Abstract
Hemoglobin Eβ thalassemia is a major public health problem in India, especially in the state of West Bengal. Various thromboembolic events are common, especially in splenectomized patients. Platelet hyperactivity most likely plays a pathogenetic role. To investigate the role of platelets in hypercoagulability, platelet aggregation tests were undertaken in the present study. Platelet-rich plasma from 30 patients with Eβ thalassemia (15 splenectomized and 15 nonsplenectomized) were studied and compared with 15 healthy participants. The 4 agonists used were adenosine 5-diphosphate, adrenaline (epinephrine), collagen, and ristocetin. The current study shows both splenectomized and nonsplenectomized patients had abnormal aggregation compared to normal healthy controls. Splenectomized patients had higher platelet aggregation than nonsplenectomized patients for all 4 agonists; but statistically significant difference among 2 groups was found only for collagen. The present study confirms a role of splenic absence in platelet hyperaggregation.
Introduction
Hemoglobin Eβ thalassemia is the commonest form of severe thalassemia in many Asian countries. 1 Hemoglobin E (HbE) is the commonest Hb variant in India with a prevalence of 7% to 50% in North Eastern region and 1% to 4% in West Bengal. 2,3 It shows remarkable clinical heterogeneity ranging from asymptomatic state to severe transfusion dependence. Severely affected patients are treated with blood transfusion and/or splenectomy in order to maintain an optimum level of Hb for normal growth and physical activities.
With continued improvement in survival, the development of a number of late effects has become increasingly apparent but the focus on hemostatic changes is relatively recent. From the hemostatic standpoint, it is characterized by hypercoagulability and an increased risk of venous and/or arterial thromboembolism. 4 Patients who undergo splenectomy are particularly prone to develop thrombosis, especially of the small pulmonary arteries. 5 Although several alterations in the blood components like increased number of circulating platelet aggregates and reduced protein C and protein S have been suggested, no clear-cut pattern of the hemostatic derangement has evolved. 6 –8
The present study was therefore undertaken to assess the derangements in basic coagulation and platelet function tests in patients with Eβ thalassemia. In this comparative study, the patients were divided into 2 groups—one group without splenectomy and the other with splenectomy. Age- and sex-matched healthy participants were taken as the control group. All 3 groups were investigated for different laboratory parameters, and the results were compared by appropriate statistical tests.
Materials and Methods
Consent taken from 30 patients with Eβ thalassemia visiting the Thalassemia Day Care Center was studied. Fifteen age- and sex-matched healthy normal individuals who consented for the study comprised the control group. Of the 30 patients, 15 were splenectomized patients and 15 were nonsplenectomized patients. Other patients with Eβ thalassemia who were seropositive for hepatitis B surface antigen, HIV antibody, or antihepatitis C virus and whose consent could not be obtained were excluded. Patients who had received a blood transfusion, fresh frozen plasma/factor transfusion, within the previous 6 weeks or had taken aspirin or anticoagulants within the previous 4 weeks were also excluded from the study. The patients or controls who were smokers or had taken some form of herbal medications were excluded from our study. Control participants had not taken any medications for at least 2 weeks.
Overnight fasting venous blood was drawn from the antecubital vein. A tourniquet was applied briefly to ease venous access and then was released. Blood was obtained using a plastic syringe. Serum was used for the determination of liver biochemical profile and serum ferritin levels. Blood anticoagulated with EDTA was used for complete blood count (CBC) and Hb typing. Blood anticoagulated with 3.2% sodium citrate (9:1) was used for coagulation tests and platelet aggregation tests. The following tests were then carried out: (1) peripheral blood smears examination by light microscopy, (2) CBC obtained from automated cell counter (sysmex, KX21; Sysmex, Kobe, Japan), (3) prothrombin time (PT), activated partial thromboplastin time (APTT), thrombin time (TT), and platelet count were done as basic coagulation tests, (4) platelet aggregation tests were done using aggregometer (Lumi-aggregometer; Chrono-Log Corporation, Havertown, PA, USA), (5) serum ferritin, serum alanine transferase (ALT), and serum albumin were estimated by standard method.
Platelet aggregation tests were done by optical methods with whole blood, for which platelet-rich plasma (PRP) was obtained by centrifuging the citrated venous blood at room temperature (20°C-25°C) for 10 minutes at 1000 rotations per minute (rpm). The PRP was carefully removed and placed in a stoppered plastic tube. Final concentration of agonists used were from Chrono-Log Corporation at the concentration of 10 μmol/L/mL adenosine 5-diphosphate (ADP), 2 μg/mL collagen, 10 μmol/L/mL epinephrine (adrenaline), and 1.25 mg/mL ristocetin. Test was done 30 minutes after PRP preparation and within 3 hours. The aggregometer was switched on 30 minutes before tests to allow heating block to warm up to 37°C. The stirring speed was then set at 900 rpm. The instrument was then calibrated by PRP as well as platelet poor plasma (PPP). The PRP (250 μL) was taken into a cuvette, which was then placed in the heating block. After 1 minute, stirrer was inserted into the plasma and transmission set to “0” on the chart recorder. The PPP-containing cuvette was then replaced and the transmission set to 100%. The PRP was allowed to warm up to 37°C for 2 minutes and then agonist was added. The change in absorbance was recorded till plateau was reached or for 5 minutes, which one was earlier. The procedure was repeated for each agonist. The results were expressed as the maximal impedance change in percentage (%). The blood sample was centrifuged at 3000 rpm for 20 minutes, and the supernatant PPP was collected in a clean plastic tube for PT, APTT, and TT by Coagulation Analyzer (CA 50; Sysmex Ltd). Complete blood count was done by automated blood cell counter (3 parts, Sysmex KX 21; Sysmex Corporation). Erba-XL 600 Automated Random Access Clinical Chemistry Analyzer (kinetic method, Mannheim, Germany) was used for serum ALT and serum albumin and TECAN ELISA, Mannedorf, Switzerland reader with washer used for serum ferritin level. The Hb typing was carried out by high-performance liquid chromatography, Biorad Variant Hb Testing System for β-thalassemia Short Programme (Bio-Rad Laboratories, Hercules, California).
Collected data were analyzed, and statistical tests were done with the help of Microsoft Excel and Win Pepi (version 404X) software. Where applicable, data were presented as the mean ± standard deviation (SD). One-way analysis of variance (ANOVA) was used to assess the difference among means of the 3 groups. If the ANOVA showed a statistically significant difference among groups, each group was paired with the others and between the groups; differences were assessed by Scheffe test. A statistically significant difference existed if the P value was <.05.
Results
Of the 30 patients, 15 were male and 15 were female (ratio, 1:1). The age range was from 5 to 36 years, with a median of 16.5 years. There were 15 controls. There were 8 male and 7 females from nonsplenectomized group with age range between 5 and 32 years with a median of 22 years. There were 8 female and 7 male from nonsplenectomized group with age range between 10 and 36 years with a median of 17 years. The basic hematologic parameters in the 3 groups are depicted in Table 1. Hemoglobin level in the control, nonsplenectomized, and splenectomized groups was 139 ± 14, 77 ± 14, and 69 ± 08 g/L respectively. The hematocrit, mean corpuscular volume, and mean corpuscular hemoglobin in the above-mentioned group are shown in Table 1. The white blood cell counts were much higher in the splenectomized patients in comparison to others. There was thrombocytosis with a platelet count of 6.1 ± 1.8 × 105/μL in the splenectomized group; much higher than that of the other 2 groups. Hemoglobin F (%) value was also much higher (33.55 ± 6.58) in the splenectomized group in comparison to the nonsplenectomized patients (18.01 ± 10.19). The transfusion requirement of patients in the nonsplenectomized group was 10 to 15 units of packed red blood cell (RBC) transfusions per year and in splenectomized group 0 to 5 units of packed red blood cell transfusions per year.
Basic Hematologic Parameters (Mean ± SD) in the 3 Groups.
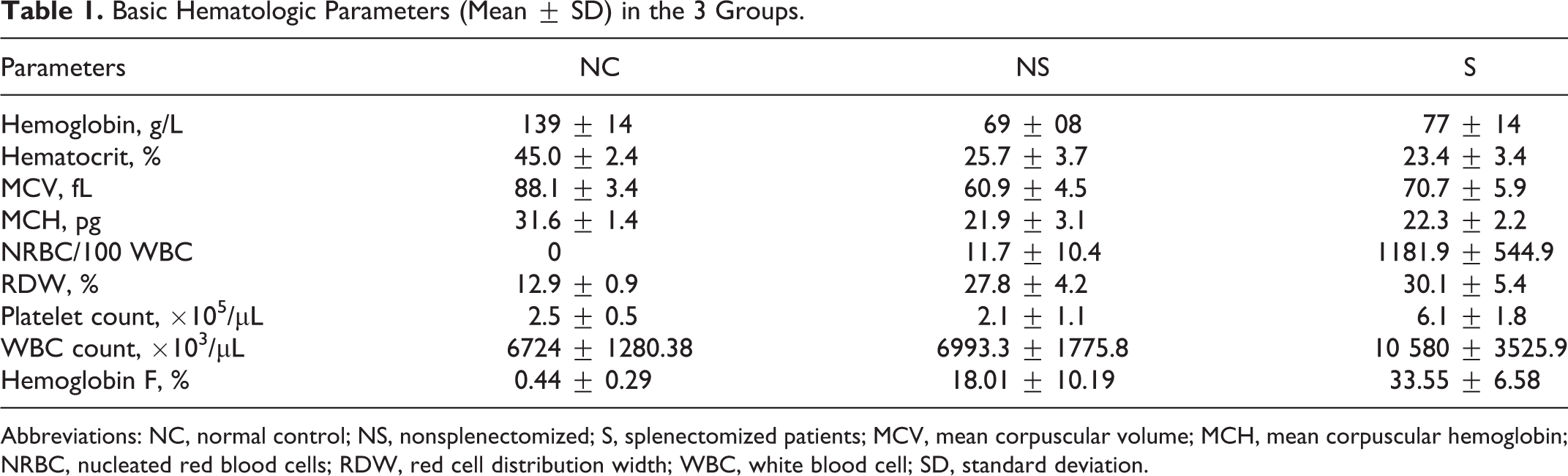
Abbreviations: NC, normal control; NS, nonsplenectomized; S, splenectomized patients; MCV, mean corpuscular volume; MCH, mean corpuscular hemoglobin; NRBC, nucleated red blood cells; RDW, red cell distribution width; WBC, white blood cell; SD, standard deviation.
Biochemical parameters (mean ± SD) in the 3 groups are shown in Table 2. There were comparable values for serum albumin, but serum ferritin and serum ALT values were much higher in the splenectomized group in comparison to the nonsplenectomized patients and the controls. As shown in Table 3, the basic coagulation test results (mean ± SD) did not show any gross variation in the 3 groups.
Biochemical Parameters (Mean ± SD) in the 3 Groups.
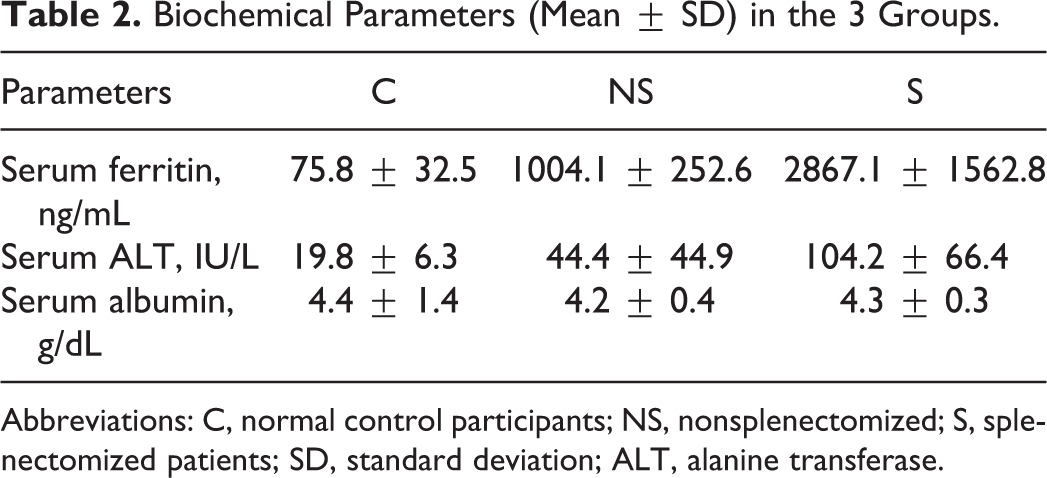
Abbreviations: C, normal control participants; NS, nonsplenectomized; S, splenectomized patients; SD, standard deviation; ALT, alanine transferase.
Basic Coagulation Test Results (Mean ± SD) in the 3 Groups.
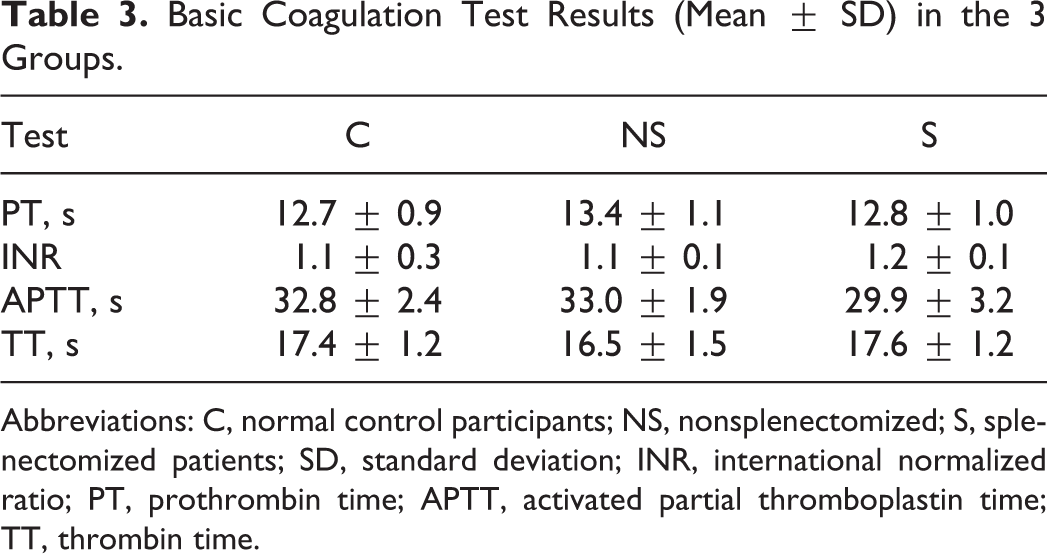
Abbreviations: C, normal control participants; NS, nonsplenectomized; S, splenectomized patients; SD, standard deviation; INR, international normalized ratio; PT, prothrombin time; APTT, activated partial thromboplastin time; TT, thrombin time.
The maximum aggregation (%) to each of the 4 agonists for all the 3 groups is shown in Table 4. Aggregation (%) to ADP, collagen, and ristocetin in the splenectomized patients is much higher in comparison to the nonsplenectomized group. As shown in Table 4, 1-way ANOVA showed statistically significant difference among the means in all 3 groups. The P values for the difference among the means of the 3 groups for response with the agonists ADP, collagen, ristocetin, and adrenaline were <.001, .013, .046, and <.001, respectively.
Mean of Maximal Percentage (%) Aggregation to Each Agonist for All 3 Groups and Comparisons of All 4 Agonists Across the Groups (by Scheffe test).a
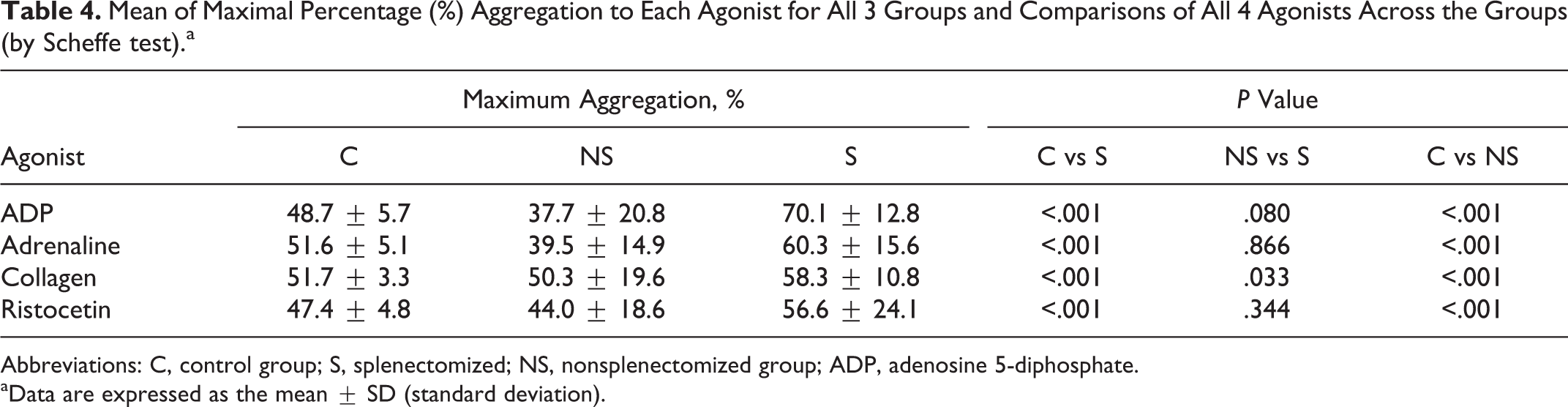
Abbreviations: C, control group; S, splenectomized; NS, nonsplenectomized group; ADP, adenosine 5-diphosphate.
aData are expressed as the mean ± SD (standard deviation).
As statistically significant difference was found among the 3 groups with all 4 agonists, each group was paired with others, and Scheffe test was carried out. As shown in Table 4, the results obtained show that statistically significant difference was seen between the splenectomized and nonsplenectomized groups, with response to collagen only (P = .033). For ADP, ristocetin, and adrenaline, the respective P values were .080, .344, and .866.
Discussion
Several studies have shown that patients with Eβ thalassemia, particularly after splenectomy, have a state of hypercoagulability and/or platelet dysfunction. 9 Among the thromboembolic manifestations of HbEβ thalassemia, deep venous thrombosis, pulmonary embolism, and cerebral thrombosis have been reported.
To investigate the role of platelets in hypercoagulability in such patients, platelet aggregation studies were carried out in PRP. The agonists used were ADP, adrenaline (epinephrine), collagen, and ristocetin. Aggregation was detected by optical methods and the maximal percentage of aggregation following addition of agonist was recorded. The mean of (%) aggregates were calculated for each agonist for the 3 groups: control, nonsplenectomy, and splenectomy.
The mean difference in platelet response across the 3 groups was statistically significant with all 4 agonists by ANOVA test. The P values for the difference among the means of the 3 groups were <.001, .013, .046, and <.001 for response to ADP, collagen, ristocetin, and adrenaline, respectively. This implies that platelet aggregation was defective across the groups. Intergroup comparison by Scheffe test showed statistically significant difference between the splenectomized and nonsplenectomized groups in response to collagen only (P .033). For ADP, ristocetin, and adrenaline, the respective P values were .080, .344, and .866, respectively.
Hence, the current study indicates that splenectomized patients had higher platelet aggregation than nonsplenectomized patients with all 4 agonists; but statistically significant difference among the 2 groups was found only for collagen. The absence of significant difference for the other 3 agonists could possibly be due to the variation in the aggregation pattern in our patients and a low sample size. However, large-scale studies are needed to address this issue.
In the present study, significant differences were observed between control and splenectomized groups with all 4 agonists, indicating aggregation abnormalities in splenectomized patients with HbEβ thalassemia compared to normal healthy controls. Similarly, statistically significant difference was seen between control and nonsplenectomized groups for all 4 agonists, indicating aggregation defects in nonsplenectomized patients compared to normal healthy controls. Hence, both splenectomized and nonsplenectomized patients had abnormal aggregation compared to the normal healthy controls.
The lower mean aggregation observed in the nonsplenectomized group in the present study could be due to the use of PRP instead of whole blood causing platelet selection during centrifugation and a low sample size.
In the study by Atichartakarn et al, 5 platelet aggregation was detected by impedance method using whole blood. They calculated the maximal impedance change after the addition of various platelet agonists. The agonists used included ADP, collagen, and ristocetin but instead of adrenaline, thrombin was used. They reported statistically significant P values for ADP (<.001), ristocetin (<.001), and thrombin (.001) but not for collagen (.08) across the groups. Among the splenectomized and nonsplenectomized groups, they found statistically significant difference in response to ADP, thrombin, and ristocetin, indicating hyperaggregation of platelets in response to the above-mentioned agonists in the splenectomy group; corresponding measurements in the nonsplenectomized and control groups showed no differences.
Visudhiphan et al 10 studied platelet aggregation in 58 cases with the same 4 agonists as the present study and reported defective aggregation in both splenectomized and nonsplenectomized groups. In the 35 nonsplenectomy cases, the platelet aggregation to ADP, adrenaline, collagen, and ristocetin was defective. In the 23 splenectomized patients, platelet aggregation in response to ADP, adrenaline, and collagen showed better results. Platelet aggregation in response to ristocetin remained defective. Opartkiattikul et al 11 studied spontaneous platelet aggregation in whole blood in 40 patients. Of them, 15 were nonsplenectomized and 25 splenectomized. The results of their study indicated increased spontaneous platelet aggregation in postsplenectomized patients with HbEβ thalassemia. Some authors have reported platelet hypoaggregation in β thalassemia and HbEβ thalassemia. 11,12 Platelet-rich plasma was employed for all such studies. This phenomenon of platelet hypoaggregation can be explained by a process of platelet selection during centrifugation in obtaining PRP. On the other hand, “spent” or “exhausted” platelets from in vivo platelet activation could also have resulted in platelet hypoaggregation. 13
Persistent thrombocytosis after splenectomy has been reported by several authors in β Thalassemia as well as HbEβ thalassemia. 5,14 In the present study, the platelet count was markedly high in the splenectomized group compared to the nonsplenectomized group (Table 1). The mean for the first group was 6.1 × 105/μL (range: 2.3-8.4 × 105/μL) unlike a normal mean of 2.1 × 105/μL (range: 1-4.6 × 105/μL) for the latter group. Similar data were reported by several workers including Premawardhena et al, 15 who reported that persistent thrombocytosis after splenectomy was common among the 109 patients they studied. Atichartakarn et al 5 reported a high mean platelet count of 6.5 × 105/μL in splenectomy patients compared to a lower mean of 2.6 × 105/μL in nonsplenectomy patients. In the study by Visudhiphan et al, 10 14 of the 23 splenectomized cases included in the study exhibited thrombocytosis. Winichagoon et al 16 reported increased circulating platelet aggregates in 71% of splenectomized and 35% of nonsplenectomized patients with Eβ thalassemia, an observation compatible with in vivo platelet activation and the existence of a hypercoagulable state. Moreover, in a subanalysis of the OPTIMAL CARE (Overview on practices in thalassemia intermedia management aiming for lowering complication rates across a region of endemicity) study, a platelet counts of >5.0 × 105/μL was an independent and significant predictor of thromboembolism in splenectomized patients with thalassemia intermedia. 17 Thus, persistent thrombocytosis after splenectomy seems to result in a hypercoagulable state contributing to pulmonary vasoocclusive disease due to thromboembolism. Although high platelet counts were encountered in splenectomized patients in the present study, there were no thrombotic episodes. Premawardhena et al 15 reported similar results with the splenectomized patients with HbEβ thalassemia in Sri Lanka. They studied 109 Sri Lankan patients with the disorder older than 5 years. Only 12 patients, of whom 10 had been splenectomized, were found to have mildly elevated pulmonary artery pressures. Unlike earlier reports from Thailand, they did not observe right heart failure associated with postsplenectomy thrombocytosis. 9
Several factors have been implicated in the pathogenesis of platelet activation. The changes appear to be the consequence of splenectomy, which may have resulted in a chronic hypercoagulable state via an excess amount of circulating phosphatylserine (PS)-exposed erythrocytes and postsplenectomy thrombocytosis. 5,8 The procoagulant surface of the PS-exposing erythrocytes can facilitate both the coagulation process and platelet activation. 18,19 Using thalassemic red cells as a source of phospholipids, enhanced thrombin generation has been demonstrated in a prothrombinase assay. 20 Thrombin at subthreshold doses can sensitize platelets to the effect of a subsequent platelet agonist. 21 Thus, platelet hyperaggregation may be due to chronic low-grade thrombin generation. However, interleukin 6, which is increased in splenectomy patients with Eβ thalassemia, can cause both increased platelet production and increased reactivity to thrombin stimulation, particularly in young platelets. 22 –24 Persistent thrombocytosis and iron excess are features of splenectomy patients with HbEβ thalassemia. Iron excess and associated oxygen free radicals have been shown to cause platelet hyperaggregation in patients with β-thalassemia major. 25 Recently, others have found evidence of chronic low-grade inflammation and endothelial cell injury in splenectomized patients with Eβ thalassemia, both of which can lead to platelet activation. 26
In patients who had undergone splenectomy, there is quantitative (thrombocytosis) and qualitative defects in platelets. So they are more prone to show platelet function defects by LTA when compared to nonsplenectomized patients who require the presence of abnormal flip-flopped RBC membrane in addition to some defect in platelets to show a significant abnormality in whole blood aggregometry, which is hindered by blood transfusions and probably that is why our patients who are nonsplenectomized have not shown significant abnormality with LTA where source of sample is PRP.
In the present study, values of PT, APTT, and TT were normal in all 3 groups and hence could not indicate the patients at risk of hypercoagulability. The failure of these tests to identify hypercoagulability in thalassemia might be due to the absence of blood cells and platelets, which may play a key role in the mechanisms responsible for thrombosis in this setting.
In conclusion, alterations in the activity of blood coagulation and fibrinolytic proteins could be detected in patients with HbEβ thalassemia, whether with or without spleen; however, the most significant changes occurred in the severe splenectomized group who, therefore, are at a higher risk of thrombosis. The present study, therefore, confirms and extends previous findings and suggests a role of splenic absence in the platelet hyperaggregation.
Footnotes
Authors’ Note
Tanushree Ghosal, Tuphan Kanti Dolai, and Prakas Kumar Mandal contributed to concept, design, literature search, clinical studies, manuscript preparation, statistical analysis, manuscript editing, and review. S. Karthik contributed to literature search, clinical studies, manuscript preparation, and editing. Anjali Bandyopadhyay contributed to concept, design, literature search, clinical studies, manuscript editing, and review. This work was carried out at the Department of Pathology, R.G. KAR Medical College, Kolkata 700004, India and the Department of Hematology, NRS Medical College, Kolkata 700014, India.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
