Abstract
Low molecular weight heparins (LMWH) represent depolymerized heparin prepared by various methods that exhibit differential, biochemical and pharmacological profiles. Enoxaparin is prepared by benzylation followed by alkaline depolymerization of porcine heparin. Upon the expiration of its patent, several biosimilar versions of enoxaparin have become available. Heparinox (Sodic enoxaparine; Cristália Produtos Químicos Farmacêuticos LTDA, Sao Paulo, Brazil) is a new biosimilar form of enoxaparin. We assessed the molecular weight and the biochemical profile of Heparinox and compared its properties to the original branded enoxaparin (Lovenox; Sanofi, Paris, France). Clotting profiles compared included activated clotting time, activated partial thromboplastin time (aPTT), and thrombin time (TT). Anti-protease assays included anti-factor Xa and anti-factor IIa activities. Thrombin generation was measured using a calibrated automated thrombogram and thrombokinetic profile included peak thrombin, lag time and area under the curve. USP potency was determined using commercially available assay kits. Molecular weight profiling was determined using high performance liquid chromatography. We determined that Heparinox and Lovenox were comparable in their molecular weight profile. Th anticoagulant profile of the branded and biosimilar version were also similar in the clot based aPTT and TT. Similarly, the anti-Xa and anti-IIa activities were comparable in the products. No differences were noted in the thrombin generation inhibitory profile of the branded and biosimilar versions of enoxaparin. Our studies suggest that Heparinox is bioequivalent to the original branded enoxaparin based upon in vitro tests however will require further in vivo studies in animal models and humans to determine their clinical bioequivalence.
Background
Enoxaparin is the most widely used low molecular weight heparin (LMWH) worldwide. Several biosimilar versions of Lovenox (enoxaparin sodium; Sanofi, Paris, France), the original branded form of enoxaparin, have become available for clinical use for the indications that Lovenox was previously approved. 1 The regulatory bodies require the biosimilar products to conform to specific product specifications demonstrating biological and structural equivalence. 2
Heparinox (Cristália Produtos Químicos Farmacêuticos LTDA, São Paulo, Brazil) represents a newer version of enoxaparin commercially available in South America. Heparinox is obtained by depolymerization of porcine mucosal heparin using benzylation followed by alkaline depolymerization, a similar process to that which is used for the manufacturing of Lovenox. 3,4 In addition to Lovenox and Heparinox, several other biosimilar enoxaparins including Versa (Eurofarma, Brazil) and Curenox (Gland Pharma, India) are also available in Brazil.
Biosimilar versions of enoxaparin have been approved in Brazil by the National Health Surveillance Agency or ANVISA (Agência Nacional de Vigilância Sanitária) on the basis of molecular and biochemical similarity to the branded product. Regulatory agencies have required that each of these products must conform with specifications set forth for the branded products as specified in QS pharmacopeia. 5,6 Various tests specified in this monograph must be performed to demonstrate comparable activities and the products must be cross-referenced against National Institute for Biological Standards and Control (NIBSC) standard. 7 –9 From a regulatory viewpoint, the Food and Drug Administration (FDA) considers LMWHs as “generic” drugs, even though they may be sourced from biological material. The European Medicine Agency (EMA) considers LMWHs as biologicals, and thus, their regulatory approval as biosimilars is different when compared to the FDA. 6,10,11
The purpose of this investigation was to compare the branded enoxaparin (Lovenox) to Heparinox using validated biologic assays and analytical methods to determine the similarities of this biosimilar enoxaparin to the branded version. These methods have been used widely by the pharmaceutical industry and reference laboratories to compare biosimilar LMWHs.
Materials and Methods
Testing Agents
Two lots of biosimilar versions of Heparinox (heparinox-lot A12306A2 and heparinox-lot A124060) were obtained from Cristália Produtos Químicos Farmacêuticos (São Paulo, Brazil). The syringes contained 20 and 40 mg respectively and were diluted in saline, to obtain a 10 mg/ml solution. Similarly, Lovenox syringes containing 30 mg were diluted to 10 mg/ml. Additional dilutions were made at 1 mg/mL and 100 µg/mL for working solutions.
Molecular Weight Profiling
The molecular weight profiling was carried out by gel permeation chromatography, HPLC (high performance liquid chromatography). The mobile phase used in all the HPLC studies was made using anhydrous sodium sulfate (0.3 M Na2SO4, pH = 5.0) and HPLC-grade water from Sigma-Aldrich Inc. (St. Louis, Missouri, USA). Thirteen standard heparin fractions (Sanofi, Paris, France) ranging in molecular weight from 51.0 kDa to 2.4 kDa were used to calibrate the GPC-HPLC instrument and to make the standard curve.
USP Potency
The USP potency was determined using commercially available assay kits in accordance to the specifications and directions provided by the manufacturer HYPHEN BioMed (Neuville-sur-Oise, France). The USP potency of the 2 lots of Heparinox and Lovenox were measured using the anti-Xa and anti-IIa assays. The results were calculated utilizing the slope ratio and fixed concentration response point methods.
Activated Clotting Time Test (Whole Blood)
Whole blood activated clotting time (ACT) using the Hemochron ACT instrument from Accriva Diagnostics, Inc. (San Diego, California, USA) was measured. All drugs were diluted to obtain a working concentration of 250 µg/mL and placed in syringes. Using a double-syringe technique, whole blood was drawn from healthy human volunteers (n = 3) into syringes to obtain a final concentration of 25 µg/mL. The blood was then mixed and transferred to the celite ACT tube and placed in the Hemochron instrument to obtain the clotting time results. Saline was used as the control.
Clot-Based Assays
The 2 lots of Heparinox and Lovenox were supplemented into citrated plasma to obtain a final concentration range of 0.0-10.0 µg/mL and the same technique was applied for NIBSC to obtain a final concentration range of 0.0-1.0 U/mL. The thrombin time (TT) and activated partial thromboplastin time (aPTT) assays were used to analyze these samples. TriniCLOT aPTT reagent was obtained from Diagnostica Stago (Parsippany, New Jersey, USA) and human alfa thrombin was obtained from Enzyme Research Laboratories (South Bend, Indiana, USA) were used. An ACL-Elite automated coagulation analyzer (Instrumentation Laboratory, Bedford, Massachusetts, USA) was used to measure both aPTT and TT.
Anti-Protease Assays
An ACL-ELITE automated coagulation analyzer (Instrumentation Laboratory, Bedford, Massachusetts, USA) was used to perform the chromogenic assays anti-FXa and anti-FIIa. The drugs were supplemented in blood bank plasma to obtain a final concentration range of 0.0-10.0 µg/mL. Anti-Xa and anti-IIa activities were determined using in-house amidolytic assays employing human thrombin or bovine factor Xa (Enzyme Research Laboratories, South Bend, Indiana, USA) and Spectrozyme TH or Spectrozyme Xa (Biomedical Diagnostics, Windsor, Nova Scotia, Canada).
Inhibition of Thrombin Generation
Fluoroskan ascent fluorimeter, calibrated automated thrombogram (CAT; Diagnostica Stago, Paris, France) was used to measure the inhibition of thrombin generation. The assay was carried out in a 96-well Immulon 2HB transparent round bottom plates. The drugs were supplemented in blood bank plasma to obtain a final concentration range of 0.0-10.0 µg/mL. The following reagents were used in this assay: the fluo-substrate, fluo-buffer, tissue factor high reagent, and a thrombin calibrator. Peak thrombin concentration, lag time, and endogenous thrombin potential (ETP) /area under the curve (AUC) were utilized to measure thrombin generation potential.
Results
Molecular Weight Profiling
The molecular weight (MW) of both Heparinox lots tested was found to be approximately 4.0 kDa, whereas Lovenox exhibited a MW of ∼4.1 kDa using the UV detector. Using the RI detector method, both of the Heparinox lots exhibited a MW around 4.3 kDa and Lovenox exhibited a MW of around 4.5 kDa. The composite results are shown in Figure 1.

Molecular weight distribution of various Heparinox in comparison to branded enoxaparin (Lovenox).
USP Potency
The 2 lots of Heparinox and Lovenox showed comparable anti-Xa and anti-IIa potencies. The USP potency of Heparinox lots A124060 and Lovenox as measured by the anti-Xa assay was 96 U/mg whereas Heparinox-lot A12306A2 showed a potency of 97 U/mg as measured by the slope ratio method. The USP potencies measured by anti-IIa assay of all drugs were comparable and ranged between 30 -32 U/mg. The USP potency measured by the point method also showed comparable potencies which ranged from 98-105 U/mg for anti-Xa and 27-29 U/mg for anti-IIa as shown in Figure 2A and B.
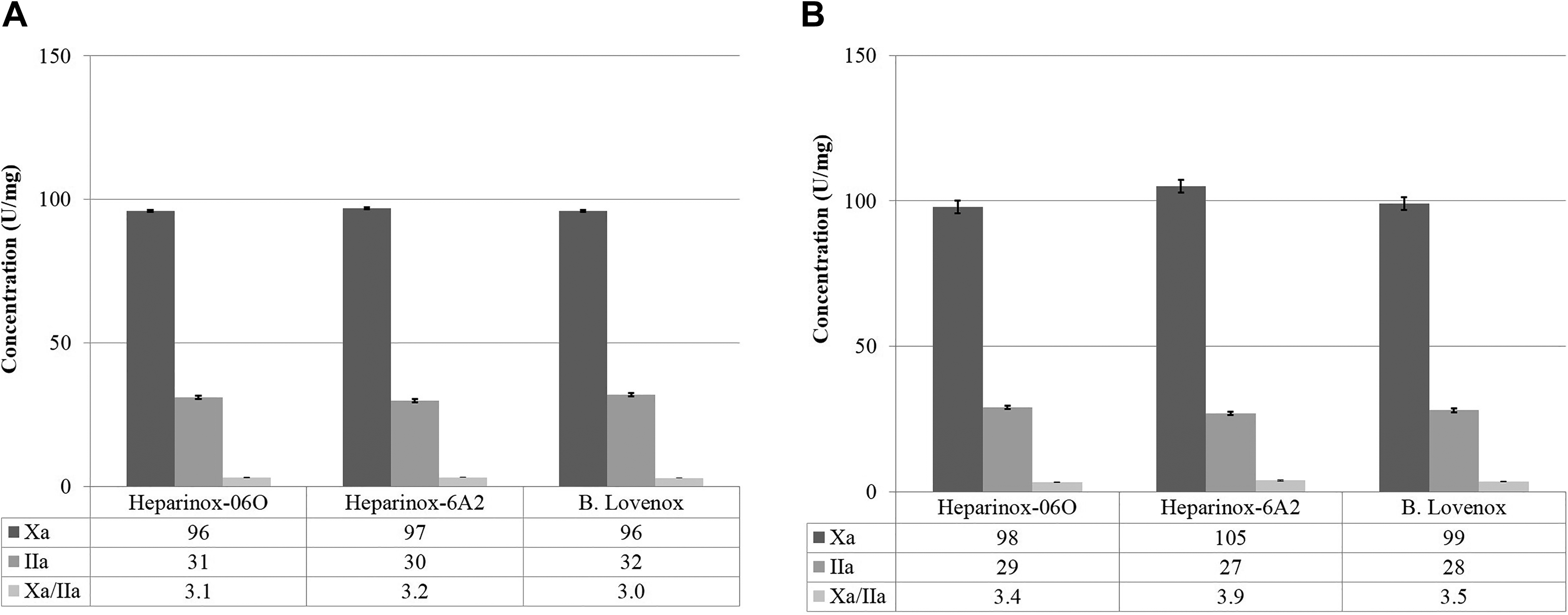
USP potency of Heparinox in comparison to branded enoxaparin (Lovenox) For Peer Review A. Points method, B. Slope method.
Activated Clotting Time Test (Whole Blood)
Lovenox, Heparinox-A124060 and Heparinox-A12306A2 at final concentration of 25 µg/ml yielded ACT values of (214.6 ± 14.6 seconds), (212 ± 1 seconds) and (215.6 ± 11.5 seconds) respectively. This demonstrates a clear anticoagulant effect when compared to a saline control value of 136.6 ± 7 seconds. When comparing Heparinox with Lovenox, the results showed a similar anticoagulant effect in the ACT assay. The ACT results are depicted in Figure 3.

Whole blood activated clotting time of various Heparinox in comparison to branded enoxaparin (Lovenox).
Clotting Assay
Figure 4 (A, B) depicts the results of clotting assays; thrombin time (TT) and activated partial thromboplastin time (aPTT). Both TT and aPTT results of Lovenox and Heparinox were comparable. Anticoagulant effects seen with all drugs were concentration dependent. The anticoagulant responses with both drugs were much stronger in the TT assay which reached a maximal response at 10 µg/mL, however the aPTT response was in the range of 75-85 seconds at this concentration.

Clotting profile of various Heparinox in comparison to branded enoxaparin (Lovenox). A. Activated partial thromboplastin time (aPTT), B. Thrombin time (TT).
Chromogenic Assay
Anti-factor Xa and anti-factor IIa assay results showed similar inhibitory responses with both lots of Heparinox and Lovenox. All agents produced a concentration-dependent inhibition of factor Xa and factor IIa as demonstrated in Figure 5. The IC50 of all drugs in anti-Xa was 2.5 µg/mL. The IC50 was much higher in the anti-IIa assays in which the Heparinox lots exhibited a value 9.4 µg/mL and 9.6 µg/mL, whereas the Lovenox showed a value of 7.6 µg/mL. The anti-Xa and anti-IIa ratios of the biosimilar and branded LMWH were comparable and in the range of 3.1-3.4.
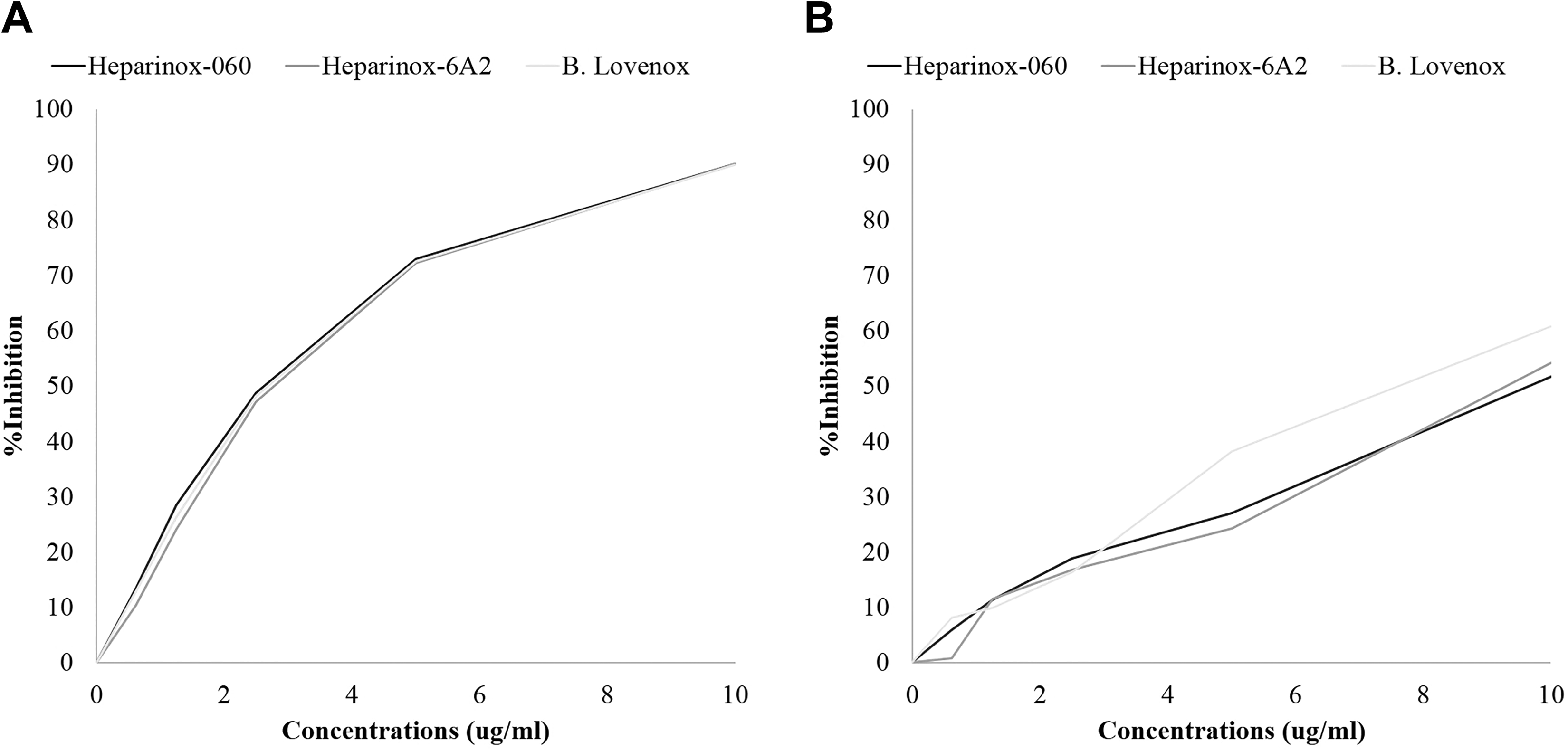
Antiprotease assay of various Heparinox in comparison to branded enoxaparin (Lovenox). A. AntiXa activity, B. Anti-IIa activity.
Inhibition of Thrombin Generation
Figure 6A-C displays the thrombokinetograms obtained by the thrombin generation on the CAT assay. All LMWHs produced a concentration-dependent inhibition of thrombin generation. Both Heparinox lots and Lovenox exhibited comparable inhibitory effects. The parameters used to measure thrombin generation are peak thrombin, AUC and lag time which are displayed in Figure 7A-C. The peak thrombin was in the range of 6.68 nM-8.37 nM at a drug concentration of 10 µg/mL, as is shown in Table 1. The peak thrombin results for all 3 drugs are comparable, with Lovenox having a slightly weaker response. In terms of lag time, as shown in Figure 4B, all drugs produced comparable responses on this parameter. Figure 7C depicts the ETP in terms of AUC. The results, again, are very comparable. The AUC values at 10ug/mL concentration ranged from 202.4 nM*min-238.8 nM*min.

Thrombokinetograms various Heparinox in comparison to branded enoxaparin (Lovenox). A. Thrombokinetogram for Heparinox-06O, B. Thrombokinetogram for Heparinox-A62, C. Thrombokinetogram for Lovenox.

Parameters of thrombin generation of various Heparinox in comparison to branded enoxaparin (Lovenox). A. Peak thrombin, B. Area under the curve (AUC), C. Lag time.
A Comparison of Heparinox and Lovenox on Thrombin Generation and Associated Parameters.

Discussion
To date, there are only a few published head-to-head comparisons of branded and generic LMWHs. 1,2,12,13 Our group has published comparable results for both efficacy and safety of biosimilar enoxaparin as compared to branded enoxaparin in the VTE prophylaxis clinical setting. 14,15 We have also compared the immunogenicity profile of biosimilar and branded enoxaparin, observing that despite small differences on sub-types of antibodies, no clinical implications were evident. 16 Our current study evaluated anticoagulant and biochemical characteristics and showed similarity between a biosimilar enoxaparin (Heparinox) as compared to its branded version (Lovenox). Both the biosimilar and the branded enoxaparin products demonstrated good comparability, indicating that both manufacturers used established and quality-assured processes. No differences were noted in the average molecular weight profiles and in the in vitro anti-FXa and anti-FIIa assays of anticoagulant potencies of the 2 enoxaparin products. Moreover, the anti-Xa/IIa ratios were comparable. No differences were observed in the activated clotting time test, aPTT, TT and chromogenic anti-Xa and anti-IIa assays.
Our analysis also showed that there were no differences in the inhibition of thrombin generation and the thrombokinetics of the inhibition of clot formation performed in the in vitro settings in plasma-based systems. Both LMWHs produced a concentration-dependent inhibition of thrombin generation. Both Heparinox lots and Lovenox exhibited comparable inhibitory effects. The parameters used to measure thrombin generation were peak thrombin, AUC and lag time. The biosimilar enoxaparin produced the equivalent inhibition of both thrombin generation and the progression of clot formation, as compared to the branded enoxaparin. These assays provide insight into the activation of the coagulation cascade, the rate of thrombin generation, the rate of fibrin clot formation, and the type and quality of fibrin clot which is eventually formed. These parameters detect a combination of the direct anti-FIIa (thrombin inhibition) activity of enoxaparin, as well as its inhibitory effect on the generation of thrombin (anti-FXa activity).
It is reasonable to project variations between branded and biosimilar enoxaparin, due to compositional differences in the oligosaccharide components of the drugs observed in previous comparative studies with different biosimilar enoxaparin. 12,17 Specific oligosaccharide sequences within the branded and biosimilar enoxaparin bind to plasma proteins and cell membranes, and the resultant effects on the activation or inhibition of biological pathways could be relevant to these findings. 18
Differences in the component oligosaccharide sequences in blood circulation at a given time may also be pertinent. In branded enoxaparin, only 30% of the oligosaccharide chains have been shown to have direct biochemical activities (evaluated by the anti-FXa and anti-FIIa assays) whereas the properties of the remaining 70% chains are not full characterized. This non-anticoagulant component of the LMWH represents a majority of the oligosaccharide chains and cannot be ignored since these chains may be responsible for mediating other biologic effects, depending on the structure of the saccharides in the chains, such as the release of tissue factor pathway inhibitor (TFPI). 19 In the current studies, no variations on the anticoagulant and biochemical characteristics were observed between the biosimilar and the branded enoxaparin. Additional structural studies using NMR and oligosaccharide analysis, and pharmacological profiling in terms of antithrombotic, bleeding and pharmacokinetic/pharmacodynamics parameters are currently in progress.
The relevance of these observed similarities in terms of therapeutic efficacy in true clinical settings is unknown. Since, Heparinox is already approved for clinical use in South America, real-world data will provide important information on both efficacy and safety of its use. Clinical trials in appropriate settings of the different indications for enoxaparin with Heparinox are in progress at this time. The results of these studies may assure health care providers the similarities in the molecular weight profile and in vitro screening of the biological properties between the branded and biosimilar enoxaparin, Heparinox, determine their in vivo effects.
Conclusion
This study demonstrates the molecular weight profile of Heparinox and Lovenox are comparable. The overall anticoagulant and antiprotease effects of Heparinox and Lovenox are also comparable. The same trend is observed in terms of the USP potency of these agents. The anti-Xa/IIa ratio for all agents are almost indistinguishable. Moreover, in the thrombin generation inhibitory assays Heparinox is comparable to Lovenox. These studies demonstrate that the newly introduced biosimilar LMWH Heparinox exhibits similar molecular profile and in vitro biological profiles in comparison to the branded enoxaparin. The data presented in this manuscript further validates the hypothesis that preclinical, molecular and biochemical profiling is sufficient to demonstrate bioequivalence of biosimilar LMWHs. The comparative studies carried out in this study can also be used to project the pharmacological behavior of these drugs in the in-vivo settings. Therefore, for the regulatory approval of biosimilar or biosimilar enoxaparins, the tests performed and summarized in this study may be sufficient for regulatory compliance.
Footnotes
Acknowledgments
The authors gratefully acknowledge the skillful assistance of the staff of the Hemostasis and Thrombosis Research Unit, Loyola University Health Science Division. We gratefully acknowledge Dr. Paul Riley of Diagnostica Stago (Paris, France) for providing us the instrument and reagents for the thrombin generation studies. We are also thankful to Ms. Erin Healy Erickson for her expert assistance in helping to prepare this manuscript. We also acknowledge the support of Dr. Eva Wojcik, Chairperson, Department of Pathology and Laboratory Medicine, and Dr. Seth Robia, Co-director of the Cardiovascular Institute for facilitating these studies.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
