Abstract
Fondaparinux (FPX), a synthesized factor Xa inhibitor, is one of the most popular anticoagulants for the prevention of postoperative venous thromboembolism (VTE). Although routine monitoring is not required, the bleeding adverse events cannot be neglected, and the measurement of anti-Xa activity is expected to be monitored. The primary purpose of this study is to evaluate the performances of 2 chromogenic assays for the detection of anti-Xa activity. Furthermore, the pharmacokinetics of FPX was examined using chromogenic assays. Anti-Xa activity was measured using 2 FPX-based chromogenic substrates (S2222 and STA-Liquid Anti-Xa). The reproducibility, detection limits, linearity, and correlations between the substrates were examined using normal plasma doped with low and high concentrations of FPX formulation. In addition, anti-Xa activity in 235 clinical samples from 164 cases treated was measured, and the pharmacokinetics of FPX was evaluated. Both of the tested substrates were capable of accurately measuring the anti-Xa activity of FPX, with a lower limit of 0.05 μg/mL and a coefficient of variation of less than 10%. The repeated administration of FPX induced a gradual but significant increase in the anti-Xa activity, which was negatively correlated with body weight and estimated glomerular filtration rate. No significant correlation between the anti-Xa activity and the occurrence of postoperative VTE or bleeding event was observed. Anti-Xa activity can be successfully determined using 2 chromogenic assays and automated biochemical analyzers. The clinical significance of anti-Xa activity monitoring should be examined in the future study.
Introduction
Venous thromboembolism (VTE) is a common cause of morbidity and mortality among patients undergoing surgery. 1 Fondaparinux (FPX), a synthetic polysaccharide parenteral anticoagulant, is one of the most popular agents used for the prevention of VTE. 2 Fondaparinux is administered subcutaneously in fixed doses and catalyzes the antithrombin-mediated inhibition of factor Xa. 3 Phase II clinical studies have identified a subcutaneous dose of 2.5 mg once daily for prophylaxis of VTE in patients undergoing major orthopedic surgery. 4 Since pharmacologic thromboprophylaxis requires a dual challenge, that is, the prevention of potentially life-threatening thromboembolism and the avoidance of bleeding complications, the accurate evaluation of the activity is preferable. For that purpose, anti-Xa activity is developed and utilized. 5 It is generally accepted that anti-Xa activity is specific for FPX in comparison with the conventional coagulation tests such as prothrombin time (PT), 6,7 and the plasma anti-Xa activities are reported to be equivalent to the gravimetric units of FPX. 8 However, the reproducibility, linearity, and correlations between the substrates and the equipment have not been examined. Therefore, the primary objective of this study was to examine the reliability of anti-Xa activity assay using the different substrates and equipment.
The second purpose of this study is to examine the kinetics of FPX in the clinical setting. Although the routine monitoring of FPX is not required, 9 considerable caution has been advocated, 10 since FPX has a long half-life (17-21 hours) 11 and no antidote available. 12 Fondaparinux is mainly excreted by the kidney and the reduced dose is recommended for the patients with a substantial reduction of renal function. 13,14 However, the bleeding adverse events caused by suspected overdose have repeatedly been reported. 15 Therefore, we tried to examine the usefulness of the monitoring to perform the preventive therapy more effectively and safely.
Materials and Methods
Reproducibility and Linearity of the Assay Procedures
Reproducibilities between-run and within-run of Coapresta 2000 (CP2000, Sekisui Medical, Tokyo, Japan) and STA-R (Roche Diagnostics, Basel, Switzerland) were analyzed using normal plasma doped with low and high concentrations of FPX formulation. To examine the within-run reproducibility, the measurements were repeated 10 times for 3 concentrations of FPX in each analyzer. Linearity of the measurements and the detection limit were examined in the samples of the FPX formulation, with decreasing concentrations prepared using control normal plasma in a stepwise dilution (1:10, 2:10, 3:10, 4:10, 5:10, 6:10, 7:10, 8:10, 9:10, and 10:10).
Assay Procedures
The chromogenic anti-Xa assay is based on the inhibitory action of FPX on Xa, which specifically cleaves para-nitroaniline (p-NA) linked to a chromogenic peptide. Increasing FPX concentrations inhibit the activity of factor Xa on the chromogenic peptide, thereby increasing the release of p-NA. Increasing FPX concentrations inhibit the activity of factor Xa on the chromogenic peptide, thereby increasing the release of p-NA [7]. The concentration of FPX is plotted against the optical density (OD) of the released p-NA, and anti-Xa activity were expressed in μg/mL. The above anti-Xa activity assay for FPX was performed using 2 FPX-based chromogenic substrates, S2222 (Sekisui Medical, Tokyo, Japan) and STA-Liquid Anti-Xa (Roche Diagnostics). CP2000 and STA-R were utilized as analyzers.
Other coagulation markers including PT, activated partial thromboplastin time (aPTT), fibrin/fibrinogen degradation products (FDP), D-dimer (DD), and fibrinogen (Fib) were measured in the same samples. Blood cell counts, estimated glomerular filtration rate (eGFR), and transaminases were also determined in the hospital laboratory.
Sample Collection
Between April 2011 to May 2012, 235 blood samples were collected from 164 patients scheduled to receive subcutaneous FPX as a prophylactic for VTE after major surgery. The postsurgical subcutaneous administration of FPX was performed as follows: 2.5 mg/body of FPX was administered daily for 5 days for the patients who had undergone lower intestinal surgery (n = 78), 1.5 mg/body was administered daily for 8 to 10 days for orthopedics patients (n = 64), and 1.5 mg/body was administered daily for 3 to 5 days for obstetrics/gynecology patients (n = 22). The patient demographics, such as age, sex, body weight, and body mass index (BMI), were obtained from the patients’ record. The study protocol was approved by the ethics committee of Juntendo University Hospital, and written informed consent was taken from all the participants.
Blood samples were obtained from the peripheral veins before the subcutaneous injection of FPX. For patients undergoing lower intestinal surgery, peripheral blood was collected at 17 hours after FPX administration on postoperative days 0, 3, 5, and 6. For the orthopedics and obstetrics/gynecology patients, peripheral blood was collected at 1 and 49 hours after the last administration and at 10, 21, and 69 hours after the last administration, respectively.
The patients were screened for deep vein thrombosis (DVT) using standard Doppler ultrasonography. 1 A diagnosis of DVT required confirmation of the presence of venous thrombosis using compression ultrasonography.
Statistical Analysis
The differences between the groups were examined using the Student t test or the Welch t test. The statistical analysis was performed using and StatFlex ver. 6.0 software (Artech Co., Ltd, Osaka, Japan). A P value of less than .05 was considered to be statistically significant.
Results
Reproducibility and Linearity
The coefficients of variation (CVs) were calculated for each component (Table 1). Both CP2000 and STA-R had CVs of less than 10% for between-run reproducibility examined on 18 separate days. As for the within-run reproducibility, STA-R was found to have a better performance than the CP2000 (CV of less than 5% for STA-R vs around 10% for CP2000).
Reproducibility of CP2000 and STA-R.
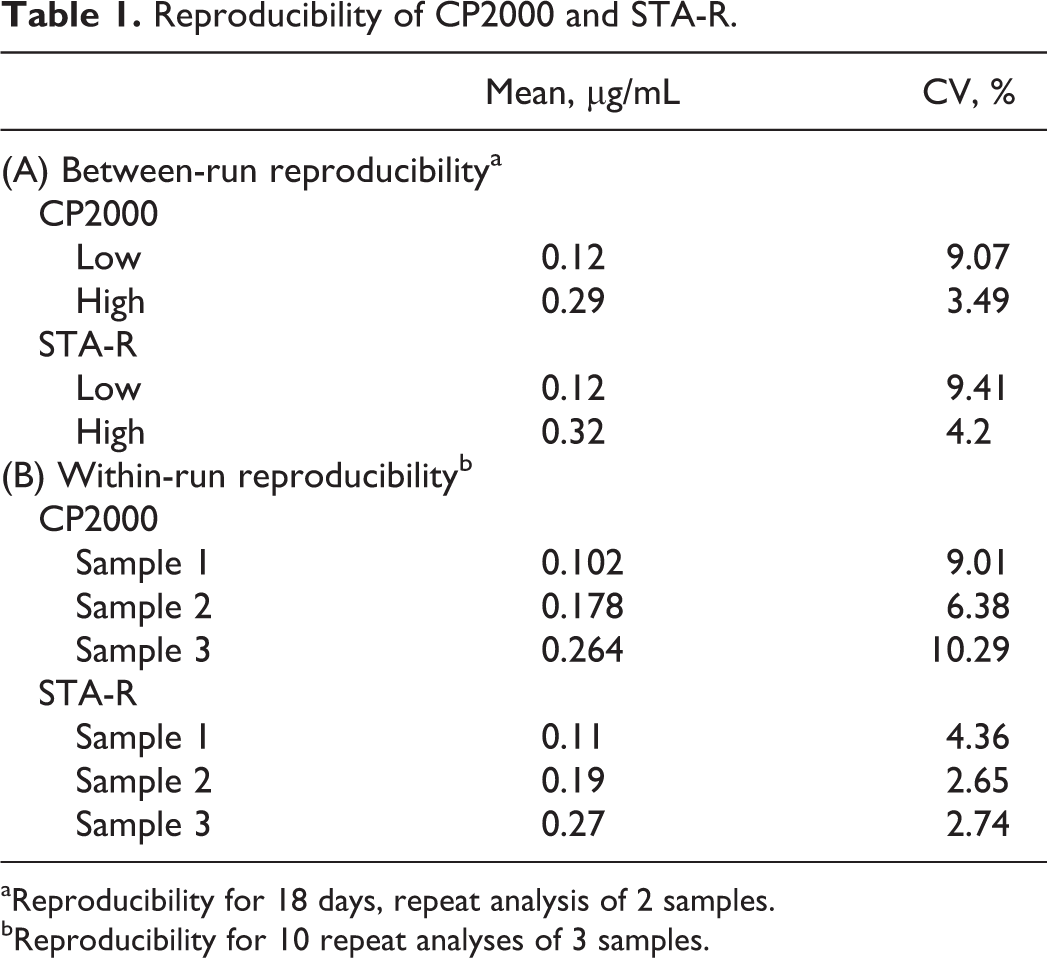
aReproducibility for 18 days, repeat analysis of 2 samples.
bReproducibility for 10 repeat analyses of 3 samples.
The linearity up to 2.06 μg/mL for the CP2000 assay and up to 1.25 μg/mL for the STA-R assay was recognized (Figure 1). The detection limits of the assays using CP2000 and STA-R were 0.042 μg/mL (CV, 19.1%) and 0.030 μg/mL (CV, 15.3%), respectively.
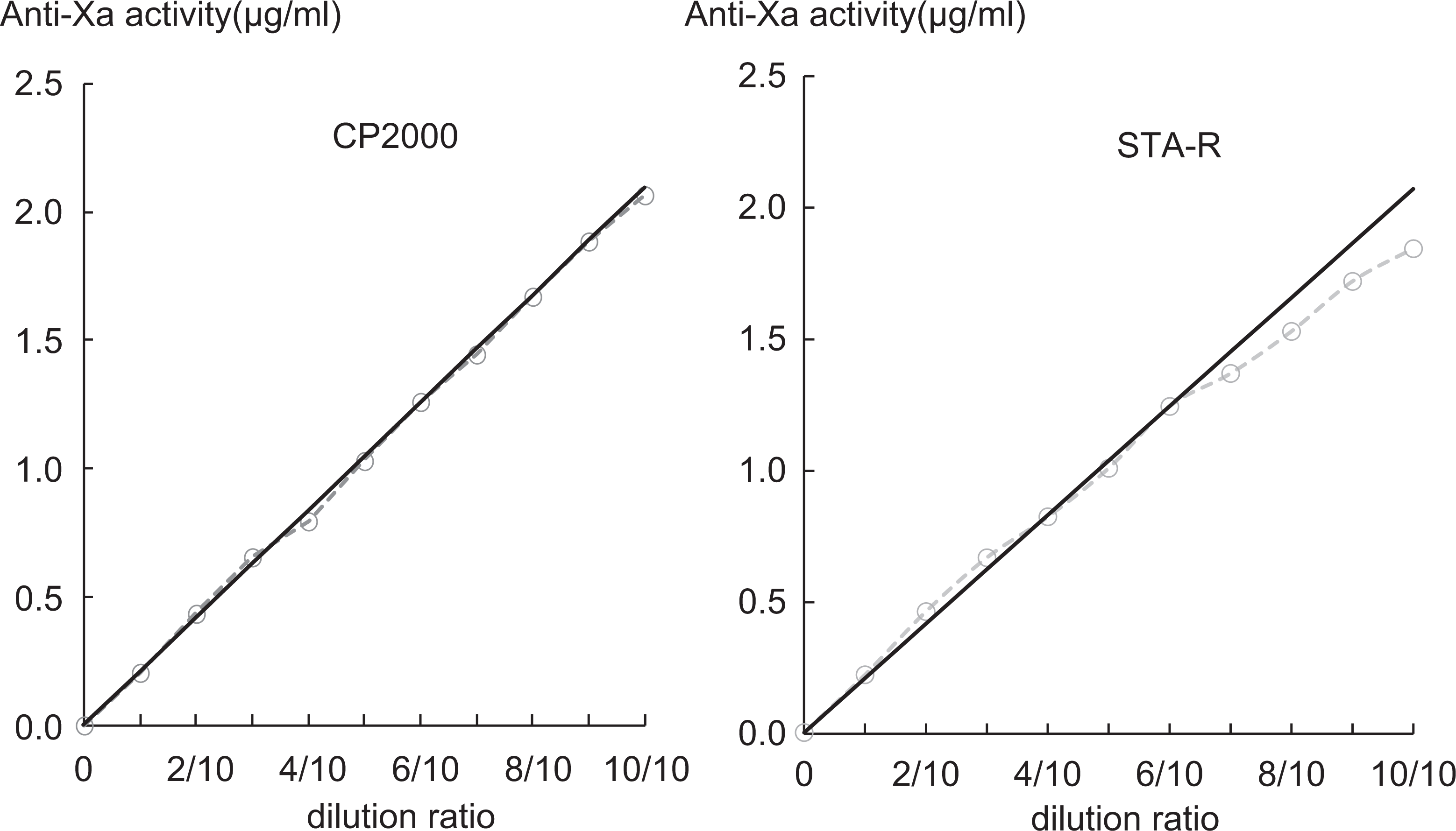
Linearity measurements for CP2000 and STA-R assays. Samples were prepared by diluting with control normal plasma in a stepwise dilution (1:10, 2:10, 3:10, 4:10, 5:10, 6:10, 7:10, 8:10, 9:10, and 10:10). Linearity was determined using duplicate specimens.
The correlation of the measurement results for anti-Xa activities using the CP2000 and STA-R assays was assessed using 235 clinical samples, and a good correlation was detected (y = 0.98x + 0.01, r = .80; P < 0.05; Figure 2).
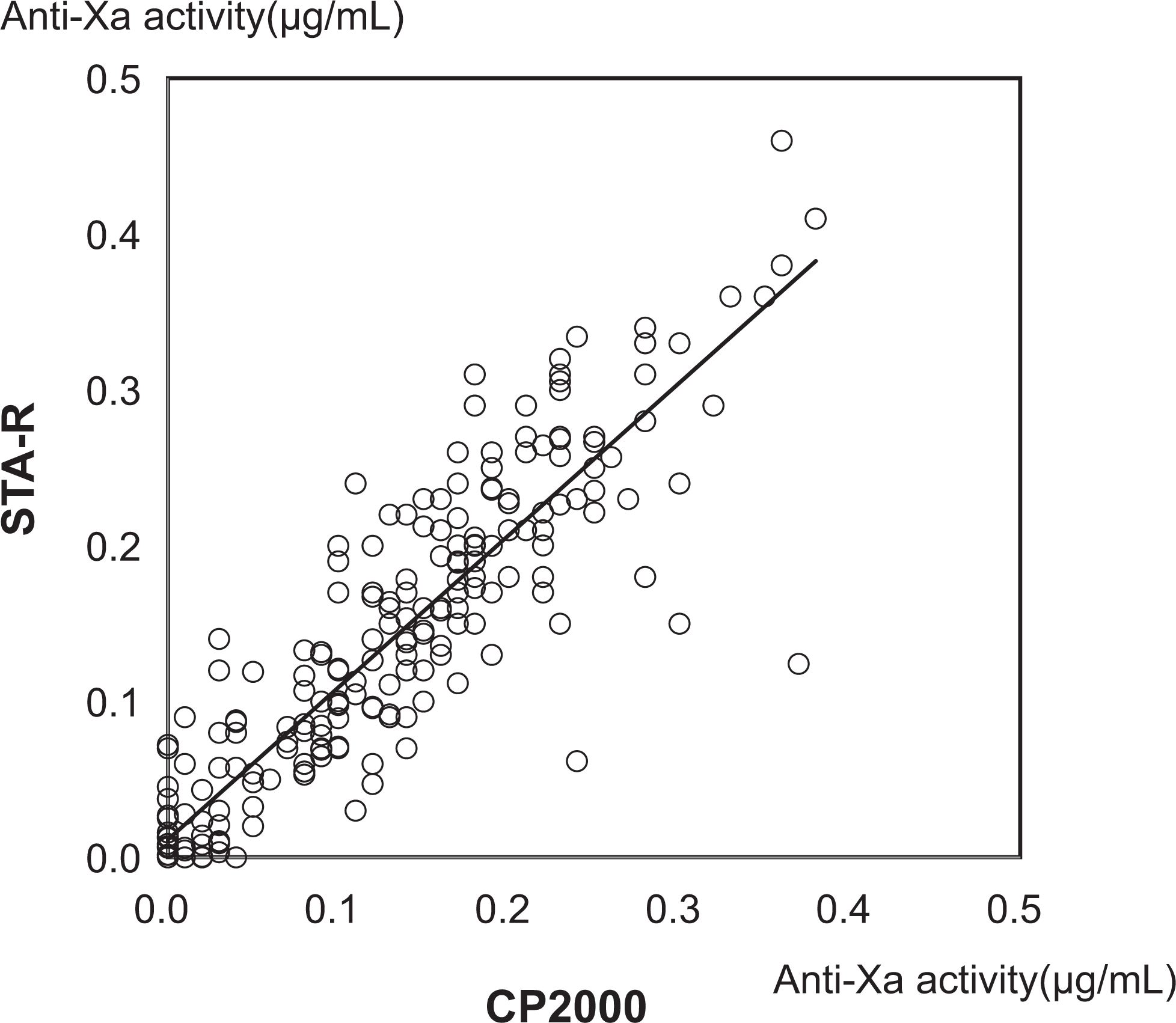
Correlation between anti-Xa activity values measured using the CP2000 and STA-R assays. The anti-Xa activity values of 235 samples from 164 patients treated with 2.5 mg or 1.5 mg of fondaparinux (FPX) daily were measured using the CP2000 and STA-R assays.
Anti-Xa Activity
The overall anti-Xa activity of FPX for the 78 patients who received repeated subcutaneous administrations of FPX (2.5 mg daily) is shown in Figure 3. A gradual but significant increase in anti-Xa activity was detected from days 0 to 5, followed by a decrease at 48 hours after the last FPX administration (day 6). A gradual increase in anti-Xa activity with time was also observed in postoperative orthopedics, obstetrics, and gynecology patients who received the continuous administration of FPX (1.5 mg daily; data not shown).
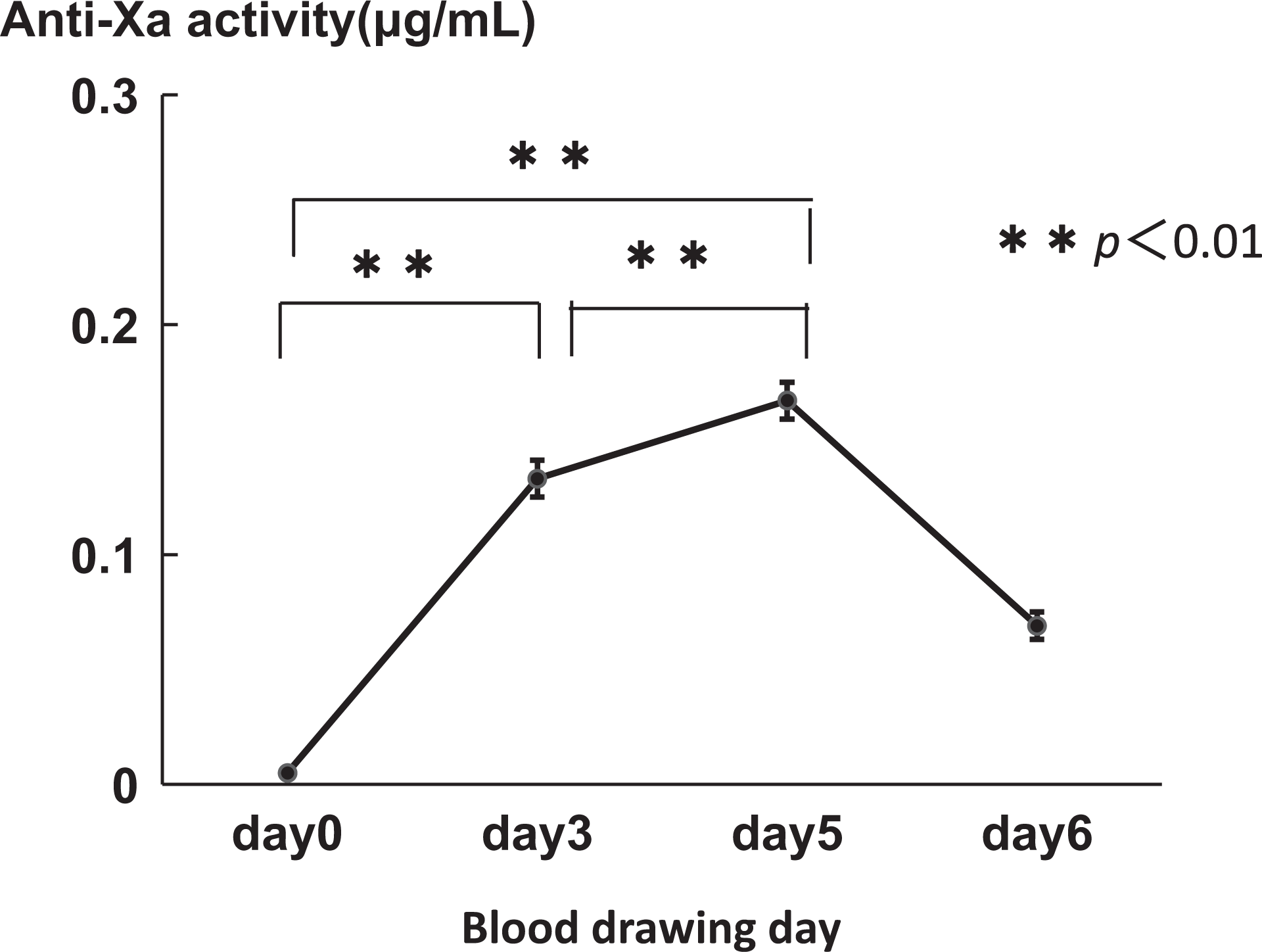
Anti-Xa activity in patients who received repeated administrations of fondaparinux (FPX). The plasma levels of the anti-Xa activity in 78 patients who received subcutaneous FPX (2.5 mg daily) after undergoing lower intestinal surgery were measured preoperatively (day 0) and postoperatively (days 3, 5, and 6). The graphs show the mean ± standard error of the mean (SEM).
The anti-Xa activity and body weight in 78 patients who received the repeated administration of FPX (2.5 mg daily) following lower intestinal surgery revealed a negative correlation (y = −0.0015x + 0.2247, r = −.352; P < .01; Figure 4A). The negative correlations between anti-Xa activity and BMI, FDP, DD, and eGFR were also observed (BMI: y = −0.0045x + 0.2358, r = −.245, P < .01; FDP: y = −0.0052x + 0.2090, r = −.416, P < .01; DD: y = −10.8710x + 4.7297, r = −.372, P < .01; eGFR: y −97.9485x + 102.1346, r = −.436, P < 0.01; Figure 4B). However, a univariate analysis indicated that age, sex, PT, aPTT, Fib, WBC, RBC, Hb, PLT, AST, and ALT were not correlated with anti-Xa activity (Table 2).
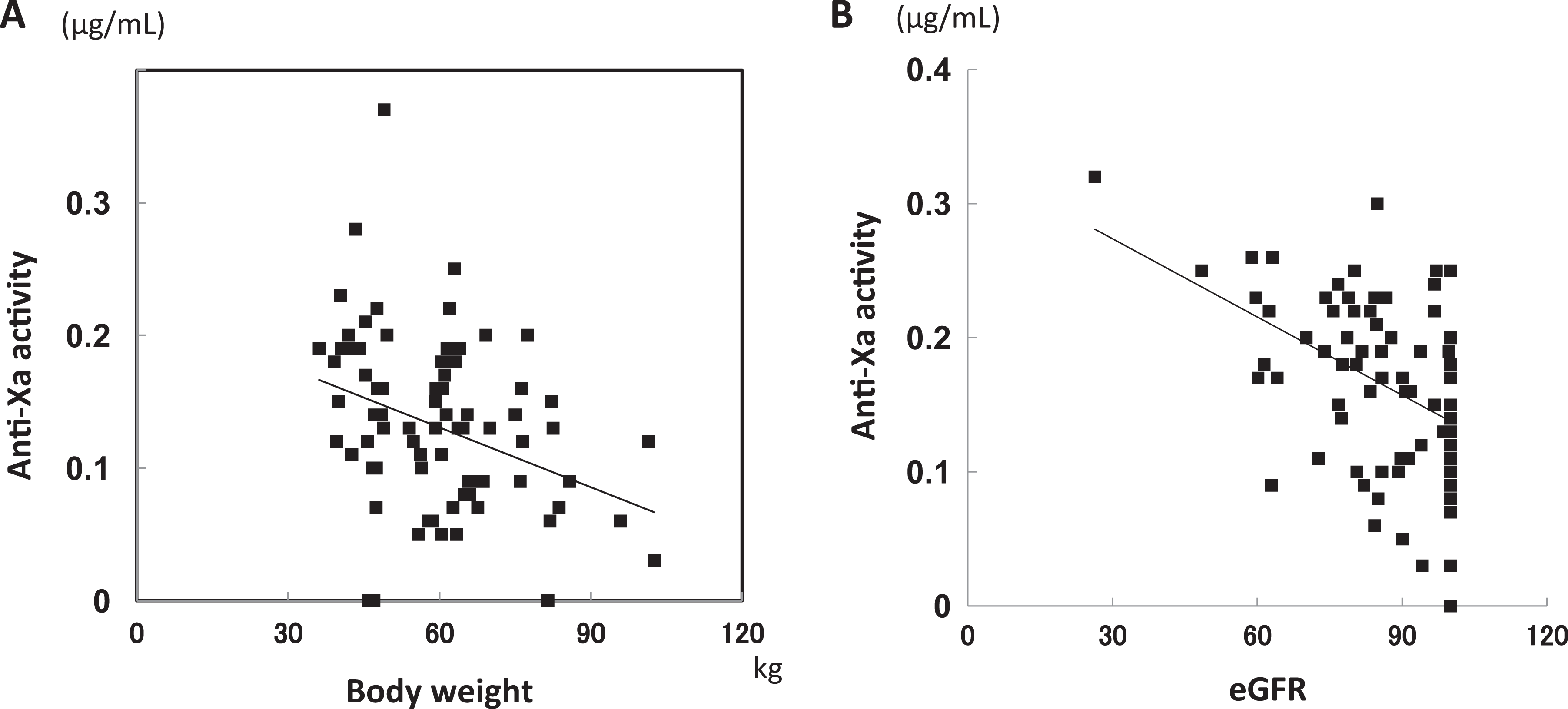
Correlation between anti-Xa activity and body weight (A) and estimated glomerular filtration rate (eGFR; B). The plasma levels of anti-Xa activity in 78 patients who received subcutaneous fondaparinux (FPX; 2.5 mg daily) after undergoing lower intestinal surgery were measured postoperatively (day 3).
Clinical Characteristics, Clinical Test Data, and Correlation With Anti-Xa Activity.a
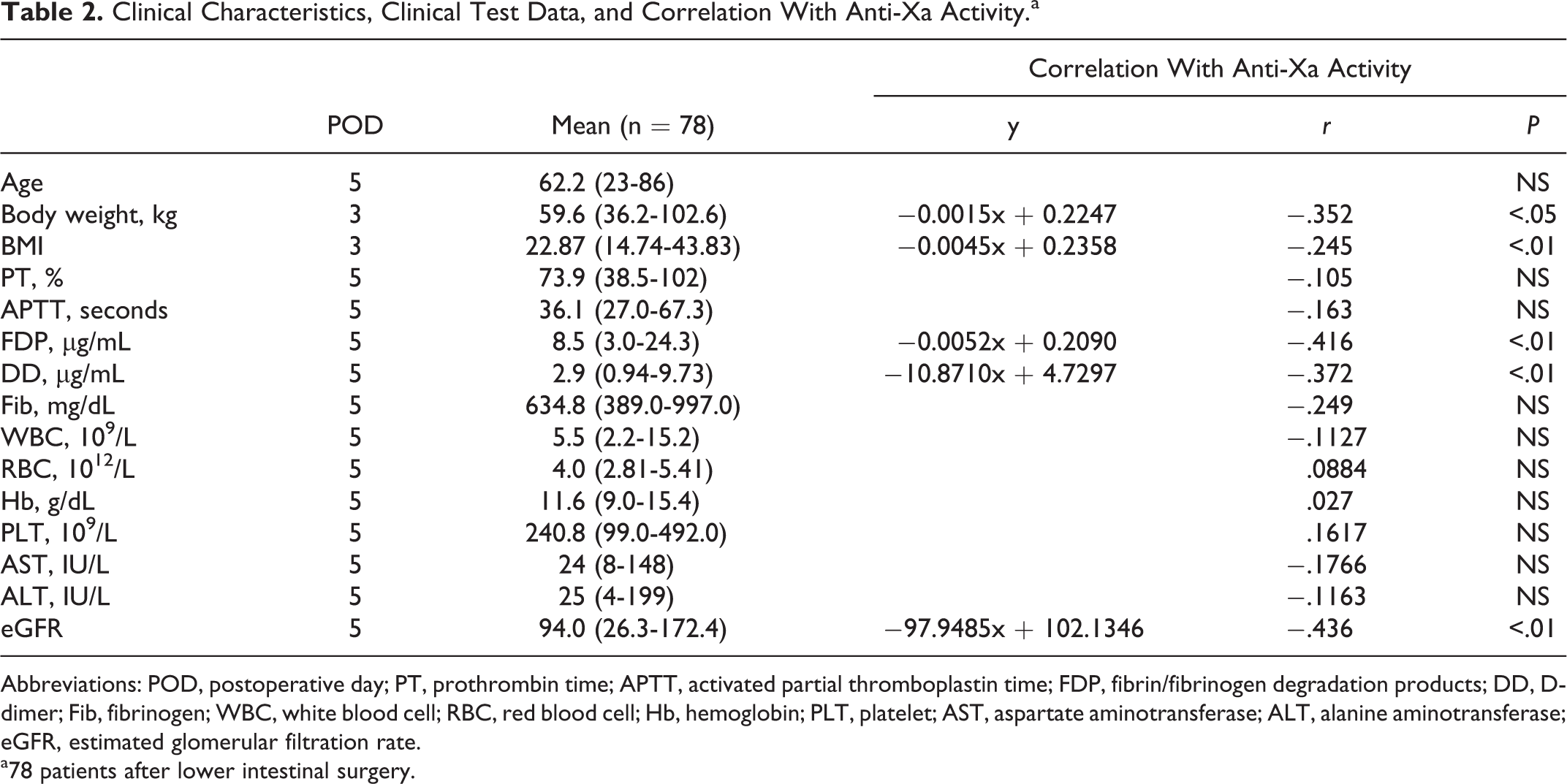
Abbreviations: POD, postoperative day; PT, prothrombin time; APTT, activated partial thromboplastin time; FDP, fibrin/fibrinogen degradation products; DD, D-dimer; Fib, fibrinogen; WBC, white blood cell; RBC, red blood cell; Hb, hemoglobin; PLT, platelet; AST, aspartate aminotransferase; ALT, alanine aminotransferase; eGFR, estimated glomerular filtration rate.
a78 patients after lower intestinal surgery.
Overall, 11.8% of the orthopedics patients who received 1.5 mg of FPX daily exhibited postoperative bleeding, and 11.5% of them experienced VTE during the observation period. Postoperative bleeding was also observed in 10.3% of the patients who underwent lower intestinal surgery and received 2.5 mg of FPX daily, included melena (n = 1), intraperitoneal bleeding (n = 2), bleeding from surgical wounds (n = 4), acute subdural hemorrhage (n = 1), and genital bleeding (n = 1). No significant correlation between the anti-Xa activity and the occurrence of postoperative bleeding was seen (Figure 5). No bleeding incidence of obstetrics/gynecology patients who received postsurgical FPX (1.5 mg daily, n = 22) was observed.
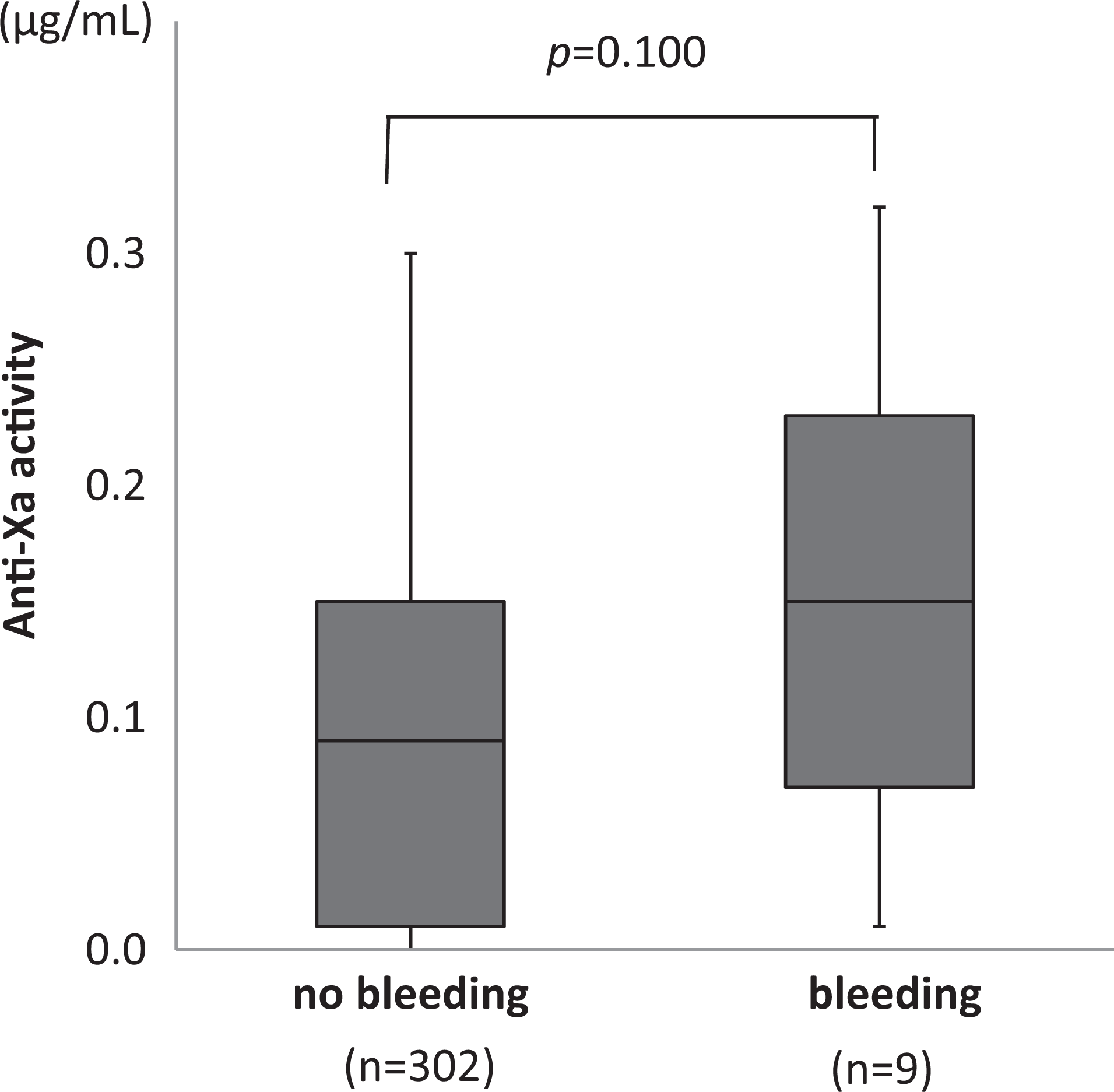
Box-and-whisker plots of the anti-Xa levels for the postoperative patients with no bleeding and bleeding incidence. Samples from 78 patients who received subcutaneous FPX (2.5 mg daily) after undergoing lower intestinal surgery were measured preoperatively (day 0) and postoperatively (days 3, 5 and 6).
Discussion
Mismetti and Laporte 13 noted 5 points regarding the necessity and reliability of the monitoring for the new anticoagulants. When there was the (1) existence of inter-individual variability (ie, variability among patients) and (2) existence of the intra-individual variability (ie, variability within the same patient), the monitoring is expected. However, it was also noted that the following 3 conditions were necessary: (3) existence of the reliable assay methods (ie, variability of the assay method is low and reproducible), (4) good correlation between the drug concentration and clinical events (efficacy and safety end points), and (5) validation (ie, useful for the decision of dose increase or decrease). To clarify the clinical usefulness of FPX laboratory monitoring, we evaluated the “assay method” using 2 commercially available chromogenic assays to determine the inhibitory activity of FPX toward factor Xa. In the next step, we assessed the “intra-individual variability,” the “inter-individual variability,” and the “correlations between the drug concentration and clinical events” in the present study.
To our knowledge, this is the first report that compares the reproducibilities and linearity of anti-Xa activities measured by 2 different substrates. The results indicate that both of the FPX-based chromogenic substrates, S2222 and STA-Liquid Anti-Xa, were capable of accurately measuring the anti-Xa activity induced by FPX, enabling the plasma concentration to be determined with a lower limit of 0.05 μg/mL. These quantitative assays were also acceptable in terms of the between-run and within-run reproducibilities, linearity, and the correlation between the tested methods, which had CVs of less than 10%. The above results confirmed that both assays were capable of reproducibly measuring the anti-Xa activity within a clinically relevant range. 16,17
As for the intra-individual variability, patients who underwent lower intestinal surgery and received 2.5 mg of FPX daily demonstrated a gradual but significant increase in anti-Xa activity. In terms of inter-individual variability, the plasma levels of anti-Xa activity showed a significant negative correlation with body weight and with BMI. We further observed a negative correlation between anti-Xa activity and eGFR, indicating the accumulation of FPX in patients with renal impairment, this observation agreed with the results of previous reports. 2,3 Thus, we think that FPX monitoring based on anti-Xa activity could provide a clinically effective means of determining the optimal dose regimen for patients with renal impairment and/or low-body weight individuals. Meanwhile, the anti-Xa activity was not affected by age, sex, cell blood counts, and liver function. Regarding coagulation tests, though FDP and DD were negatively correlated to anti-Xa activity, both PT and aPTT were not as reported before. 18
With regard to the relevance to the clinical events, some studies have reported that there were no differences in the anti-Xa activity in patients with and without postoperative VTE or bleeding. 14,16,17 Similarly, no significant associations between anti-Xa activity and the occurrence of postoperative bleeding or VTE were recognized in our study. This finding can probably be explained by the fact that thrombotic events and adverse events are induced by multiple factors, and the anti-Xa activity is just one of them. In addition, since the samples were collected at a low point in the concentration profile, they might not have been linked to the adverse events. 19 At any rate, a single test of anti-Xa activity would not be sufficient to predict such clinical symptoms.
On the other hand, the results from other laboratories have indicated the potential benefit of the monitoring in patients with a risk of bleeding, such as those with renal dysfunction or a low body weight. 7,12,14,20 Further validation of the benefits of anti-Xa activity monitoring in these patients is required.
The present study had several limitations. First, the patients’ background was heterogeneous and the interventions performed were not uniform. Second, only the trough levels were measured, and the peak levels were not evaluated.
A clinical interest in identifying reliable and readily available laboratory assays for measuring anti-Xa activity is not limited to studies on FPX. The knowledge reported in this study may also be applicable to novel oral Xa inhibitors. 12,21,22 Since the interassay variability observed with different reagents can reportedly be reduced by mathematical modeling, 23 the standardization of anti-Xa assays across reagents and methods can be expected.
In summary, the present investigation demonstrated that anti-Xa monitoring using chromogenic assay is simple, reproducible, and reliable. In addition, the assay procedure of pharmacodynamic parameters for FPX by automated biochemical analyzers is feasible.
Footnotes
Acknowledgments
The authors wish to thank Dr Mayumi Idei for statistical support.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
