Abstract
Aim:
To determine the utility of platelet (PLT) indices for the prediction of recurrent pregnancy loss (RPL).
Methods:
In all, 208 patients who experienced 2 or more first trimester spontaneous abortions and 95 controls who had no abortions were studied. The hematological markers, including plateletcrit (PCT) and neutrophil (Neu) to lymphocyte (Lym) ratio (NLR), were measured. Thrombophilia genetic tests for factor V Leiden mutation, prothrombin G202I0A mutation, and methylenetetrahydrofolate reductase C677 T and A1298C mutations were performed.
Results:
The PLT count, PCT, white blood cell count, red cell distribution width (RDW), Lym and Neu count, and NLR were significantly higher in patients with RPL than in controls. The RDW, PLT, and PCT values were higher in the low-risk group, whereas mean PLT volume values were lower than the high-risk group values.
Conclusion:
Plateletcrit is a low-cost, widely available marker for prediction of RPL in patients with a history of at least 1 abortus.
Introduction
Recurrent pregnancy loss (RPL) is defined by the World Health Organization as 3 or more miscarriages before 20 weeks of gestation. 1 The etiology of RPL is undefined in 50% of cases and in the remaining cases can be divided into the following 3 categories: definite, probable, and doubtful. 2 Parental chromosomal abnormality, uterine anomalies, polycystic ovary syndrome, antiphospholipid syndrome, and factor V Leiden (FVL) play a definite role in cases with RPL. 2 Probable associations with RPL include uncontrolled thyroid disease or diabetes, T-helper 1 cell cytokine bias, increased natural killer (NK) cell cytotoxicity, and maternal human leucocyte antigen levels. 2
During hemostatic disturbances in pregnancy, the thrombophilia risk is increased, which causes obstetric complications such as abortions, intrauterine fetal death in early pregnancy, and preeclampsia or placental abruption in the second or third trimester. The increase in prothrombotic factors and decrease in antithrombotic factors result in modification of the hemostatic balance in placental vessels, causing insufficient placental perfusion and fetomaternal circulation. 3 The most common hereditary thrombophilias causing thrombosis are the FVL G169IA mutation, prothrombin (PT) G202I0A mutation, and methylenetetrahydrofolate reductase (MTHFR) mutations. 4
The management of thromboembolic diseases can be more successful if activation of the coagulation cascade is detected early. Platelet (PLT) volume is directly related to increased PLT synthesis 5 that, along with increased PLT distribution width (PDW), causes PLT aggregation. However, plateletcrit (PCT), which is a measure of the mean platelet volume (MPV) × PLT count, may be more predictive of PLT aggregation.
After a spontaneous recurrent abortion, decidual inflammation and decidual fibrin deposition are detected during immunopathological evaluation of the implantation site. 6 Polymorphisms in cytokines such as tumor necrosis factor-α and interferon-γ trigger thrombotic and inflammatory processes in maternal blood vessels, causing abortions. 7 The normal physiology of pregnancy requires the presence of sterile inflammation for successful implantation of the embryo. However, persistent and uncontrolled inflammatory responses can damage placental growth, antenatal development, and maternal health. Increased PLT counts and PLT indices have been related to the severity of infection and suggested as acute-phase reactants. 8 Since RPL is correlated with the inflammatory process, PLT indices may be used for prediction of RPL. Thus, in this study, we evaluated changes in PLT indices in patients with an RPL history.
Materials and Methods
Study Enrollment
This retrospective study was performed by reviewing the obstetric history of 16 500 reproductive age women admitted to gynecology outpatient clinics at Bülent Ecevit University Hospital, Zonguldak, Turkey, from August 2011 to December 2013. The study was approved by the local ethics committee of Bülent Ecevit University. The study group included 208 patients who experienced 2 or more first trimester spontaneous abortions. Of these 208 patients, 153 (73.6%) had 2 abortions and 55 (26.4%) had 3 or more consecutive pregnancy losses. The control group included 95 age-matched healthy patients who had not had an abortion and were admitted for other gynecologic problems. This study was approved by the local ethics committee of Bülent Ecevit University. The inclusion criteria of the study group were >18 years, a history of 2 or more consecutive pregnancy losses with no other systemic illness, the absence of any uterine anomaly, no sign of lower genitourinary infection, and the presence of hereditary thrombophilia. Diabetic or hypertensive women, patients with coagulation defects, history of deep vein thrombosis or pulmonary thromboembolism, use of medications affecting PLT functions such as aspirin, nonsteroidal anti-inflammatory drugs, oral contraceptives, hormonal treatments, anti-PLT, and anticoagulant drugs were excluded. Women with at least 1 successful pregnancy and without a history of miscarriage, intrauterine growth retardation, stillbirth, or preeclampsia, systemic disorders, or venous thromboembolism were included as control group.
Complete blood counts (CBCs) of the groups were compared. Patients were instructed not to consume alcohol or tobacco or engage in vigorous exercise for 24 hours. Fasting blood samples at least 12 weeks after the last abortus and during the early follicular period were acquired to rule out pregnancy. A Beckman Coulter LH 780 analyzer (Beckman Coulter, Miami, Florida) was used for the analyses of 22 parameters. White blood cell (WBC) count, 4.8 to 10.8 × 103/mm3; red blood cell count, 4.2 to 6.1 × 106/mm3; hemoglobin (HGB), 12 to 18 g/dL; hematocrit, 37% to 52%; mean cell volume (MCV), 80 to 99 fL, red cell distribution width (RDW), 11.5 to 15.5; PLT, 130 to 400 × 103/mm3; MPV, 7.4 to 10.4 fL, PCT; lymphocyte count (Lym#), 1.3 to 2.9 × 103/mm3; and neutrophil count (Neu#), 2.2 to 4.8 × 103/mm3 were recorded and Neu:Lym ratio (NLR) calculated. The uterus was evaluated by bimanual examination and radiologic examination by ultrasonography and hysterosalpingography, thyroid function tests, glucose, prolactin, and progesterone levels, as well as infectious causes of RPL, such as Chlamydia trachomatis, were ruled out in all study group and healthy patients.
Hereditary thrombophilia genetic tests for FVL, PT G20210A, and MTHFR C677T/A1298C gene polymorphisms were analyzed by polymerase chain reaction. The results of the thrombotic factors were used to classify the study group into low and high risk for thrombophilia groups. The low-risk group included 170 patients heterozygous for FVL or PT heterozygous or hetero/homozygous for MTHFR C677T/A1298C mutations. The high-risk group included 38 patients homozygous for PT or FVL mutations or heterozygous for PT and FVL mutations. Additional thrombotic risk factors, such as protein C, protein S deficiency, activated protein C resistance, and homocysteine levels or autoimmune causes including antiphospholipid antibodies, anticardiolipin antibodies, or lupus anticoagulant causes were eliminated as causative factors.
Statistical Analysis
Statistical analyses were performed using the SPSS ver. 18.0 software (SPSS Inc, Chicago, Illinois). Distribution of data was determined by the Kolmogorov-Smirnov test. Continuous variables were expressed as means ± standard deviations or medians (minimum-maximum), and categorical variables as frequencies and percentages. Continuous variables were compared by an independent sample t test or Mann-Whitney U test, and categorical variables using Pearson chi-square (χ2) test or Fisher exact χ2 test. P < .05 was considered to indicate statistical significance for all tests.
Results
A total of 208 patients with the history of RPL and genetic thrombophilia was compared with 95 participants in the control group who had not experienced a pregnancy loss. The demographic characteristics and obstetric history of all study participants are listed in Table 1. The median age of the control group was 34 years, which was significantly higher than that of the RPL group (P < .001). In the RPL group, the number of gravida was higher, although the number of live births was lower (P < .001).
Demographic Properties of the RPL Group Having Genetic Thrombophilia and Control Group.a
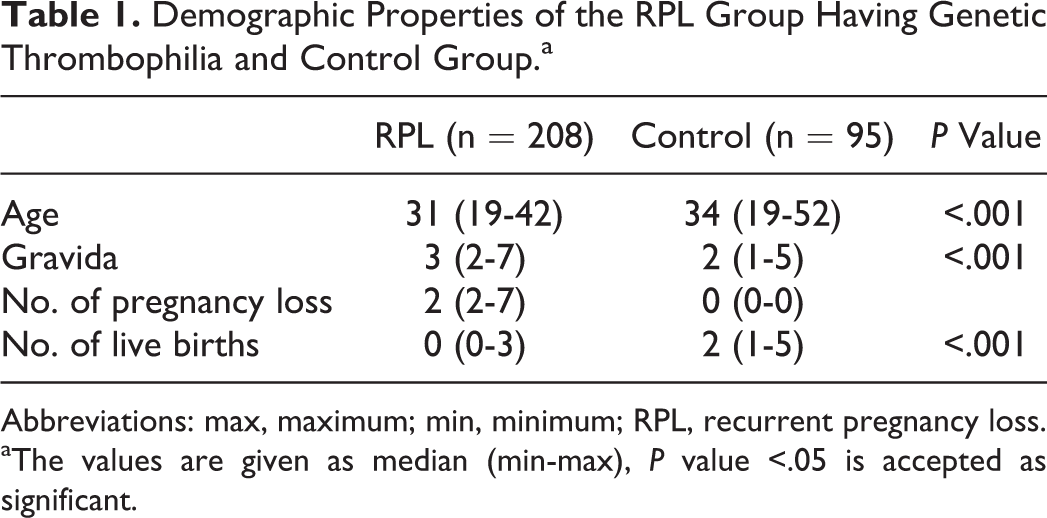
Abbreviations: max, maximum; min, minimum; RPL, recurrent pregnancy loss.
aThe values are given as median (min-max), P value <.05 is accepted as significant.
The frequencies of thrombophilic gene mutations in the low- and high-risk groups are listed in Table 2. Of the 208 patients in the low- and high-risk groups, 186 (89.4%) had 1 or 2 genetic mutations but only 22 (10.6%) had 3 mutations. Of the 170 patients in the low-risk group, 54.2% were heterozygous for MTHFR C677, and no patient was homozygous for PT G202I0A or FVL. There were 21 (55.2%) of the 38 patients in the high-risk group homozygous for PT G202I0A, and 17 (44.7%) were homozygous for FVL. There was no significant difference in the demographic characteristics of the low- and high-risk groups in terms of age, gravida, number of fetal losses, and number of live births.
The Frequencies of the Thrombophilia Gene Mutations in Low- and High-Risk RPL Group.
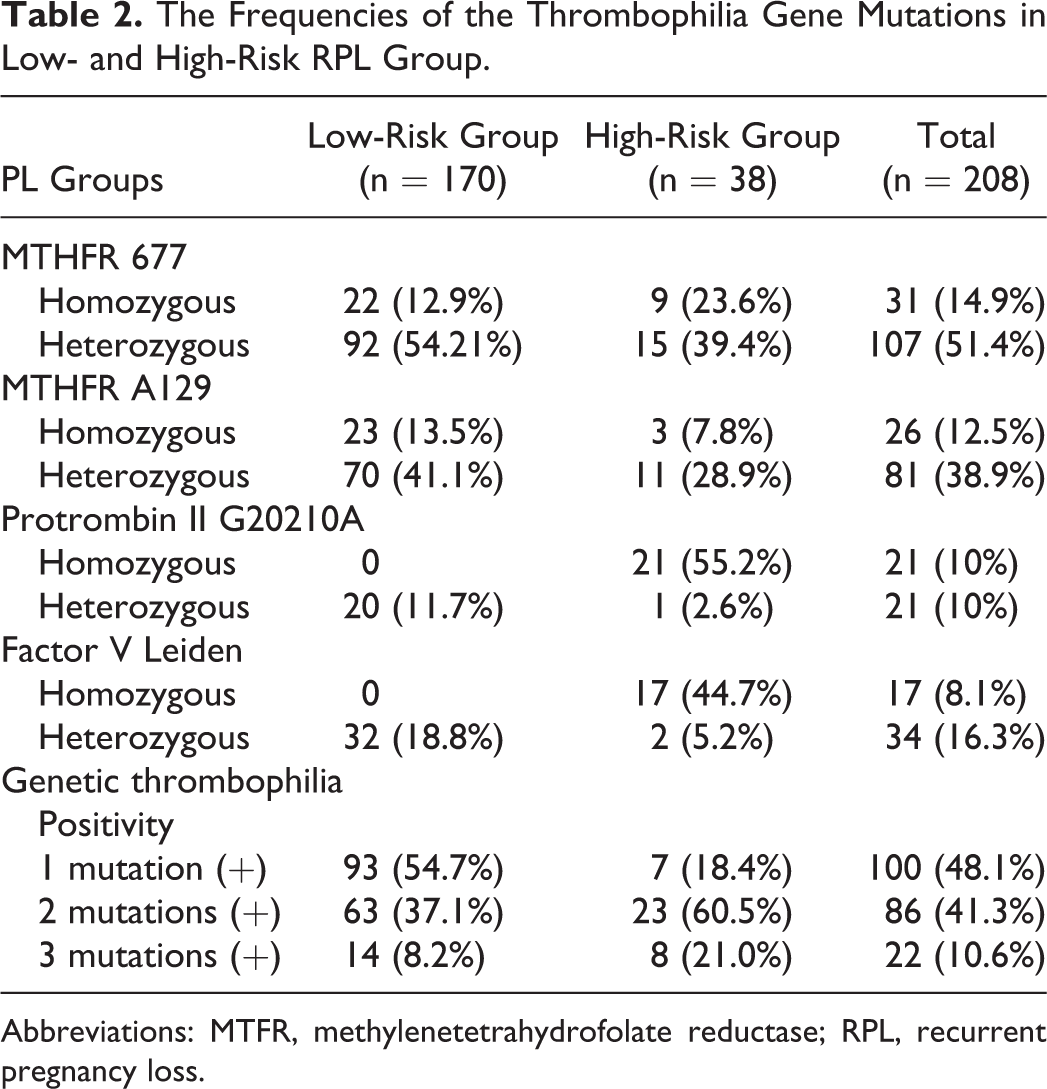
Abbreviations: MTFR, methylenetetrahydrofolate reductase; RPL, recurrent pregnancy loss.
Laboratory Results
Complete blood count data are shown in Table 3. The HGB, HTC, and MCV values were not significantly different between the RPL and control groups, although WBC, RDW, PLT, and PCT values in patients with RPL were significantly higher than those of patients in the control group (P < .001). In addition, MPV values were lower (P < .001), whereas Lym# (P = .034) and Neu# (P < .001) were significantly higher in the RPL group than in the controls. The NLR was also higher in the RPL group than in the controls (P < .001; Table 3).
Hematological Values of RPL and Control Groups.a
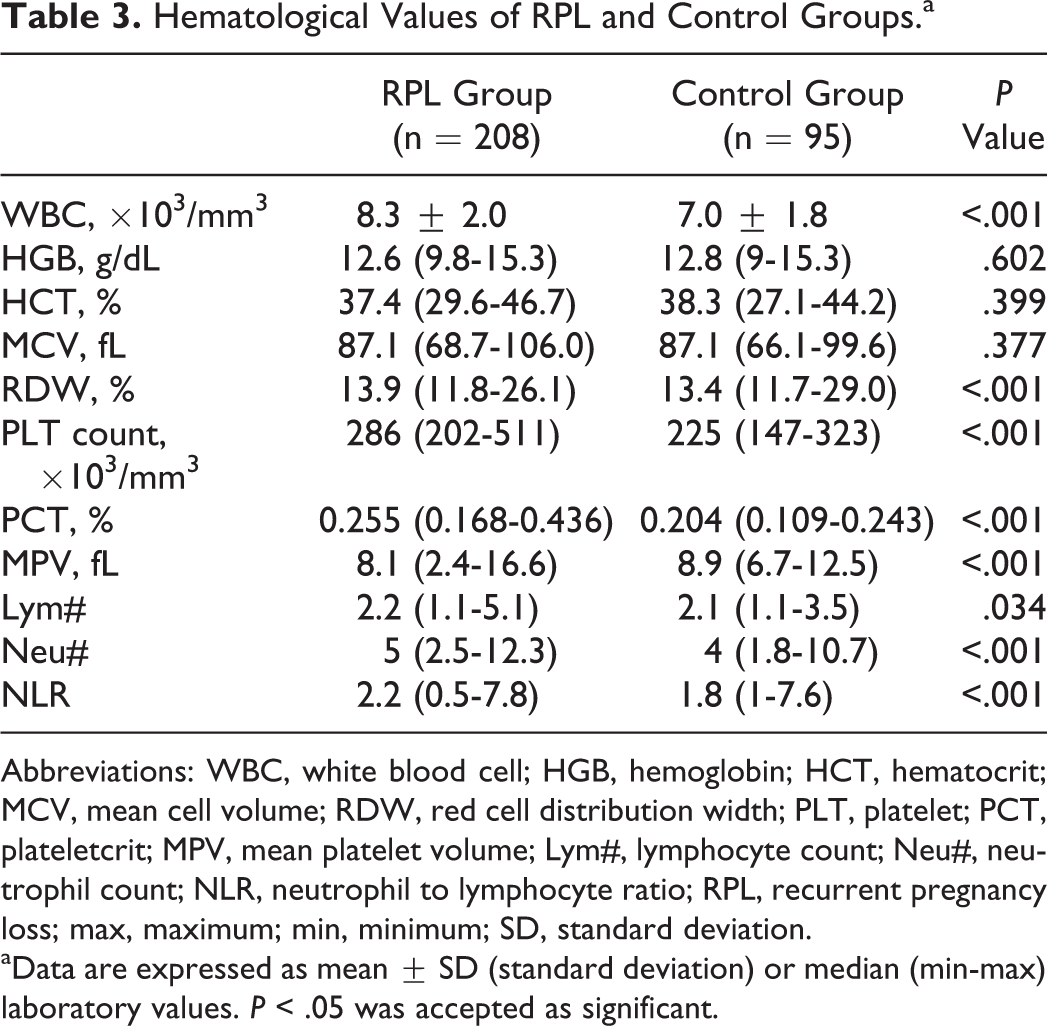
Abbreviations: WBC, white blood cell; HGB, hemoglobin; HCT, hematocrit; MCV, mean cell volume; RDW, red cell distribution width; PLT, platelet; PCT, plateletcrit; MPV, mean platelet volume; Lym#, lymphocyte count; Neu#, neutrophil count; NLR, neutrophil to lymphocyte ratio; RPL, recurrent pregnancy loss; max, maximum; min, minimum; SD, standard deviation.
aData are expressed as mean ± SD (standard deviation) or median (min-max) laboratory values. P < .05 was accepted as significant.
The blood count data of low- and high-risk patients for thrombophilia with RPL are shown in Table 4. The HGB, HTC, MCV, and Lym# and Neu# of the low- and high-risk groups were not significantly different. The NLR in the low- and high-risk groups was not significantly different. The RDW, PLT, and PCT values in the low-risk group were higher, whereas the MPV values were lower than those of the high-risk group (Table 4).
Hematological Values of RPL and Control Groups.a
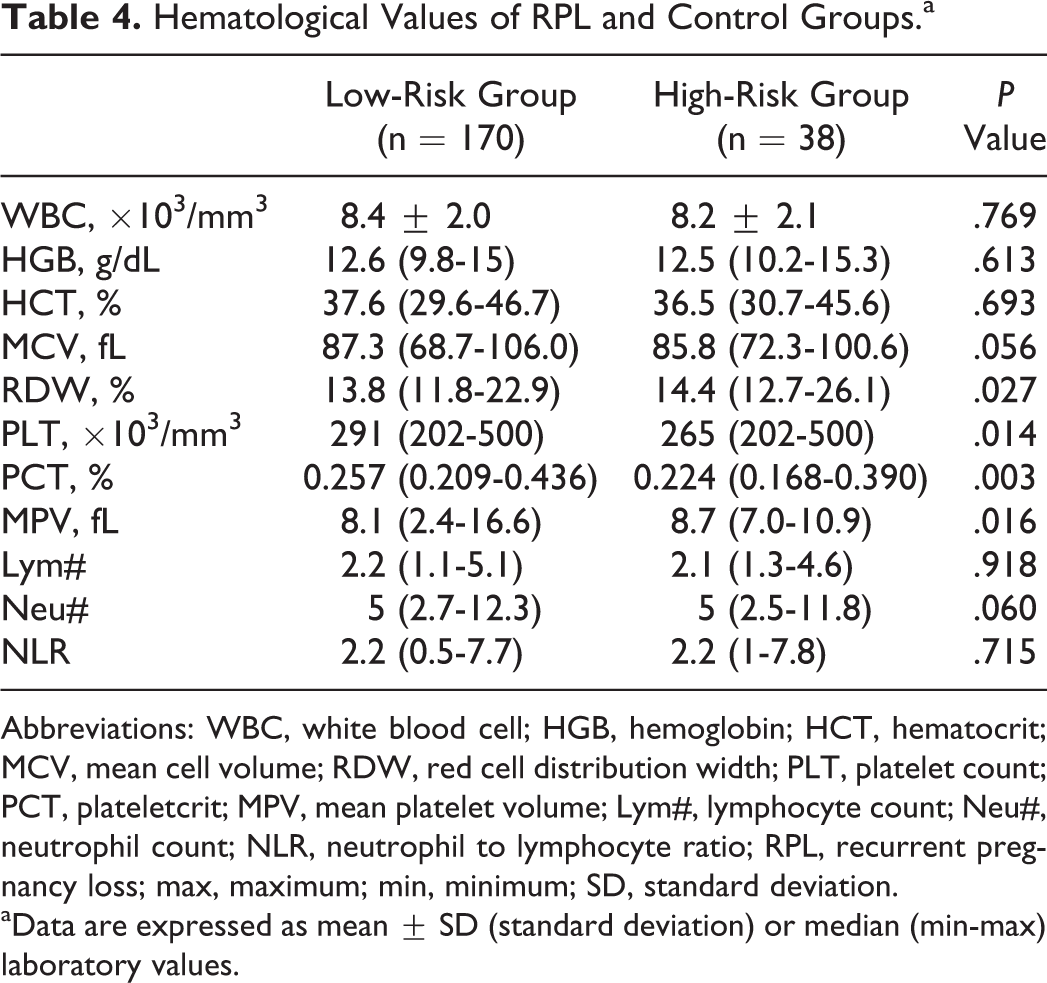
Abbreviations: WBC, white blood cell; HGB, hemoglobin; HCT, hematocrit; MCV, mean cell volume; RDW, red cell distribution width; PLT, platelet count; PCT, plateletcrit; MPV, mean platelet volume; Lym#, lymphocyte count; Neu#, neutrophil count; NLR, neutrophil to lymphocyte ratio; RPL, recurrent pregnancy loss; max, maximum; min, minimum; SD, standard deviation.
aData are expressed as mean ± SD (standard deviation) or median (min-max) laboratory values.
Discussion
Pregnancy is a hypercoagulable state in which the concentrations of coagulant factors, such as factors II, VII, VIII: C, X, α 1-antitrypsin, increase and those of natural anticoagulants, such as protein C, protein S, and antithrombin III, decrease. 9,10 In the case of inherited or acquired thrombotic tendency, an exaggerated hemostatic response may exist during pregnancy. The coexistence of thrombotic tendency and coagulation stimulators induced by pregnancy result in various pregnancy complications, such as RPL. Fibrin deposition and fibrinoid necrosis in the decidual bed as well as thrombi in intervillous spaces occur in RPL, causing fetal hypoperfusion and subsequent fetal loss. 11 During miscarriages, thrombocytic functions change, returning to normal levels 12 weeks after the pregnancy loss. 12 Environmental and hormonal factors, pregnancy, and miscarriage can alter PLT functions. To minimize the effect of these factors on PLT function, blood samples should be obtained during the early follicular period of the menstrual cycle at least 12 weeks after miscarriage, 13 as in our study patients.
In the pathogenesis of RPL, inflammation and coagulation disorders are proposed to have important role, since immunopathological evaluation of the abortus material at the placental implantation site reveals inflammation and fibrin deposition in the decidua and thromboembolism in decidual vessels. 14 Normal pregnancy requires the presence of sterile inflammation for successful embryonic implantation. However, if implantation becomes uncontrolled and placental growth persists, antenatal development and maternal health may deteriorate. 15 Increased NK cells in uterine tissue secrete cytokines that act on the uterus, and embryo loss occurs due to ischemia caused by the activation of vascular endothelial cell procoagulants, resulting in thrombosis and inflammation. 16,17
This study addressed the relationship between recurrent abortions and PCT and PLT indices, which are thought to serve as inflammatory markers for various diseases. Platelet indices have been investigated as inflammatory markers for patients undergoing bypass surgery. Plateletcrit has been suggested as a marker of saphenous vein graft disease, an important cause of morbidity and mortality of patients with coronary artery disease. 18 In addition, changes in PLT are correlated with the severity of infections. 19 Yazici et al reported a correlation between MPV and rheumatoid arthritis. 20 Mete Ural et al stated that the PDW increased with recurrent miscarriage. 21 These results support our finding that the PLT and PCT values were significantly increased in patients with RPL.
We also investigated NLR as a marker of inflammation. The NLR values were significantly higher in the low- and high-risk thrombophilia groups than in the controls. Our results are compatible with other studies that used NLR as a marker of inflammation in carcinomas, spontaneous preterm labor, and invasion of gestational trophoblastic disease. 22 –24
During pregnancy, the thrombotic tendency is increased by changes in coagulation factors or an alloauto immune response to the fetal graft. 15 The association of RPL with both acquired and inherited thrombophilia has been reported, 25,26 although the data remain contradictory. The current guidelines recommend thrombophilia screening tests for people with personal history of venous thromboembolism or first degree relative to a high-risk thrombophilia. 27 Testing inherited thrombophilia in women with recurrent pregnancy loss is not recommended since the benefit of anticoagulant therapy on reducing recurrent fetal loss is unclear. However, in a recent review of Davenport et al, the authors made a survey on physicians obstetricians and reproductive endocrinologists. The survey suggested that around 40% of physicians may screen thrombophilia contrary to current guidelines. These physicians agree that evidence is not adequate to make a definitive association between inherited thrombophilia and pregnancy outcomes. 28
American College of Obstetricians and Gynecologists (ACOG) recommends screening FVL, PT G20210A, protein C And S, and antithrombin III. 27 Most physicians order these tests but greater than 40% of physicians also order MTHFR and homocysteine levels, although not recommended according to the ACOG. However, the screening of MTHR may have been based on the association of MTHFR polymorphisms with neural tube defects. 29 In our study, we also screened MTHFR. Regarding the role of hereditary thrombophilia in RPL, Karata et al suggested that heterozygous and homozygous mutations of MTHFR C677, but not PT and FLV, were significantly different in patients with RPL. 30 Habibovic et al found that the frequencies of the FVL, PT G20210A, and MTHFR C677 T mutations were similar in RPL and control participants. 31 A meta-analysis by Rey et al reported that FVL was associated with early- and late-recurrent fetal loss and late nonrecurrent fetal loss. 32 Ocak et al evaluated polymorphisms in 495 women and found that MTHFR heterozygosity occurred the most frequently. In our study, we also evaluated the inherited thrombophilia pattern in recurrent fetal loss. In low-risk patients, the most common mutation was in the MTHFR 677 gene, while 50% of the genetic mutations were in the PT in high-risk patients. Because of their high cost and the fact that genetic testing for thrombophilia is not covered for normal patients without RPL, control patients with no RPL history were not screened for thrombophilia. However, we classified patients in the RPL group as low or high risk. The PC and PCT values were higher in the low-risk patients, likely attributable to the high number of low-risk cases.
Higher PLT indices have been reported to increase the risk of thrombosis. Gîrleanu et al stated that PLT indices contributed to the prethrombotic state in liver cirrhosis and that larger PLTs might play a specific role in thrombosis, despite thrombocytopenia. 33 Akpek et al reported that increased levels of MPV and PCT were associated with spontaneous echocontrast in patients with mitral stenosis. 34 Spontaneous echocontrast occurs due to the aggregation of cells in blood in situations of stasis or low blood flow velocity. 34 Likewise, in our study, PCT and MPV were significantly increased in patients with RPL. As our knowledge of thrombotic tendency in RPL increases, these economical and easily measurable PLT indices can be used for prediction of fetal loss. In our country, hereditary thrombophilia genes are not screened for unless the patient has a history of 3 or more miscarriages. Lesser known markers, such as PCT, PLT, and MPV, in a CBC panel can be used to predict fetal loss in patients with a history of only 1 abortus. A CBC panel including 8 important parameters, that is, WBC, HGB, thrombocyte count, PCT, MPV, PDW, Neu#, and Lym#, costs only 1€, whereas genetic screening for FVL, PT, and MTHFR mutations costs ˜137€, which is not reimbursed by insurance.
In conclusion, PLT indices are cost-effective, easily measurable, and practical markers for the prediction of RPL. Additional multicenter trials should be performed to confirm the relationship between PLT indices and RPL.
Footnotes
Authors’ Note
Oner Aynıoglu contributed to conception, design, acquisition of data, revised the article, and approved the final version. Hatice Isık contributed to design, acquisition and analysis of data, drafted the article, and approved the final version. Ahmet Sahbaz contributed to design of data, revised the article, and approved the final version. Mehmet I. Harma contributed to interpretation of data, revised the article, and approved the final version. Metin Isık contributed to interpretation of data, revised the article, and approved the final version. Furuzan Kokturk contributed to the analysis of data, revised the article, and approved the final version. In this article, the language has been checked by at least 2 professional editors, both native speakers of English. For a certificate, see ![]() .
.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
