Abstract
Background: Prophylaxis for venous thromboembolism after total knee replacement (TKR) is standard of care. Two enoxaparin regimens are approved for thromboprophylaxis, one in the United States (30 mg twice daily [bid]) and another in the rest of the world (40 mg once daily [qd]). Data on frequency of utilization of these 2 regimens at discharge from US hospitals after TKR are not available. Methods: We conducted a retrospective/descriptive analysis of the PharMetrics claims database to estimate the frequency of utilization of enoxaparin 40 mg qd compared to 30 mg bid after discharge for TKR from US hospitals. Results: Of the 44 552 TKR patients identified, 7198 had an outpatient claim for enoxaparin within 14 days postoperatively. The 40 mg strength of enoxaparin was prescribed more commonly (~51%) than the 30 mg strength (~46%). Conclusions: Enoxaparin 40 mg qd is prescribed in approximately the same proportion of patients as the current Food and Drug Administration–approved regimen of 30 mg bid.
Introduction
Venous thromboembolism (VTE) is a devastating disease, with an estimated 275 000 new cases per year in the United States. 1 Major orthopedic surgery leads to a high risk of postoperative VTE, particularly after total knee replacement (TKR). Venous thromboembolism prophylaxis after TKR is standard of care, although guideline recommendations vary.2–5 The American College of Chest Physicians consensus guidelines recommends routine pharmacologic prophylaxis with low-molecular-weight heparin (LMWH), fondaparinux, or adjusted-dose warfarin. 3 Recommendations for pharmacologic prophylaxis from the American Academy of Orthopaedic Surgeons vary depending on the patient’s risk of pulmonary embolism and major bleeding but include aspirin, LMWH, fondaparinux, and warfarin. 5
In much of the world, enoxaparin, a LMWH, is the most commonly used agent. In the United States, prophylaxis patterns are more widely variable. One analysis of a US retrospective claims database found that 91% of patients received some form of pharmacologic prophylaxis after major orthopedic surgery. In the hospital, patients received enoxaparin only (35%), warfarin only (34%), or other anticoagulants (31%). The rate of prophylaxis at hospital discharge dropped to 54%, with the majority of patients receiving either warfarin (57%) or enoxaparin (34%) alone. 6
Two enoxaparin regimens are commonly used for thromboprophylaxis after TKR. The first, approved by the US Food and Drug Administration (FDA), is 30 mg twice daily (bid) begun postoperatively. 7 The second, commonly used in Europe and the rest of the world, is 40 mg once daily (qd) begun 12 hours preoperatively. 8 As new oral anticoagulant medications are being developed and local regulatory standards must be met for approval, it is common to conduct studies of thromboprophylaxis in TKR versus both regimens.9–14
Although only one dosing regimen for enoxaparin is FDA approved for TKR in the United States, it is likely that clinicians would use both regimens. No data are currently available on utilization of the 40 mg enoxaparin regimen compared to the 30 mg regimen in the United States. Therefore, the objective of our study was to estimate the frequency of utilization of enoxaparin 40 mg qd compared to 30 mg bid after discharge for TKR from US hospitals.
Methods
Data Source
This study was conducted using the PharMetrics integrated health care paid claims database. This database represents approximately 18 million patients from 20 health plans across the United States. Plans in this database provide both medical and pharmacy benefits for its members usually on a fee for service basis; plans with capitated benefits still provide individual claims. Each claim in the database contains a unique encrypted patient identifier that is used to assemble a longitudinal record of medical and pharmacy services for each member. Eligibility, year of birth, and gender are available for all the members in the database (members missing this information were excluded). Participants in PharMetrics are representative of the national commercially insured population on a variety of demographic measures including age, sex, geographic region, and health plan type.
Data available for each medical claim include dates of service, location of service, International Classification of Diseases, Ninth Revision, Clinical Modification (ICD-9-CM) diagnosis codes, physician specialty, and procedure codes in the Physicians' Current Procedural Terminology and the Centers for Medicare and Medicaid (CMS) provided Common Procedure Coding System. Pharmacy claims include the drug dispensed in National Drug Code (NDC) format, the date of dispensing, and the quantity and number of therapy days dispensed.
Patient Population and Index Event
The study population consisted of all adult patients (age 18+) with at least one procedure code of TKR surgeries identified by a claim with a current procedure terminology procedure code, 27 447 (total knee arthroplasty), 27 487 (revision of total knee arthroplasty), and 27 488 (removal of total knee prosthesis) during a hospital stay occurring between January 2007 and June 2009. The date of the procedure was considered the index date.
Drug Exposure
Treatment with enoxaparin, warfain, clopidogrel, and LMWH was identified by NDC in the pharmacy claims data within the 15-day period postadmission for TKR surgery. Enoxaparin strength was obtained from the NDC information. We used the first enoxaparin claim in this period for analysis. Patients with no pharmacy claims during this period were excluded since we could not determine whether these patients remained in the hospital or were sent to other care facilities.
Descriptive Analyses
Patient variables such as age (at index date) and geographic region were tabulated and compared to US 2000 Census data to assess the representativeness of the sample population. We used descriptive analyses to compare utilization patterns of enoxaparin 40 mg qd and 30 mg bid. To evaluate whether patients on multiple medications used different dosing of enoxaparin, we sequentially excluded patients who also filled a prescription for warfarin and clopidogrel. To assess differences in enoxaparin dosing by categories of age and geographic region, we performed chi-square tests. All analyses were performed using SAS 9.1.
Results
Between January 1, 2007 and June 30, 2009, we identified 44 552 adults with TKR. Of these patients, 29 096 (65.3%) had an outpatient claim for antithrombotic therapy within 14 days of surgery (Figure 1 ). Forty-three percent of patients were greater than or equal to 65 years of age. Distribution of patients was geographically representative of the US population (Table 1 ).
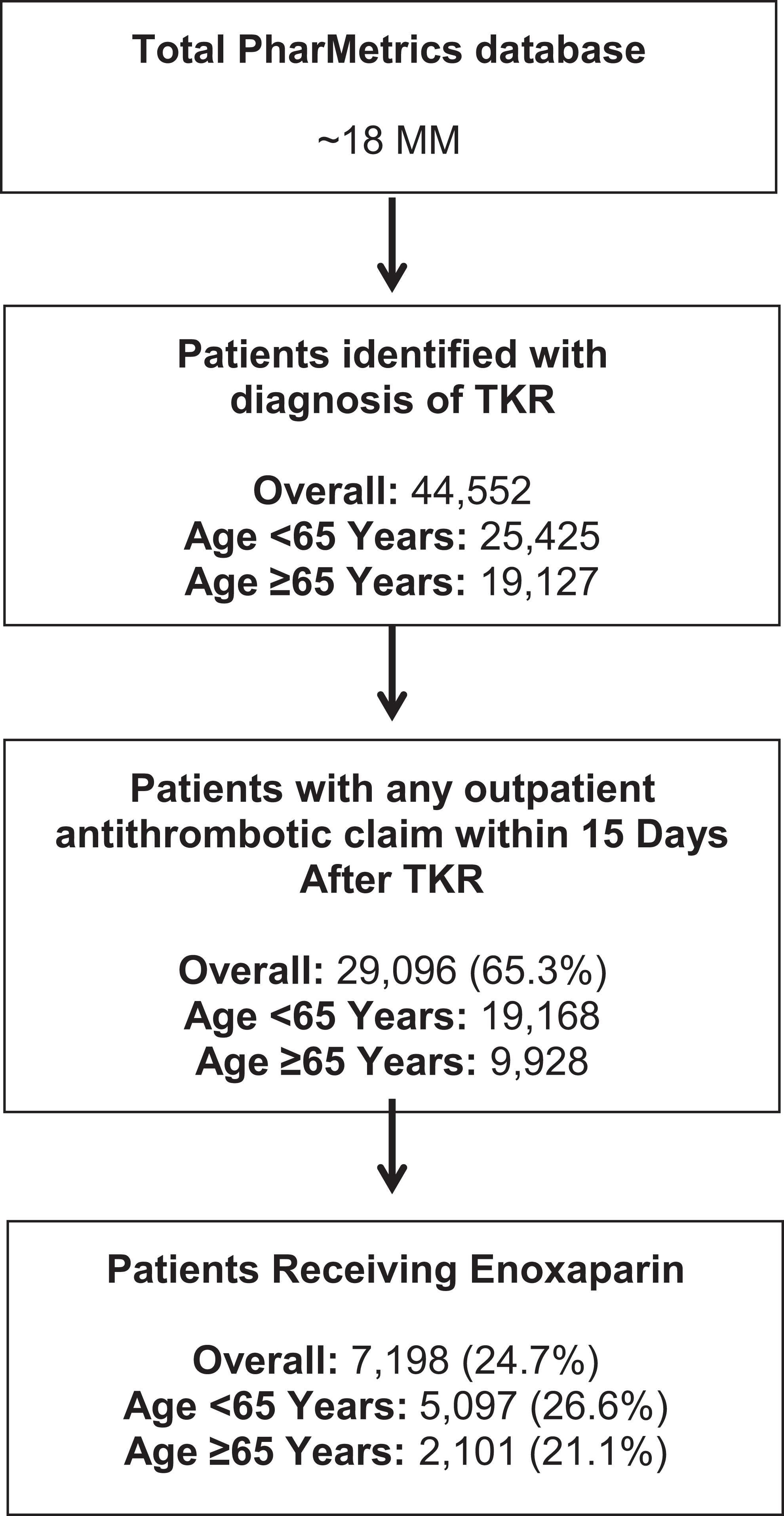
Patient flow.
Patient Demographics
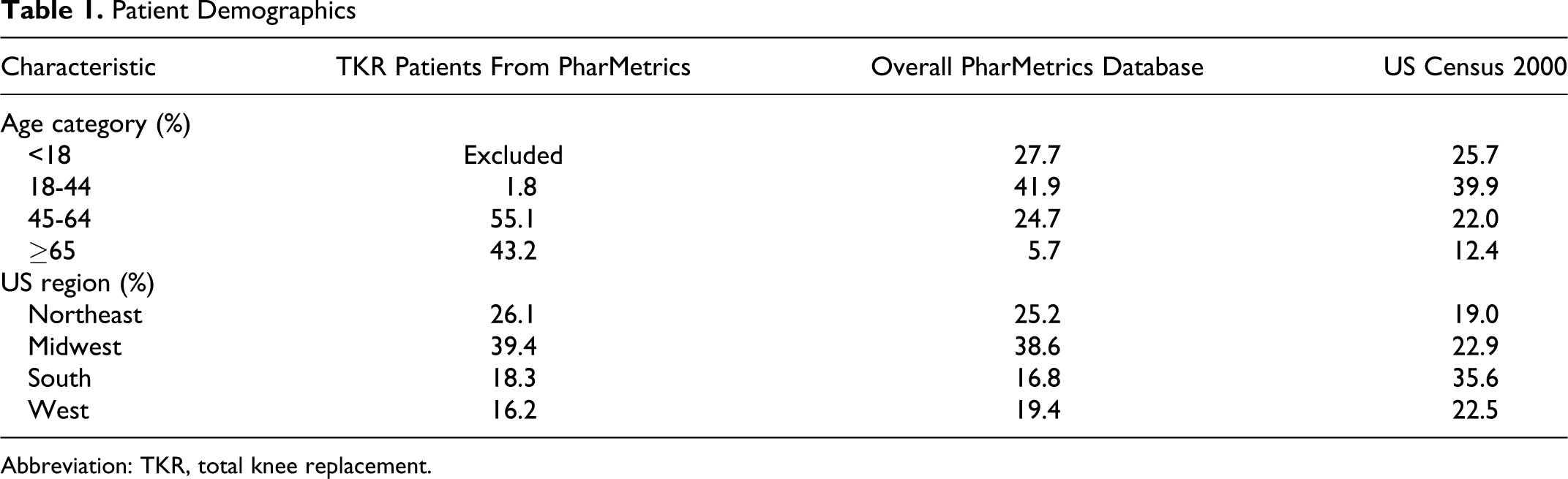
Abbreviation: TKR, total knee replacement.
Of the patients with TKR that received an outpatient prescription claim for an antithrombotic therapy, enoxaparin was identified in 7198 (24.7%) patients. Claims for enoxaparin were less common in patients 65 years or older (21.1%) compared to younger patients (26.6%). The 40 mg strength of enoxaparin was prescribed more commonly (~51%) than the 30 mg strength (~46%). Choice of enoxaparin strength did not differ by age (chi-square = 2.41, P = .2997; Figure 2 ).
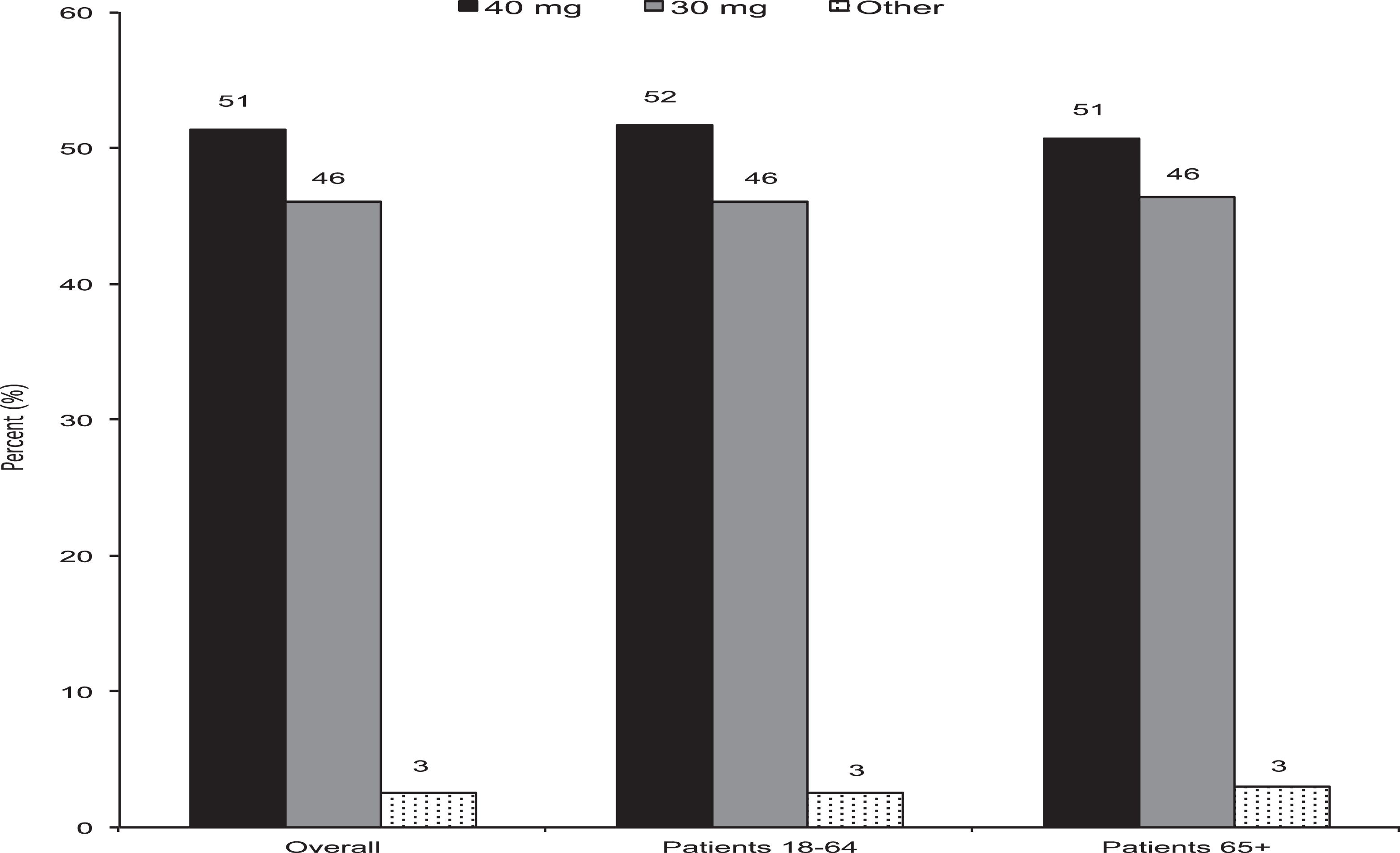
Enoxaparin dose by age category. Chi-square test for differences in enoxaparin dosing by age: category chi-square = 2.41, P = .2997.
Enoxaparin prescribing patterns varied according to geographic region (chi-square = 78.89, P < .0001; Figure 3 ). The 40 mg dose of enoxaparin was more common in all regions except the Midwest. This preference for utilization of the 40 mg dose of enoxaparin appeared most pronounced in patients from the south.
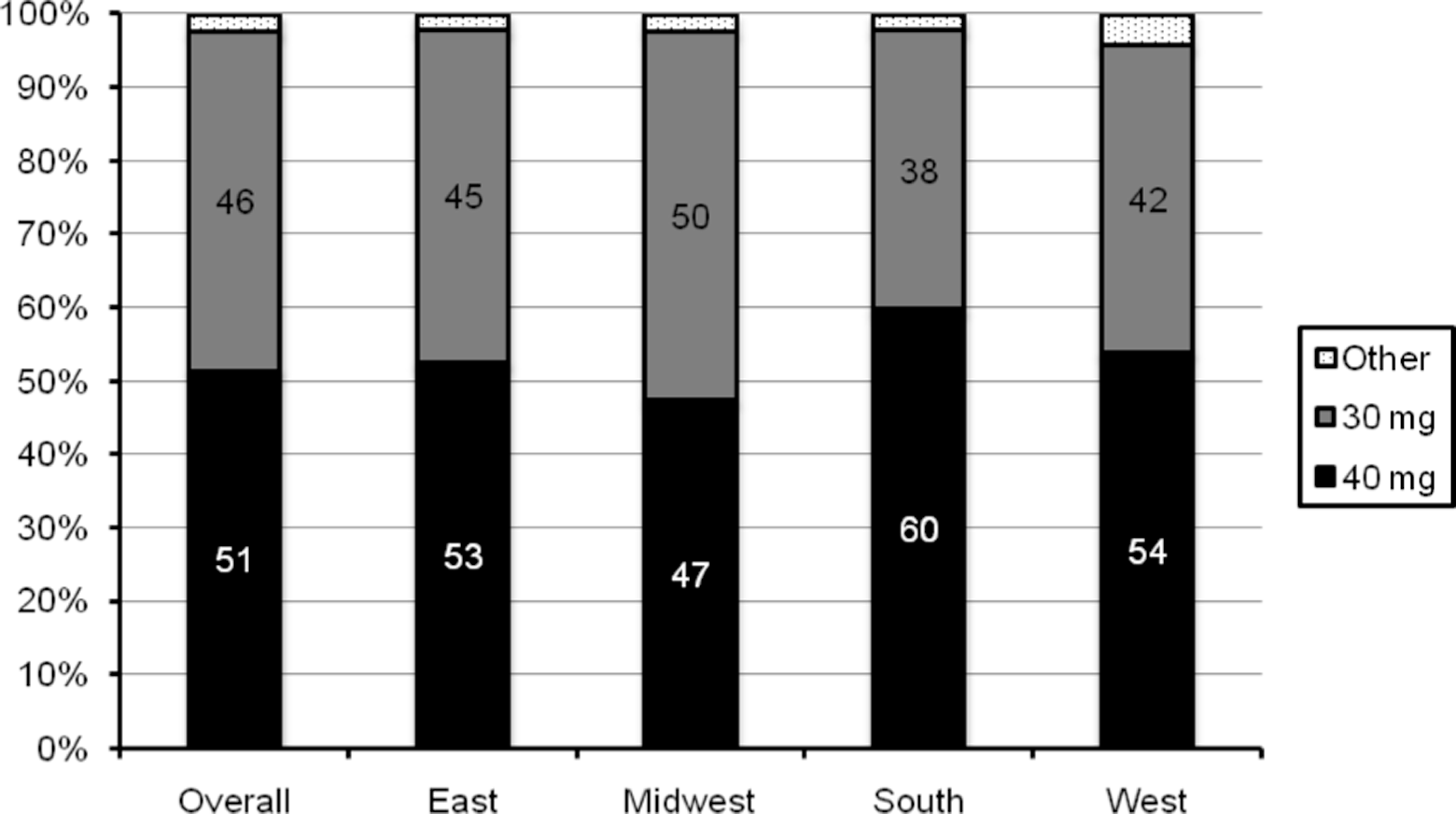
Enoxaparin dosing by geographic region. Chi-square test for differences in enoxaparin dosing by region: chi-square = 78.89, P < .0001.
A small number of patients had claims for concomitant warfarin (n = 399) or clopidogrel (n = 35) along with the claim for enoxaparin. When patients with claims for warfarin or clopidogrel were removed from the analysis, similar proportions of patients received the enoxaparin 40 mg regimen (Figure 4 ).
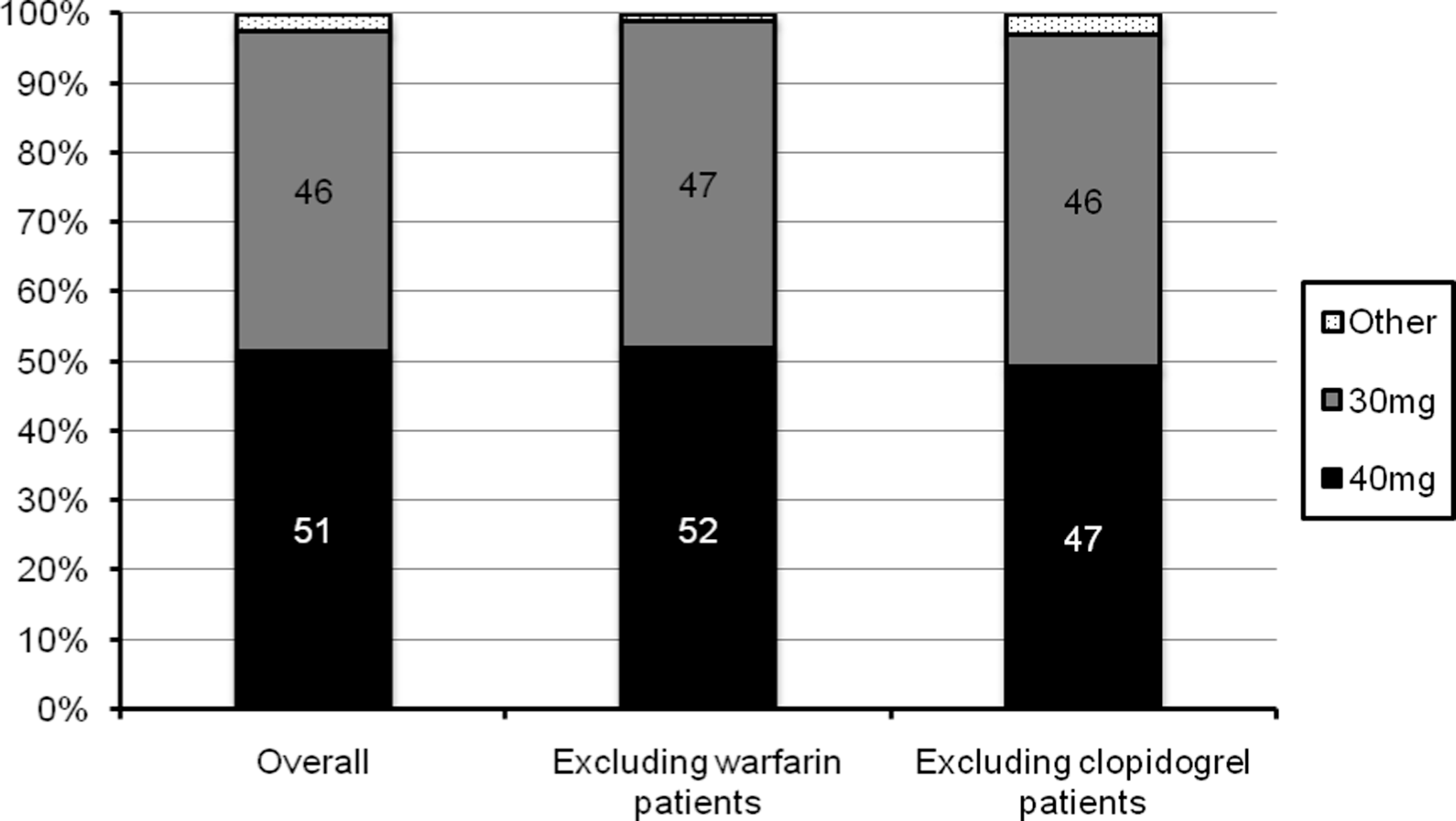
Enoxaparin dosing post-TKR in patients receiving concomitant warfarin or clopidgrel. TKR indicates total knee replacement.
Claims for concomitant statins during the postadmission period were low (7.8% overall) in our cohort of knee replacement patients. The highest proportion of statin claims occurred in the midwest (4%) and the lowest percentage in the west (0.8%). Data on concomitant use of aspirin in our cohort were unavailable.
Discussion
In a large US prescription database, we found that 40 mg of enoxaparin was the most commonly used dose for thromboprophylaxis after discharge for TKR. This pattern was consistent across categories of age and in most geographic regions. Although the 40 mg dose of enoxaparin is not FDA approved for thromboprophylaxis after TKR in the United States, there are a variety of reasons why it might be commonly used. First, the 40 mg dose is widely recognized in international clinical practice guidelines and is standard practice throughout the rest of the world. 3 Additionally, subcutaneous administration of enoxaparin is inconvenient for patients after discharge, and the once daily administration of 40 mg may be preferred by clinicians over twice daily administration of 30 mg in order to improve patient compliance.
Claims for enoxaparin in our cohort were somewhat less frequent in elderly patients compared to younger patients (11% vs 20%). The claims database we used does not capture patients who are discharged to long-term care facilities. Approximately 20% to 37% of unilateral or bilateral knee replacement patients in the United States are discharged to such facilities. 15 Since it is likely that elderly patients would more often require long-term care than younger patients, it is possible that enoxaparin use in the elderly patients is simply underrepresented in our cohort. Importantly, age did not influence the choice of enoxaparin dose in our study.
Lack of an effect of age on enoxaparin dosage patterns is not surprising given that thromboprophylaxis regimens are often included in standard order sets. With standard order sets, dosing choices are made at the institutional level and are less subjected to interpatient variability. This is further reinforced by the consistency of enoxaparin dosing patterns when patients with concomitant warfarin or clopidogrel were excluded.
A limitation of our study is that the claims data included only represent filled prescriptions that were paid by health plans and included in the PharMetrics database. The database does not capture unfilled prescriptions and is unable to distinguish whether or not the patient took the medication according to their physician’s instructions. The postsurgical time period for assessing medication claims was relatively short, so statin use may be underrepresented. Additionally, since aspirin is available in the United States without a prescription, data on its use are not available in the PharMetrics database. Although these limitations are common to administrative claims studies, these large databases represent an important source of information on drug utilization patterns due to their geographic span and utility for examining large numbers of patients inexpensively.
Overall, 65% of patients in our study had some outpatient claim within 15 days of TKR. It is possible that some patients received prescriptions for thromboprophylaxis that were not captured in the claims database (eg, Medicare). For example, aspirin is recommended in some US guidelines as an option for thromboprophylaxis after TKR. 5 However, its use would not be captured in the claims database we used. Due to our inability to capture all potential outpatient thromboprophylaxis regimens in this cohort, we only sought to examine patterns of enoxaparin prescribing after TKR and not to examine overall rates of thromboprophylaxis after TKR.
Conclusion
In conclusion, we found that the 40 mg qd and 30 mg bid regimens of enoxaparin were used in approximately equal proportions of US patients discharged after TKR. Choice of dosage regimen did not appear to be influenced by patient age or by concomitant use of warfarin or clopidogrel. A variety of factors such as cost, patient convenience, and supportive recommendations by international consensus guidelines may influence the frequency of use of a non-FDA-approved prophylaxis regimen by US clinicians.
Footnotes
Authors’ Note
All the authors are (or were at the time the research was conducted) employees of Bristol-Myers Squibb.
Declaration of Conflicting Interests
The authors declared no conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: sponsored by Bristol-Myers Squibb and Pfizer, Inc.
