events. 5 However, traditional agents are associated with a number of limitations. The parenteral route of administration renders these agents inconvenient for long-term use, and UFH is also associated with the risk of heparin-induced thrombocytopenia. 5 The VKAs are very effective, orally administered anticoagulants, but they have a narrow therapeutic window and numerous food–drug and drug–drug interactions, and they require routine coagulation monitoring and dose adjustment. 6 Poor adherence, discontinuation of therapy, and suboptimal treatment of VTE can arise from these challenges, which place patients at risk 7,8 ; therefore, it is important for hospital physicians to start effective treatment and to ensure that this continues in the outpatient setting. Effective treatment of VTE reduces the risk of future complications such as VTE recurrence and secondary complications, such as postthrombotic syndrome and chronic thromboembolic pulmonary hypertension. 9
Rivaroxaban is a highly selective, oral, direct factor Xa inhibitor that was developed to address some of the limitations of the traditional anticoagulants. Rivaroxaban and other direct oral anticoagulants, such as apixaban and dabigatran, have undergone, or are being studied in, phase III clinical trials for the treatment of VTE and prevention of VTE recurrence (AMPLIFY study of apixaban; clinicaltrials.gov NCT00643201). 10 –12 However, rivaroxaban is currently the only direct oral anticoagulant approved in the European Union and the United States for the treatment of acute DVT and PE and secondary prevention of DVT and PE. Approval of rivaroxaban for this indication was based on the results of the EINSTEIN DVT, EINSTEIN PE, and EINSTEIN EXT phase III clinical studies. 13,14 Rivaroxaban is also approved for the prevention of VTE in patients after elective hip or knee replacement surgery and for the prevention of stroke and systemic embolism in patients with nonvalvular atrial fibrillation. 15,16
This review critically assesses aspects of the use of rivaroxaban for the treatment of VTE, including its use in specific clinical settings (such as perioperative management and treatment of bleeding) and in special populations (such as patients with renal or hepatic impairment). Most recommendations are given in accordance with the European and United States labels for the use of rivaroxaban for the treatment of VTE, although in some cases they are based on a review of the literature and on expert opinion.
Rivaroxaban Pharmacology
Rivaroxaban dose dependently inhibits factor Xa activity 17,18 and has a rapid onset of action similar to that of the injectable LMWHs; rivaroxaban reaches maximum plasma concentrations 2 to 4 hours after dosing and has a half-life of 5 to 9 hours in healthy, young individuals and 11 to 13 hours in healthy, elderly individuals (≥60 years of age; Table 1). 15,16,19 –21 Population-based pharmacokinetic analyses derived from phase II clinical data indicated that rivaroxaban exhibited predictable pharmacokinetic and pharmacodynamic properties in patients after major orthopedic surgery and in patients treated for DVT. 22 –24 Therefore, rivaroxaban can be administered at fixed doses, and patients do not require routine anticoagulation monitoring. The resulting simplified management of anticoagulation with rivaroxaban compared with standard therapy may encourage hospital physicians to use such regimens increasingly in clinical practice.
Characteristics of Rivaroxaban and Dosing for VTE Treatment.
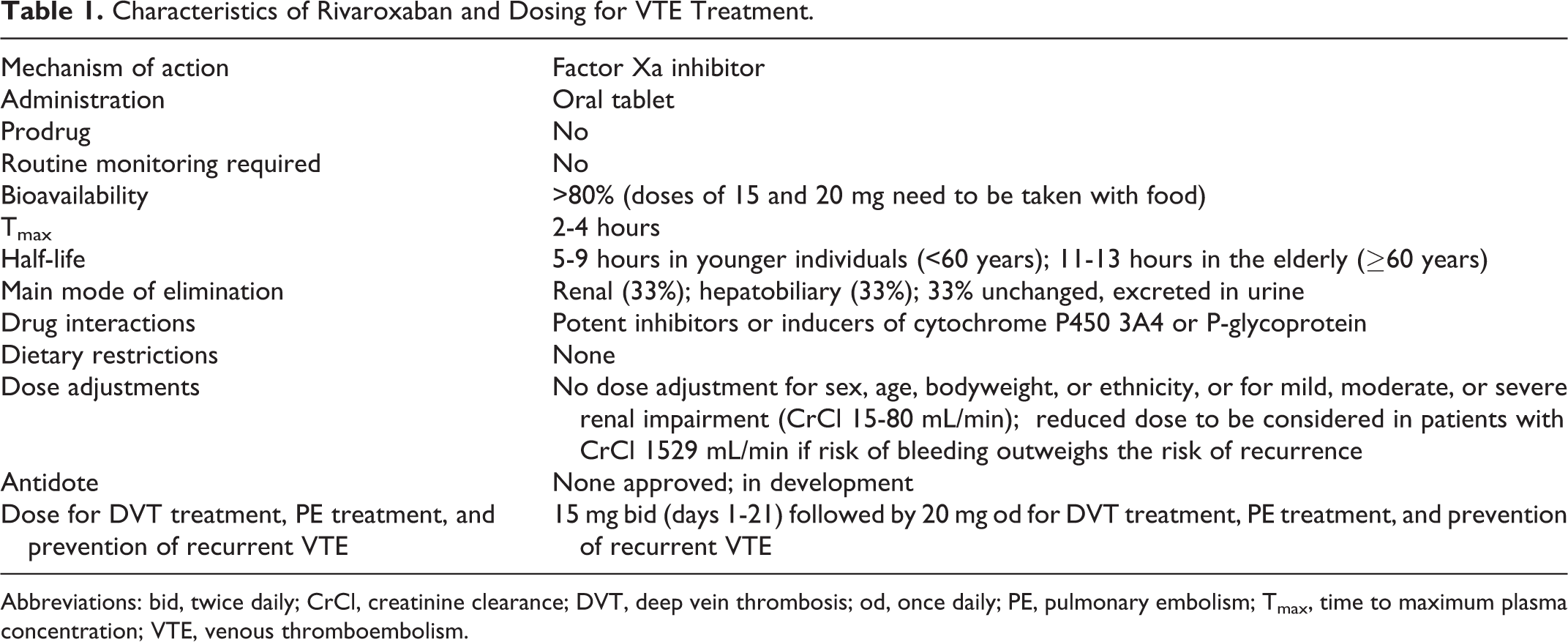
Abbreviations: bid, twice daily; CrCl, creatinine clearance; DVT, deep vein thrombosis; od, once daily; PE, pulmonary embolism; Tmax, time to maximum plasma concentration; VTE, venous thromboembolism.
Rivaroxaban at a dose of up to 10 mg has high oral bioavailability (80%-100%), irrespective of fasting or fed conditions. 15,16,19 Oral bioavailability of the 20 mg dose is reduced to 66% under fasting conditions; therefore, doses of rivaroxaban greater than 10 mg, that is, the 15 and 20 mg doses, must be taken with food to ensure high oral bioavailability (Table 1). 15,16,25 Dose-escalation studies with single and multiple doses in healthy individuals demonstrated that doses up to a total daily dose of 80 mg were well tolerated and not associated with an increased risk of bleeding compared with placebo. 19,20
Rivaroxaban is metabolized by the cytochrome P450 (CYP) isoforms CYP3A4 and CYP2J2 and also by CYP-independent mechanisms. 15,16,18 Approximately two-third of the orally administered dose undergoes degradation to inactive metabolites in the liver; half of this is then eliminated renally and the other half by the hepatobiliary route. The remaining one-third of the drug is eliminated as unchanged, active drug in the urine. 15,16,26,27 Therefore, when treating a patient with renal or hepatic impairment, the hospital physician must consider the severity of impairment before prescribing rivaroxaban (refer to subsequent section on use of rivaroxaban in special populations).
Potential Drug Interactions
In routine clinical practice, many patients in whom anticoagulation therapy is indicated take additional medications for existing comorbidities. Rivaroxaban has been assessed in a number of clinical studies for potential changes in pharmacokinetic parameters and/or changes in safety profile when administered with other commonly used medications. Overall, rivaroxaban has a low propensity for drug–drug interactions. 28 However, owing to rivaroxaban being a substrate of both CYP3A4 and P-glycoprotein, drugs that are strong inducers or inhibitors of CYP3A4 and/or P-glycoprotein will alter the pharmacokinetics and pharmacodynamics of rivaroxaban when coadministered. Therefore, systemic treatment with azole-antimycotics (such as ketoconazole, itraconazole, voriconazole, and posaconazole) or HIV protease inhibitors (such as ritonavir) is not recommended, because these drugs increase the plasma concentration of rivaroxaban (by up to 1.7-fold) and may increase the risk of bleeding (Table 2). 15,16 Strong CYP3A4 inducers (such as rifampicin, phenytoin, carbamazepine, phenobarbital, or St John wort) may reduce the plasma concentration of rivaroxaban and, therefore, should be used with caution. 15,16
Guidance for Use of Rivaroxaban With Comedications.
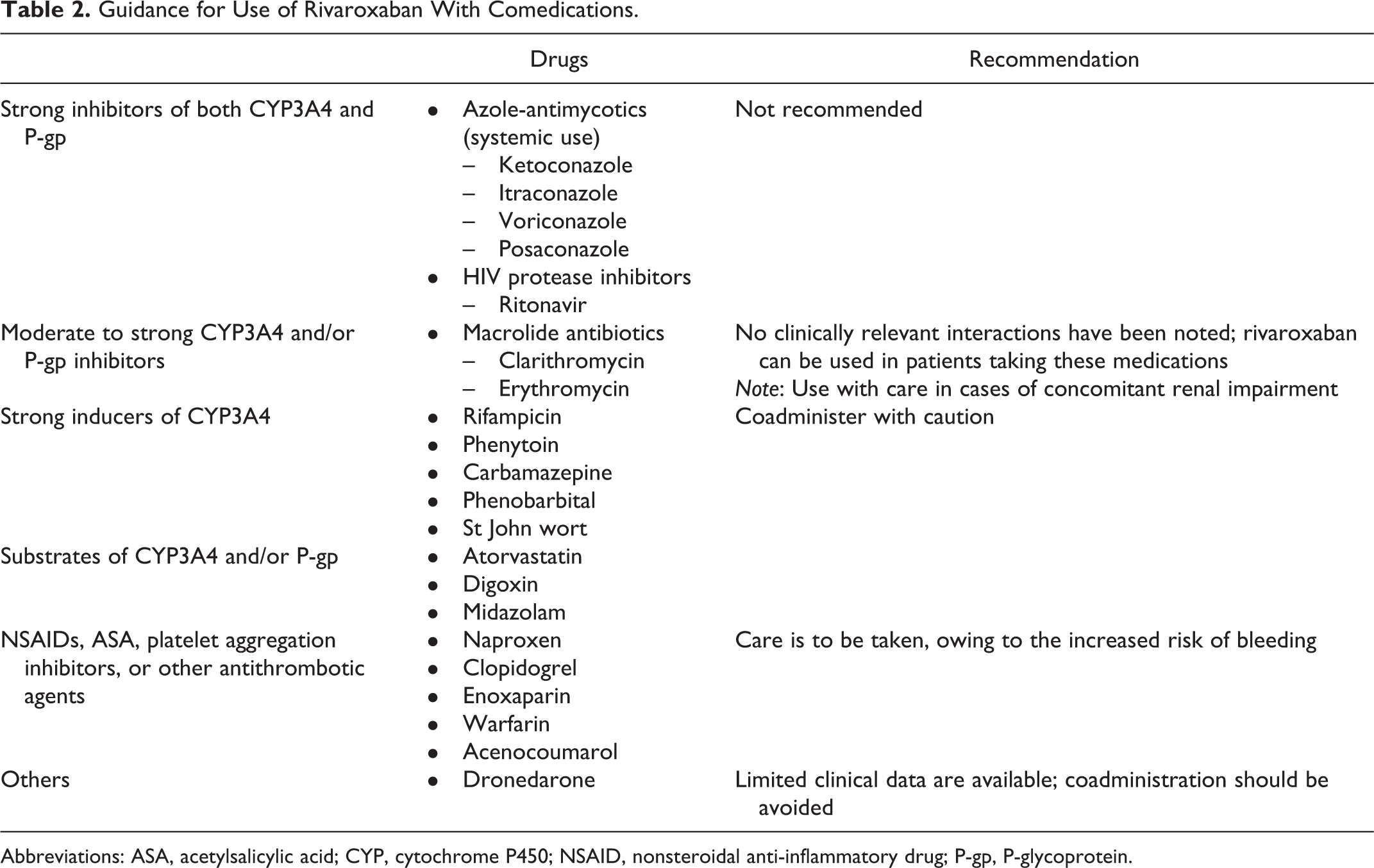
Abbreviations: ASA, acetylsalicylic acid; CYP, cytochrome P450; NSAID, nonsteroidal anti-inflammatory drug; P-gp, P-glycoprotein.
Other drugs that have the potential to increase the risk of bleeding, such as other anticoagulants, nonsteroidal anti-inflammatory drugs, acetylsalicylic acid, or platelet aggregation inhibitors, should be used with caution in patients receiving rivaroxaban. 15,16 In a pooled analysis of 4 phase III trials that evaluated rivaroxaban for the prevention of VTE after total hip or knee replacement surgery, concomitant use of rivaroxaban with nonsteroidal anti-inflammatory drugs or acetylsalicylic acid resulted in a nonsignificant increase in bleeding events; a similar increase was seen in patients in the enoxaparin arm. 29 Another phase III trial that evaluated rivaroxaban in addition to dual antiplatelet therapy for the prevention of recurrent events in patients with a recent acute coronary syndrome event demonstrated that the addition of rivaroxaban significantly increased the risk of major bleeding compared with dual antiplatelet therapy alone. 30
Some patients who have cardiac arrhythmias receive dronedarone to maintain normal heart rhythm; there are only limited data on the coadministration of rivaroxaban and dronedarone and, therefore, rivaroxaban should not be used in patients receiving dronedarone. 15
Dosing Regimen
The dosing regimen of rivaroxaban for the treatment of DVT and PE (15 mg twice daily for 21 days followed by 20 mg once daily; Table 1) was the culmination of two dose-finding studies. 31,32 Thrombus regression at 21 days was greater with twice-daily regimens of rivaroxaban compared with once daily regimens. 31 The bleeding profile was better for once daily regimens of rivaroxaban than twice-daily regimens over the 12-week treatment period. These results, together with earlier studies of ximelagatran and idraparinux, which showed that recurrence rates of VTE were high during the first few weeks of treatment, 33,34 formed the rationale for the dosing regimen chosen for the EINSTEIN phase III studies of rivaroxaban. This dosing regimen has since been approved for the treatment of acute DVT and PE, and the prevention of recurrence. 13,14
No dose adjustment is necessary in patients with mild, moderate, or severe renal impairment (creatinine clearance [CrCl] 15-80 mL/min). 15,35 A reduction in the dose from 20 to 15 mg once daily should be considered if the patient’s risk of bleeding outweighs the risk of recurrent DVT and PE. Rivaroxaban is not recommended in patients with CrCl <15 mL/min. 15
The EINSTEIN Program
The standard of care available to hospital physicians for anticoagulation after diagnosis of acute VTE has been a dual-drug approach with heparin or LMWH overlapping with and followed by a VKA. 4 The EINSTEIN DVT and EINSTEIN PE studies compared the efficacy and safety of the single-drug approach with rivaroxaban with the current dual-drug approach of LMWH/VKA. EINSTEIN DVT and EINSTEIN PE were open-label, randomized, event-driven, noninferiority trials that shared a similar design and compared the efficacy and safety of rivaroxaban with the dual-drug approach of enoxaparin and VKA for 3, 6, or 12 months; patients in the rivaroxaban group received 15 mg twice daily for the first 21 days followed by 20 mg once daily (Table 3). 13,14 EINSTEIN DVT enrolled patients with acute, symptomatic DVT (without symptomatic PE), and EINSTEIN PE enrolled patients with acute, symptomatic PE (with or without symptomatic DVT).
Summary of Outcomes of EINSTEIN DVT and EINSTEIN PE.
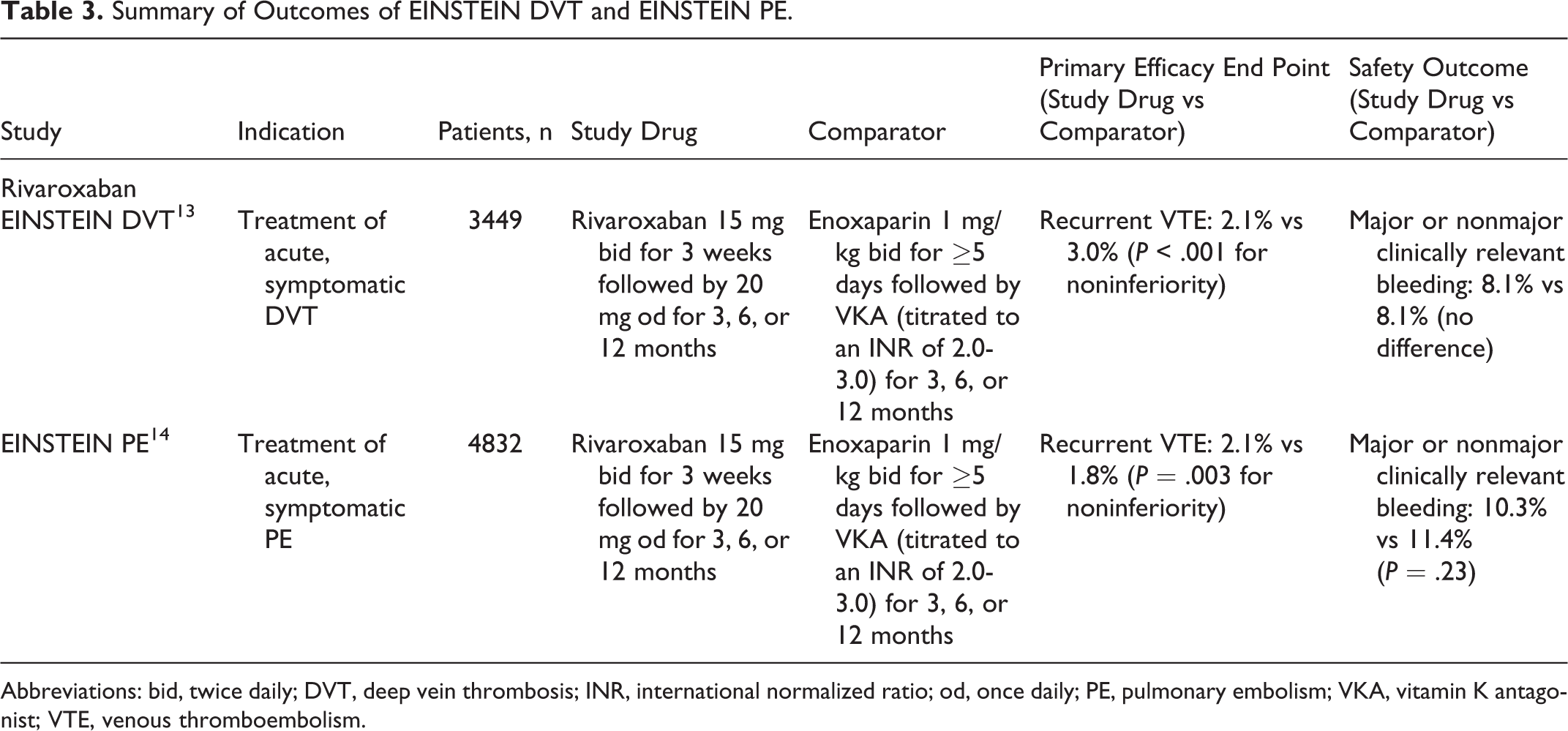
Abbreviations: bid, twice daily; DVT, deep vein thrombosis; INR, international normalized ratio; od, once daily; PE, pulmonary embolism; VKA, vitamin K antagonist; VTE, venous thromboembolism.
In both the trials, the primary efficacy end point was recurrent VTE. Results demonstrated noninferiority of rivaroxaban compared with enoxaparin/VKA for the prevention of recurrent VTE in EINSTEIN DVT (2.1% vs 3.0%; P< .001) and EINSTEIN PE (2.1% vs 1.8%; P= .003). The principal safety outcome, defined as major or nonmajor clinically relevant bleeding, occurred at a similar rate in both study arms in EINSTEIN DVT (8.1% in both arms) and in EINSTEIN PE (10.3% vs 11.4%; P= .23). In EINSTEIN PE, major bleeding events occurred at a significantly lower rate in the rivaroxaban arm compared with the standard therapy arm (1.1% vs 2.2%; P= .003). 13,14
EINSTEIN EXT was a double-blind, randomized, event-driven superiority study that compared rivaroxaban with placebo for an additional 6 or 12 months in patients who had completed 6 to 12 months of treatment for VTE (acute DVT or PE). Rivaroxaban demonstrated superior efficacy to placebo for the primary efficacy end point of recurrent DVT/PE (1.3% vs 7.1%; P< .001). The principal safety outcome was major bleeding, which occurred at a similar rate in both treatment groups (4 of 602 patients in the rivaroxaban group vs none of 594 patients in the placebo group; P= .11). These results demonstrated that rivaroxaban was very effective for the extended treatment of VTE, reducing the rate of VTE by 82% while not significantly increasing the rate of major bleeding. Compared with placebo, rivaroxaban provided a significantly improved net clinical benefit (2.0% vs 7.1%; P< .001). 13
Measuring the Anticoagulant Effect
The routine coagulation monitoring necessary with VKA therapy is not required with rivaroxaban. This can be seen as an advantage to patients who find regular clinic appointments inconvenient and can reduce the costs associated with running these clinics. However, routine monitoring may provide comfort and reassurance to some patients and enable health care professionals to monitor and encourage patient compliance with treatment.
There may be situations in daily clinical practice when it is helpful to measure the coagulation status of a patient receiving rivaroxaban. Such situations include patients presenting with a thrombotic or hemorrhagic event while receiving anticoagulation therapy; prior to emergency surgery; after a potential overdose; or in patients with deteriorating renal function. 15 Routinely used clotting assays, such as those used for conventional INR monitoring, are not suitable for measuring rivaroxaban plasma concentrations because rivaroxaban interferes with these assays. Therefore, chromogenic antifactor Xa assays and prothrombin time (PT) assays have been investigated (Table 4). 36,37 Chromogenic antifactor Xa assays were found to be suitable for measuring a wide range of rivaroxaban plasma concentrations (20-660 ng/mL; peak to trough range after therapeutic doses) when using rivaroxaban calibrators and controls. 36 These assays are now commercially available for clinical use but they require specialized clinical laboratories. In contrast, the PT assay is a well-established clotting assay that can be performed in almost all clinical laboratories. 37 However, there are marked variations in the response sensitivity of different PT reagents 38,39 and, therefore, a sensitive PT reagent, such as Néoplastine Cl Plus (Diagnostica Stago, Asnières-sur-Seine, France), should be used in conjunction with rivaroxaban calibrators to assess hemostatic status after rivaroxaban intake. 37,40,41 In summary, chromogenic antifactor Xa assays and PT tests performed with rivaroxaban-sensitive reagents and calibrators and controls for rivaroxaban can both be used to assess rivaroxaban plasma concentration if necessary, but routine monitoring is not required.
Commercially Available Assays for Measuring Anticoagulant Effect of Rivaroxaban.a

Abbreviations: INR, international normalized ratio; PT, prothrombin time.
aLaboratory monitoring of rivaroxaban anticoagulant effect is not routinely necessary.
Overdose and Management of Bleeding
The risk of major bleeding with rivaroxaban was shown to be low and similar to that of standard therapy in the phase III EINSTEIN studies. 13,14 However, if severe bleeding or overdose does occur in a patient taking rivaroxaban or any other anticoagulant, reversal methods may be required. The effect of VKAs can be specifically reversed by the administration of vitamin K (although this reversal is not immediate) and the antidote for heparins is protamine sulfate. There are currently no specific antidotes available for the non-VKA oral anticoagulants, including rivaroxaban. However, owing to the relatively short half-life of rivaroxaban, this may be less of a requirement than is the case for other anticoagulants such as warfarin (mean half-life of 40 hours). Nevertheless, in such situations in patients receiving rivaroxaban, delaying the next dose, temporarily interrupting treatment, or applying local mechanical compression will usually be sufficient to manage these events. In these situations, the use of activated charcoal may also be considered to reduce absorption of rivaroxaban if it was ingested within the past 4 hours. Depending on the severity and location of the bleeding event, other symptomatic treatment strategies may also be considered, including surgical intervention, fluid replacement and hemodynamic support, and blood product or component transfusion (Figure 1). 15,16
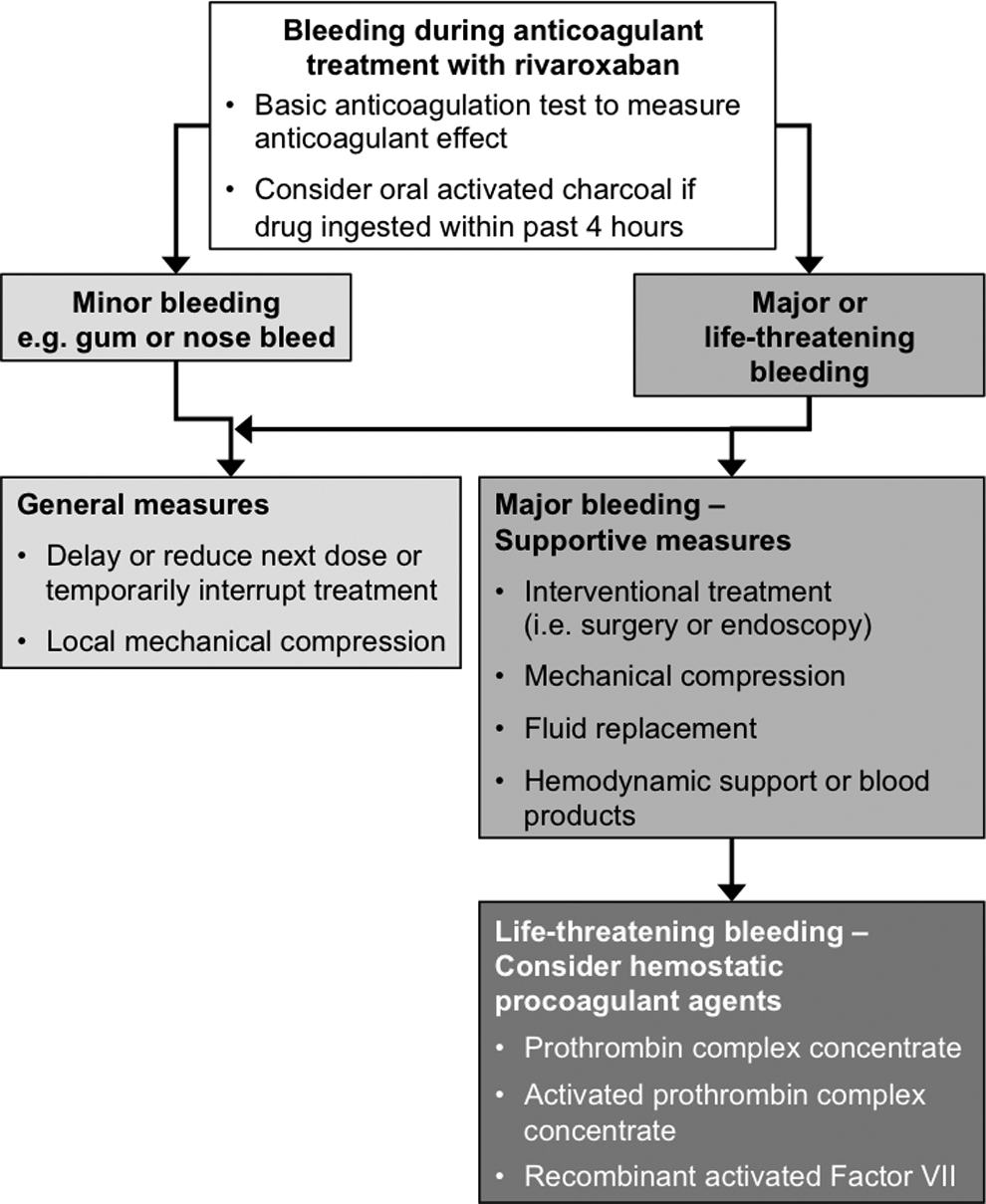
Flowchart for the management of bleeding in patients treated with rivaroxaban.
Administration of hemostatic procoagulant agents—prothrombin complex concentrate (PCC), activated PCC (factor VIII inhibitor bypassing activity [FEIBA]), and recombinant activated factor VII [rFVIIa])—should also be considered (Figure 1). 15,16,41 –43 In a randomized, double-blind, placebo-controlled, phase I study with healthy individuals, rivaroxaban-induced prolongation of the PT and inhibition of endogenous thrombin potential were immediately and completely reversed with PCC (50 IU/kg). 42 In a rabbit model of bleeding and arterial thrombosis, PCC and rFVIIa partially reversed some coagulation parameters, including endogenous thrombin potential. 44 Further studies are required to demonstrate whether PCC, FEIBA, or rFVIIa may be used as a standard antidote for rivaroxaban in clinical practice. However, it is important to note that clinical experience with these measures is limited to date.
Compliance
Noncompliance with anticoagulation therapy can result in patients being at increased risk of a thrombotic or bleeding event. In patients taking warfarin, suboptimal compliance may result in the INR not being maintained within the therapeutic range of 2.0 to 3.0. A meta-analysis published in 2006 showed that patients who received oral anticoagulation (mainly warfarin) spent 63.6% of time within the therapeutic range. 45 A similar time in therapeutic range of 62.7% (ranging from 57.8% in the first month to 72.7% in month 11) was reported in EINSTEIN PE for patients in the warfarin group, whereas adherence to rivaroxaban was more than 80% in 94.2% of the patients. 14 The simplicity of oral dosing with rivaroxaban without the need for monitoring may improve patient compliance, although the lack of monitoring may mean that compliance is more difficult to measure in clinical practice or that poor compliance is not identified, leading to an increased risk of thrombosis.
Switching Strategies
After surgery, patients often experience nausea or vomiting and may not be able or willing to take oral anticoagulation. In these situations, administration of a parenteral anticoagulant may be advantageous. However, it may still be preferable to switch to an oral anticoagulant at patient discharge to allow more convenient administration outside of the hospital setting. 46 In other cases, the patient may have problems with compatibility or adherence to the prescribed anticoagulant and the hospital physician may consider switching to another agent.
When switching from one anticoagulant to another it is important to ensure continued adequate anticoagulation while minimizing the risk of bleeding. For patients with a condition that is well controlled by VKA or heparin therapy there is no need to switch treatment options. However, if a change in anticoagulation therapy is indicated, switching patients who are being treated for DVT, PE, or for prevention of recurrent VTE from VKA or heparin to rivaroxaban or vice versa is a relatively straightforward process. 15,16 When switching from a VKA to rivaroxaban, VKA therapy should be stopped and rivaroxaban initiated when the INR reaches ≤2.5. However, consensus guidance recently highlighted the importance of considering the type of VKA owing to variations in their half-lives. 41 The VKA therapy should be stopped and the INR monitored to establish when rivaroxaban may be administered. For example, in the case of warfarin or acenocoumarol, the INR should be <3.0, whereas rivaroxaban therapy should be initiated when the INR is ≤2.5 for phenprocoumon, which has a longer half-life. However, in patients with a higher risk of bleeding, rivaroxaban should be administered when the INR is 2.0 to 2.5. 41 When switching from rivaroxaban to a VKA, the VKA and rivaroxaban should initially be given concomitantly until an INR ≥2.0 is reached, at which time rivaroxaban can be stopped. During the period when patients are receiving both the VKA and rivaroxaban, the INR should be measured 24 hours after the last dose of rivaroxaban and prior to the next dose, because rivaroxaban falsely elevates INR values. When switching from continuous intravenous heparin to rivaroxaban, rivaroxaban should be started at the time of discontinuation. When switching from subcutaneous heparin or LMWH to rivaroxaban, rivaroxaban should be started 0 to 2 hours before the next scheduled dose of heparin/LMWH. Conversely, when switching from rivaroxaban to heparin or LMWH, the first dose of the parenteral agent should be administered at the time of the next scheduled rivaroxaban dose. 15,16
Patients Undergoing Invasive Procedures (Elective and Urgent)
If patients require an invasive procedure or surgical intervention, rivaroxaban therapy should be discontinued—ideally for at least 24 hours before the intervention—and restarted when adequate hemostasis has been reestablished. This applies to patients who are scheduled to undergo surgery. 15,16,41,47 However, if emergency surgery is needed, no further rivaroxaban tablets should be taken and, if possible, surgery should be delayed until the half-life of rivaroxaban (5-9 hours for younger patients; 11-13 hours in elderly patients) has elapsed. In this scenario, the PT test with the use of a rivaroxaban-sensitive reagent (such as Néoplastine CI Plus) or a chromogenic antifactor Xa assay should be used to measure rivaroxaban activity in plasma. If the procedure cannot be delayed, the treating physician should assess the increased risk of bleeding against the urgency of the intervention. After the intervention, rivaroxaban can be restarted as soon as possible, provided that the clinical situation allows and that adequate hemostasis has been established. It is noteworthy that, owing to the fast onset of action of rivaroxaban, it is not necessary for patients to receive bridging therapy with parenteral UFH or LMWH before the procedure. In the event of bleeding, management strategies as previously described in this review and detailed in the Summary of Product Characteristics and Prescribing Information should be employed. 15,16
Use in Special Populations
Populations in Whom Rivaroxaban Should Not Be Used
Pregnancy is known to increase the risk of VTE. However, because of the potential for reproductive toxicity, the intrinsic risk of bleeding, and evidence that rivaroxaban crosses the placenta, rivaroxaban is contraindicated during pregnancy, and women of child-bearing potential should avoid becoming pregnant during treatment with rivaroxaban. 15 There is also evidence from animal studies that rivaroxaban is secreted into milk. 15,16 Therefore, rivaroxaban is contraindicated in lactating women; breast feeding should either be discontinued or rivaroxaban therapy stopped.
Unlike LMWH or VKAs, rivaroxaban is not recommended for use in patients below 18 years of age, because its safety and efficacy have not been established in the pediatric patient group.
Although approved for both the treatment of DVT and PE, rivaroxaban is not approved for treatment of VTE in other locations, such as superficial vein thrombosis or acute upper extremity DVT. Fondaparinux or LMWH are recommended in these indications 4 ; however, clinical trials are underway to evaluate the efficacy and safety of rivaroxaban in superficial vein thrombosis (clinicaltrials.gov NCT01499953) and in patients with cancer with upper extremity blood clots (clinicaltrials.gov NCT01708850).
The European Society of Cardiology (ESC) recommends thrombolytic therapy or embolectomy for the treatment of patients with life-threatening, high-risk PE (those who present with shock and/or hypotension) 48 ; however, the safety and efficacy of rivaroxaban have not been established in these patients. Rivaroxaban should, therefore, not be used in hemodynamically unstable patients with PE or those requiring thrombolysis or embolectomy. 15
Weight and Age
Treating patients with rivaroxaban at extremes of bodyweight (<50 or >120 kg) is not a problem. Extreme bodyweight has only a small influence on rivaroxaban plasma concentrations, and no dose adjustment is necessary in these patients. 15,16 This finding was supported by subgroup analyses of the EINSTEIN studies, which showed that efficacy and safety outcomes were consistent irrespective of weight category. 13,14
Elderly patients present to hospital physician with additional problems with respect to VTE treatment. Age is a major risk factor for VTE, and it is important to note that elderly patients are more likely to have renal impairment compared with their younger counterparts. Impaired renal function can lead to slower clearance of drugs and their metabolites, resulting in increased risk of bleeding. Results in healthy elderly individuals showed higher plasma rivaroxaban concentrations than in younger individuals, which was mainly because of reduced (apparent) total and renal clearance 21 ; however, no dose adjustment is necessary in these patients. A pooled subgroup analysis with respect to relative efficacy and safety in prespecified age categories (<65, 65-75, and >75 years) of the EINSTEIN data supports this finding. 13,14,49
Renal Impairment
One-third of the administered dose of rivaroxaban is eliminated via the kidneys as unchanged, active drug; therefore, renal impairment decreases clearance of rivaroxaban and, in turn, increases rivaroxaban plasma exposure. 15,16
For the treatment of DVT and PE and prevention of recurrent VTE (rivaroxaban 15 mg twice daily for 21 days and 20 mg once daily thereafter), no dose adjustments are necessary in patients with mild (CrCl 50-80 mL/min), moderate (CrCl 30-49 mL/min), or severe (CrCl 15-29 mL/min) renal impairment, although caution is needed in patients with severe renal impairment. 15,41 It should be noted that for patients with CrCl <30 mL/min, these recommendations are based on limited clinical data and pharmacokinetic modeling because such patients were excluded from the EINSTEIN program. 13,14,35 Use of rivaroxaban is not recommended in patients with CrCl <15 mL/min. 15,16
Hepatic Impairment
Hospital physicians may have concerns about prescribing anticoagulants to patients with hepatic impairment because these patients frequently have an increased risk of bleeding. 50 Because rivaroxaban is metabolized in the liver, the pharmacokinetics of rivaroxaban will change with increasing hepatic impairment. Rivaroxaban is recommended in patients with mild hepatic impairment without coagulopathy (Child-Pugh A) but is contraindicated in patients with hepatic disease when associated with coagulopathy (including cirrhotic patients classed as Child-Pugh B and C) and a clinically relevant bleeding risk. It may be used with caution in cirrhotic patients with moderate hepatic impairment (Child-Pugh B), if it is not associated with coagulopathy. 15
Patients With Cancer
Patients with cancer have a 4- to 7-fold overall increased risk of VTE compared with patients without malignancies; approximately 15% of patients with cancer will experience a venous thromboembolic event. 51 There are only limited efficacy and safety data available for rivaroxaban in the treatment of VTE in this patient group. The proportion of patients who had active cancer at the time of enrollment in the EINSTEIN program was similar in the rivaroxaban and standard therapy groups (EINSTEIN DVT, 6.8% vs 5.2%; EINSTEIN PE, 4.7% vs 4.5%). The relative efficacy and safety of rivaroxaban in these patients were similar to those of LMWH overlapping with and followed by VKA therapy. 13,14 However, more data are needed in this subgroup, particularly in comparison with extended treatment with LMWH alone; the preferred treatment recommendation of the American College of Chest Physicians (ACCP) for patients with cancer and VTE. 4 Rivaroxaban is contraindicated in patients with malignant neoplasms who have a high risk of bleeding. 15
Real-World Experience With Rivaroxaban
Clinical experience with rivaroxaban has accumulated over the 5 years since it was approved for the prevention of VTE after elective hip or knee replacement surgery, and there are now data from real-world studies and registries that support the efficacy and tolerability of rivaroxaban demonstrated in phase III clinical trials. 52,53 Results from a prospective registry of patients receiving direct oral anticoagulants for extended VTE treatment or stroke prevention in daily care also demonstrated low rates of adverse events and low rates of treatment discontinuation in the first 180 days of rivaroxaban therapy. 54,55 However, as with all new drug treatments, after approval and use in large numbers of patients, rare side effects may come to light. Hair loss is one such side effect which has been evaluated in a large prospective registry in both rivaroxaban- and dabigatran-treated patients. 56 In patients receiving long-term anticoagulation with rivaroxaban or dabigatran, the incidence of hair loss as a spontaneously reported side effect was 4.4 per 100 patient years. 56 However, this side effect is also a common complaint in patients receiving other anticoagulants such as VKAs, and it is also associated with LMWH. Therefore, hair loss is a side effect reported with anticoagulant use and not rivaroxaban use specifically.
Conclusions
The EINSTEIN program demonstrated the noninferior efficacy and similar safety of oral rivaroxaban as a single-drug approach compared with dual-drug standard therapy. These results, together with the predictable pharmacokinetics and pharmacodynamics of rivaroxaban, the wide therapeutic window, short half-life, and no requirement for routine coagulation monitoring, constitute a simplified treatment strategy compared with traditional anticoagulants. Based on the EINSTEIN DVT, EINSTEIN PE, and EINSTEIN EXT data, rivaroxaban was approved in the European Union and the United States for the treatment of acute DVT and PE and for the prevention of recurrent VTE; the approval represents a landmark shift in anticoagulation care in this clinical setting. However, it is important to underline that clinicians in their daily practice should develop a good familiarity with rivaroxaban, especially with respect to its use in specific clinical settings and special populations.
Footnotes
Acknowledgments
The authors would like to acknowledge Claudia Wiedemann who provided medical writing services with funding from Bayer HealthCare Pharmaceuticals and Janssen Scientific Affairs, LLC. The sponsor had no involvement in the writing of the manuscript or in the decision to submit the manuscript.
Declaration of Conflicting Interests
Davide Imberti has received honoraria or consulting fees from Daiichi Sankyo, Bayer, Boehringer Ingelheim, Pfizer, Kedrion, and Sanofi-aventis; he has no direct or indirect conflicts of interest regarding this manuscript. Raffaella Benedetti has no conflicts of interest.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
