Abstract
Objectives:
Patients with a history of heparin-induced thrombocytopenia (HIT), who require subsequent anticoagulation, have limited options. Rechallenge with unfractionated heparin (UFH) has been reported but may be associated with a risk of recurrence of HIT. The objective of this study was to determine the safety of heparin reexposure in patients with a history of HIT.
Methods:
Using several search terms, all cases of heparin reexposure in patients with HIT indexed in MEDLINE (English language only) by June 2014 were reviewed. The bibliography of each relevant article was searched for additional reports. In cases of multiple reexposures, each reexposure was identified as a separate instance of reexposure during analysis.
Results:
A total of 136 patients with a history of HIT had 141 instances of heparin reexposure. Cardiac (76%) and vascular surgeries (11%) were the most common indications. Antiplatelet factor 4/heparin antibodies were positive in 63% of evaluable cases before reexposure. Preexposure plasma exchange (11%) and postexposure nonheparin anticoagulants (63%) were frequently utilized. Complications with heparin reexposure included recurrence of HIT (2.1%, 95% confidence interval 0.73%-6.07%) and bleeding (2.1%).
Conclusion:
Intraoperative heparin reexposure in patients with a history of HIT has a small risk of developing HIT recurrence. The use of preexposure plasma exchange in patients with positive antiplatelet factor 4/heparin antibody and postexposure nonheparin anticoagulants arguably may have reduced the risk of recurrence of HIT.
Keywords
Introduction
Heparin-induced thrombocytopenia (HIT) is an immune-mediated complication of heparin therapy, associated with the formation of antiplatelet factor 4/heparin antibodies. Nonheparin anticoagulants such as argatroban, bivalirudin, lepirudin, danaparoid, and fondaparinux have been proposed as alternatives but concerns about their efficacy and safety, reliability on renal or hepatic excretion for reversal, and lack of availability in some centers may limit their use. 1,2 Patients with a history of HIT, who require a cardiopulmonary bypass, pose a challenge regarding their anticoagulation. Although bivalirudin 3 and argatroban 4,5 have been used in patients with HIT, the quality of data is not high enough to consider these agents as validated options. Rechallenge with heparin has a risk of recurrence of HIT, but unfractionated heparin (UFH) is still preferred by surgeons during cardiopulmonary bypass because of their familiarity with the drug, short half-life of the drug, easy monitoring, and reversibility with protamine sulfate. 2 A number of reports describe successful heparin reexposure during cardiopulmonary bypass. However, small sample size and a lack of systematic review of these cases have prevented better understanding of the potential complications. We have analyzed all published cases of heparin reexposure to determine the safety of heparin reexposure in patients with HIT and to understand various strategies utilized to reduce the recurrence of HIT.
Methods
This is a retrospective review of published cases of heparin reexposure in patients with HIT. One of the authors (PD) reviewed all cases of heparin reexposure in patients with HIT published and indexed in English language in MEDLINE by June 2014. Search terms included ((((heparin[Title/Abstract]) OR heparin induced thrombocytopenia[Title/Abstract]) OR HIT[Title/Abstract])) AND (((exposure[Title/Abstract]) OR re-exposure[Title/Abstract]) OR heparin re-exposure[Title/Abstract]). Case reports, clinical trial, comparative study, controlled clinical trial, observational study, and randomized controlled trial were included. The bibliography of each relevant article was searched for additional related reports. Inclusion criteria included patients who had a prior history of HIT, as reported by the authors of the report, and a subsequent exposure to heparin products. A total of 27 articles met the inclusion criteria, which were included in this study (Figure 1).
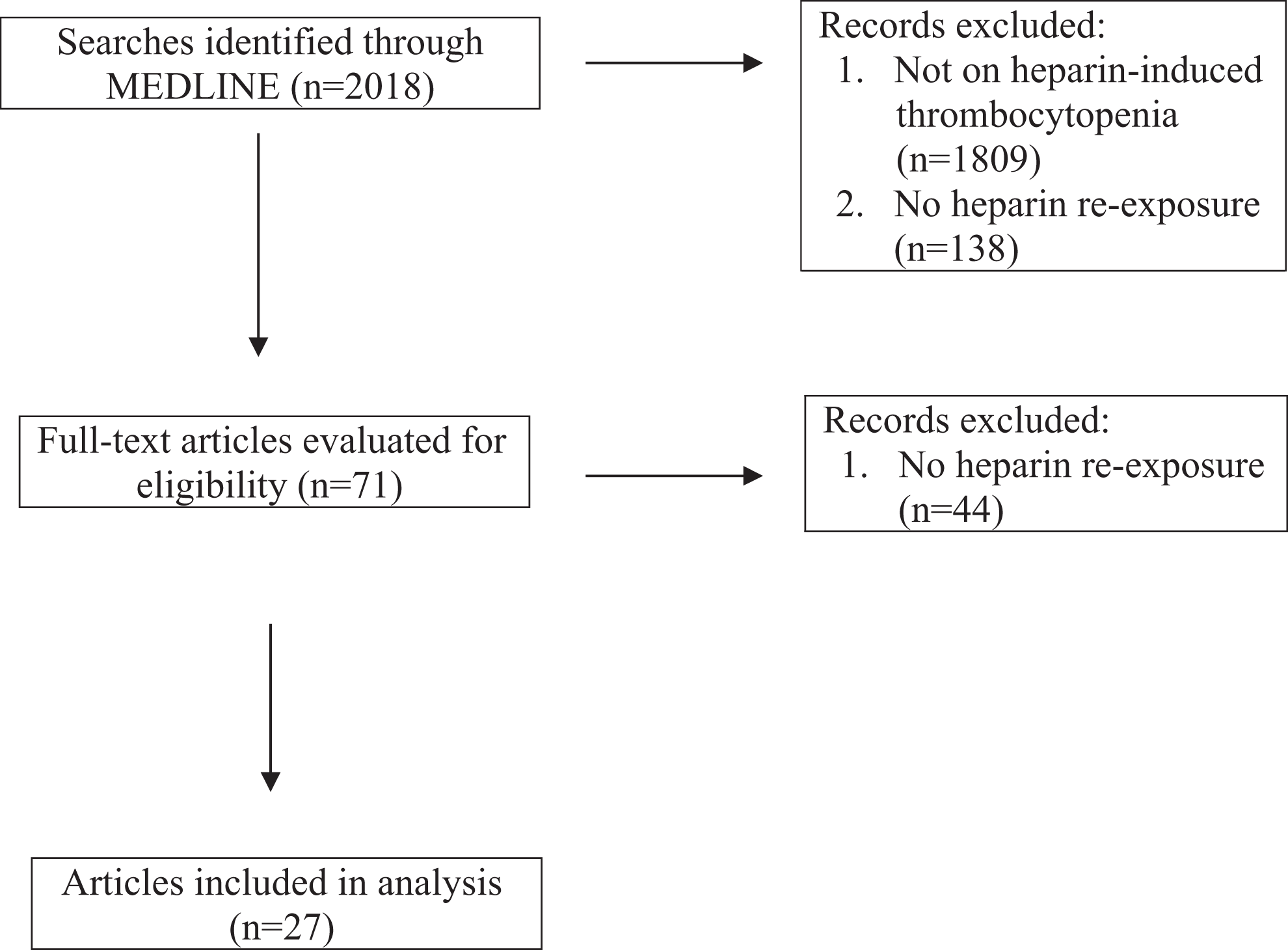
Flow diagram for selection of the articles.
The diagnosis of HIT was based on the clinical probability or 4T scoring system and laboratory tests. The reported 4T score was categorized as low (a score of 0 to 3), medium (4 to 5), and high (6 to 8) based on established criteria. 6 We calculated the score in 10 cases, where the criteria for scoring system were available but the score was not reported. 7 –15 Laboratory tests included enzyme-linked immunosorbent assay (ELISA), serotonin release assay (SRA), or other tests such as platelet aggregation studies. The exposure to either UFH or low-molecular-weight heparin (LMWH) in patients with a prior history of HIT was considered reexposure to heparin product, regardless of the time interval from the diagnosis. In 2 cases, heparin was used multiple times for repeated cardiovascular surgeries after an initial diagnosis of HIT. 8,16 Each reexposure was identified as a different instance of reexposure during analysis. Statistical analysis comprised of descriptive statistics including calculation of the frequency of distribution of different variables.
Results
We identified a total of 136 patients with a history of HIT with 141 instances of heparin reexposure. 1,7 –32 The study population had a median age of 56 years (4 weeks-87 years) and 67% males. Regarding the original HIT diagnosis, UFH (98%) and nadroparin (2%) 19 were the causes of HIT. Thrombotic complications occurred in 23% (n = 32) of cases during their index episode of HIT, comprising arterial in 15% (n = 20), 11,16,20,22,27,29,32 venous in 4% (n = 6), 8,16,29,32 and both arterial and venous in 4% (n = 6). 12,14,16,18 The pretest probability (4T) score was not available in 73% (n = 100) of cases. We calculated the score in 10 cases, where criteria for the scoring system were mentioned. 7 –15 The 4T score, where available, was high in 78% (n = 28) of cases and moderate in 22% (n = 8). Immunological studies were not available in 10% (n = 13) of cases. When available, platelet aggregation studies (59%), ELISA (29%), SRA (2%), and both SRA and ELISA (10%) were performed for diagnosis.
Cardiac (76%) 1,7 –9,13,15,16,20 –22,25 –30,32 and vascular (11%) 16,17,32 surgeries were the most common indications for heparin reexposure (Table 1). The other reasons (11%) included hemodialysis (n = 9), 10,11,19,31 thromboembolism (n = 2), 12,18 medical treatment (n = 3), and inadvertent use (n = 3). 32 Nadroparin (a LMWH) was used in 1 case to treat HIT that developed after the use of heparin. 14 The interval between HIT and heparin reexposure was not available in 62% of cases. Where available, 66% of the cases were reexposed to heparin after 3 months of HIT diagnosis, whereas 11%, 8%, and 15% were reexposed within 1 week, between 1 week to 1 month, and 1 month to 3 months of HIT diagnosis, respectively. Antiplatelet factor 4/heparin antibody, not available in 13% of instances (n = 19), was positive in 63% of the remaining instances (n = 77) at the time of heparin reexposure. 1,8,13,14,16,17,19 –22,27,30 Unfractionated heparin was used alone 1,8 –11,13,15,16,18,20 –24,26 –32 or in addition to LMWH 7,12 in 93% of reexposures, whereas LMWHs such as enoxaparin 17,25 or nadroparin 14,19 alone were used in 7% of cases.
Review of Patients With Heparin-Induced Thrombocytopenia With Heparin Reexposure.
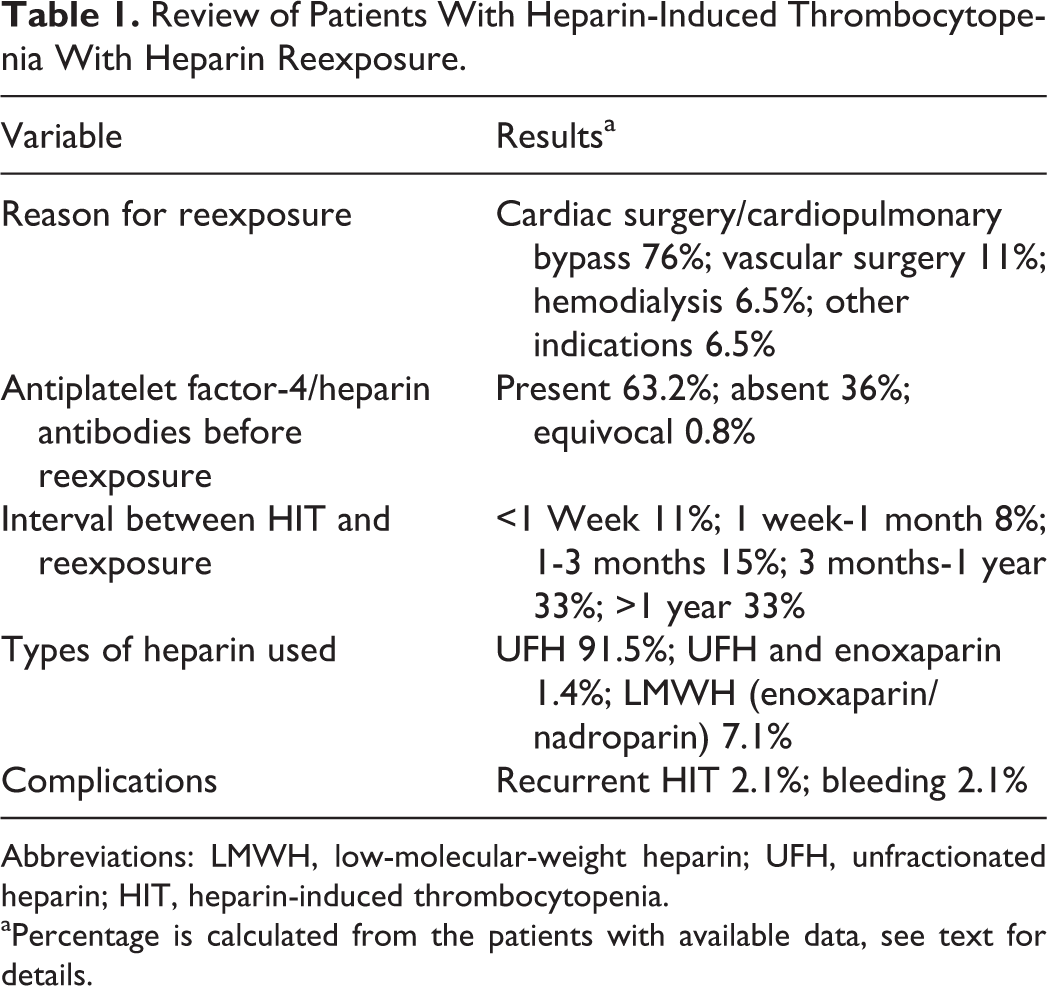
Abbreviations: LMWH, low-molecular-weight heparin; UFH, unfractionated heparin; HIT, heparin-induced thrombocytopenia.
aPercentage is calculated from the patients with available data, see text for details.
The patients received various additional interventions along with heparin. Sixteen (11%) patients underwent plasma exchange, 1,8,9,15,30 which lowered the level of antiplatelet factor 4/heparin antibodies to negative or very low before heparin reexposure. Of 16 patients, 3 underwent plasma exchange multiple times, that is, 1 patient underwent plasma exchange 3 times (once before each cardiopulmonary bypass) 8 and the other 2 had it multiple times before cardiopulmonary bypass. 9,15 Among the patients who underwent preexposure plasma exchange, 3 patients were reexposed to heparin within a week of HIT diagnosis, 8,30 9 within 2 weeks, 1 1 within a month, 15 and 3 within 3 months. 1,9
Nonheparin anticoagulants such as bivalirudin, 1,15 fondaparinux, 16 danaparoid, 16 r-hirudin, 20 –22,28 argatroban, 27 lepirudin, 13,26,27,29,30 warfarin, 1,15,16 acenocoumarol, 14 and phenprocoumon 26 were used singly or in combination in 63% of patients. These drugs were mainly used for postsurgical anticoagulation. Other utilized drugs included antiplatelet drugs such as aspirin with or without clopidogrel, 9,16,31 alteplase, 18 tirofiban, 13,20 –22,26 and aprotinin. 26 Perioperative r-hirudin, along with intraoperative tirofiban and heparin, was used in 43% (n = 58). 20 –22 Protamine was administered postoperatively in 6% (n = 8) of cases 7,9,13,17,25,28 –30 to reverse the effects of heparin.
With heparin reexposure, 4.2% had complications which included recurrence of HIT (n = 3, 2.1%) 12,16,18 and bleeding (n = 3, 2.1%). 7,25,27 The risk of recurrence of HIT was 2.1% (95% confidence interval 0.73%-6.07%). 33 Among the patients with HIT recurrence, 1 patient was reexposed to UFH within a week of HIT diagnosis and shortly after platelet recovery with LMWH, thus this is more appropriately considered worsening of HIT, rather than HIT recurrence. 12 The other 2 patients were initially diagnosed with HIT more than 5 years ago and tested negative for antiplatelet factor 4/heparin antibody prior to heparin reexposure. 16,18 One of the patients developed pulmonary embolism, thrombi in aorta and common iliac artery, and eventually died; however, HIT recurred in this patient following a full course of heparin. 18 Among the patients with bleeding, heparin was used in bonded circuit in 1 patient during cardiopulmonary bypass along with preoperative as well as intraoperative enoxaparin and postoperative protamine. 7 Bleeding subsided after transfusion of fresh frozen plasma and platelets in this patient. In another patient, argatroban was used perioperatively along with intraoperative heparin during cardiac transplantation. 27 The bleeding stopped in this patient after revision of surgery. In the third patient, enoxaparin was used intraoperatively with protamine at the end of aortic valve replacement; postoperative bleeding stopped after surgical exploration and use of a fibrin sealant compound. 25
Discussion
Our review determined a total of 136 patients with a history of HIT with 141 instances of heparin reexposure. Cardiovascular surgeries accounted for the majority of cases, which may highlight cardiovascular surgeons’ preference of UFH because of aforementioned reasons. Recurrence of HIT following intraoperative UFH reexposure was 2.1% (95% confidence interval 0.73%-6.07%). This is similar to the reported incidence of first episode of HIT, which is 0.2% to 5% of adults treated with heparin. 2 In our review, 2 patients with HIT recurrence were reexposed to heparin after more than 5 years of initial diagnosis, when they tested negative for antiplatelet factor 4/heparin antibody. 16,18 Thrombocytopenia recurred in 1 patient who was reexposed to UFH within a week of HIT diagnosis and shortly after platelet recovery with LMWH. 12 This is considered as a worsening of preexisting HIT rather than a recurrent episode of HIT.
In a well-designed study by Warkentin et al evaluating 20 patients with a previous history of HIT who were reexposed to heparin (17 for surgical and 3 for medical indications), 65% of 17 patients with intraoperative heparin reexposure at a mean of about 4.4 years post-HIT developed ELISA seroconversion; 73% of these patients demonstrated SRA positivity. The median time to detection of antiplatelet factor 4/heparin antibody was 7 days. One (5%) of the 20 patients developed HIT 7 days after intraoperative heparin reexposure for cardiac surgery; HIT occurred as a result of newly regenerated antiplatelet factor 4/heparin antibody while on fondaparinux prophylaxis (included in this review). The authors concluded that the patients with previous HIT may be at an increased risk of developing recurrent platelet-activating antiplatelet factor 4/heparin antibody, such antibody formation follows the time line of a typical HIT but the risk of HIT is low with intraoperative heparin reexposure. 16 This case demonstrates an example of delayed-onset HIT, 34 which occurred after the cessation of heparin use. Whether the ELISA is weakly or moderately positive before the reexposure is irrelevant to this type of HIT reoccurrence. 16 Other types of HIT recurrence may include typical-onset HIT recurrence 18 and rapid-onset HIT. 12 The rapid-onset HIT may result as an abrupt recurrence of HIT in a patient who is given heparin prior to disappearance of platelet-activating heparin-dependent antiplatelet factor 4/heparin antibody. The typical-onset HIT recurrence 18 reflects recurrent formation of HIT antibodies after receiving a full course of heparin (this requires ongoing heparin for pathogenicity). The other 2 patients 12,18 in this review demonstrate these latter 2 types of HIT recurrence.
These different types of HIT recurrence raise important treatment implications. First, repeat heparin should be avoided in patients with persistence of platelet-activating antiplatelet factor 4/heparin antibody, which is likely to be the case if heparin rechallenge is done soon after recovery from HIT. 12 A full course of heparin, to treat venous thromboembolism or for any other indication, is not acceptable in a patient with a previous well-documented history of HIT. Prolonged exposure in such patients can be fatal 18 and is not necessary due to the availability of other nonheparin anticoagulants. Patients with a prior history of HIT may develop delayed-onset HIT recurrence even with the use of intraoperative or short-course heparin for cardiovascular surgery. Although the risk of such recurrence is low, as shown in our review, it can occur. 16 Therefore, it is important to at least monitor postoperative platelet count for 7 to 10 days; any development of a new, otherwise unexplained platelet count drop that begins 5 to 10 days after heparin reexposure should trigger a concern for delayed-onset HIT.
The time interval between HIT diagnosis and the reexposure is considered an important factor so as to allow the HIT-related immune response to decrease over time. 32 In the presence of pathogenic antiplatelet factor 4/heparin antibody, HIT can recur within 24 hours of heparin reexposure (rapid-onset HIT), as discussed previously. Antiplatelet factor 4/heparin antibodies are reported to be negative in 60% of cases after more than 100 days of HIT 32 and heparin reexposure is considered relatively safe in the absence of antiplatelet factor 4/heparin antibody. 2 The risk of recurrent HIT may remain modest, even with positive antiplatelet factor 4/heparin antibodies, if the antibodies do not activate platelets (negative functional assays). 16 In our study, one-third of patients were reexposed to heparin within 3 months of HIT diagnosis and nearly two-thirds of evaluable patients had antiplatelet factor 4/heparin antibodies at the time of heparin reexposure. Despite this, the risk of HIT recurrence was low. This may be the result of plasma exchange or the use of alternative nonheparin anticoagulants, as discussed further subsequently, or because of the presence of nonpathogenic or low-titer antiplatelet factor 4/heparin antibody. Although time is a crucial factor to allow for disappearance of HIT antibodies, it may not be an important issue for determining risk of recurrent HIT in a patient who had remote HIT (arbitrarily, more than 3 months earlier). This is exemplified by the 2 patients who developed recurrent HIT after many years had elapsed since the initial diagnosis of HIT. 16,18 Further, patients may develop a delayed-onset HIT recurrence as a result of newly regenerated antiplatelet factor 4/heparin antibody. 16
In patients without heparin exposure in previous 100 days, about 5 to 7 days may be necessary for antibody formation after heparin reexposure. Unless heparin is present when antibody forms, the risk of HIT recurrence is low but not zero because of the possibility of delayed-onset HIT. Thus, limiting the heparin use to intraoperative anticoagulation may reduce the possibility of HIT recurrence. 16 The majority of patients in this study were reexposed to heparin for a short time period. In patients with a history of HIT, who are reexposed to heparin, postoperative anticoagulation, if needed, should be achieved with nonheparin anticoagulant. 16 Direct thrombin inhibitors, indirect factor Xa inhibitors, and vitamin K antagonists were frequently utilized as alternative anticoagulants in our study. Many of these agents are useful in the management of HIT. 2 These 2 factors may account for relatively small risk of HIT recurrence noted in this study. However, the use of postoperative anticoagulants may be associated with an increased risk of bleeding, hence they should be utilized only when needed and under close monitoring.
Plasma exchange has been utilized as a treatment of HIT. It is believed to reduce antiplatelet factor 4/heparin antibody titers through filtration and centrifugation, as well as a result of the replacement of plasma with fluids such as albumin. 35 Improvement in platelet count 8,9,36,37 and rapid resolution of thrombosis 38,39 after plasma exchange has also been reported in a few patients with HIT. One study (n = 44) reported the use of early plasma exchange (within 4 days of the onset of thrombocytopenia) to possibly decrease 30-day mortality in comparison to late or no plasma exchange. 40 However, this study had multiple flaws including lack of reliability of diagnosis of HIT, longer time to discontinuation of heparin in the late plasma exchange group, and lack of faster platelet recovery or lower thrombotic rate in early versus no plasma exchange groups. Plasma exchange is associated with potential complications such as hemodynamic instability, hypocalcemia, alkalosis, hypokalemia, and infection. 1 In our study, 16 patients underwent plasma exchange before heparin reexposure to prevent HIT and none of them had any complication. 1,8,9,15,30 However, the efficacy of plasma exchange in treating patients with HIT has only been demonstrated by case series hence not established. The use of plasma exchange as a treatment of HIT is currently a category III recommendation (optimum role not established; decision making should be individualized) by American Society for Apheresis guidelines. 41 Although plasma exchange may (or may not) have reduced the risk of HIT recurrence in our study population, further well-designed studies are needed to evaluate the role of plasma exchange in prevention of HIT recurrence (as well as initial treatment of HIT). In this context, recently, Warkentin et al 42 demonstrated that plasma exchange can remove sufficient antiplatelet factor 4/heparin antibody to achieve negative SRA. ELISA (Enzyme-linked immunosorbent assay) may remain positive despite a negative SRA. Given the goal of achieving a negative SRA before heparin reexposure, monitoring of SRA rather than ELISA can limit the number of plasma exchange.
Our retrospective review has several limitations. Unsuccessful cases are less likely to be reported than successful cases, thus the review may significantly underestimate the risks of HIT recurrence or other complications. The diagnosis of HIT requires the presence of intermediate to high pretest probability and high-titer immunoglobulin G antiplatelet factor 4/heparin antibody or positive functional tests such as SRA. Given the retrospective nature of the study, the accuracy of HIT diagnosis could not be verified in many cases. Important information such as the presence and titer of antiplatelet factor 4/heparin antibody prior to reexposure, which are important determinants of the risk of recurrence of HIT, was missing in many cases. However, to our knowledge, this is the first systematic analysis of all published cases of heparin reexposure in patients with a history of HIT, which provides information useful for patient education and decision-making process.
Conclusion
Our study indicates that the risk of recurrent HIT with intraoperative heparin reexposure in patients with a history of HIT is small but possible among patients who develop platelet-activating antiplatelet factor 4/heparin antibody. Additionally, patients may be at a risk of delayed-onset HIT, thus requiring monitoring of platelet count for 7 to 10 days after heparin reexposure. The use of a full course of heparin can result in typical-onset HIT with potential fatal complications, hence should be avoided. Despite this, if appropriate precautions are taken, the actual risk of HIT recurrence with intraoperative heparin use is modest and may be similar to the risk of HIT in a patient without a history of HIT. Preexposure plasma exchange in patients with positive antiplatelet factor 4/heparin antibody and the use of postexposure nonheparin anticoagulants arguably may have played a role in reducing the reoccurrence of HIT. This study cannot confirm a low risk of HIT recurrence in patients with positive functional test or high-titer antiplatelet factor 4/heparin antibody at the time of exposure.
Footnotes
Author’s Note
This article was presented as an abstract at the 56th American Society of Hematology 2014 Annual Meeting held on December 8, 2014, San Francisco, California.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
