Abstract
Introduction and Aim:
Expression and peptide immunoreactivity of apelin messenger RNA have been described in a variety of tissues, including gastrointestinal tract, adipose tissue, brain, kidney, liver, cardiovascular system, and lungs. This study aimed to investigate the possible involvement of the endogenous apelin in the pathophysiological events that occur in patients with pulmonary embolism (PE).
Materials and Methods:
In total, 53 patients with PE and 35 healthy volunteers were included the study. This cross-sectional study was conducted at a tertiary care university hospital and among patients diagnosed as having PE. The control group consisted of healthy volunteers who applied to hospital for a routine checkup examination. Serum apelin 13 levels were measured in both the groups and their results were compared.
Results:
The median ages were 57 and 53 years, and female–male ratios were 30/23 and 20/15, in the PE and control groups, respectively. The mean serum apelin 13 levels were found to be significantly higher in the PE group (76.94 ± 10.70 ng/mL) than in the control group (50.01 ± 7.13 ng/mL; P < .001).
Conclusion:
This study demonstrated that apelin 13 levels are elevated in patients with PE. These results suggest that apelin may be a novel biomarker and a potential therapeutic target in patients with acute PE in the future.
Introduction
A variety of adipokines have been reported in the recent years that contribute to many different systemic processes. One of these novel adipokines, apelin, has become popular in the past decades. Expression and peptide immunoreactivity of apelin messenger RNA (mRNA) have been described in a variety of tissues, including gastrointestinal tract, adipose tissue, brain, kidney, liver, cardiovascular system, and lungs. 1 –4 Interaction between apelin and its receptor APJ may have pathophysiologic effects in those tissues. Apelin-12, -13, -17, and -36 all originate from a common 77 amino acid precursor and are all bioactive forms of this adipokine. 5 It has been suggested that apelin 13 is the final active product of apelin, as it is more resistant to enzymatic cleavage. 6,7 However, it has not been determined which peptide is the best target for investigation. Pulmonary embolism (PE) is a common clinical entity and remains poorly understood, despite advanced diagnostic methods. Impaired gas exchange, hypoxia in particular, cannot be explained solely on the basis of mechanical obstruction of the pulmonary arteries. 8,9 Different mediators have been reported that contribute to chemical vasoconstriction and gas exchange abnormalities, similar to serotonin and thromboxane in patients with PE. 8,9
We hypothesized that serum concentrations of apelin may be correlated with PE, with this adipokine serving as a lung-derived marker in patients with PE. To date, only animal studies have examined apelin’s involvement in acute PE. 10
In an acute PE, apelin may possess either protective vasodilator or vasoconstrictor effects within the pulmonary vessels. 11 This study aimed to investigate the possible involvement of the endogenous apelin within the pathophysiological events in patients with PE.
Material and Methods
Patients and Setting
In total, 53 patients with PE and 35 healthy volunteers were included the study. This cross-sectional study was conducted at a tertiary care university hospital and among patients diagnosed as having PE. Patients with PE were diagnosed between January 2013 and August 2013 by computed tomography pulmonary angiography. There were 4 high-risk, 32 intermediate-risk, and 17 low-risk patients with PE according to the European Society of Cardiology data.
12
Additional routine diagnostic laboratory tests were performed, including echocardiography,
Patients having any clinical and laboratory abnormalities were excluded from the control group. Venous blood samples were collected before beginning anticoagulation. In order to measure apelin, serum was separated from the blood corpuscles by centrifugation at 5000g for 10 minutes and kept frozen at −80°C until the analysis was carried out. The BMI was calculated as the weight in kilograms divided by the height in meters squared (kg/m2).
The Measurement of Biochemical Markers
Serum samples were obtained as described previously. Serum levels of troponin I was measured the by Immulite 2500 (Siemens Medical Solutions, Holliston, Massachusetts), with enzyme-labeled chemiluminescent immunometric assay method (reference interval 0-0.04 ng/mL). Serum BNP concentrations were determined using a chemiluminescent immunoassay method (Immulite 2500; Siemens Medical Solutions, with a reference level of 0-100 pg/mL).
Determination of Serum Apelin Concentrations by Enzyme-Linked Immunosorbent Assay
Serum samples were obtained as described earlier. The group assignment of all samples was blinded, and all samples were tested in technical duplicates. The amount of apelin in each sample was detected using a human apelin 13 enzyme-linked immunosorbent assay (ELISA) Kit (Hangzhou Eastbiopharm Co, Ltd; Cat no. CK-E11153, assay range 0.5 ng/L to 200 ng/mL, Hangzhou, China). Assays were performed according to the manufacturer’s instructions.
Statistical Analysis
Data analysis was carried out using a SPSS (Version 15.0; SPSS Inc, Chicago, Illinois). The Kolmogorov-Smirnov test was used to test for normality of continuous variables’ distribution. Data characterized by a normal distribution were expressed as mean and standard deviation. Variables without normal distribution were expressed as median with range. Student test was used in normally distributed continuous data and Mann-Whitney U test in not normal distributed variables for comparing the 2 groups. Categorical variables were compared using the chi-square or Fisher exact test. The Pearson or Spearman correlation analyses were performed for correlation analyses. A P value less than .05 was accepted as statistically significant.
Results
The median ages were 57 and 53 years in the PE and control groups, respectively. The difference was not statistically significant (P = .233). Female–male ratios were 30/23 and 20/15, and mean BMI values were 24.95 ± 2.99 and 26.25 ± 3.21 in the PE and control groups, respectively. There was no difference between the 2 groups in terms of gender distribution and mean BMI values (P = .960 and P = .846, respectively). The baseline laboratory parameters of patients with PE are given in Table 1. The mean serum apelin 13 levels were found to be 76.94 ± 10.70 and 50.01 ± 7.13 ng/mL in the PE and control groups, respectively. The difference was statistically significant (P < .001; Figure 1). There were no correlations between serum apelin levels and BNP levels (P = .108, r = −.224), troponin I levels (P = .225, r = −.170), oxygen saturation (P = .789, r = .038), partial oxygen pressure (P = .545, r = .085) in arterial blood gas analysis, and pulmonary artery maximum pressure in echocardiography (P = .096, r = .231).
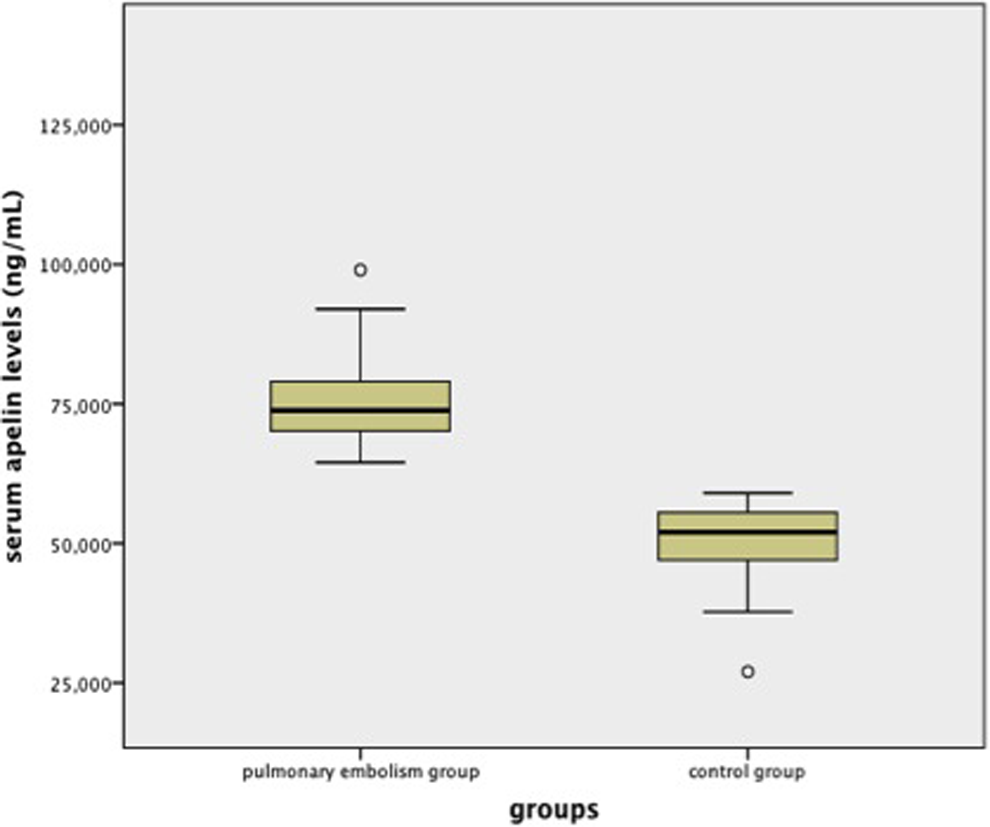
Box plot showing serum apelin levels between patients with pulmonary embolism and control group.
Baseline Laboratory Parameters of Patients With Pulmonary Embolism.a
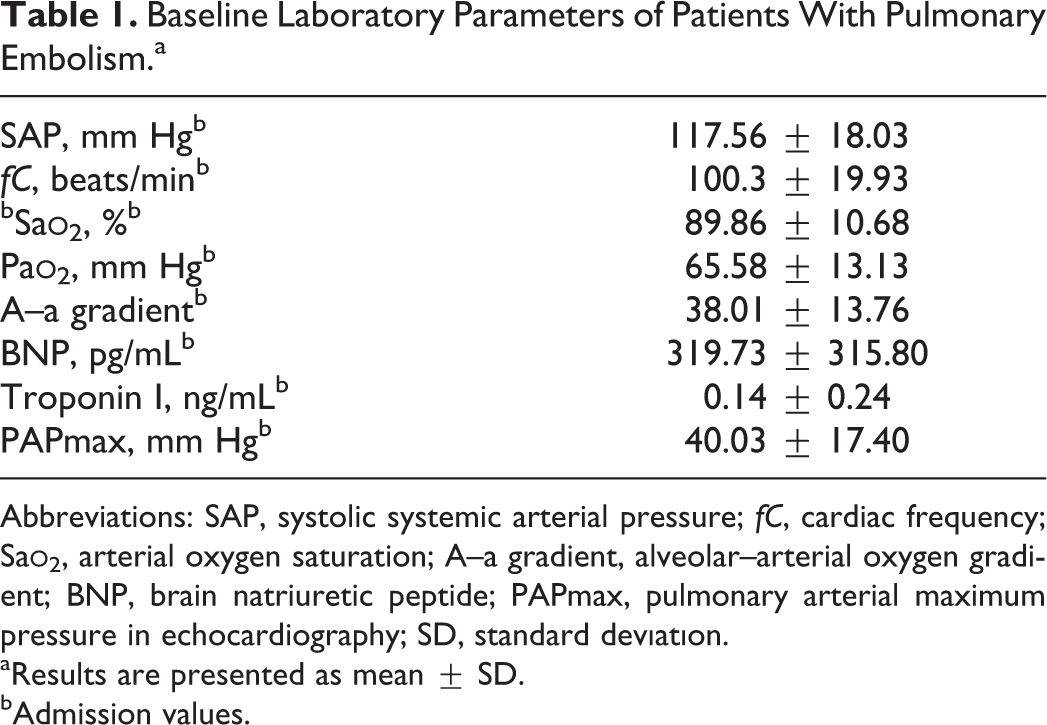
Abbreviations: SAP, systolic systemic arterial pressure; fC, cardiac frequency; Sa
aResults are presented as mean ± SD.
bAdmission values.
Discussion
This is the first study to investigate the serum apelin 13 levels in patients with PE. The results of the study showed that PE is associated with increased serum apelin 13 levels compared to healthy volunteers.
Recently, it has become apparent that adipose tissue is an active endocrine and paracrine organ that releases several bioactive mediators that produce metabolically active proteins, such as apelin, leptin, resistin, and adiponectin, termed adipocytokines. 13 Those substances influence bodyweight homeostasis, inflammation, insulin resistance, and diabetes. 14 However, their precise role in PE has not been fully elucidated. In 1993, O’Dowd et al identified a gene with a similar identity to the angiotensin II type 1 receptor and called it APJ. 15 This receptor was assumed to be an orphan, until 1998 when Tatemoto et al identified a selective endogenous ligand named apelin. 1 Expression of apelin has been reported in large vessels, coronary arteries, and endothelial cells. 16 The vascular endothelial apelin expression is particularly high in pulmonary tissue. 4 Immunohistochemical studies were performed to further characterize the presence of apelin and APJ in the pulmonary tissue. 17 The highest apelin mRNA expression was reported in the lungs; these data suggest that the pulmonary system may be a major source of circulating apelin. 4 Apelin and APJ are synthesized in both the pulmonary artery and the airway. 18 This document suggests that apelin may directly affect the vasomotor and bronchomotor tones and thus influence pulmonary arterial pressure and gas exchange by altering regional ventilation and perfusion. 18 The effect of apelin on vasomotor tone is complicated, as it can cause differential changes in pulmonary and systemic arterial pressures under different physiological and pathological conditions. 10 However, the molecular mechanism as to how apelin affects arterial pressure in various disease states is still unclear. Apelin seems to be a positive inotropic agent and a potent vasodilator via a nitric oxide (NO)-dependent mechanism 2,7,19 but also has been reported to have a direct vasoconstrictive action in both the endothelium-denuded arteries and the veins by inducing phosphorylation of the myosin light chains in vascular smooth muscle cells. 20 A previous study demonstrated that administration of acute apelin in vivo causes NO-mediated arterial vasodilation but does not appear to affect peripheral venous tone. 21 The effect of apelin in pulmonary arteries has been examined in only a few in vitro studies. 22,23 Sheikh at al suggested that the apelin-APJ pathway allows for systemic endothelial monitoring of tissue perfusion. 24 Recently, some reports were published about the prophylactic role of apelin in chronic pulmonary hypertension and the in vitro effect of apelin on pulmonary vasomotion. 25 The changes in plasma apelin levels in heart failure and pulmonary hypertension have brought forward the idea that apelin may serve as a biomarker for these diseases. 26 All of these studies suggest that the blood apelin levels may change in PE and affect prognosis. In our study, we found significantly increased serum apelin levels in patients with PE compared with healthy participants. However, we did not perform a prognostic evaluation in this study.
Acute right heart failure due to the abrupt increase in pulmonary arterial pressure is the principal cause of death in PE. 27 The current treatment of PE is focused on removing the mechanical obstruction in the pulmonary vessels. There is accumulating experimental evidence suggesting that pharmacological blockade of pulmonary vasoconstriction attenuates the hemodynamic changes associated with PE. 27,28 A transgenic mouse expressing higher amounts of adiponectin in the blood was protected in a murine arterial thrombosis model, whereas the adiponectin knockout mice had enhanced thrombus formation. 29 A previous study reported that there was no obvious role of the endogenous apelin—in the pulmonary arterial wall in the regulation of the pulmonary vascular tone in the acute phase of PE. 22 In another study, the level of endogenous apelin in the pulmonary arterial wall did not change significantly, whereas the expression of apelin in the bronchial epithelium was upregulated in the early stage of PE. 10 In the acute phase of PE, many vasoactive substances are released, and apelin may have an antagonistic effect on the vasoconstrictors via NO and prostanoid mechanisms. In addition, apelin may play a role by inhibiting reactive oxygen species and increasing constitutive NO synthase (NOS) and decreasing inducible NOS activities. 25,30 Despite all of these data, the net effects of apelin on the vascular system and its mechanisms remain unclear.
The importance of apelin in the pathophysiology of hypoxic conditions has been a topic of discussion in recent years.
24,31
However, the effect of hypoxia on apelin regulation in human adipocytes has yet to be clarified. Hypoxia has been shown to increase apelin expression in endothelial cells, adipocytes, and murine lung tissue.
24,32,33
The decrease in arterial pH and Pa
We observed for the first time elevated levels of apelin 13 in patients with PE. In conclusion, the findings of this study intrigue idea for the possible regulatory role of apelin in PE. More experiments directly in PE model are essential to reveal the substantial role of apelin in the pathogenesis of PE. We hope that subsequent studies will establish background data on further investigations of the molecular mechanisms of the apelin in PE.
Limitations
Serum apelin 13 levels have not been compared according to clinical severity of PE due to the limited number of patients.
Footnotes
Authors’ Note
Drs Selimoglu Sen and Kaplan were involved in the literature search, data collection, study design, analyzing the data, and approval of the manuscript. Dr Abakay was involved in analyzing the data, drafting, and reading. Drs Sezgi, Taylan, and Yilmaz were involved in data collection and analyzing the data. Drs Abakay and Tanrikulu were involved in manuscript preparation and review of manuscript. Dr Tanrikulu was involved in coordination, design, and interpretation of the study and drafting. The cost of the Apelin 13 ELISA kit was beared by the authors.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
