Abstract
Effects of unfractionated heparin and low-molecular-weight heparins (LMWHs) on human microvascular endothelial cell sprouting (tube formation assay) in vitro were determined. Antiangiogenesis efficacy of commercially available LMWHs tinzaparin and enoxaparin in the chick chorioallantoic membrane (CAM) model of growth factor-induced angiogenesis was compared. The LMWH tinzaparin was fractionated into different molecular weight (MW) pools by size exclusion chromatography; they inhibited CAM angiogenesis depending on their MW distribution, with optimal inhibition at 8 to 12 kDa and no inhibition at <2 kDa. Tinzaparin demonstrated greater antiangiogenesis efficacy than enoxaparin (P < .001); these CAM results correlated with the endothelial tube formation assay results (P < .001, tinzaparin vs enoxaparin). These data point to the variable antiangiogenesis efficacy of different LMWHs as a function of MW and perhaps other structural differences. Our hypothesis confirmed a relationship between lower release of tissue factor pathway inhibitor by lower MW fractions of tinzaparin or enoxaparin leading to reduced antiangiogenesis efficacy.
Keywords
Introduction
Low-molecular-weight heparins (LMWHs) have been shown to be at least as effective and safe for initial anticoagulation as compared with unfractionated heparin (UFH) in individuals with acute venous thromboembolism (VTE) and have become the focus of much interest in the setting of VTE in patients with cancer. Angiogenesis, the formation of new blood vessels, is vital for the maintenance of vascular integrity. Although angiogenesis is normally tightly regulated by multiple cytokines and growth factors, pathologic angiogenesis is associated with a variety of disorders, including cancer. Agents with antiangiogenic activity may have therapeutic value in disease states associated with pathologic angiogenesis.1,2
The clinical benefits of UFH and LMWHs for the treatment of thrombotic disorders such as deep vein thrombosis and pulmonary embolism are well established.3,4 Several clinical trials have also shown that treatment with heparin or LMWHs improves the survival of patients with cancer.5–14 However, these effects of LMWH are only evident in certain subpopulations of patients with cancer, such as those with better prognosis. The potential anticancer effects of heparin and LMWHs may be mediated, in part, by mechanisms that interfere with tumor growth and metastasis. As demonstrated by in vitro studies, LMWHs inhibit both fibroblast growth factor 2 (FGF-2) and vascular endothelial growth factor (VEGF)-mediated angiogenesis.15,16 The relative potency against FGF-2 versus VEGF was not determined. One intriguing possibility is that LMWHs promote antiangiogenic activity through the release of endothelial tissue factor pathway inhibitor (TFPI), which is an important endogenous inhibitor of tissue factor/factor VIIa (TF/VIIa).15,17–19
Tinzaparin is the only LMWH that is manufactured by enzymatic digestion of UFH by heparinase. In contrast, enoxaparin and other LMWHs are prepared by different methods of chemical depolymerization. Furthermore, tinzaparin has a higher average molecular weight (MW) and a higher content of molecules, in the MW range above 8000 Da, than enoxaparin. The present study was carried out to investigate the antiangiogenesis efficacy of heparin and LMWHs against FGF-2- or VEGF-induced angiogenesis.
Materials and Methods
Reagents
The LMWHs tinzaparin and enoxaparin were obtained as commercially available samples. All other general reagents not specified were obtained from Sigma Co. (St Louis, Missouri).
Preparation of Tinzaparin MW Fractions
Tinzaparin was fractionated by size-exclusion chromatography on a Sephadex G50 column using 50 mmol/L ammonium acetate as eluent. Five different pools of fractions were collected covering the MW ranges of ≥10, 10 to 8, 8 to 6, 6 to 4, and 4 to 2 kDa. The corresponding average MWs of the different pools were 12.6, 8.6, 7, 5, and 3.1 kDa, respectively. The pools were freeze-dried and then re-dissolved in water. The compounds were isolated by precipitation with ethanol and dried. The characteristics of the compounds are detailed in Table 1 .
Characteristics of Tinzaparin Molecular Weight Fractions
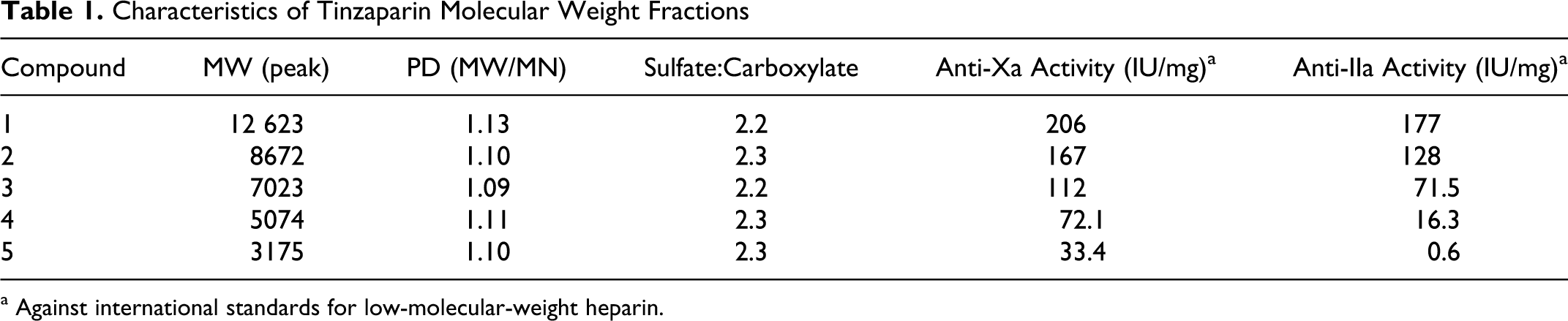
a Against international standards for low-molecular-weight heparin.
In Vitro Human Microvascular Endothelial Cell Sprouting Assay
The endothelial cell (EC) sprouting assay was performed as described previously.20,21 Briefly, 80% confluent human dermal microvascular endothelial cells (HDMECs; passage 5-10) were collected and mixed with gelatin-coated Cytodex-3 beads at a ratio of 40 cells per bead. Cells plus beads (150-200 beads per well for a 24-well plate) were suspended in 5 mL of endothelial basal medium (EBM) + 15% normal human serum (HS) and mixed gently every hour for the first 4 hours, after which the mixture was cultured overnight in a CO2 incubator. Ten mL of fresh EBM + 15% HS were added and the cells were cultured for another 3 hours. Before experiments, the EC-bead cultures were checked by mixing 100 μL of the EC-bead culture with 500 µL of phosphate-buffered saline (PBS) in the well of a 24-well plate. The number of beads was counted in order to calculate the EC-bead concentration. The EC-beads were used for experiments within 48 hours.
A solution of fibrinogen (1 mg/mL) in EBM medium was prepared with or without angiogenic factors and test compound (heparin or LMWHs). As a positive control, 30 ng/mL VEGF + 25 ng/mL FGF-2 alone (no test compound added) was used. The EC-beads were washed twice with EBM and then added to the fibrinogen solution and mixed gently. Each condition was performed in triplicate. Human thrombin (2.5 μL and 0.05 U/μL) was added to 1 mL of fibrinogen solution and then 300 μL of this mixture was immediately transferred to each well containing EC beads. The fibrinogen solution polymerized in 5 to 10 minutes. After 20 minutes, EBM containing 20% of normal HS and 10 μg/mL aprotinin was added to the wells. The plate was incubated in a CO2 incubator. The EC invasion of the gel and tube formation was typically evaluated after approximately 48 hours.
Chick Chorioallantoic Membrane Model of Angiogenesis
The chick chorioallantoic membrane (CAM) model was first described by Auerbach et al 22 and was carried out as implemented in our laboratory.23–31 Briefly, 10-day-old fertilized chicken eggs were incubated at 37°C and 55% relative humidity. In the dark (with the help of a candling lamp), a hypodermic needle was used to puncture a small hole in the shell covering the air sac. A second hole was punctured on the wider side of the egg above the avascular area of the embryonic membrane. An artificial air sac was created below the second hole by applying gentle vacuum to the first hole, using a small rubber squeeze bulb. The vacuum caused the separation of the CAM from the shell. An approximate 1.0 cm 2 window was cut in the shell over the dropped CAM with the use of a mini-drill. The underlying CAM was accessed through this small window. Filter disks (Filter paper #1; Whatman International, Kent, UK) were soaked in 3 mg/mL cortisone acetate solution (95% ethanol and water) and then air-dried under sterile conditions. For the positive controls, sterile filter disks were saturated with FGF-2 (1 μg/mL) or VEGF (1.5 µg/mL); negative control disks were saturated with PBS.
Using sterile forceps, 1 filter/CAM was placed on the CAM through the window cut in the shell. The window was sealed with Highland brand transparent tape. After 24 hours, 10 μL of test compound were added to the CAM topically. After 48 hours, CAM tissue directly beneath the filter disk was harvested and placed in a 35-mm Petri dish. Ten eggs per treatment were used. For digital imaging and analysis, CAM sections in Petri dishes were examined using an SV6 stereomicroscope (Carl Zeiss, Thornwood, NY) at ×50 magnification. Digital images were captured using a 3-CCD color video camera system (Toshiba America; New York, New York) and the images were analyzed using Image-Pro Plus software (Media Cybernetics, Bethesda, MD). The number of vessel branch points within a circular region superimposed on the area of the filter disk were counted for each section. A detailed description and utilization of angiogenesis-related models can be found in several recent publications.23–31
Results
Differential Inhibition of Angiogenesis by Heparin and LMWH
In an in vitro EC sprouting assay of angiogenesis, in the presence of added human plasma proteins (ie, HS), tinzaparin at concentrations of 3.0 and 10.0 μg/mL demonstrated greater potency in inhibiting FGF-2- and VEGF-mediated HDMEC sprouting as compared to enoxaparin (Figure 1A ). In contrast, heparin was ineffective in inhibiting angiogenesis in the presence of human plasma proteins (Figure 1A). The IC50 of tinzaparin was significantly lower (P ≤ .01) than enoxaparin or heparin, whereas there was no difference in the IC50 values of enoxaparin and heparin for inhibition of EC sprouting in vitro.
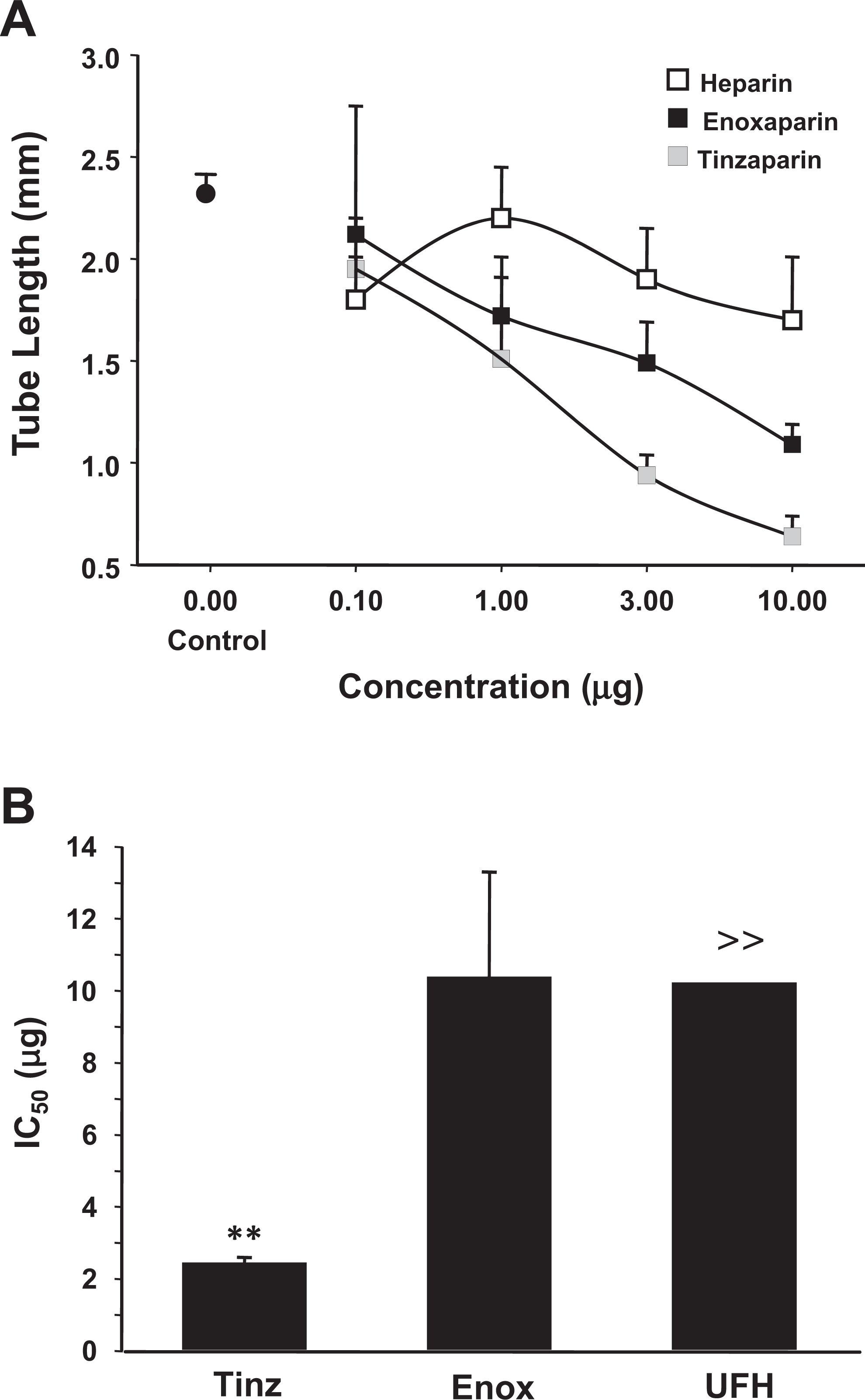
Effect of unfractionated heparin versus the LMWHs tinzaparin and enoxaparin on human microvascular EC sprouting induced by VEGF + FGF-2 in the presence of human plasma proteins. A, Tube length. B, IC50 (µg) values. >> An inhibitory concentration of 40-45% was achieved at 10 μg for UFH. Data represent the mean ± SD; n = 5 for tinzaparin versus enoxaparin, **P < .01. LMWHs indicates low-molecular-weight heparin; EC, endothelial cell; VEGF, vascular endothelial growth factor; FGF-2, fibroblast growth factor 2; SD, standard deviation.
To confirm the results of the EC tube formation assay, we examined the effects of LMWH on angiogenesis in the CAM. The CAM model of angiogenesis is a well-characterized ex ovo system that offers the advantage of being able to directly and quantitatively compare multiple pro- or antiangiogenic compounds and doses in a more physiological setting. We first examined the effects of different heparin fractions on FGF-2-induced angiogenesis. Tinzaparin was fractionated into different average MW subfractions using size-exclusion chromatography (Table 1). In the CAM system, all 3 MW fractions tested inhibited angiogenesis in a dose-dependent manner (Figure 2 ). The higher MW fractions of 12 000 average MW and 8000 average MW demonstrated comparable antiangiogenesis efficacy, and both of the higher MW fractions were significantly more potent than the lower MW fraction of 3175 average MW (Figure 2). These results provided evidence for the differential effects of heparin MW fractions on angiogenesis and suggested that the action of LMWHs in inhibiting FGF-2-induced angiogenesis is mediated predominantly by relatively higher MW molecules in the range of 8000 to 12 000 Da.
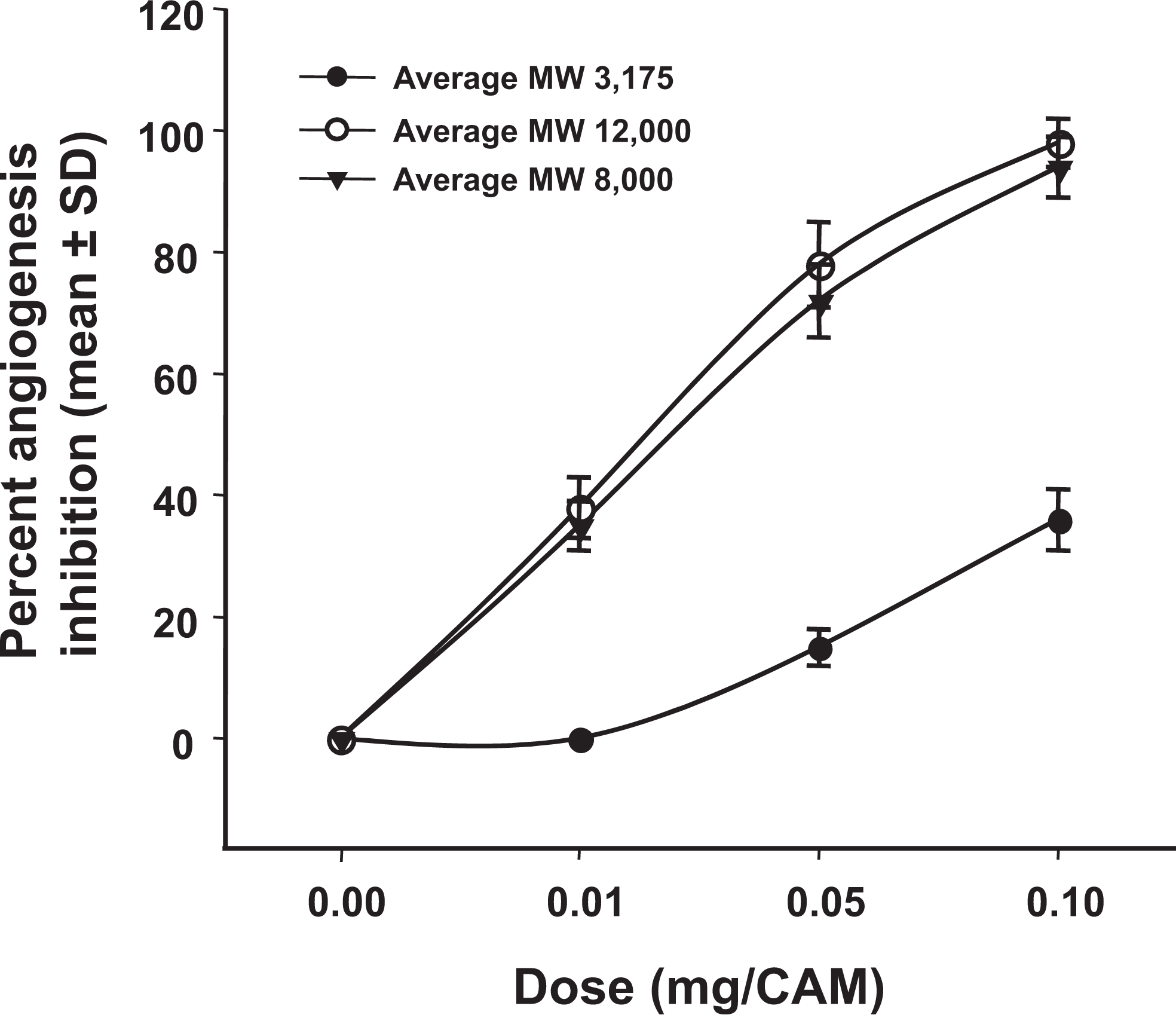
Antiangiogenesis efficacy of different molecular weight pools of tinzaparin in the CAM model system. Angiogenesis was quantified as the number of vessel branch points in the treated area of the CAM. Results are expressed as percentage inhibition of FGF-2-induced angiogenesis. Data represent mean ± SD, n = 8 per condition. CAM indicates chick chorioallantoic membrane; SD, standard deviation.
Differential Effects of Heparin Fractions on FGF-2- and VEGF-Induced Angiogenesis
We next compared the antiangiogenesis efficacy of tinzaparin and enoxaparin. Both tinzaparin and enoxaparin inhibited FGF-2-induced angiogenesis in a dose-dependent manner over a dose range of 0 to 30 μg/CAM (Figure 3A and B ). Tinzaparin, which contains relatively higher MW fractions as compared to enoxaparin, was significantly more potent than enoxaparin, with an IC50 of 0.25 ± 0.05 µg versus 3.805 ± 0.2 µg for enoxaparin (P ≤ .001; Figure 3C).
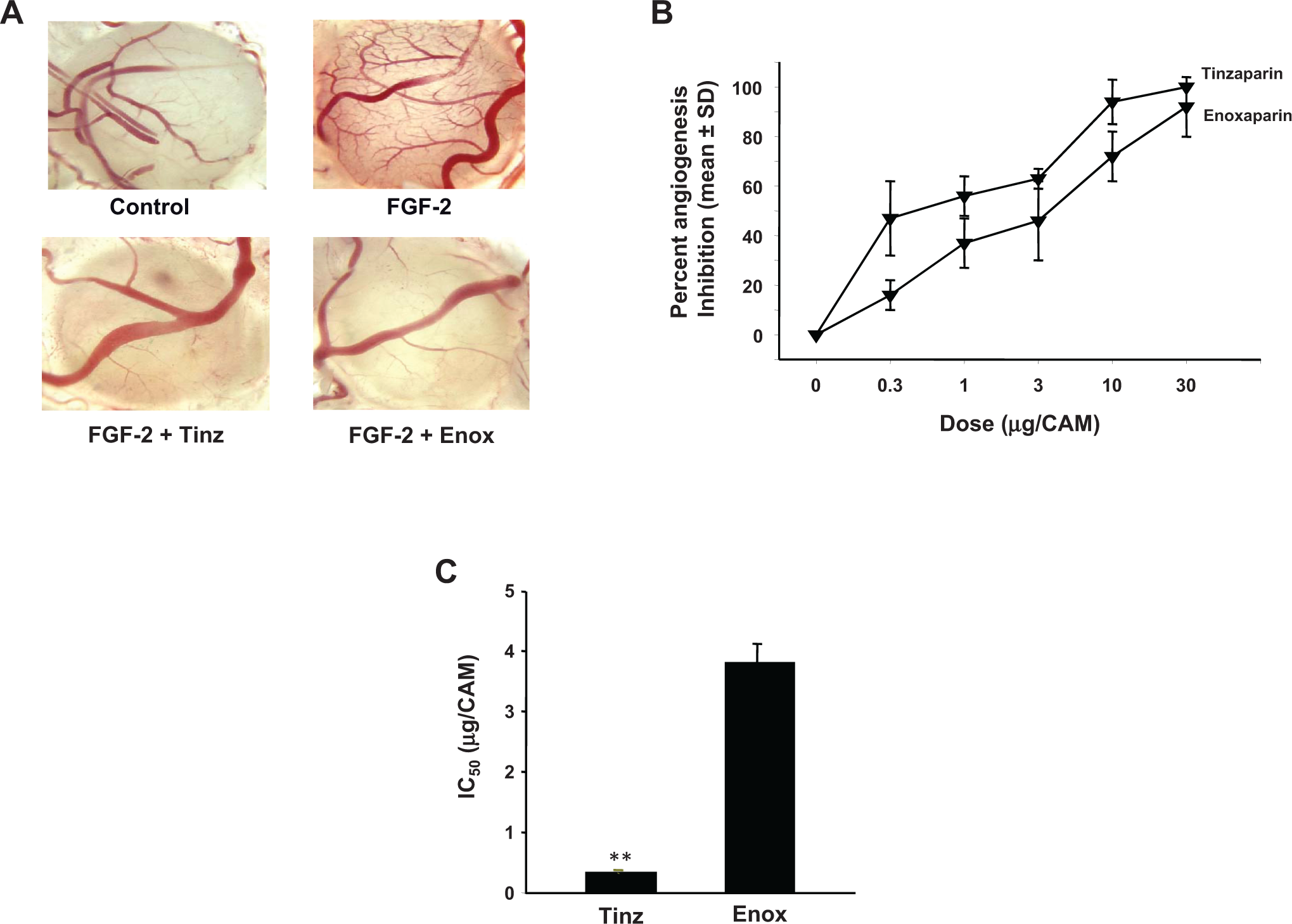
Antiangiogenesis efficacy of tinzaparin versus enoxaparin in FGF-2-induced angiogenesis in the CAM. A, Representative microscope images of CAMs treated with FGF-2 (top right) or PBS (top left) as positive and negative controls, respectively, or FGF-2 plus tinzaparin or enoxaparin (bottom left and right panels, respectively). B, Inhibition of angiogenesis by tinzaparin and enoxaparin is dose dependent. (C) IC50 (µg) values for tinzaparin and enoxaparin for FGF-2-induced angiogenesis. Data represent the mean ± SD, n = 8. **P < .001. [del] CAM, chick chorioallantoic membrane; SD, standard deviation; FGF-2, fibroblast growth factor 2; PBS, phosphate-buffered saline.
Similarly, tinzaparin and enoxaparin inhibited VEGF-induced angiogenesis in a dose-dependent manner; although relatively higher doses (nearly 10-fold higher) of both agents were required for maximal inhibition of VEGF-induced angiogenesis in the CAM system (compare Figures 3B and 4A ). As with FGF-2, tinzaparin was more potent than enoxaparin (P ≤ .001) in inhibiting VEGF-induced angiogenesis (Figure 4B). The mean IC50 for inhibition of VEGF-induced angiogenesis was 22 ± 4 µg for tinzaparin and 140 ± 11 µg for enoxaparin. These results indicated that tinzaparin is more potent than enoxaparin in inhibiting both FGF-2- and VEGF-induced angiogenesis and that both agents are less effective in counteracting the angiogenic signals mediated by VEGF as compared to FGF-2.
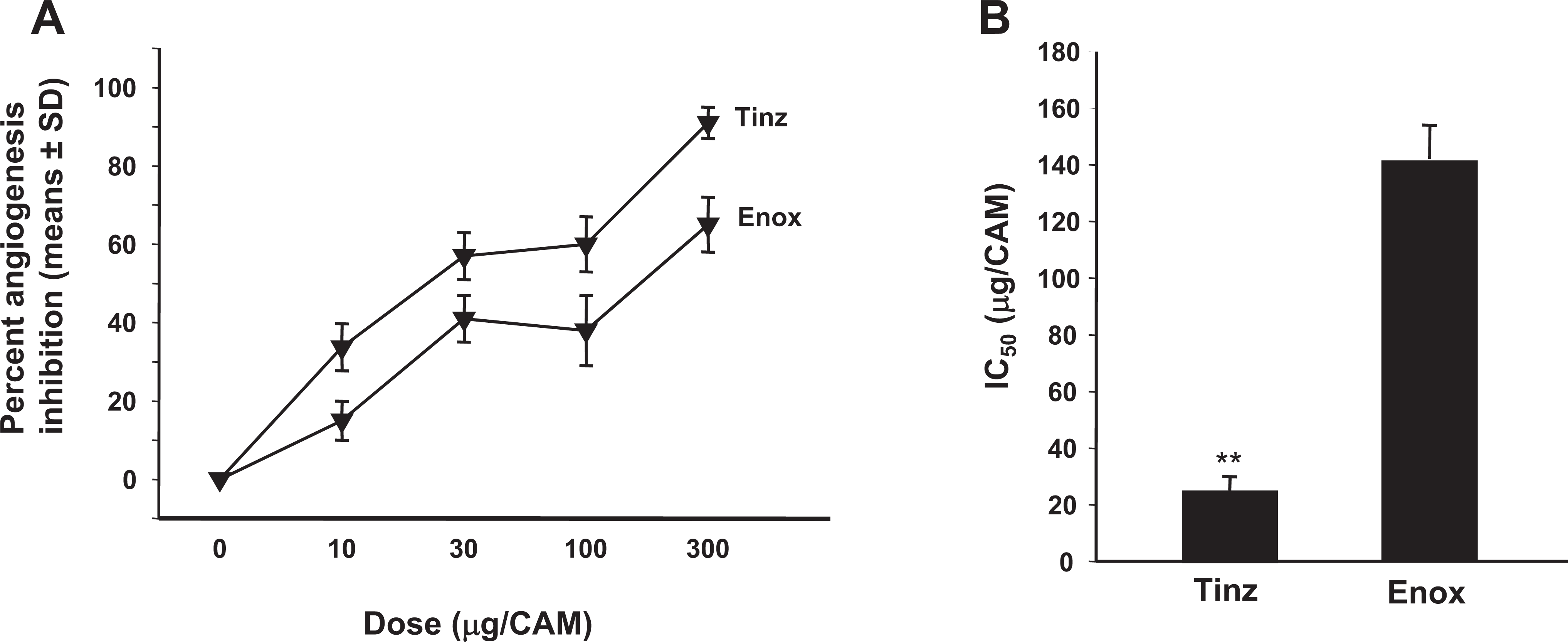
Antiangiogenesis efficacy of tinzaparin versus enoxaparin in VEGF-induced angiogenesis in the CAM. A, Inhibition of VEGF-induced angiogenesis by tinzaparin and enoxaparin is dose dependent. B, IC50 (µg) values for VEGF-induced angiogenesis. Data represent the mean ± SD, n = 8. **P < .001. VEGF indicates vascular endothelial growth factor; CAM, chick chorioallantoic membrane; SD, standard deviation.
Discussion
Angiogenesis is a critical determinant of tumor growth and the development of metastasis. Heparin and LMWH have been shown in a variety of in vitro and in vivo models to be effective modulators of angiogenesis. Meta-analysis of clinical trials comparing heparin with LMWH for the treatment of VTE in patients with cancer indicates improved survival associated with the administration of an LMWH.
Heparin and LMWHs have multiple activities, predominantly antithrombotic effects and stimulation of the release of endothelial TFPI32,17,19 that could potentially contribute to the observed clinical benefits of these agents. As the mechanisms of heparin and LMWHs become more fully elucidated, they may be better exploited to optimize drug therapy in the clinical setting and, perhaps, expand the indications for these agents. The use of LMWHs to stimulate the release of TFPI may have important implications particularly in the treatment of cancer.
The involvement of the vascular system in malignancy encompasses not only angiogenesis but also systemic hypercoagulability and a prothrombotic state, and there is increasing evidence that pathways of blood coagulation and angiogenesis are reciprocally linked. Tumor growth and spread are dependent on angiogenesis, and preclinical studies have demonstrated that LMWH fragments exert potent antiangiogenic effects that are fragment-mass-specific and angiogenesis-type-specific. In the current study, we observed differential mass-specific and growth factor-specific (ie, basic FGF [bFGF] or VEGF) activities of commercially available LMWHs in 2 different model systems, the EC tube formation assay and the CAM model of angiogenesis. Additional mechanistic and comparative pharmacodynamic studies of the LMWHs and LMWH fractions using these model systems are warranted.
Our results indicate that tinzaparin may be particularly suited for additional investigation as a potential antineoplastic agent. The MW distribution of tinzaparin is slightly greater than other commercially available LMWHs. Importantly, the MW profile of LMWHs has been shown to affect the release of TFPI. 33 Single administration of a therapeutic dose of tinzaparin resulted in a rapid, sustained, 5-fold increase in plasma levels of TFPI in healthy individuals. Molecular weight fractions less than 2000 Da had little effect on the release of TFPI, and fragments within the 2000 to 8000 Da range appear to be primarily responsible for the potent and sustained increases in TFPI plasma levels. 34
The current results are also in agreement with previous studies in our laboratory in which we investigated the antiangiogenic activity of tinzaparin. Tinzaparin or recombinant TFPI inhibited the growth of colon carcinoma, human fibrosarcoma, and human lung carcinoma in the CAM tumor implant model. 34 In addition, subcutaneous administration of tinzaparin prevented tumor-associated coagulopathy and lung seeding in the B16 melanoma model of metastasis. 35 Similar data were shown through increasing TFPI levels in the same model. 36 The relationship between heparin’s average MW and inhibition of angiogenesis is due to the level of EC TFPI released, which we described in our earlier report. 34 The smaller the heparin fraction, the lower the release of TFPI and the lower the level of angiogenesis inhibition. The smallest anticoagulant heparin fragment, the pentasaccharide (Fondaparinux), has no effect on EC TFPI release and no significant inhibition of angiogenesis. Because tinzaparin has relatively higher MW fractions and higher sulfate/carboxylate ratio, it has higher endothelial TFPI release and higher level of inhibition of angiogenesis as shown in this report.
In contrast, dalteparin was shown to stimulate angiogenesis, 37 which is not in agreement with other reports.38,39 We and others have shown that heparin itself has a biphasic effect on angiogenesis depending on the concentration, which might be the case with dalteparin in the study done by Norrby and Nordenhem. 37 In this current report and in our previous reports, we showed a monophasic inhibitory effect by tinzaparin at the various doses tested.
In conclusion, LMWHs exhibit differential antiangiogenesis efficacy, which is a function of the structure and molecular chain length of the preparation. The LMWHs have greater in vivo antiangiogenesis efficacy as compared to UFH and are more effective against bFGF-induced angiogenesis than VEGF-induced angiogenesis in the CAM model. Tinzaparin has clinical potential in the treatment of disorders supported by pathologic angiogenesis such as tumorigenesis and tumor metastasis.
Footnotes
Acknowledgments
We thank Laura O’Connor and Seema Mohamed for technical assistance.
Declaration of Conflicting Interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported in part by the Pharmaceutical Research Institute.
