Abstract
Abnormal cellular adhesion is one of the primary causes of vaso-occlusive crisis in sickle cell disease (SCD). Levels of intercellular adhesion molecule 1 (ICAM-1) and P-selectin are upregulated, resulting in increased adhesion of leukocytes and sickle red blood cells (RBCs) to endothelium. This study compares the inhibitory effect of a sulfated nonanticoagulant heparin (S-NACH) derivative with a low-molecular-weight heparin, tinzaparin, on the adhesion of sickle RBCs to endothelium. The S-NACH exhibits minimum effects on hemostasis and bleeding and interferes with the binding of pancreatic cancer cells to endothelial cells via P-selectin. We show by static binding assay that pretreatment of both erythrocytes and endothelial cells with S-NACH significantly inhibits the increased adhesion of sickle RBCs to endothelial cells. The S-NACH treatment also decreases the higher plasma levels of (adhesion biomarkers) ICAM-1 and P-selectin in SCD mice. This investigation signals further research into the potential use of S-NACH in treating vaso-occlusions with minimal bleeding events in patients with SCD.
Keywords
Introduction
Sickle cell disease (SCD) is one of the most commonly inherited hemoglobinopathies, caused by a mutation in the gene that encodes for β-globulin; valine is substituted for glutamic acid at the sixth position on the β-globulin chain. In the United States, 2 million Americans of African descent are carriers for this mutation, and approximately 72 000 people are affected by SCD. 1 –3 Clinically, SCD is characterized by thrombosis, chronic hemolytic anemia, and vaso-occlusive crisis. 1,4
Vaso-occlusion occurs due to erythrocyte sickling caused by hemoglobin (Hb) polymerization and interactions between proadhesive population of sickle cells and the endothelial cells of the vasculature. 5,6 Almost every aspect of hemostasis, such as platelet function, the procoagulant and anticoagulant, and fibrinolytic systems, is altered in SCD leading to the view that SCD is in a “hypercoagulable state.” 7 On the surface of sickle red blood cells (RBCs), increased expression of adhesion molecules such as integrin α4β1, CD36, band 3 protein, sulfated glycolipid, Lutheran protein, phosphatidylserine, and integrin-associated protein make them adhesive to bind to P-selectin, E-selectin, vascular cell adhesion molecule 1 (VCAM-1), CD36, and integrins present on endothelial cells. Treating endothelial cells by specific agonists increases their adhesion and causes cell contractions, which leads to the exposure of extracellular matrix proteins, such as thrombospondin (TSP), laminin, and fibronectin. Plasma proteins such as von Willebrand factor and TSP also participate by bridging the adhesive interactions. 8
Pain associated with vaso-occlusion experienced by patients with SCD varies tremendously in frequency and severity. We have previously shown that both during the steady state and pain crisis, the plasma levels of several chemokines and cytokines such as tumor necrosis factor α (TNF-α), interleukin 1β (IL-1β), IL-6, IL-8, monocyte chemoattractant protein 1, macrophage inflammatory protein 1α, and interferon-γ were significantly increased compared to the plasma of healthy donors.
9
The plasma levels of various growth factors including human vascular endothelial growth factor, human basic fibroblast growth factor, and human hepatocyte growth factor showed a sustained 2- to 3-fold increase along with the biomarker
Pathophysiologic processes such as oxidative stress and inflammation are downstream byproducts of Hb S polymerization, transduced through secondary pathways of hemolysis and vaso-occlusion. 10 Strategies for sickle cell management can be broadly categorized into (1) strategies that attempt to modify the clinical severity of the disease (increase HbF level [hydroxyurea and valporoic acid], reduce dehydration within RBC, prevent RBC membrane damage, prevent abnormal RBC adhesion to other cells [heparin derivatives and antiselectin], vasodilators [NO and others], suppress oxidative stress, inflammation, complement activation, RBCs hemolysis, and hypercoagulation [heparin derivatives]) and (2) strategies that attempt to cure the disease (bone marrow transplantation, gene therapy, or pharmacological intervention that regulate gene expression). A strategy that has impact on these multifactorial pathophysiological processes might provide an effective means of improving the management of SCD.
Current treatment strategies for SCD provide supportive care and anticoagulant medications. Supportive care includes blood transfusions, bone marrow transplantation, and prolonged hydroxyurea treatment. Although there is strong evidence for disease management using supportive care, it is not without serious adverse effects and lack of patient compliance due to the duration of treatment. To manage hypercoagulation and to reduce the episodes of vaso-occlusion, antiplatelet molecules and anticoagulant medications such as heparin are usually considered in order to reduce the episodes of vaso-occlusion. 11
Heparin has been shown to block cell adherence in certain types of tumor, TSP-independent sickle cell adherence, and coagulation processes that are active in SCD. 8,12,13 In an uncontrolled study, reduction in the frequency of sickle cell pain crises was observed upon prophylactic administration of heparin. 14 The demonstration that heparin can inhibit the flow adherence of sickle cells to thrombin-treated human vascular endothelial cells via P-selectin indicates that heparin can also inhibit the P-selectin-dependent adhesion of sickle cells. 8 Although heparin can be administered therapeutically to prevent thrombosis, the effects of natural or unfractionated heparin are more unpredictable, 15 and therefore various methods of heparin depolymerization were used to manufacture low-molecular-weight heparin (LMWH). 16 Because of its ease of administration, lack of need for patient monitoring, longer half-life, predictable pharmacokinetics, and anticoagulant effects, the LMWH tinzaparin is recommended for initial treatment of deep vein thrombosis. 17 A double-blind, randomized, placebo-controlled study confirmed the role of tinzaparin in the management of acute painful vaso-occlusive crisis in SCD, with 2 minor bleeding events. 18 –20 Given its renal clearance, the use of LMWH needs to be monitored closely in patients susceptible to renal dysfunction and in patients with bleeding disorders.
Currently, the use of heparins in the management of vaso-occlusion episodes in SCD is thus limited due to associated bleeding events. We developed a sulfated nonanticoagulant heparin (S-NACH) with low- to no-systemic anticoagulant activity that could be safely administered to patients in higher doses if necessary. 21 We compared the effects of subcutaneously administered S-NACH versus an LMWH (enoxaparin), both dosed at 10 mg/kg in mice, on the coagulation time and time to clot initiation using whole blood with a Sonoclot analyzer (Sienco, Inc., Morrison, Colorado) and thromboelastography. Although enoxaparin significantly prolonged the clotting time and the time to clot initiation, the effects of S-NACH were not different from control. In addition, the activity of factors Xa and IIa was assessed in the mice blood plasma. Although administration of enoxaparin upregulated the activities of both factors Xa and IIa, administration of S-NACH did not increase the activities of these factors compared to control mice plasma. However, both enoxaparin and S-NACH caused similar tissue factor pathway inhibitor (TFPI) release from endothelial cells in vitro. 21 Thus, S-NACH is a nonanticoagulant-oxidized heparin that is devoid of antithrombin binding and inhibition of systemic antithrombin-dependent coagulation factors such as factor Xa and factor IIa but has an optimal releasing capacity of endothelial TFPI. 21,22 We also showed that S-NACH can interfere with the P-selectin-dependent binding of cancer cells to endothelial cells, both in vitro and in vivo. 23
In this study, we test the hypothesis that S-NACH via its antithrombotic effects and by interference with the P-selectin-dependent binding of sickled RBCs to endothelial cells will decrease the adhesion of sickle RBCs to the endothelial cells. By in vitro static adhesion assay, we show here for the first time that S-NACH decreases the increased adhesion of sickle RBCs to endothelial cells. In vivo treatment of an SCD transgenic mouse model with S-NACH significantly lowered the levels of intercellular adhesion molecule 1 (ICAM-1) and P-selectin, molecules that promote adhesion between RBCs and leukocytes to endothelial cells.
Materials and Methods
Reagents
Tinzaparin was obtained from Leo Pharma Inc (Ballerup, Denmark), and S-NACH was synthesized at Rensselaer Polytechnic Institute, as described before. 21 In brief, S-NACH was prepared by fragmenting porcine mucosa heparin by treating periodate-oxidized heparin with sodium hydroxide, followed by reduction with sodium borohydride and acid hydrolysis. Gradient polyacrylamide gel electrophoresis analysis showed a mixture of heparin fragments. The average molecular weight of S-NACH is 4000 Da, and the average molecular weight of tinzaparin is 6500 Da.
Cell Culture
Gelatin was purchased from Sigma-Aldrich (St Louis, Missouri). Human umbilical vein endothelial cells (HUVECs), endothelial cell growth medium (EGM) complete media, trypsin/EDTA, trypsin neutralizing solution, and HEPES-buffered saline solution were obtained from Lonza (Allendale, New Jersey). Tissue culture supplies including flasks and well plates were obtained from Celltreat (Shirley, Massachusetts). Using gelatin-coated culture flasks, HUVECs were cultured in EGM complete media supplemented with 2 mL bovine brain extract and passaged when 80% confluent using trypsin/EDTA solution according to the manufacturer’s instructions. Cells from passages 2 to 6 were used for adhesion assay experiments. Cells were subcultured in a 96-well plate, precoated with collagen, with 20 000 cells per well, and left for 24 hours at 37°C in a CO2 (5%) incubator to form a confluent monolayer.
Packed RBCs Separation
Blood collection from humans with or without SCD was approved by the institutional review boards of Albany Medical Center (Albany, New York) and the Albany College of Pharmacy and Health Sciences. Informed consents were obtained. Normal and sickle blood samples were collected in EDTA tubes from the antecubital vein of healthy volunteers and patients with homozygous sickle cell disease. The RBCs were separated from whole blood by centrifugation (1000
PKH67 Labeling of RBCs
Normal and sickle RBCs were labeled using the PKH67 green fluorescent cell linker kit according to the manufacturer's instructions (MIDI67; Sigma-Aldrich). Briefly, 50 µL (50 × 106 cells) of single RBCs were incubated with 4 μL of PKH67 dye in 2 mL diluent C, provided in the kit, for 5 minutes at room temperature with periodic mixing. The staining was stopped by adding 2 mL of 1% BSA for 1 minute, and the cells were centrifuged (1000
Static Adhesion Assay
The HUVEC monolayer was grown to reach at least >90% confluence to prevent the nonspecific adhesion of RBCs to the gelatin-coated wells. The experiment was repeated 3 times to include 4 groups in sextuplicate: medium only (negative control), TNF-α only (positive control), TNF-α + tinzaparin, and TNF-α + S-NACH. Previously, we tested for the inhibitory effects of S-NACH and tinzaparin on the adhesion of pancreatic cancer cells to the endothelial layer of umbilical cord vein at 1, 5, 10, and 40 µg/mL concentrations and found that tinzaparin inhibited the adhesion of pancreatic cells in a dose-dependent manner, while S-NACH attenuated the cancer cell adhesion by 60% to 75% at 5, 10, and 40 µg/mL. 23 Therefore, here we tested the effects of S-NACH and tinzaparin on binding of RBCs to endothelial cells at the maximum dose of 40 µg/mL. All endothelial cells except the negative control group were treated with TNF-α (10 ng/mL) for 1 hour and rinsed with PBS, followed by treatment with either tinzaparin or S-NACH. In parallel, RBCs were incubated with treatment compounds (tinzaparin and S-NACH) for 45 minutes at 37°C. 25 The media were removed, and 200 µL of labeled RBCs (2 × 106 cells) were added to each well and incubated at 37°C for 45 minutes. Nonadherent RBCs were removed with PBS wash, and then 150 µL of complete medium was added to each well for plate reading using a BioTek microplate reader (BioTek, Winooski, Vermont) at excitation 490 nm and emission 535 nm. 26 In order to quantitate endothelial-adherent RBCs by the fluorescence plate reader, serial 2-fold dilutions of PKH67-labeled normal RBCs were prepared from 2 × 106 to 0.16 × 106 RBCs/well. The number of bound PKH67-labeled RBCs in each sample was calculated from the corresponding standard RBC curve using Excel software (Microsoft, Redmond, Washington).
Animal Studies
The study was conducted at the animal facility of the Veteran Affairs (VA) Medical Center, Albany, New York, in accordance with and approved by the VA IACUC institutional guidelines for humane animal treatment and according to the current guidelines. Both wild-type and SCD mice were maintained under pathogen-free conditions and housed under controlled conditions of temperature and humidity and 12-hour light/dark cycle with ad libitum access to water and food. Mice were allowed to acclimatize for 5 days prior to the start of study.
Transgenic SCD model (
To determine the proper dosing, we relied on our previous studies where we observed a 50% mortality rate in mice treated with 10 mg/kg of tinzaparin and no mortality in animals treated with 5 mg/kg of tinzaparin or 20 mg/kg of S-NACH for 28 days, 27 and we found no significant difference in the bleeding times between animals treated for a week with 10 or 20 mg/kg of S-NACH (Alyahya, 2014). 28 Therefore, in this study, we decided to treat the animals with the well-tolerated doses of tinzaparin at 5 mg/kg and S-NACH at 20 mg/kg to maximize interference with P-selectin-dependent binding of sickle RBCs to endothelial cells.
The SCD mice were randomly divided into 3 groups of 4 animals each. One group of each type was treated subcutaneously with tinzaparin (5 mg/kg), 1 group with S-NACH (20 mg/kg), and 1 group with physiological saline, daily for 7 days. The wild-type animals served as control. Blood samples were taken from the animals by retro-orbital venipuncture into citrated microhematocrit tubes, both before and an hour after treatment, and centrifuged immediately (800
Measurement of P-Selectin and ICAM-1
Levels of adhesion biomarker ICAM-1 were measured in mice plasma using the Bio-Plex 200 system (Bio-Rad, Hercules, California). The assay kit (171-304080M) was obtained from Bio-Rad, and the experiment was conducted following the provided instructions. The assay principle is based on the concept of sandwich enzyme-linked immunosorbent assay (ELISA) where magnetic beads are covalently bound to the capture antibodies that react to the targeted biomarker. Second detection antibodies with streptavidin–phycoerythrin conjugate complete the sandwich complex. P-selectin levels in the plasma samples were quantified using specific ELISA assay (R&D Systems, Minneapolis, Minnesota) according to the manufacturer’s instructions.
Statistical Analysis
Results are presented as the means ± standard deviations comparing experimental and control groups.
Results
Sickle RBCs Exhibit Strong Adherence to HUVECs
To compare the adhesion of sickle RBCs and normal RBCs to HUVECs, RBCs were isolated from respective blood samples. The PKH67 labeling of RBCs was uniform, intense, with no leaking and no cytotoxicity to the cells (Figure 1A and B). The standard curves of PKH67-labeled RBCs demonstrated good linearity over the range of RBCs per well measured (
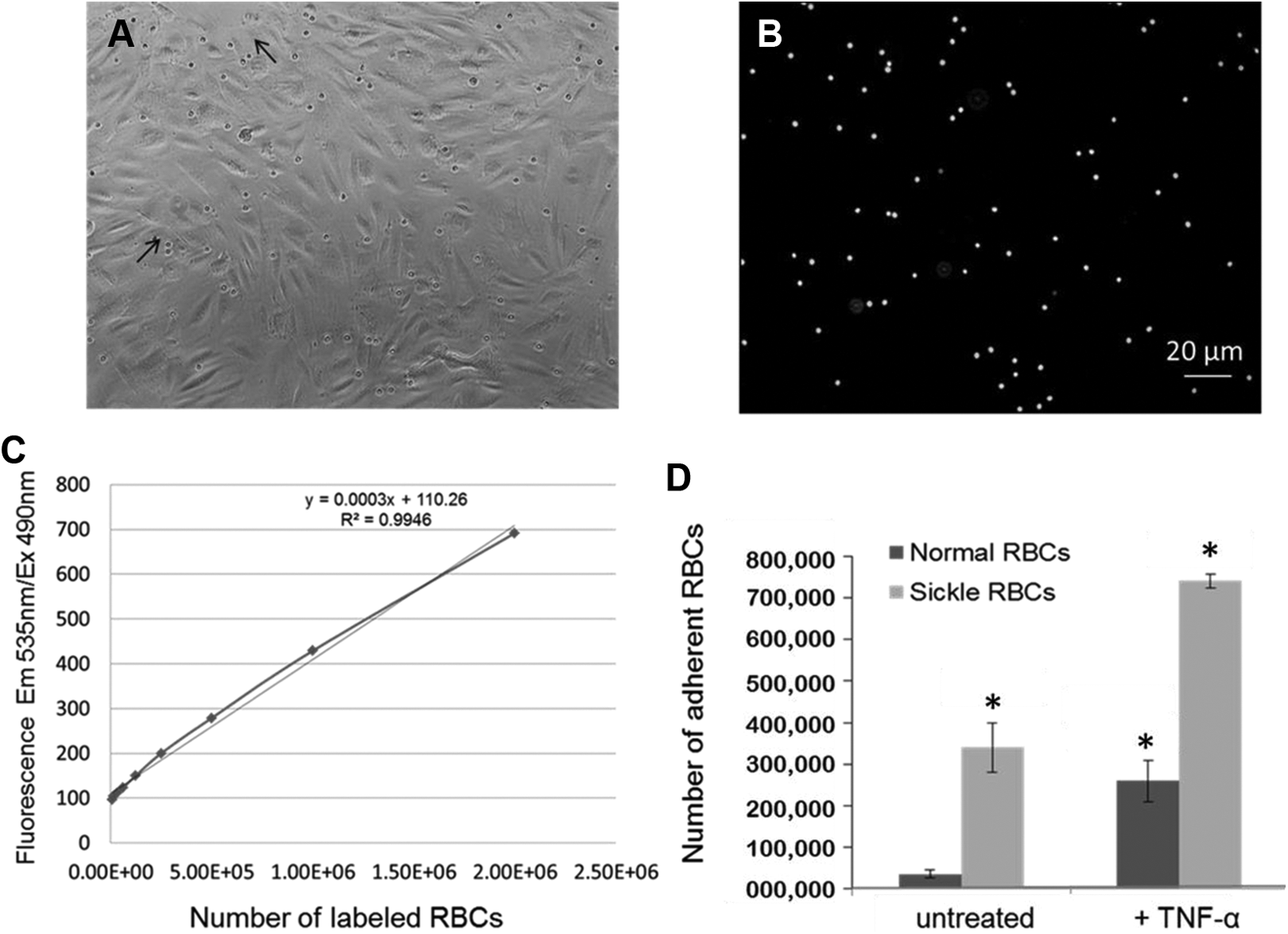
Sickle RBCs adhere more than normal RBCs to the HUVECs monolayer. A, The HUVECs monolayer is >90% confluent to prevent RBCs from adhering to the base of the wells. Small rounded cells on top of HUVECs are bound RBCs, as indicated by arrows. B, Bound RBCs labeled with PKH67 dye. Note that all RBCs show intense labeling with no leaking of the dye onto HUVECs. C, Standard curve of PKH67-labeled RBCs, demonstrating good linearity over the range of RBCs per well (
S-NACH Inhibits the Binding of Sickle RBCs to HUVECs
After confirming that sickle RBCs exhibit increased adherence to the HUVECs by static adhesion assay, we tested the effects of S-NACH on the adherence of sickle RBCs to HUVECs. As shown in Figure 2A, normal RBCs showed increased binding to HUVECs treated with TNF-α and remained unaffected even after treatment with tinzaparin and S-NACH. However, the increased adherence of sickle RBCs to the TNF-α-treated HUVECs decreased significantly by 5-fold (
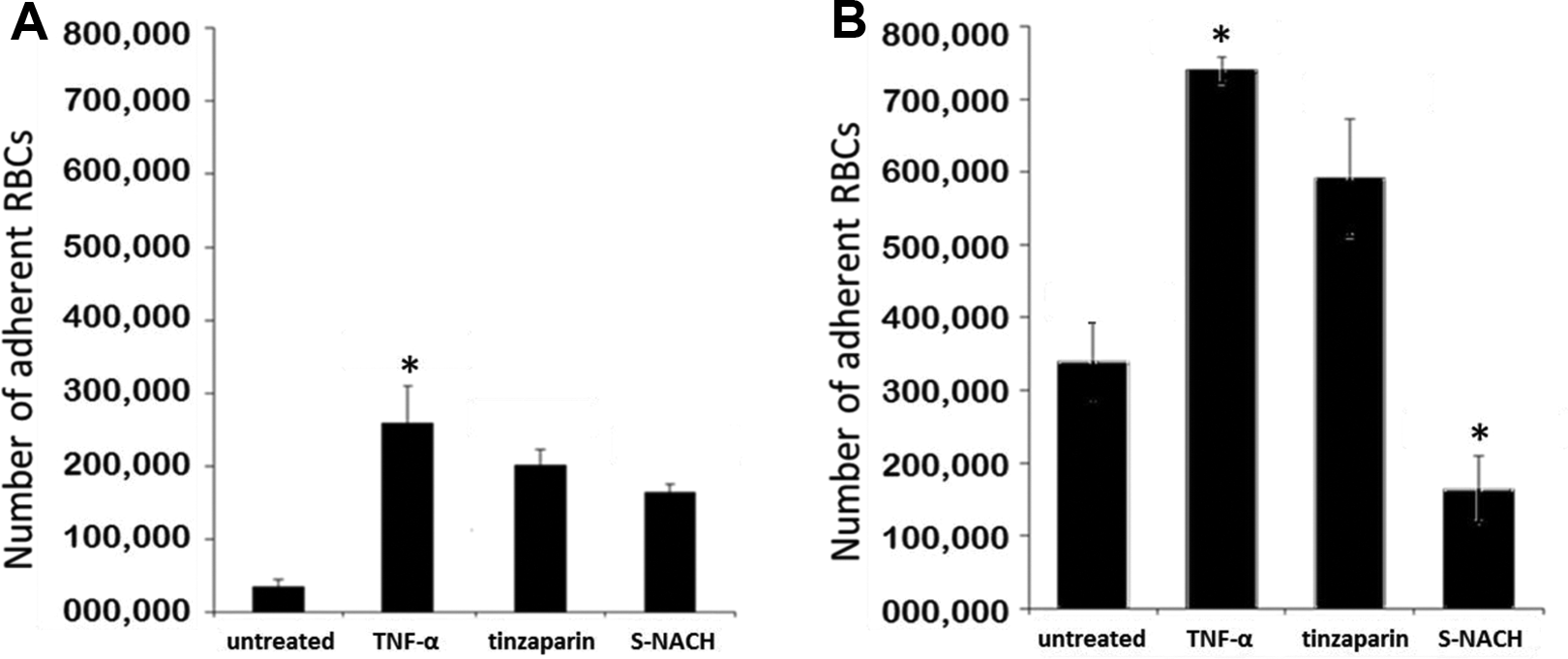
The S-NACH can effectively decrease the adhesion of sickle RBCs to the endothelial layer. A, The effect of treatment compounds on the adhesion of healthy RBCs to the endothelial layer. Healthy RBCs do not show significant adherence to the HUVECs, while they bind 10-fold more to HUVECs treated with TNF-α (
Increased Levels of Soluble ICAM-1 in the Plasma of SCD Mouse Model Are Reversed by S-NACH
We compared the plasma ICAM-1 levels between healthy wild-type mice and the SCD transgenic mice. The SCD mice showed significantly higher levels of ICAM-1 in their blood plasma (
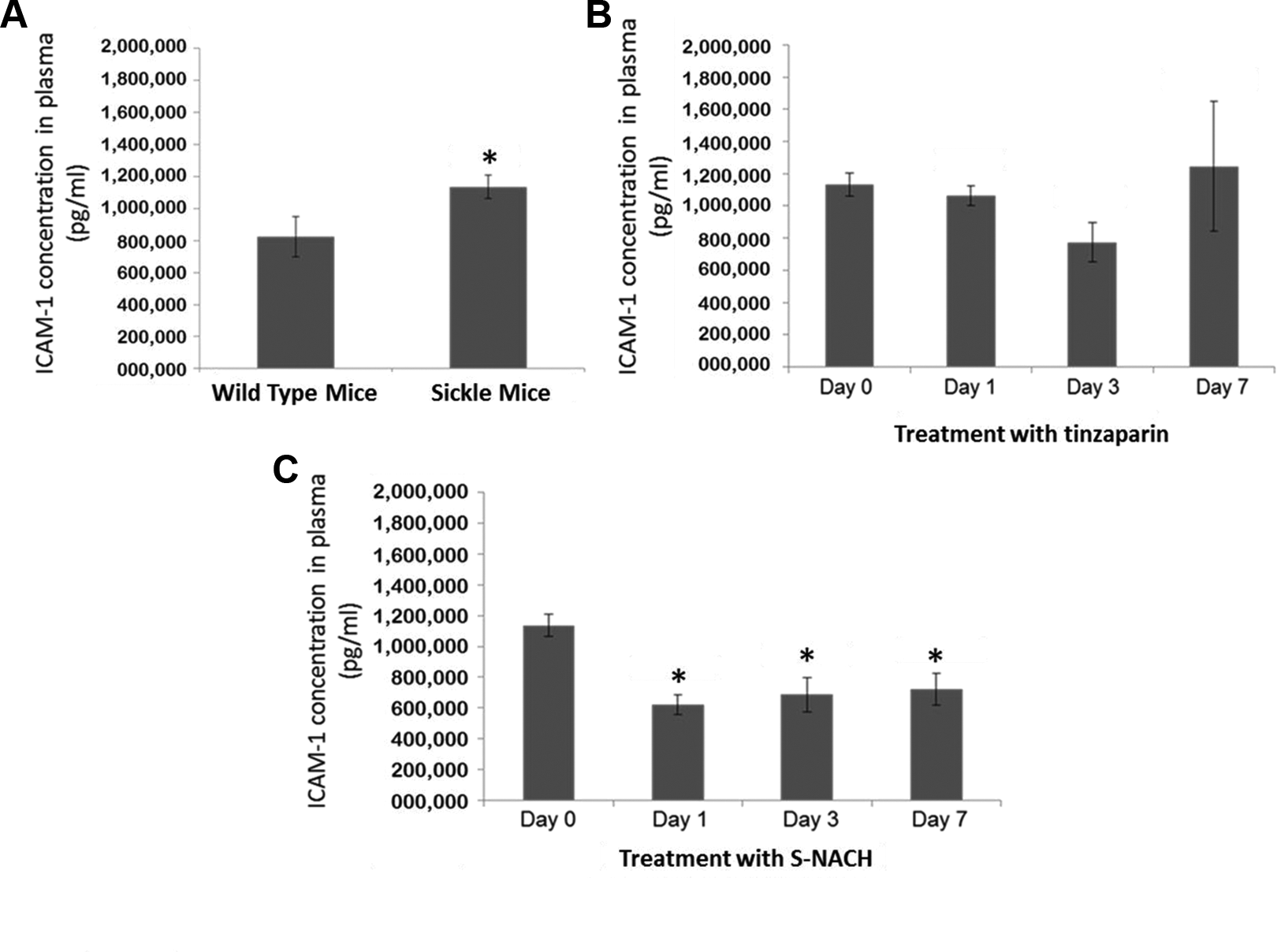
The S-NACH treatment decreases the ICAM-1 levels in the plasma of sickle mice. A, Increased ICAM-1 levels in the plasma of transgenic sickle cell mice (n = 4) in comparison to wild-type mice (n = 4). Compared with healthy wild mice, the plasma of transgenic sickle mice showed significantly higher levels of ICAM-1 (
Increased Levels of Soluble P-Selectin in the Plasma of SCD Mouse Model Are Reversed by S-NACH
Since administration of S-NACH lowered the levels of ICAM-1, we next tested the levels of P-selectin in the plasma of the mice treated with S-NACH and tinzaparin. We first compared the P-selectin levels between the plasma of wild-type mice and SCD mice. The plasma of SCD mice showed significantly higher levels of P-selectin (
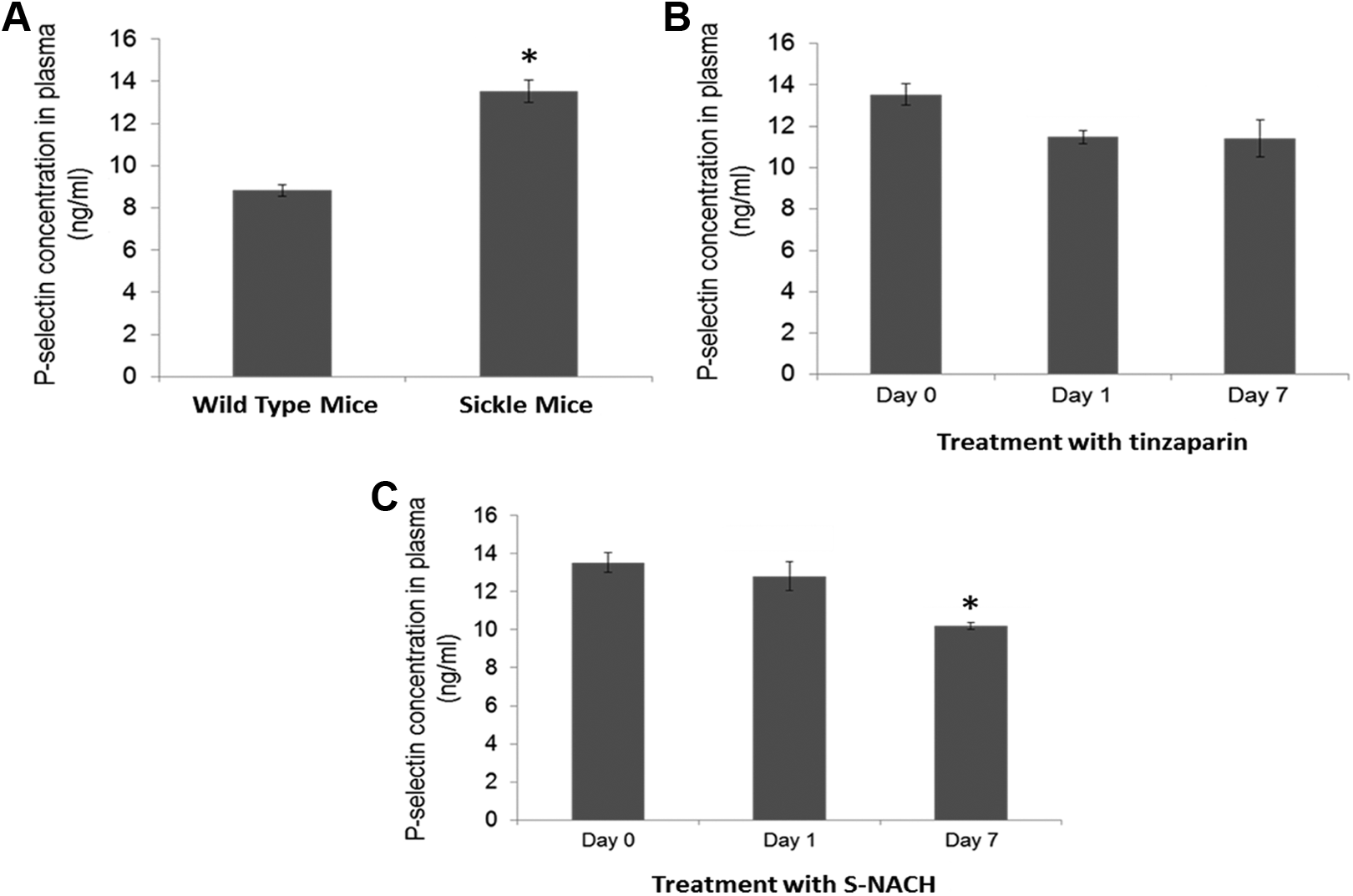
The S-NACH treatment decreases the P-selectin levels in the plasma of sickle mice. A, Increased P-selectin levels in the plasma of transgenic sickle cell mice (n = 4) in comparison to wild-type mice (n = 4). Compared with healthy wild mice, the plasma of transgenic sickle mice showed significantly higher levels of P-selectin (
Discussion
Using an in vitro static adhesion assay and an SCD mouse model, we show that S-NACH can inhibit the in vitro adhesion of sickle RBCs to endothelial cells and lower the levels of biomarkers ICAM-1 and P-selectin in the plasma of sickle transgenic mice. In the static adhesion assay, sickle RBCs showed higher adhesion to endothelial cells, compared to normal RBCs, consistent with previously published findings. 29 Addition of TNF-α to the endothelial cells increased the expression of adhesion molecules, enabling increased adhesion of RBCs to even higher levels. This mimics the in vivo condition in SCD where there are elevated levels of inflammatory biomarkers including TNF-α. 9 This increased adhesion of sickle RBCs to endothelial cells treated with TNF-α was significantly inhibited by treatment with S-NACH. Although treatment with tinzaparin also decreased the adhesion of sickle cells to TNF-α-treated endothelial cells, the inhibition was not as significant as S-NACH. One of the major advantages of the static adhesion assay we have developed is the use of nonradioactive dye (PKH67) to label RBCs. Labeling of cells with the PKH67 green fluorescent dye occurs through its long aliphatic carbon tails, which bind to the lipid bilayer of the cell membrane. 30 This avoids the use of radioactive label sodium chromate (51Cr), which requires special handling and has limitations.
In vitro static adhesion assays and in vivo flow analysis of RBCs in wild-type, P-selectin knock-out mice and sickle cell mice by computer-assisted intravital microscopy indicate that sickle RBCs show greater adherence to the endothelial cells, due to increased expression of P-selectin in SCD.31,32 Soluble ICAM-1 levels are significantly increased in patients with SCD, and ICAM-1 has been suggested to be one of the factors that promote increased adhesive events between the blood vessel endothelium and red and white cells in the initiation of vaso-occlusive crisis SCD. Levels of ICAM-1 in the plasma of patients with SCD were reduced by treatment with hydroxyurea, possibly reflecting reduced endothelial activation in patients taking hydroxyurea. 33 In addition, the biomarkers ICAM-1 and P-selectin have been well established to reflect the process of leukocytes–endothelial adhesion, and we wanted to test whether their levels are altered by test compounds that affect the adhesion of erythrocytes to endothelium. Both ICAM-1 and P-selectin in the basal state were significantly higher in transgenic mice than wild mice, as expected. At dosages that were well tolerated by the animals, treatment with tinzaparin appeared to lower the P-selectin levels, and treatment with S-NACH reduced the levels of both ICAM-1 and P-selectin levels consistently. Indeed these data corroborate with our finding that pretreatment of mice with S-NACH significantly reduces the levels of TNF-α, IL-1, and exotoxin within the first hour of lipopolysaccharide treatment (unpublished results). Mechanistically, we suggest that S-NACH exhibits local antithrombotic activity through the release of TFPI with minimal effect on hemostasis and bleeding and inhibits the adherence of sickle cells to endothelial cells via P-selectin. This study has its limitations, and useful data would be added if the static adhesion assay was performed at various time points on the blood of treated animals and if the treatment time of mice was extended beyond 7 days.
Abnormal cellular adhesion to the endothelial layer of blood vessels is one of the primary initiating factors in the development of vaso-occlusive crises in SCD. 28 This abnormal adhesion is mediated through different adhesion molecules on leukocytes, sickle erythrocytes, and endothelial cells.8,34 Some of these adhesion molecules including E-selectin, P-selectin, ICAM-1, and VCAM-1 exist as soluble forms circulating in plasma, in addition to their presence on the cellular surface. The shedding of these membrane-bound adhesion molecules into plasma is elevated during inflammatory conditions due to cytokines activation of the cells, but the mechanism of shedding is still largely unknown. 35 Using targeted therapies that inhibit the abnormal adhesion may enable control of the harmful side effects of inflammation and vascular injury and prevent painful crisis.
In conclusion, by using an in vitro adhesion assay and an SCD mouse model, we show that S-NACH can effectively inhibit the adhesion of sickle RBCs to endothelial cells and decrease the levels of ICAM-1 and P-selectin in the plasma of SCD mice. These results in combination with minimum associated bleeding with S-NACH treatment even at higher concentrations paves the way as a proof of concept that S-NACH has the potential to be used in the treatment of vaso-occlusion crisis in patients with SCD. This study also opens an area of research to study the effects of S-NACH on other factors of SCD pathogenesis such as inflammation, hypercoagulation, and nitric oxide depletion.
Footnotes
Acknowledgments
We appreciate Dr Kelly Keating (Pharmaceutical Research Institute [PRI] at the Albany College of Pharmacy and Health Sciences) for her excellent editing of this manuscript. We would like to thank Mr Alexander Durant in the Animal Laboratory of the Albany Stratton VA Medical Center, (Albany, NY) for maintenance of animals and Dr Robert J. Linhardt (Rensselaer Polytechnic Institute, Rensselaer, NY) for supplying S-NACH.
Authors’ Note
Abdulelah Alshaiban and Vandhana Muralidharan-Chari contributed equally to this work.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: S. A. Mousa holds a US patent on S-NACH. 22
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
