Abstract
Angelica shikokiana is a Japanese medicinal plant that is used traditionally in several ailments of cardiovascular diseases. However, there is no report regarding its anticoagulant or antiplatelet activities. So this study was designed to screen for such activities (anticoagulant by prothrombin time [PT], activated partial thromboplastin time, and thrombin time assays and antiplatelet activities against adenosine 5′-diphosphate [ADP] and arachidonic acid-induced platelet aggregations) for the methanol extract of the aerial part (Angelica methanol extract [AME]), its isolated coumarins, flavonoids, and flavonoid metabolites. The AME had potent anticoagulant and antiplatelet activities, and the flavonoid compounds were evidenced to be responsible for such activities. Among coumarins compounds, hyuganin C showed significant prolongation of only PT, while other coumarins were inactive. Similarly, hyuganin C and bergapten were the only active coumarins against ADP-induced platelet aggregation. Compared to the parent compounds, colonic metabolites of the flavonoids had similar anticoagulant and antiplatelet activities, while glucuronides showed sharp decreases in all studied activities. This is the first report showing that the medicinal plant A shikokiana has potent antiplatelet and anticoagulant activities.
Introduction
Hemostasis is a crucial physiological process, which ensures that bleeding stops and blood vessel wall closes following vascular injury. The four major components are the vascular system, platelets, blood coagulation factors, and fibrinolysis and ultimate tissue repair. Normal hemostasis requires an exquisite balance of platelets, endothelial cells, and coagulation factors to maintain a resting nonthrombotic state, however, responds instantly to vascular damage. 1,2 Although platelet activation and subsequent thrombus formation at sites of vascular injury are crucial for normal hemostasis, they also play fundamental roles in atherothrombosis and subsequently cardiovascular disease such as myocardial infarction and stroke. 3 –5
Despite the continued efforts to control cardiovascular risk factors, cardiovascular disease remains the leading cause of death worldwide, in both developed and developing countries, according to the most recent World Health Organization report. 6 For more than 5 decades, antiplatelet and anticoagulant drugs have been the major players in this clinical setting. Although their efficacies are well established, the deleterious life-threatening side effects of these drugs have also been well documented, 7,8 which prompt for the investigations of natural products as alternatives. An exploratory trial toward identifying new anticoagulants and antiplatelet from medicinal plants is worthwhile. 9
The perennial herb Angelica shikokiana (Apiaceae) is a Japanese medicinal plant known as Inutouki or Yamaninjin and is used as a substitute drug for ginseng roots. 10 Traditionally, soluble preparations from stems and leaves are used for several complications of cardiovascular diseases such as hyperlipidemia, diabetes, atherosclerosis, and hypertension. 11 Our previous study 12 revealed that the ethanol extract of A shikokiana leaves and stems had potent anti-inflammatory, antioxidant, and antilipase activities. Recently, we have isolated 3 coumarins, hyuganin E, isoepoxypteryxin, and isopteryxin, and 5 flavonoids, kaempferol, luteolin, quercetin, kaempferol-3-O-glucoside, and kaempferol-3-O-rutinoside, from the methanol extract of the aerial part. 13 In our continuous upsurge in searching for novel treatments from medicinal plants, we aimed to screen for the potential, anticoagulant, and antiplatelet activities of A shikokiana and its previously isolated coumarin and flavonoid compounds. To the best of our knowledge, there is no previous report about such activities from A shikokiana.
Materials and Methods
Preparation of Plant Material and Isolated Compounds
The dried powder of the aerial part of A shikokiana (voucher specimen No. Y-1) was obtained from Joy Plus (Kurume, Japan). Twenty grams of the dried powder of the aerial part of A shikokiana was extracted with methanol (150 mL × 3) at room temperature (RT; 25°C) to prepare Angelica methanol extract (AME). The extract was concentrated on a rotary evaporator, and the percentage of yield was 8.4%. The following compounds: isoepoxypteryxin, isopteryxin, hyuganin E, quercetin, luteolin, kaempferol, kaempferol-3-O-glucoside, and kaempferol-3-O-rutinoside were isolated as described previously. 13 Bergapten, psoralen, angelicin, and hyuganin C were isolated, and the chemical structures were elucidated from 1H- and 13C-NMR and 2-dimensional spectra recorded using a Bruker DRX 600 NMR spectrometer (Bruker Daltonics Inc, Billerica, Massachusetts) as mentioned in the supplementary material. The structure of the tested compounds is shown in supplementary materials, Figure S1.
Commercially available flavonoid metabolites, colonic metabolites, p-hydroxy phenyl acetic acid (p-HPAA) and 3,4-dihydroxyphenylacetic acid (DOPAC), 14 –17 and glucuronide conjugates, 18 kaempferol-3-O-glucuronide, quercetin-3-O-glucuronide, luteolin-7-O-glucuronide, phosphate-buffered saline (PBS), and acetylsalicylic acid (ASA), were purchased from Sigma Chemical Company (St Louis, Missouri). The AME and compounds were dissolved in dimethyl sulfoxide (DMSO) to prepare stock solutions which were finally diluted with PBS to obtain working ones (the final concentration of DMSO was less than 1%).
The blood coagulation assays reagents were purchased from Siemens Healthcare Laboratories (Siemens Medical Solutions, Malvern). These reagents used were Thromborel S for prothrombin time (PT) assay, Pathromtin SL, and calcium chloride solution 0.025 mol/L for activated partial thromboplastin time (APTT), test thrombin reagent for thrombin time (TT), control plasma normal, and control plasma high (pathological). For platelets aggregation, arachidonic acids (AAs) and adenosine 5′-diphosphate (ADP) were purchased from Chrono-log Corporation (Chicago, Illinois).
Blood Sampling and Plasma Preparation
The blood used for the experiments was obtained from healthy resting volunteers (n = 25, aged 18-30 years) who signed an informed consent before starting the research. They had no history of hematological diseases (related to, or associated with platelet and/or coagulation disorders), or cardiovascular disorders, with no drug history for at least 14 days before the blood withdrawal, all were nonsmokers with no medical history of chronic diseases, and have refrained from caffeine ingestion on the day of testing. Additionally, there was no history of herbal remedies, garlic, alcohol, and excess vegetable intake.
Venous blood samples were collected by aseptic venipuncture from the cubital vein with the citrated blood collection system, with minimal stasis. Blood of 1.8 mL was delivered into the citrated plasma Blood Collection system sterile vacutainer tube containing 200 µL sodium citrate (3.2%) in a 9:1 (v/v) ratio (0.129 mol/L) solution as anticoagulant (9 parts of blood with 1 part of anticoagulant; Vacutest, Kima s.r.l., Italy). From the citrated blood samples, platelet-rich plasma (PRP) and platelet-poor plasma (PPP) were prepared. The PRP was prepared by slow-speed centrifugation (MPW 350R; MED Instruments, Warsaw, Poland), at 170g for 10 minutes at RT without application of the brake. After centrifugation, the PRP was collected and the remaining blood was centrifuged at 2000g for further 15 minutes at RT for preparation of PPP, then was pooled, centrifuged again, and stored at −70°C. All these procedures were in accordance with the guidelines of the British Committee for Standards in Hematology on the laboratory aspects of assays used in hemostasis and thrombosis 19 and the laboratory investigation of blood platelets function. 20
In Vitro Anticoagulant Activity
To evaluate the extrinsic and the intrinsic pathways of coagulation, PT and APTT were measured, respectively. For the final stage of coagulation, TT was evaluated. The tests were conducted in a fully automated coagulometer (Sysmex CA-560; Sysmex, Kobe, Japan). Normal citrated PPP (50 µL) was preincubated for 5 minutes at 37°C with different concentrations of AME (1.25, 2.5, 5, 10, and 20 µg/mL, final concentration) and compounds (1.25, 2.5, 5, 10, and 20 µmol/L) and then PT, APPT, and TT assays were done according to the manufacturer and the coagulometer protocols. Saline solution with 1% DMSO and heparin (final concentration of 10 µg/mL for PT and 0.5 µmol/L for APTT and TT) were used as negative and positive controls, respectively.
In Vitro Platelet Aggregation Assay
Light transmittance platelet aggregometry is based on spectrophotometric monitoring of the turbid PRP sample after addition of various agonists. Agonist-induced platelet aggregation results in decreased turbidity and increased light transmittance. 21 The PRP was obtained freshly by centrifugation of whole citrated blood sample as mentioned previously. For PRP, the platelet count was performed to ensure that the platelet concentration was in the range of ∼250 × 109/L and was immediately used for the experiments. The PRP (480 µL) was incubated for 5 minutes at 37°C in the aggregation cuvette with different concentrations of samples and an aggregation agonist, and 5 μL of ADP or AA (final concentrations in the incubation media is 10 μmol/L for ADP and 0.5 mmol/L for AA, respectively) was added to the mixtures. The resulting aggregation was monitored and recorded for 6 minutes using the Dual-channel Lumi-aggregometer (Chrono Log 540-VS Platelet Aggregometer; Chrono-log Corporation) with constant stirring at 1000 rpm and expressed as the percentage change in light transmission. For each sample, the measurement was done in less than 1 hour after sampling. The PBS with 1% DMSO and ASA (final concentration 10 µg/mL) were used as negative and positive controls, respectively. The results were expressed as percentage of inhibition of agonist-induced platelet aggregation, which was calculated as follows: percentage inhibition (%) = [1 − (platelet aggregation of sample/platelet aggregation of control)] × 100%.
Statistical Analysis
All variables were tested for normality with the Kolmogorov-Smirnov test. Normally distributed continuous variables were expressed as means ± standard deviations of 3 measurements. Statistical analysis was performed by 1-way analysis of variance followed by the Dunnet test or Student t test as appropriate using GraphPad Prism version 5.0 (GraphPad Software, San Diego, California). P value <.05 was considered statistically significant.
Results
Anticoagulant Activities of AME, Coumarins, Flavonoids, and Its Metabolites
Figure 1 shows the activity of AME, hyuganin C, isolated flavonoids, and their colonic (DOPAC and p-HPAA) and glucuronide (quercetin, kaempferol, and luteolin glucuronides) metabolites on PT, APTT, and TT tests. The AME showed dose-dependent and significant (P < 0.001) prolongation of PT, APTT, and TT at concentrations of 5, 10, and 20 μg/mL. Among isolated coumarins, only hyuganin C, at 10 and 20 μmol/L, showed significant prolongation of only PT in a dose-dependent manner, while it had no activities on APTT or TT. Other isolated coumarins showed no anticoagulant activities on PT, APTT, or TT tests as shown in Table 1.
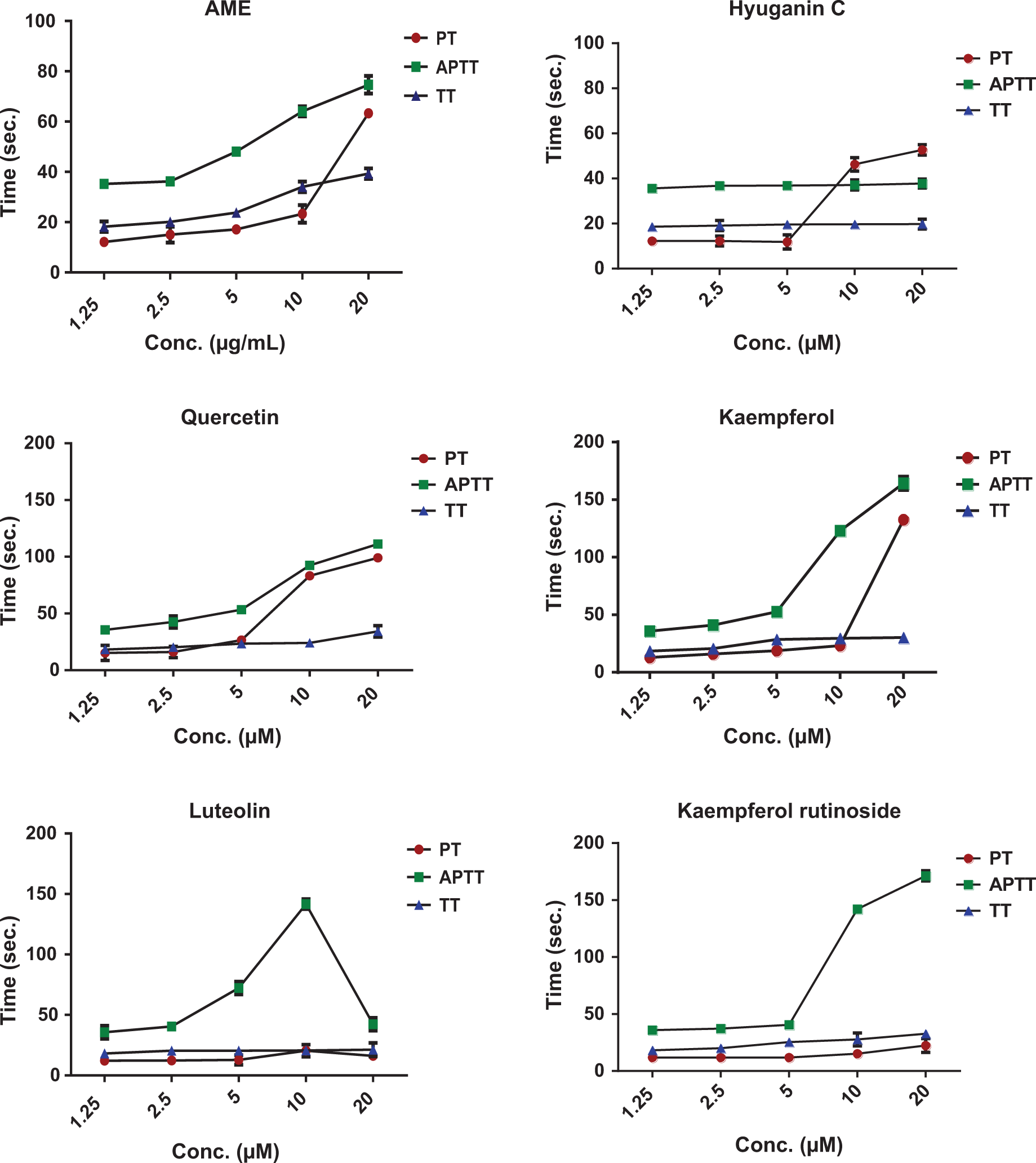
Anticoagulant activities of AME, active coumarin (hyuganin C), flavonoid compounds and their metabolites.
Anticoagulant Activity of Inactive Coumarins.a
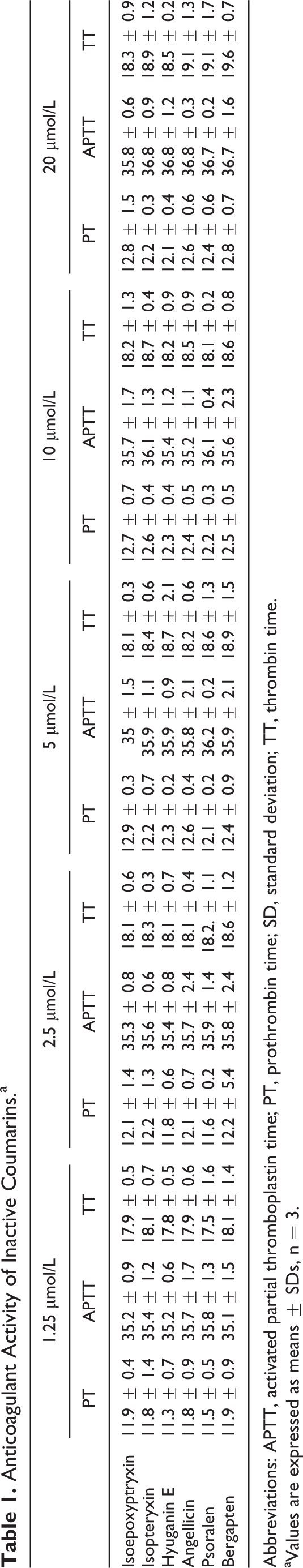
Abbreviations: APTT, activated partial thromboplastin time; PT, prothrombin time; SD, standard deviation; TT, thrombin time.
aValues are expressed as means ± SDs, n = 3.
Isolated flavonoids quercetin, kaempferol, kaempferol-3-O-glucoside, and kaempferol-3-O-rutinoside induced significant prolongation of PT, APTT, and TT at concentrations of 10 and 20 μmol/L. Kaempferol at a concentration of 20 μmol/L had the highest activity on PT prolongation (132.6 ± 3.01 seconds) compared to the normal PT control (12.08 ± 0.12 seconds). Kaempferol-3-O-rutinoside showed the strongest activity on APTT, which was prolonged to 171.2 ± 2.9 seconds at 20 μmol/L. Quercetin at 20 μmol/L concentration had the strongest activity on TT (34.2 ± 3.2 seconds), compared to the normal TT control (17.98 ± 0.11 seconds). Luteolin significantly prolonged PT only at 10 μmol/L to 20.42 ± 1.9 seconds while decreased its value to 16.16 ± 1.4 seconds at 20 μmol/L. Similarly, it prolonged APTT at 2.5, 5, and 10 μmol/L to 40.5 ± 2.3, 72.2 ± 3.4, and 141.6 ± 2.6 seconds, respectively, but at 20 μmol/L, APTT decreased to 42.24 ± 1.7 seconds.
To gain some insights into the activity of the active form of flavonoids in vivo, we studied the effect of colonic and glucuronide metabolites on PT, APTT, and TT. Kaempferol and quercetin glucuronides showed a sharp decrease in the activity of PT, APTT, and PT when compared with their parent compound’s activity. For example, at 20 μmol/L, glucuronidation decreased PT of quercetin and kaempferol by 74.74 ± 2.2 and 108.3 ± 0.8 seconds, respectively. Quercetin and kaempferol glucuronides showed similar effects on APTT and TT. At 10 μmol/L, glucuronidation of luteolin decreased PT and APTT by 6.92 ± 1.3 and 103.7 ± 4.5 seconds, respectively. Little effect was observed on TT of luteolin, which decreased only 2.4 ± 0.8 seconds by glucuronidation. Colonic metabolites showed similar or higher activities than their parent compounds. p-HPAA and DOPAC showed dose dependent and significant prolongation of PT, APTT, and TT at 2.5, 5, 10, and 20 μmol/L. Table 2 lists the values of PT, APTT, and TT of negative (pooled human plasma treated with sample vehicle) and positive controls.
The PT, APTT, and TT Values of Negative and Positive Controls in Anticoagulant Assays.a
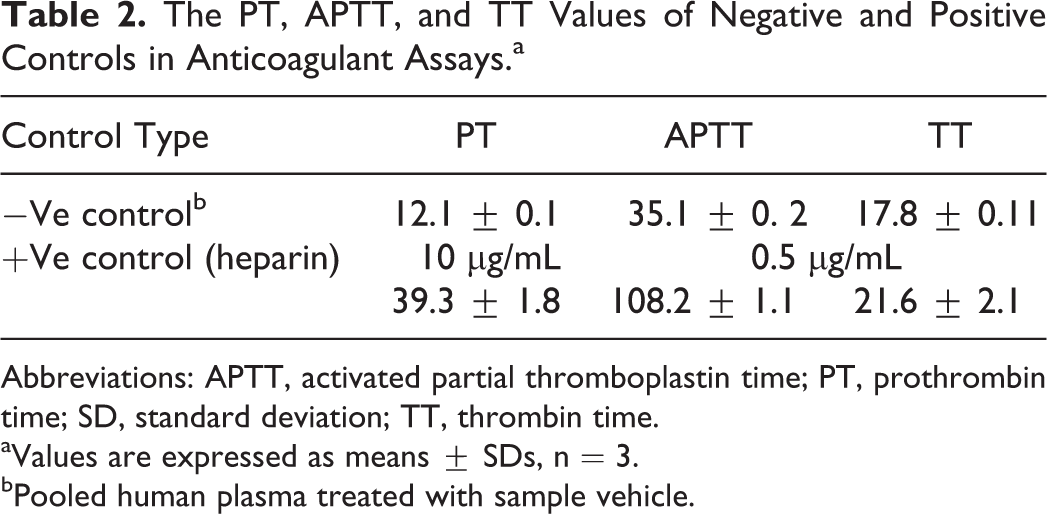
Abbreviations: APTT, activated partial thromboplastin time; PT, prothrombin time; SD, standard deviation; TT, thrombin time.
aValues are expressed as means ± SDs, n = 3.
bPooled human plasma treated with sample vehicle.
Antiplatelet Activities of AME, Coumarins, Flavonoids, and Its Metabolites
Table 3 demonstrates the percentages of the inhibitions of agonist-induced platelet aggregation by AME, its coumarins, flavonoids, and flavonoid metabolites. The AME inhibited platelet aggregation significantly against both ADP and AA at all tested concentrations in a dose-dependent manner. In addition, hyuganin C and bergapten from isolated coumarins showed preferential inhibition of ADP-induced platelet aggregation. The strongest activity was achieved by the flavonoid compounds. Quercetin and kaempferol inhibited platelet induced aggregation by both ADP and ADP at all tested concentrations in a dose-dependent manner. Luteolin, kaempferol glucoside, and rutinoside significantly inhibited the aggregation induced by ADP and AA at the higher concentrations of 5 and 10 μmol/L.
% Aggregation Inhibition of AME and Isolated Coumarins, Flavonoids, and Flavonoid Metabolite Against Agonist (ADP or AA)-Induced Platelet Aggregation.a,b
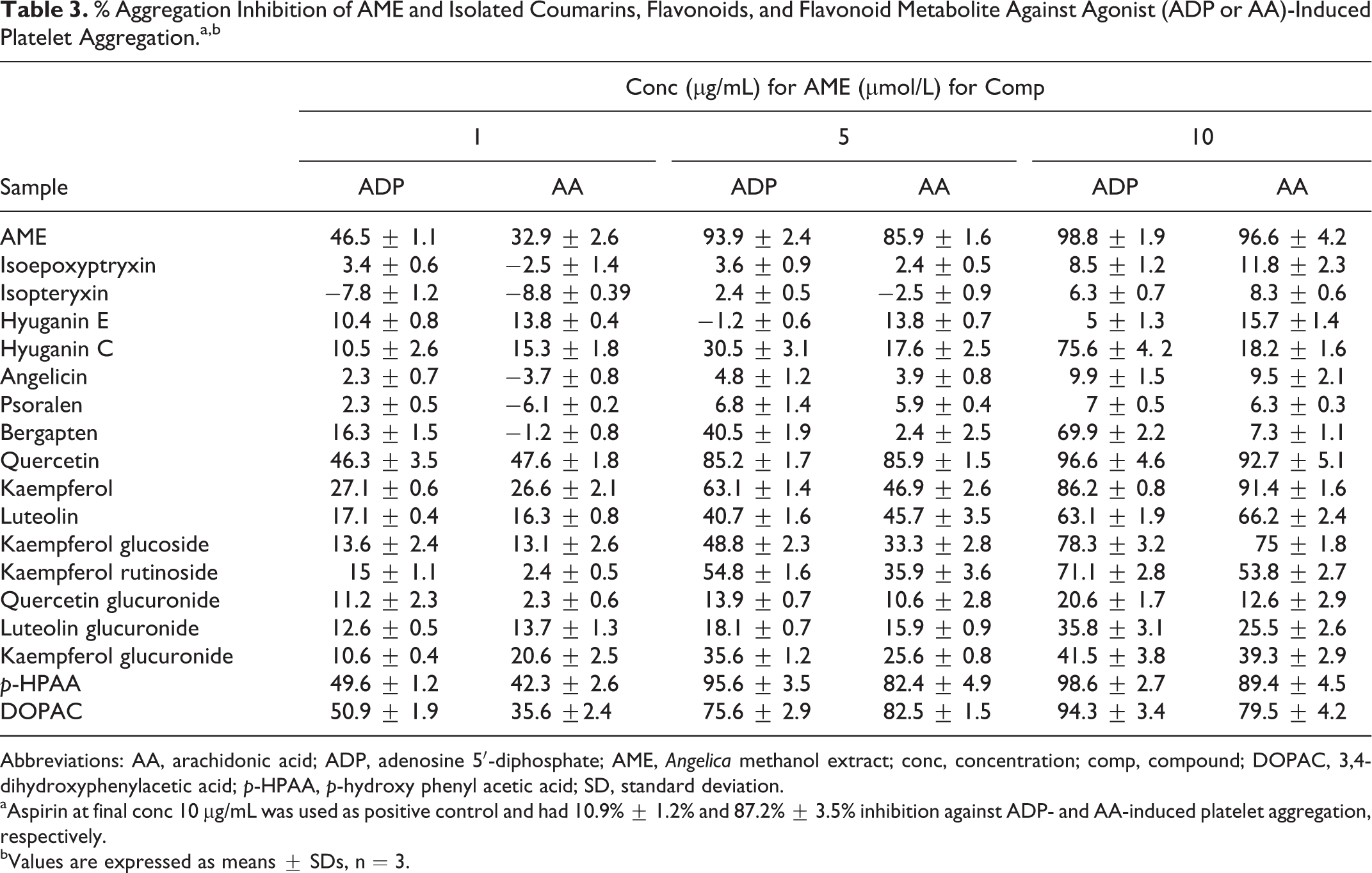
Abbreviations: AA, arachidonic acid; ADP, adenosine 5′-diphosphate; AME, Angelica methanol extract; conc, concentration; comp, compound; DOPAC, 3,4-dihydroxyphenylacetic acid; p-HPAA, p-hydroxy phenyl acetic acid; SD, standard deviation.
aAspirin at final conc 10 µg/mL was used as positive control and had 10.9% ± 1.2% and 87.2% ± 3.5% inhibition against ADP- and AA-induced platelet aggregation, respectively.
bValues are expressed as means ± SDs, n = 3.
Glucuronidation of flavonoid aglycones had different degrees of decreasing the inhibitory activity of the parent compounds. Quercetin was the most affected aglycon by glucuronidation. Quercetin-3-O-glucuronide showed about 5 and 7 times decrease in the inhibitory activity of quercetin against ADP- and AA-induced aggregation, respectively, at 10 μmol/L and did not show any significant inhibition at all tested concentrations. However, kaempferol-3-O-glucuronide showed only 1.8 to 2.3 times decrease in the inhibitory activity of kaempferol and showed significant inhibition of platelet aggregation induced by both ADP and AA at 5 and 10 μmol/L concentration. Similarly, the inhibitory activity of luteolin was decreased by glucuronidation by 1.5 and 2.6 times against ADP- and AA-induced aggregation, respectively, and induced only significant activity at 10 μmol/L.
The activity of colonic metabolites, DOPAC, and p-HPAA was comparable to their parent aglycones. Both metabolites showed significant inhibitory activity at all tested concentrations. Moreover, at a concentration of 5 and 10 μmol/L, p-HPAA showed stronger inhibitory activity against ADP-induced platelet aggregation than the most active aglycone, quercetin.
Discussion
Angelica shikokiana is commonly used in Japanese traditional medicine. One of its traditional indications is cardiovascular disease. However, its possible association has not been elucidated yet. A previous study showed that the coumarins isolated from A shikokiana had antihyperlipidemic effects by enhancing adrenaline-induced lipolysis in fat cells. However, the effects of A shikokiana on the hemostatic system have not been reported. So, the present study was designed to investigate the anticoagulant and antiplatelet activities of the methanol extract of A shikokiana as well as its isolated compounds to find a clue to its traditional use for cardiovascular diseases.
Hemostasis includes two succeeding processes: platelet aggregation and activation of the coagulation cascade. 1,2 Accordingly, the screening of platelet and the coagulation activation may be a useful tool to explore antithrombotic agents. 7,9 In this study, anticoagulant assays showed that AME could significantly prolong coagulation parameters, PT, APTT and TT, indicating its ability to inhibit both extrinsic, intrinsic, and the common pathways of the coagulation cascades. Moreover, our results revealed that the active compounds responsible for these activities may be the flavonoid compounds, which showed stronger activities than coumarins for all anticoagulant assays. All tested flavonoids showed a dose-dependent increase in PT, APTT, and TT except luteolin which at 20 μmol/L strongly decreased PT and APTT after increasing their times at 10 μmol/L. A possible explanation is that while luteolin exhibits anticoagulant effects at low concentration, it may stimulate some prothrombotic mechanisms at higher concentrations.
The prolongation of PT, APTT, and TT by quercetin may be related to direct competitive thrombin inhibition 22 and the inhibition of coagulation factor X by binding to its active sites. 23 Kaempferol-3-O-rutinoside showed the strongest and the highest activity on APTT, indicating its ability to inhibit the coagulation factors in the intrinsic pathway preferentially.
Although common coumarins are widely used as anticoagulants as warfarin and dicoumarol, 24,25 we identified only hyuganin C, among the isolated coumarins, to possess an anticoagulant activity evidenced by significant prolongation of PT. The inactivity of other coumarins may be due to lacking the minimal structural requirement for the anticoagulant activity, 4-hydroxy-3-substituted coumarins ring. 26 This is the first report of the anticoagulant activity of hyuganin C, and its mechanism of action needs further evaluation.
Platelets can be activated by some agonists such as ADP, AA, and thrombin. This induces intracellular signaling by activating multiple G protein-mediated pathways to activate platelet shape change, degranulation, aggregation, and subsequent thrombus formation. 27,28 In this study, AME could potently inhibit platelets aggregates induced by either ADP or AA in a dose-dependent manner. Our results showed that the activity related mainly to flavonol compounds, quercetin and kaempferol, that showed higher activities than other flavonoids and the active coumarins, hyuganin C and bergapten. Further, the antiplatelet activity of quercetin and kaempferol is stronger than that of luteolin and kaempferol glycosides that may implicate a significant role for the presence of free C3-OH in the antiplatelet activity.
The ADP binds to two G-protein-coupled receptors: P2Y1 and P2Y12. Binding of ADP to the P2Y1 receptor induces shape change and initiates primary wave platelet aggregation through calcium mobilization. 29,30 The P2Y12 receptor is considered to be the major ADP receptor and responsible for full platelet aggregation through the inhibition of adenyl cyclase. 31
On the other side, AA is the precursor of thromboxane A2 (TXA2) within platelets. Arachidonic acid is converted to TXA2 by cyclooxygenase and thromboxane synthase. 32 The TXA2 is a potent inducer of platelet aggregation causing granule release, more TXA2 generation, and then sustained glycoprotein (GP) IIb-IIIa activation. 33
The antiplatelet activity of quercetin may be related to the inhibition of phosphoinositide-3-kinase (PI3K)/Akt signal transduction and Ca2+ ions mobilization 34 and the inhibition of agonist-induced GPIIb/IIIa activation and P-selectin expression, which attenuates the platelet granule exocytosis. 35 In addition, the strong antioxidant activity of quercetin and kaempferol may account for indirect inhibitory activity on platelet aggregation through scavenging reactive oxygen species (ROS). The ROS stimulate the inhibition of the protein tyrosine kinases, which stimulate phosphorylation of some proteins such as SyK and FaK family proteins that induce the initial signal of platelets aggregation. 36,37 Moreover, quercetin inhibits the platelet TxA(2) signaling pathway and antagonize TxA(2) receptors (TP) in platelets through impairment of platelet tyrosine phosphorylation and extracellular signal-regulated kinase 1/2 activation. 38
After flavonoid-rich diet intake, only flavonoid metabolites could be detected in human plasma or urine due to extensive metabolism. 39 However, most of the reported biological activities were contributed to the parent flavonoid compounds. Thus, we aimed to evaluate the activities of the available metabolites of flavonoid compounds that exhibited higher activities as anticoagulant and antiplatelet than coumarins to reflect the actual effectiveness inside the body. Colonic metabolites, DOPAC and p-HPAA, had similar activities to their parent compounds, whereas glucuronide conjugates showed a sharp decrease in both activities compared to the parent compounds.
Conclusion
This study is the first—to the best of our knowledge—to report the anticoagulant and antiplatelet activities of A shikokiana extract and its bioactive compounds. The methanol extract of the aerial part of A shikokiana exhibited potent anticoagulant and antiplatelet activities. Moreover, the isolated flavonoid compounds showed higher anticoagulant and antiplatelet activities than the isolated coumarins. Among coumarin compounds, hyuganic C had a moderate anticoagulant activity while both of bergapten and hyuganin C compounds exhibited moderate antiplatelet activities against ADP-induced platelet aggregation. Further studies of the possible in vivo activities, as well as, the specific mechanisms of action are warranted that, ultimately, may open the door for the search for new anticoagulant or antiplatelet drugs.
Footnotes
Authors’ Note
All authors contributed equally to this work.
Acknowledgments
We would like to thank Joy Plus (Y. Tateyama), Fukuoka, Japan, for offering the plant powder.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The project was partly supported by the Department of Clinical Pathology, Mansoura Faculty of Medicine, Mansoura University, Egypt.
