Abstract
Venous thromboembolism is known to be a complex interaction of genetic and acquired factors leading to thrombosis. JAK2V617F mutation is believed to contribute to a thrombophilic phenotype, possibly through enhanced leukocyte–platelet interactions in myeloproliferative neoplasms (MPNs). Several studies have focused on the importance of screening for JAK2V617F mutation in patients with splanchnic venous thrombosis (VT) for the detection of nonovert MPNs. The role of JAK2V617F mutation in VT outside the splanchnic region is still widely unsettled. The primary aim of this study was to find out the prevalence of JAK2V617F mutation in patients with deep venous thrombosis (DVT), its clinical significance as a prothrombotic risk factor, and its possible interactions with other genetic thrombophilic risk factors. A total of 148 patients with idiopathic, symptomatic DVT were evaluated. Median age of presentation was 32 years (range 15-71 years) with a sex ratio of 1.3:1. Overall, the most common genetic prothrombotic factor was factor V Leiden mutation, found in 10.8% (16 of 148) of patients who also showed strong association with increased risk of thrombosis (odds ratio 5.94, confidence interval 1.33-26.4, P = .019). Deficiencies in protein C, protein S, and antithrombin were seen in 8 (5.4%), 10 (6.7%), and 8 (5.4%) patients, respectively. It was observed that the frequency of JAK2V617F mutation was lower in Indian patients, and it also showed weaker association with risk of thrombosis, at least in cases of venous thrombosis outside the splanchnic region.
Introduction
Venous thromboembolism is known to be a complex interaction of genetic and acquired factors leading to thrombosis. 1 The “gain-of-function” mutation in the exon 14 of Janus kinase 2 is an established marker in bcr-abl negative myeloproliferative neoplasms (MPNs). 2,3 It causes constitutive activation of the Janus kinase/signal transducers and activators of transcription (JAK-STAT) signaling pathways and mediates cytokine-independent stimulation of granulocytes and enhanced leukocyte–platelet interactions and the spontaneous formation of endogenous erythroid colonies. 4 Some studies claim that these mechanisms might contribute to a thrombophilic phenotype, but it is still unclear whether JAK2V617F mutation is associated per se with hypercoagulability. 5 Several studies have focused on the importance of screening for JAK2V617F mutation as an initial test in patients with splanchnic venous thrombosis (VT) for the detection of an underlying MPN. In patients presenting with extrahepatic portal vein obstruction and Budd Chiari syndrome, incidence rates of JAK2V617F mutation have been found to be as high as 70% to 90% in overt MPNs and 20% to 30% in nonovert MPNs. 6,7 The role of JAK2V617F mutation in VT outside the splanchnic region is still widely unsettled. Especially in the Indian population, the prevalence rate of JAK2V617F mutation in patients with nonsplanchnic VT (NSVT) has varied remarkably from <1% to as high as 6%. 4,8 The primary aim of this study was to find out the prevalence of JAK2V617F mutation in Indian patients with deep venous thrombosis (DVT), its clinical significance as a prothrombotic risk factor, and its possible interactions with other genetic thrombophilic risk factors.
Materials and Methods
Patients and Controls
This prospective study involved patients with idiopathic, symptomatic, single episode of DVT who presented to the hematology outpatient department during the 2-year period from January 2011 to December 2012.
A detailed history was taken from all the patients regarding the presence of known circumstantial predisposing factors of venous thromboembolism such as surgery, trauma, immobilization, pregnancy, oral contraceptive use, liver or renal disease, hyperlipidemia, diabetes, and so on. Patients with any of these situational risk factors were not incorporated in the final study in order to obtain a more reliable evaluation of the prevalence of various genetic thrombophilic risk factors. Samples were also not collected from patients with DVT already on oral anticoagulants or those with a past or family history of thrombotic events. Apart from this, only those patients with DVT in whom acquired prothrombotic risk factors such as antiphospholipid syndrome (APLA), paroxysmal nocturnal hemoglobinuria (PNH), hyperhomocystinemia, or overt MPN were ruled out by routine laboratory testing, that is, (a) negative for lupus anticoagulant by 2 assays—lupus-sensitive Activated partial thromboplastin time (APTT) and dilute Russel viper venom test, (b) negative for PNH by CD55/CD59 Gel card method, (c) normal serum homocysteine levels, and (d) normal hemogram and differential count were selected in the final cohort of patients for uniformity of results.
A total of 148 patients who satisfied all the above-mentioned criteria were selected for the final study. Of these patients, 64 (43.2%) were females and the remaining 84 (56.8%) were males, with a sex ratio of 1.3:1. Median age at presentation in these patients was 32 years (range 15-71 years). Diagnosis of VT was confirmed in all these patients by imaging techniques such as duplex ultrasonography, venography, computed tomography, or contrast-enhanced magnetic resonance imaging.
One hundred healthy controls chosen from among the friends or partners of patients referred to the coagulation laboratory for thrombophilic screening were selected as part of a larger comprehensive project. Written informed consent was obtained from both the patients and the controls, and ethical clearance was obtained from the Institutional Ethical Committee prior to the conduct of the study. Median age of the controls was 29 years (range 15-65 years), and the male:female ratio was 1:1. None of the healthy controls had a family history of thrombosis or presence of any predisposing risk factors for thrombosis or any other comorbidities.
Laboratory Testing
Blood samples for thrombophilia testing were collected in 3.2% trisodium citrate (ratio 1:9), centrifuged at 3500 rpm for 10 minutes, and the platelet-poor plasma so obtained was aliquoted immediately and preserved at −70°C. All the plasma samples were tested within 2 months of collection. Special care was taken to ensure that samples were not collected during the acute thrombotic episodes to avoid false lower levels of natural anticoagulants. Antigen activity of protein C (PC) and protein S (PS) was determined by sandwich enzyme-linked immunosorbent assay using commercially available kits (Corgenix, Inc, Denver, Colorado). The normal reference range for PC and PS per kit was 72% to 130% and 60% to 135%, respectively. Quantification of antithrombin (AT) was done by chromogenic assay using STACHROM ATIII kit on automated STA Compact Analyzer (Diagnostica Stago, France). Normal reference range for AT was 80% to 120% per the kit. All samples showing borderline results were repeated, and only those samples giving consistent results on both occasions were considered as deficient.
Total genomic DNA was isolated from peripheral blood leukocytes using commercially available kits (Bioserve, Beltsville, USA). Mutational analysis for factor V Leiden (FVL) and prothrombin G20210A was performed by polymerase chain reaction (PCR)–restriction fragment length polymorphism method using MnlI and HindIII restriction enzymes, respectively. 9 The existence of C677T methylene tetrahydro-folate reductase (MTHFR) polymorphism was determined by PCR and Hinf1 digestion. 9 Presence of JAK2V617F mutation was assessed by DNA tetra-primer–based amplification refractory mutation system-(ARMS) PCR method exactly as described by Jones et al. 10 All samples were initially screened by ARMS-PCR technique (due to its high sensitivity), and the positive samples were reanalyzed by a second technique, allele-specific oligo-nucleotide PCR as described by Baxter et al, for validation of results. 11
Statistical Analysis
Data were presented in median and frequency percentage. Odds ratio (OR) for VT associated with various thrombophilic markers and the corresponding 95% confidence intervals (CIs) were calculated using logistic regression, adjusted for age and sex. P values < .05 were considered as significant.
Results
Demographic Characteristics of 150 Patients With Single Episode of Deep venous thrombosis.
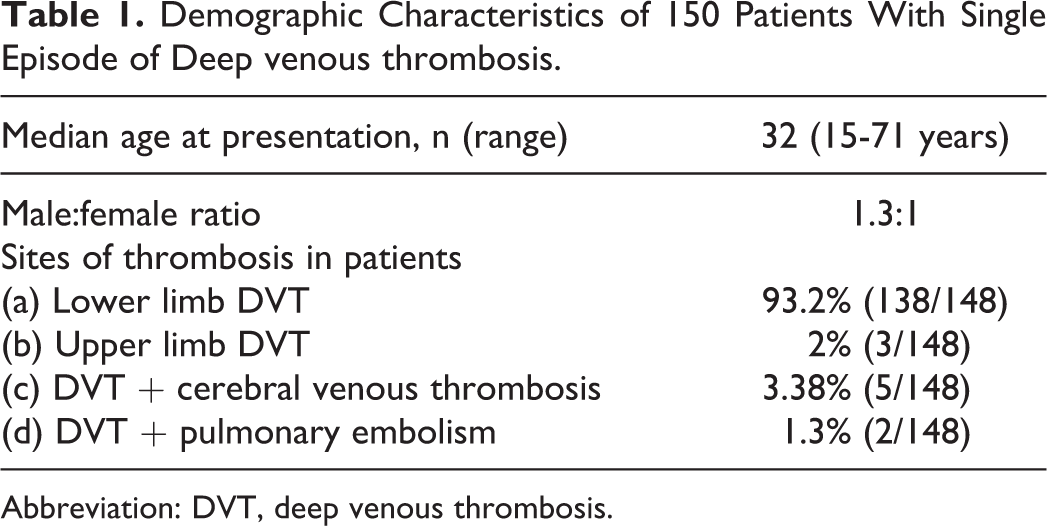
Abbreviation: DVT, deep venous thrombosis.
Prevalence Rates of Thrombophilic Factors and Their Relative Risk for Thrombosis in Patients With Deep Venous Thrombosis.
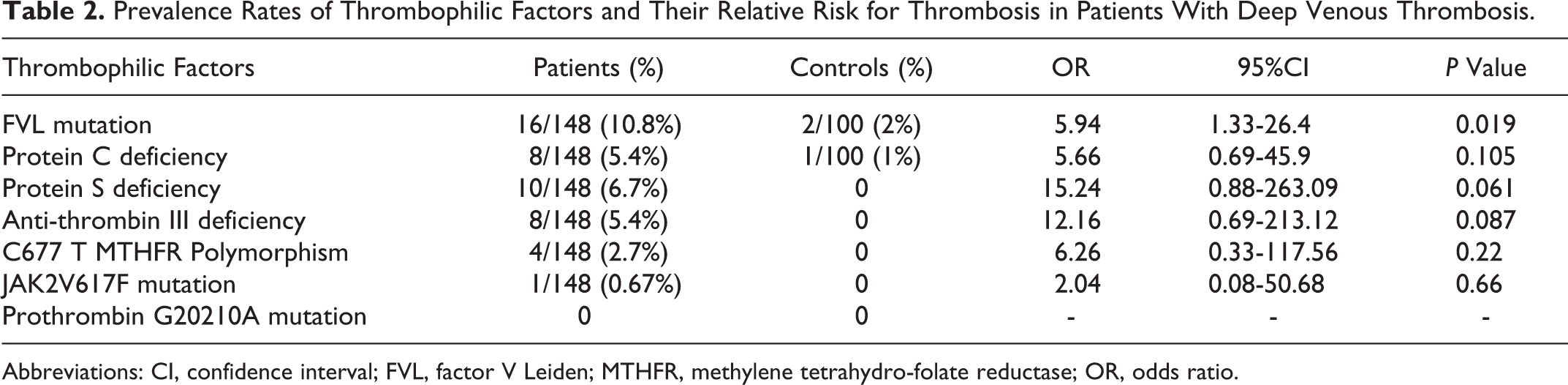
Abbreviations: CI, confidence interval; FVL, factor V Leiden; MTHFR, methylene tetrahydro-folate reductase; OR, odds ratio.
In this study, only 1 (0.67%) patient was found to be positive (heterozygous) for the JAK2 V617F mutation. That patient was a 35-year-old male with normal peripheral blood counts, that is, hemoglobin = 12.4 g/dL, white blood cell = 9.4 × 103/L, and platelet count = 412 × 109/L with normal differential count. He also showed concomitant deficiency of PS and did not progress to overt MPN even after a follow-up of over 2 years (30 months). No significant association was found between JAK2V617F mutation and other thrombophilic factors. Also, none of the healthy control individuals enrolled in this study showed the presence of JAK2V617F mutation. Apart from this, no cause could be identified in 68.3% of these patients with VT, despite detailed thrombophilic workup.
Discussion
Both inherited and acquired conditions account for a significant proportion of all cases with NSVT in randomly selected group of patients. But despite extensive laboratory workup, in as many as 60% to 65% of selected patients, no etiology can be defined. 5 This corroborates well with the observations of many other studies and reinstates the fact that VT has a diversified spectrum of etiology.
Factor V Leiden mutation describes a G1691A nucleotide transition resulting in an R506Q amino acid missense mutation. It was found only in 2% of our general population which is concordant with the other ethnic studies involving the Asian and African regions but much lower in comparison to the European and North American population where incidence rates of 5% to 10% have been observed. 12 According to various studies, FVL accounts for 20% of all consecutive cases of VT and up to 50% of all thrombophilic families among the caucasians and in the Middle-East. 13 Unlike them, a much lower overall prevalence of FVL mutation was observed among our patients with VT (10.8%). Moreover, FVL mutation showed a stronger association (5- to 6-fold) with increased risk of thrombosis (OR 5.94, CI 1.33-26.4, P = .019) and can be considered as an independent thrombophilic risk factor for VT.
Prothrombin G20210A gene variants were not found in any of the patients or the controls in this study. In Western population, the frequency has been found to be around 1% to 3% in healthy individuals, 6% to 8% in patients with VT, and as high as 18% in those with a positive family history of VT. However, in Indian patients, its role as a thrombophilic factor still remains controversial due to paucity of the prothrombin gene polymorphism. 9,12,14
Prevalence rates of PC, PS, and AT in this study were higher in comparison to the data from most other published studies worldwide which may be due to variations in ethnic distribution of patients. 12 Few other published Indian studies have also documented relatively higher prevalence rates of these thrombophilic risk factors in Indian patients. 15,16 Also, 2.7% of patients showed homozygosity for the MTHFR C677T polymorphism in the presence of normal homocysteine levels, and no significant association with increased risk of thrombosis was found (P = .22). This finding is in concordance with the results of most other published studies that have proclaimed that MTHFR C677T polymorphism in the absence of hyperhomocystinemia is not a risk factor for VT. 17,18
Screening for JAK2V617F mutation has been carried in several retrospective cohort of patients with arterial thrombosis or VT outside the splanchnic region in which no signs of overt MPN were present. In this study, a lower prevalence of JAK2V617F mutation was found in Indian patients (0.67%) and also, none of the controls showed this mutation. This was in concordance with few other studies worldwide who found that this mutation was either absent or was present at a very low frequency (1%-2%) in patients with NSVT. 4,19 –22 On the contrary, some other studies have demonstrated prevalence rates as high as 5.7% in patients with DVT 5 and 5.9% in patients with CVT without overt MPN. 8 In healthy controls, marked variation was observed in the frequency of JAK2V617F mutation ranging from <1% according to a Chinese study 23 and 3.3% in an Egyptian study. 5
Moreover, the strength of its contribution and potential interactions with other thrombophilic factors also remain to be elucidated. In this cohort, JAK2V617F mutation was present in the patient with concomitant deficiency in PS, which by far has been considered as an independent thrombophilic risk factor (OR 15.24; CI 0.88-263.1; P = .06). Attributing JAK2V617F as a causative mechanism in such a scenario where the patient did not develop overt signs of myeloproliferation even after a follow-up of over 2 years needs further validation through comprehensive studies with longer follow-up durations. Also, issues as to whether patients with VT having JAK2V617F mutation represent early-phase myeloproliferative disorder (MPD) or if there is a need for addition of cytoreductive medication apart from oral anticoagulants require further discussion. It can be substantiated by the fact that in a larger study by Pardanani et al, 21 none of the JAK2V617F-positive patients developed an overt MPD or recurrent thrombosis after a median follow-up of 40 months.
Conclusion
JAK2V617F mutation is found at a very low frequency in Indian patients. Also, it appears to be a weak prothrombotic risk factor, especially in cases of VT outside the splanchnic region. It can therefore be concluded that screening for the JAK2V617F mutation should not be recommended as part of screening for DVT in routine practice as the yield is very low and may not be cost effective.
Footnotes
Acknowledgments
We are thankful to Mr Suresh and Ms Davinder for technical assistance in performing the tests.
Author Contributions
NS organized and analyzed the data and drafted the manuscript. AS provided the clinical data and performed experimental studies. SS coordinated the study and participated in its design. AU performed statistical analysis. AA, MM, and RS provided valuable clinical inputs and reviewed the article. All authors read and approved the final manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
