Abstract
Background:
The impact of fat distribution, muscle mass, and muscle strength on no-reflow and severity of coronary artery disease in patients with ST-segment elevation myocardial infarction (STEMI) remains unclear.
Objective:
To investigate association between muscle strength and fat and muscle mass and severity of coronary atherosclerosis.
Methods:
We included 218 patients with STEMI who had undergone primary percutaneous coronary intervention. We evaluated the no-reflow phenomenon in infarct-related artery and calculated Gensini scores from initial angiograms as indicative of coronary atherosclerosis severity. The patients were divided into 2 groups as patients with no-reflow and with thrombolysis in myocardial infarction grade 3 flow and patients with low (<55) Gensini and with high (≥55) Gensini. Patients’ total fat, muscle mass, visceral fat mass, and muscle strength were measured via body composition analyzer and handgrip dynamometer.
Results:
High Gensini patients had a greater body mass index (BMI) and lower handgrip strength and more visceral fat (P = .05, P = .017, and P < .001, respectively). The patients with no-reflow had significantly lower handgrip strength and more visceral fat (both, P < .001). In multivariate regression analysis, visceral fat and handgrip strength were associated with high no-reflow rate and high Gensini scores in patients with STEMI (P = .001, P = .014, P = .022, and P = .010; respectively).
Conclusion:
Increased visceral fat and lower handgrip strength may be related to increased no-reflow rate and coronary plaque burden in STEMI. Visceral fat and muscle strength may be better prognostic markers than weight, BMI, total fat, and muscle mass in coronary artery disease.
Introduction
Obesity has been considered a risk factor for type 2 diabetes, hypertension, lipid disorders, coronary artery disease (CAD), and cancer and is associated with increased mortality rates. 1,2 However, in the past decade, there has been increasing evidence that obese patients have lower all-cause and cardiac mortality than patients with normal weight, named as “obesity paradox.” 3 In addition, overweight and obese patients have better survival rates for peripheral artery disease, heart failure, venous embolism, and stroke. 4 Similarly, the obesity paradox is manifest in the outcomes of the patients with ST-segment-elevation myocardial infarction (STEMI). 5,6 While body mass index (BMI) is the most widely used tool for the assessment of obesity, on its own, the BMI is an inadequate tool because patients are not homogenous in their ratio of fat to muscle mass or distribution of fat tissue. Therefore, measures of fat, muscle mass, and fat distribution may be more accurate prognostic markers. 7,8
Muscle strength is a significant predictor of all-cause mortality and hospitalization, independent of muscle mass, inflammatory markers, and comorbidities in the older patients. 9 In addition, muscle strength is a prognostic marker inversely associated with increased mortality for patients with heart failure, but its prognostic value for patients with STEMI has not been clarified. 10
The no-reflow phenomenon and increased atherosclerotic burden have been considered 2 major prognostic markers associated with worse short- and long-term outcomes in STEMI. 11,12 In our study, we investigate the relationship between fat and muscle mass, visceral fat and muscle strength, and no-reflow rate and coronary atherosclerotic burden reflecting Gensini score in patients with STEMI who had undergone primary percutaneous coronary intervention (pPCI).
Methods
In this prospective study, we included 218 consecutive patients with STEMI admitted to our hospital between January and December 2014. All patients with STEMI underwent pPCI. Shortly after pPCI, body compositions and hand grip strengths of all patients were calculated during hospitalization.
For the purposes of this study, patients had to satisfy the following criteria before being diagnosed with STEMI: (1) acute myocardial infarction (AMI) symptoms lasting at least 30 minutes and (2) concurrent ST-segment elevation of at least 1 mm (0.1 mV) in 2 or more contiguous leads. All patients received acetylsalicylic acid plus clopidogrel, prasugrel, or ticagrelor (loading doses 300 mg, 600 mg, 60 mg, and 180 mg, respectively) prior to coronary intervention. Unfractionated heparin 100 U/kg was initially administered to extend the activated clotting time 200 seconds. Femoral access was preferred for pPCI; however, for bilateral femoral artery occlusion, radial artery was used per physician preference.
Exclusion criteria were the presence of a previous pacemaker or implantable cardiac defibrillator, previous myocardial infarction, urgent bypass operation, and any infectious, autoimmune disease and fever. In addition, the patients with radial access were excluded because it might affect handgrip strength.
Two experienced interventional cardiologists, blinded to the study protocol and each other’s interpretation, conducted the angiographic analysis. The interventional cardiologists calculated the post-pPCI Thrombolysis In Myocardial Infarction (TIMI) flow grade in infarct-related artery (IRA) and severity of coronary atherosclerosis, using Gensini scoring system.
The no-reflow phenomenon was defined as final TIMI flow grade <3. TIMI flow grade 0 was defined as the absence of any antegrade flow, TIMI flow grade 1 was defined as weak antegrade coronary flow with incomplete filling of the distal coronary bed, TIMI flow grade 2 was defined as delayed antegrade flow with complete filling of the distal coronary bed, and TIMI flow grade 3 was defined as normal flow filling the distal coronary bed completely. 13
The CAD severity was assessed using the Gensini score. 14 This method ranks the stenosis of the lumen of coronary arteries thus 1 (1%-25%), 2 (26%-50%), 4 (51%-75%), 8 (76%-90%), 16 (91%-99%), and 32 (total occlusion). The score is then multiplied by a factor that represents the importance of the lesion’s location in the coronary artery system. The multiplication factor for the left main lesion is 5, 2.5 for the proximal left anterior descending artery (LAD) and proximal circumflex artery (Cx) lesions, 1.5 for a mid-LAD lesion, and 1 for distal LAD, mid/distal Cx, and right coronary artery lesions. For any other branch, the multiplication factor is 0.5. 14
In our study, Gensini scores were calculated from initial angiograms. For scoring of IRA, IRA had a total occlusion with thrombus, it was accepted as a total occlusion (32 points) or if it had a stenosis of 91% to 99%, 16 points was given as Gensini scoring system. In the similar studies using scoring systems reflecting atherosclerotic burden, the scores were calculated mostly from initial angiograms in STEMI. 15,16
We divided the patients into 2 groups according to CAD severity as determined by Gensini scores, with a low Gensini (<55) and high Gensini (≥55) group. There is no cutoff value predicting cardiac outcomes for Gensini scoring system in the literature. It is a linear scoring system correlated with CAD severity and poor cardiac outcomes. 15 We choose the score of 55 because it could divide into statistically comparable 2 groups.
Intra- and interobserver variables were calculated from samples of 50 randomly selected patients. Intra- and interobserver variables for no-reflow rate were 1% and 3%, respectively, and for high Gensini score were 0% and 1%, respectively.
Patients were evaluated via echocardiogram (Philips IE33 Matrix, Philips Healthcare, the Netherlands) within 1 week post-AMI. Left ventricular ejection fraction (EF) was regarded as an indicator of systolic function and calculated by modified Simpson method.
Muscle strength was calculated by the same cardiologist via handgrip dynamometer (model SH5001, Saehan Corporation, Mansan, South Korea). The patients squeezed the handgrip 3 times as hard as possible with dominant hand. The mean value of these 3 measurements was accepted as the handgrip strength.
Patients’ total skeletal mass weight, total fat mass weight and percentage, visceral fat weight, BMI, and baseline metabolic rate (BMR) were calculated by body composition analyzer (Tanita, body composition analyzer type BC-418MA, Tokyo, Japan). Calculations were performed by the same physician 3 times in separate days postdischarge from the coronary care unit and while hemodynamically stable during hospitalization. The patients were in fasting and dehydrated in the morning during calculation. Ethical approval for this study was obtained from the institutional ethics committee.
Statistical Analysis
All analyses were performed using SPSS 20.0 (released 2011, IBM statistics for Windows version 20, IBM Corp, Armonk, New York). All data are presented as mean ± standard deviation, unless otherwise stated. Comparisons of parametric values between the 2 groups was performed using an independent samples t test. The comparisons of nonparametric values between the 2 groups were performed using the Mann-Whitney U test. Categorical variables were compared using a chi-square test. Logistic regression analysis was used to assess the no-reflow and the severity of CAD. Those variables with P < 0.1, by univariate analysis, were included in the stepwise multivariate logistic regression analysis model, and the respective odds ratios (ORs) were calculated with 95% confidence intervals (CIs). A receiver–operating characteristic curve (ROC) was constructed to determine visceral fat, handgrip cutoffs, for the no-reflow and the severity of CAD. A 2-tailed P < .05 was considered statistically significant.
Results
We first included 222 consecutive patients with STEMI who had undergone pPCI for our prospective study. Four patients were excluded. Two patients with cardiogenic shock died in the catheter laboratory during pPCI, 1 patient died during hospitalization due to mechanical complications, and another patient had undergone urgent bypass, so the body compositions of these patients were not calculated. The mean age was 56.8 (range 32-84) years and 79% of the patients were male. The no-reflow rate was 19%, and the mean Gensini score was 55 (range 16-136). The baseline clinical characteristics, biochemical parameters, fat, and muscle mass and muscle strength of the patients are presented in Table 1.
Comparison of the Patients With STEMI in Baseline Clinical, Angiographic, and Biochemical Characteristics; Fat and Muscle Mass; and Muscle Strength in Terms of No-Reflow and Gensini Score.
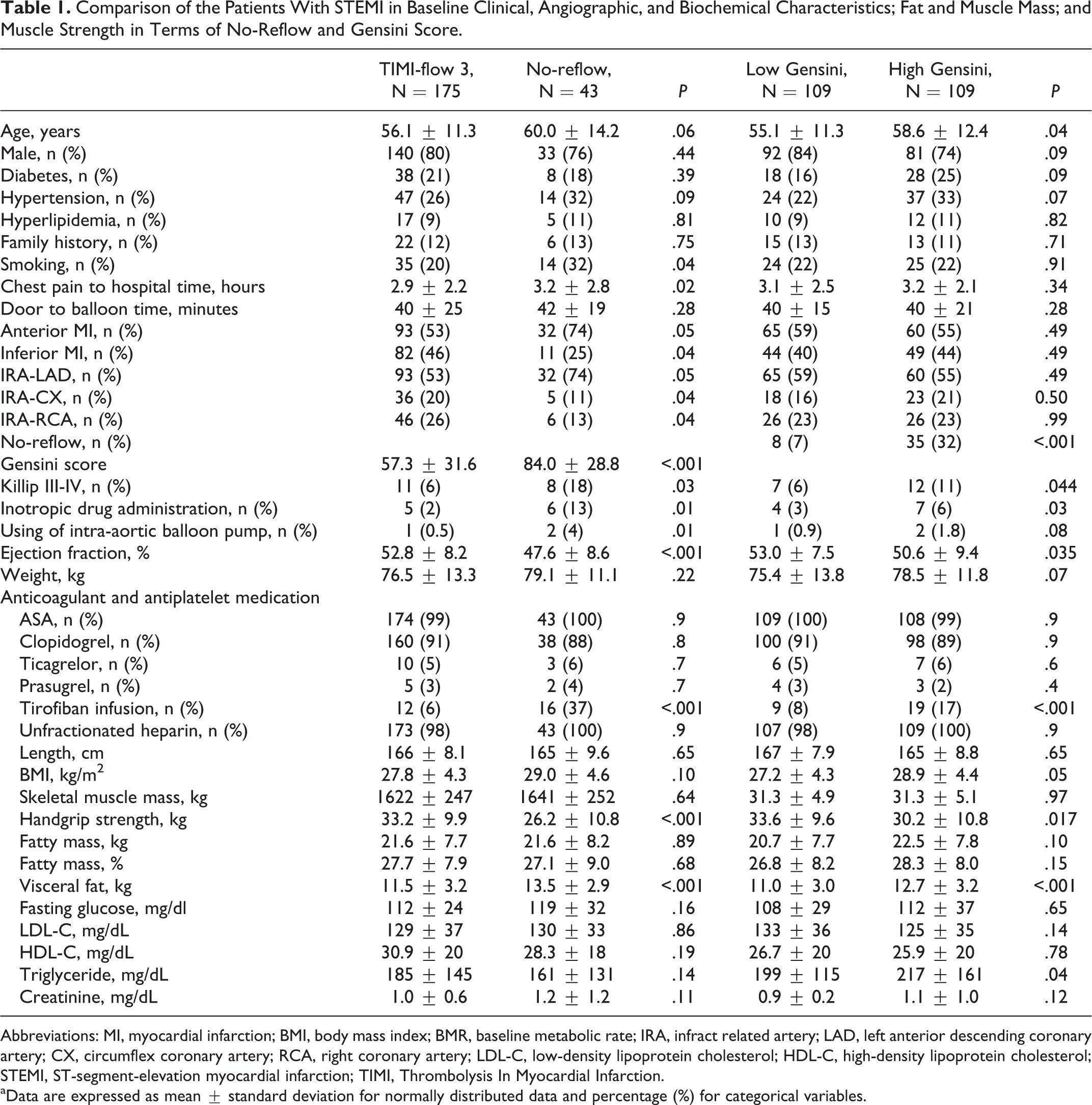
Abbreviations: MI, myocardial infarction; BMI, body mass index; BMR, baseline metabolic rate; IRA, infract related artery; LAD, left anterior descending coronary artery; CX, circumflex coronary artery; RCA, right coronary artery; LDL-C, low-density lipoprotein cholesterol; HDL-C, high-density lipoprotein cholesterol; STEMI, ST-segment-elevation myocardial infarction; TIMI, Thrombolysis In Myocardial Infarction.
aData are expressed as mean ± standard deviation for normally distributed data and percentage (%) for categorical variables.
There was no difference in BMI, BMR, total fat, and muscle mass between the patients with no-reflow and TIMI 3 flow (all P > .05). However, the patients with no-reflow had significantly lower handgrip strength and more visceral fat (both P < .001, Table 1).
We used the ROC curve analysis to identify a cutoff value for the predictors of no-reflow. We found that the visceral fat >11.5 kg was related to no-reflow with a sensitivity of 74% and a specificity of 55% (area under curve: 0.686, 95% CI: 0.609-0.764; P < .001). The handgrip strength <23.5 kg was related to no-reflow with a sensitivity of 78% and a specificity of 52% (area under curve: 0.324, 95% CI: 0.226-0.421; P < .001; Figure 1).
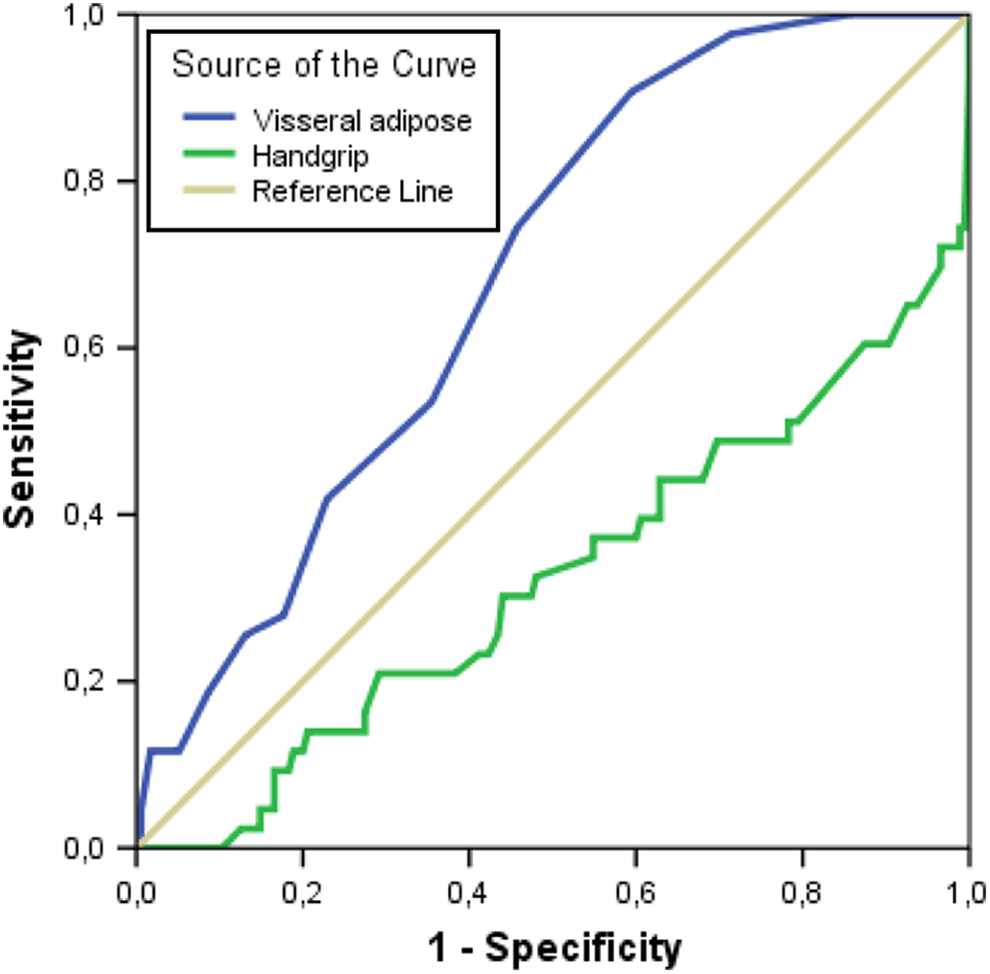
Receiver–operating characteristic curve (ROC) analysis of visceral fat and handgrip strength for predicting no-reflow. The visceral fat > 11.5 predicted no-reflow with a sensitivity of 74% and a specificity of 55% (area under curve (AUC): 0.686, 95% confidence interval [CI]: 0.609-0.764; P < .001) and the handgrip strength < 23.5 predicted with a sensitivity of 78% and a specificity of 52% (AUC: 0.324, 95% CI: 0.226-0.421; P < .001).
In multivariate regression analysis, visceral fat, handgrip strength, and EF were independently associated with no-reflow in pPCI (P = .001, P = .014, and P = .006, respectively; Table 2).
Multivariate Regression Analyses of Variables Related to No-Reflow.
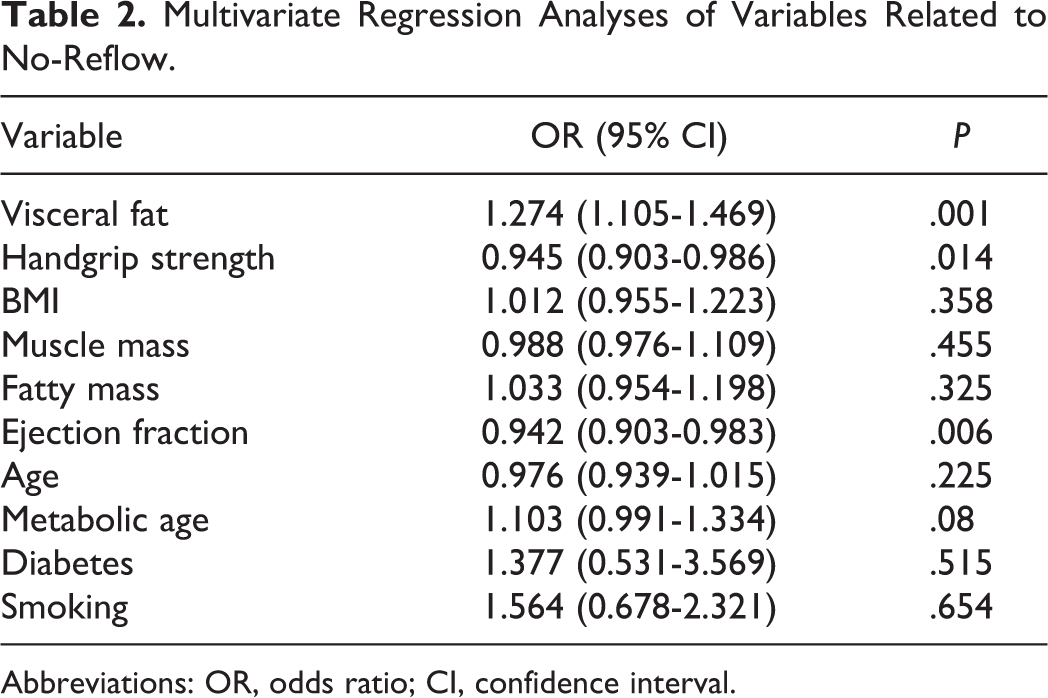
Abbreviations: OR, odds ratio; CI, confidence interval.
Comparison of fat and muscle mass and muscle strength between patients with low and high Gensini scores revealed no difference in BMR, fat, and muscle mass (all P > .05). Patients with high Gensini scores had more BMI, lower handgrip strength, and more visceral fat (P = .05, P = .017, and P < .001, respectively; Table 1).
We used ROC curve analysis to identify a cutoff value to be related to high Gensini scores. We found that having greater than 11.5 kg of visceral fat was associated with a high Gensini score, with a sensitivity of 61% and a specificity of 59% (area under curve: 0.644, 95% CI: 0.571-0.717; P < .001). Furthermore, a handgrip strength less than 28.6 kg was associated with a high Gensini score with a sensitivity of 68% and a specificity of 52% (area under curve: 0.406, 95% CI: 0.331-0.482; P = .017; Figure 2).
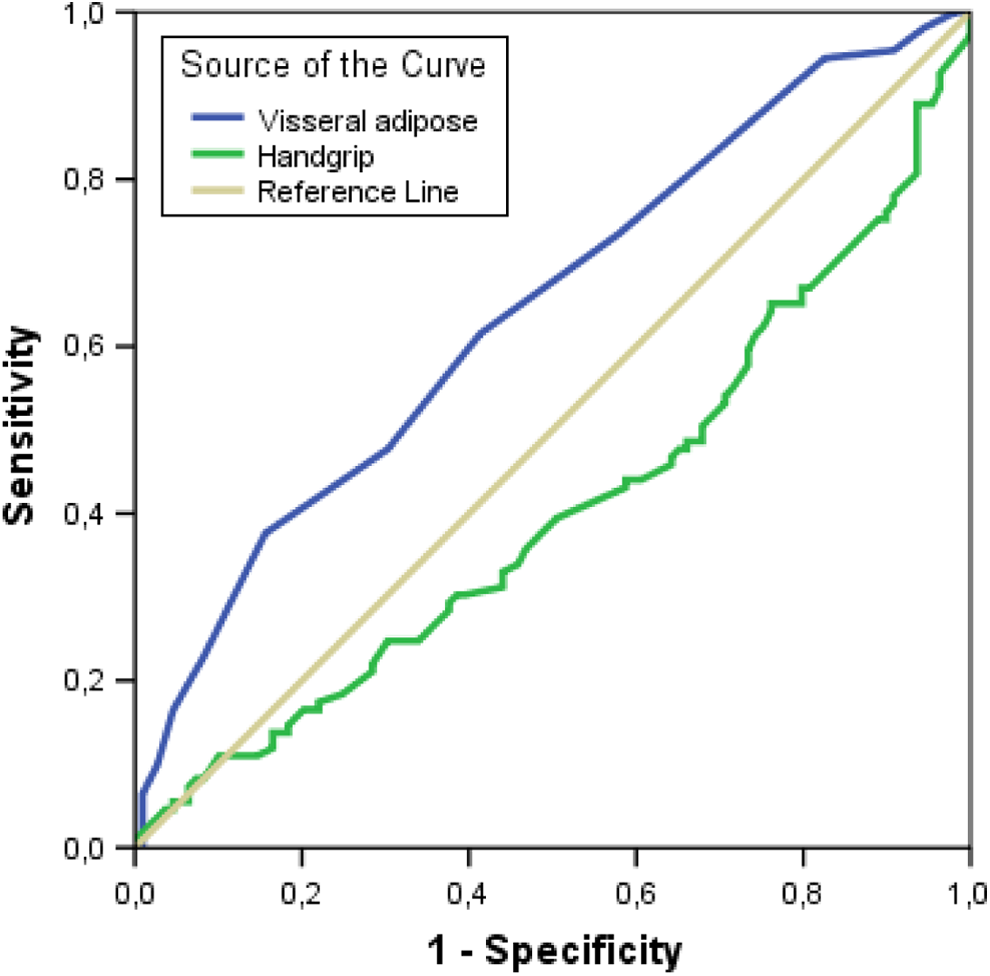
Receiver–operating characteristic curve (ROC) analysis of visceral fat and handgrip strength for predicting no-reflow. The visceral fat > 11.5 predicted high Gensini score with a sensitivity of 61% and a specificity of 59% (area under curve (AUC): 0.644, 95% confidence interval [CI]: 0.571-0.717; P < .001) and the handgrip strength < 28.6 predicted high Gensini score with a sensitivity of 68% and a specificity of 52% (AUC: 0.406 95% CI: 0.331-0.482; P = .017).
In multivariate regression analysis, visceral fat and handgrip strength were independently associated with high Gensini scores in patients with AMI (P = .022 and P = .010 respectively; Table 3).
Multivariate Regression Analysis of Variables Related to High Gensini Scores.
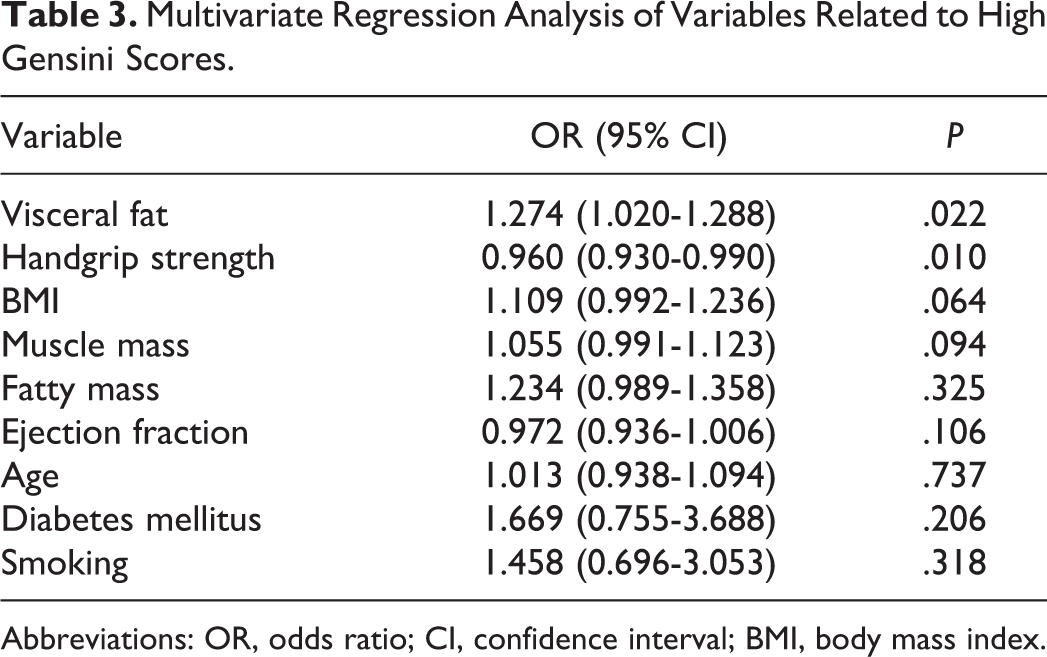
Abbreviations: OR, odds ratio; CI, confidence interval; BMI, body mass index.
Discussion
We evaluated the relationship between fat and muscle mass, muscle strength and no-reflow rate, and severity of coronary atherosclerosis in the patients with STEMI who underwent pPCI. We found that more visceral fat and lower muscle strength were significantly related to both no-reflow and CAD severity. In addition, there was no association between total fat and muscle mass and no-reflow and CAD severity in the patients with STEMI.
No-reflow is a significant prognostic marker related to short- and long-term poor cardiac outcomes in pPCI. 17 Moreover, Gensini scoring system reflecting severity and extensiveness of coronary atherosclerosis is another prognostic marker correlated with poor cardiac outcomes. 15 In our study, the patients with no-reflow and high Gensini scores had lower EF and admitted more with Killip Class 3 and 4 compared to the patients with TIMI 3 flow and low Gensini scores. Multiple angiographic scoring systems, such as Gensini, Syntax, CASS-50, and CASS-70, have been used to describe coronary atherosclerotic burden. 12 Although intravascular ultrasound (IVUS) remains the gold standard, angiographic scoring systems are homogeneously consistent with one another and with IVUS-derived atherosclerotic plaque burden. 12
Increased visceral fat, as an ectopic fat mass, results from increased free fatty acids and triglycerides because of saturated lipid storage capacity in subcutaneous adipose tissue due to chronic high caloric intake. 18 Visceral fat is associated with a higher risk of lipid disorders, type 2 diabetes, cardiovascular disease, cancer, and death. 19 Obese patients with a similar BMI and lower visceral fat are classified as metabolic healthy obese, and they have lower cardiovascular and all-cause mortality compared to obese patients with more visceral fat and similar BMI. Lower visceral fat is associated with higher insulin sensitivity and lower blood pressure, fasting glucose, triglycerides, fibrinogen, and higher high-density lipoprotein (HDL) levels. 19
There is lacking data on impact of increased visceral fat on no-reflow rate in STEMI. Bakirci et al found that increased epicardial fat was associated with impaired coronary flow in patients with myocardial infarction. 20 Okura et al showed that increased pericardial fat volume was associated with severity of CAD in patients with preserved EF. 21 Morricone et al showed that increased visceral fat correlated with high Gensini scores in patients with CAD. 22
Visceral fat induces insulin resistance and inflammation. Inflammatory markers, including C-reactive protein (CRP) and tumor necrosis factor (TNF), and mediators such as interleukin (IL) 6 and IL-1 and monocyte chemoattractant protein 1, are increased. In addition, adiponectin levels are decreased, and leptin resistance is developed. All these pathophysiologic mechanisms contribute substantially to accelerated atherosclerosis in visceral obesity. 23 –25
In addition, visceral fat mass secrets atherogenic adipocytokines, such as plasminogen activator inhibitor 1, adipocyte fatty acid-binding protein, lipocalin 2, chemerin, visfatin, vaspin, and resistin. 26 High-density lipoprotein decreases and becomes less dense due to increased hepatic lipase activity and cholesteryl ester transfer protein. Instead of having an anti-inflammatory effect, this dysfunctional HDL may actually contribute toward inflammation and the accelerated atherosclerotic process. 27
Muscle mass and muscle strength are independent prognostic markers for mortality in older patients with chronic diseases, including heart failure, renal failure, and cancer, in addition to middle-aged healthy men. 10,28,29 The impact of muscle strength and muscle mass on no-reflow and CAD severity of the patients with STEMI has not been clarified in the literature. Although the amount of muscle mass correlates with muscle strength, they are 2 different entities and strength is the better prognostic marker. 29,30 Muscle strength substantially depends on the size, number, and contractility of fibers; collagen content; and fat infiltration. Muscle strength, easily measured by handgrip, shows muscle quality and is more of a protective marker than muscle mass in terms of mortality. 30,31 Greater handgrip strength reflects better nutritional and physical status. Handgrip strength is routinely measured in patients with heart failure in Japan. 10 Low muscle strength was more strongly associated with aging, protein-energy wasting, physical inactivity, inflammation, and mortality than low muscle mass in patients with renal failure. 32 In our study, muscle mass did not relate to no-reflow and severity of atherosclerosis, but lower muscle strength was significantly associated with them.
Lower muscle strength is related to lower growth hormone, testosterone, and vitamin D levels; increased cortisol levels; and insulin resistant. 33 In addition, CRP levels increase when muscle mass and strength are lower. 34 Lower strength may be the result of visceral obesity, leading to increased adipokines and proinflammatory cytokines. 34 Like an endocrine organ, the skeletal muscle mass secrets hormone-like cytokines, named myokines. These myokines including IL-6, IL-8, IL-15, brain-derived neurotrophic factor, leukemia inhibitory factor, follistatin-like 1, and fibroblast growth factor-21 are produced by the skeletal muscle. 35 These myokines, increased inflammation, and hormonal status may be underlying pathophysiological mechanisms of increased atherosclerotic burden, increased cardiovascular events, and other metabolic diseases.
We measured the fat and muscle mass with a body composition analyzer using a bioelectrical impedance analysis technique. Bioelectrical impedance analysis is an approved method for estimating body composition, in particular fat and muscle mass, in nutrition, hemodialysis, gerontology, and sports medicine. 36 In addition, our body composition analyzer (Tanita, type BC-418 M) was validated and used to analyze body composition of the patients in many studies. 37,38
We used handgrip for estimating muscle strength. Handgrip strength is correlated with quadriceps strength, measured via isokinetic dynamometry, and is useful in estimating mortality risk. 30
Although the cutoff values of visceral fat and handgrip strength had relatively lower specificity and sensitivity, they were statistically significant. In addition, we used the ROC curve analysis to show a statistically significant relationship between lower handgrip strength and increased visceral fat and no-reflow and high Gensini score.
However, we did not aim to clarify any possible mechanisms of no-reflow and CAD severity and muscle strength, fat, and muscle mass. Our study emphasizes that visceral fat mass and muscle strength are significantly associated with no-reflow and high Gensini scores. Our findings cannot contribute to the treatment of STEMI but support that visceral fat and muscle strength may have a prognostic value in STEMI. Visceral fat and muscle strength may be important tools in primary and secondary prevention in STEMI.
Thus, the measuring visceral fat and muscle strength may be 2 useful methods for estimating cardiac outcomes. In addition, decreasing visceral fat and increasing muscle strength may protect patients under risk of CAD.
Our study is limited by, for example, the relatively low number of patients and absence of short- and long-term outcomes. However, our study has shown a new perspective for ongoing arguments about the impact of BMI, fat mass, visceral fat, muscle mass, strength, nutrition, and exercise on cardiovascular outcomes.
Conclusion
Increased visceral fat and lower handgrip strength are related to no-reflow rate and severity of coronary atherosclerosis in the patients with STEMI. Visceral fat and muscle strength may be better prognostic markers than BMI, total fat, and muscle mass.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
