Abstract
Safety issues have been raised about dabigatran. We aimed to investigate the occurrence of safety outcomes in patients who had atrial fibrillation and a risk of stroke. We analyzed 439 patients prescribed dabigatran (n = 220) or warfarin (n = 219). Ischemic stroke occurred in 15 (6.8%) patients in the warfarin group versus 5 (5.2%) patients in the 110-mg group versus 1 (0.8%) patient in the 150-mg dabigatran group (P = .015). Intracranial hemorrhage occurred in 6 (2.7%) patients in the warfarin group versus 3 (2.4%) patients in the 150-mg dabigatran group (P = .104). Death from any cause occurred in 10 (4.6%) patients in the warfarin group versus 1 (1.0%) patient in the 110-mg dabigatran group (P = .005). Dabigatran was associated with less ischemic stroke and death from any cause than warfarin. Dabigatran may be a better option for stroke prophylaxis, where recommended monitoring with warfarin is suboptimal.
Introduction
Atrial fibrillation (AF) is the most common sustained tachyarrhythmia and is associated with a hypercoagulable state resulting in a significantly increased risk of preventable stroke. Current recommendation is to use antithrombotic therapy in all patients with AF except those with lone AF. Warfarin is known to have multiple drug–drug and drug–food interactions. It has a narrow therapeutic window that requires frequent blood monitoring to reduce increased risk of bleeding. In 2009, the pivotal review of the randomized evaluation of long-term anticoagulation therapy (RE-LY) trial established the oral thrombin inhibitor dabigatran to be at least as safe and effective as warfarin in preventing stroke in AF. 1 It is ideal for patients who are unwilling to adhere coagulation monitoring or in whom time in therapeutic range (TTR) with warfarin is suboptimal, despite frequent monitoring. Dabigatran has been subsequently marketed extensively throughout the world. Since dabigatran’s approval, there have been persistent concerns about its safety. Therefore, in this retrospective observational open label study, we assessed some selected outcomes such as ischemic stroke, intracranial hemorrhage, and major extracranial hemorrhage in adults diagnosed with nonvalvular AF, who were new users of dabigatran or warfarin.
Patients and Methods
Study Design
The study design was retrospective. The study population consisted of 220 patients who were newly started on dabigatran and were recruited from 3 different centers from March 2012 to December 2013. Dabigatran-treated patients were identified from the electronic patient record at 3 centers. This cohort was compared to a randomly selected historical control group consisting of 219 patients with initiated warfarin treatment for at least 6 months. Those warfarin patients were randomly selected from all centers, matching the number of dabigatran patients from each center, by our statistician who was blinded to clinical characteristics of the patients and with the aid of a statistical software package. Inclusion of the study population with inclusion and exclusion criteria is shown in Figure 1. All patients in both the groups were previously untreated. It was assumed that the dabigatran and warfarin treatment was initiated at time of prescription. Patient characteristics included age, gender, weight, CHA2DS2-VASc score, HAS BLED score, estimated glomerular filtration rate (eGFR), treatment duration, comorbidities, medication use, and rate of dabigatran discontinuation and were extracted from the patients’ medical records or via telephone interviews. The CHA2DS2-VASc score is a measure of the risk of stroke in which congestive heart failure, hypertension, diabetes mellitus, vascular disease, an age of between 65 and 74 years, and female sex are each assigned 1 point and an age of 75 years or older and previous stroke or transient ischemic attack or thromboembolism are each assigned 2 points; the score was calculated by summing all the points for a given patient. 2 The CHA2DS2-VASc score ≥2 determines risk of stroke, and oral anticoagulants are recommended according to the guideline. The HAS BLED score is a measure of the bleeding risk in which hypertension, renal or liver disease, stroke, bleeding, labile INR, an age of 65 years or older, and the use of medications or alcohol are each assigned 1 point; the score was calculated by summing all the points for a given patient. 2 Creatinine clearance was calculated according to the Cockcroft-Gault method. The efficacy outcomes included ischemic stroke, pulmonary embolism, myocardial infarction (MI), and death from any cause, which resembled those in the RE-LY study. The safety outcomes included intracranial hemorrhage, major extracranial hemorrhage, and minor bleeding as noted in the electronic patient record and as well as assessed by a telephone interview. This study was approved by the medical ethics committee of the participating university and was conducted in accordance with Helsinki Declaration.
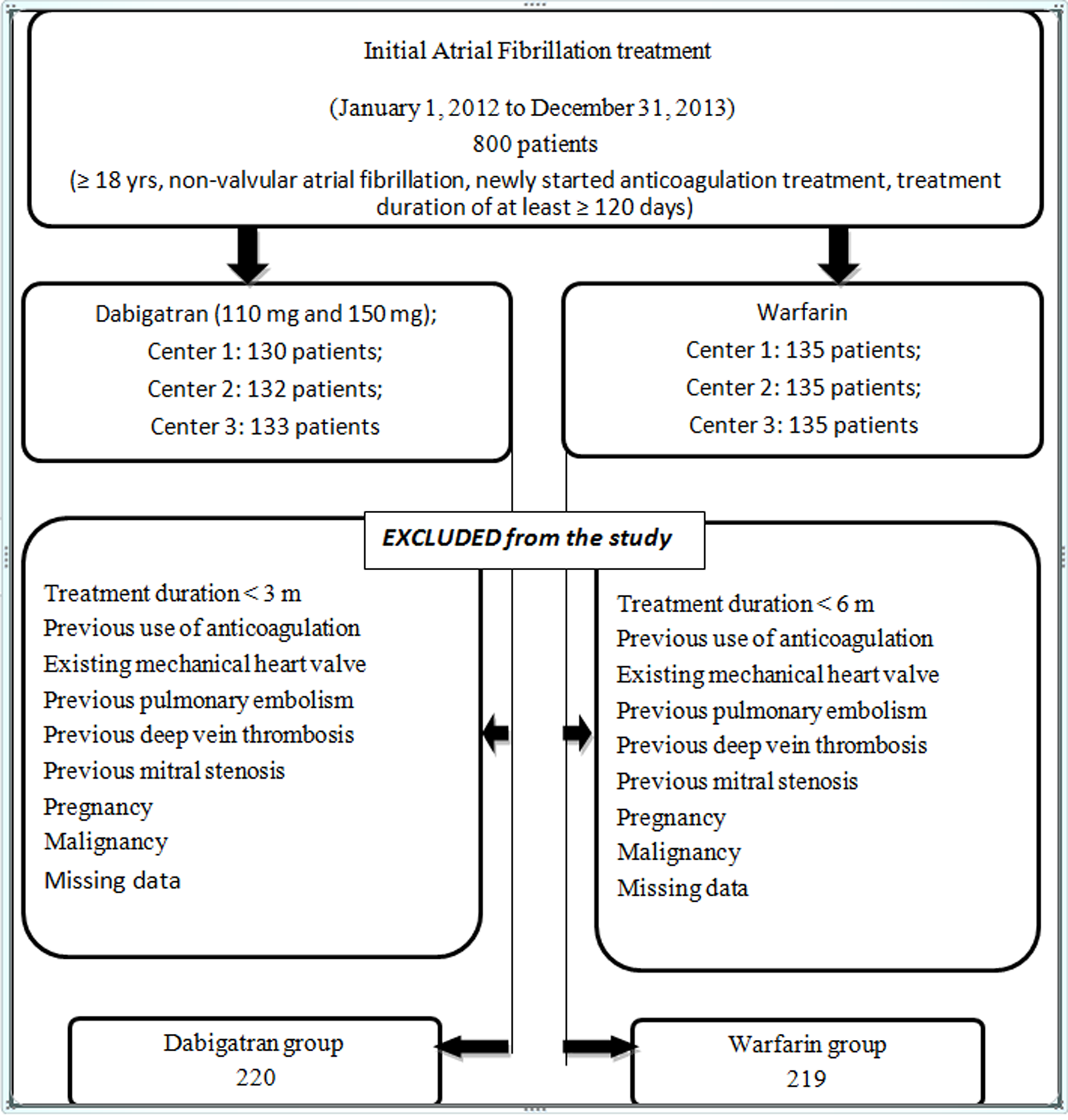
Inclusion of the study population.
Statistics
A statistical software package (SPSS 18.0; Chicago, IL) was used to perform all analyses. Continuous data are reported as median (min–max) with the lower and upper quartiles, and categorical data are reported as percentages. Since our data were not normally distributed, Kruskal-Wallis 1-way analysis of variance was used to compare the differences of the groups. Post hoc Mann-Whitney U test with Bonferroni correction test was used when the Kruskal-Wallis test determined a significant difference. Chi-square test was used for categorical variables. Statistical significance was described as P < .05.
Results
Baseline characteristics according to the treatment group of 439 patients are listed in Table 1. The median duration of the follow-up period was 15 months in the warfarin group and 6 months in the dabigatran group. The TTR in the warfarin group was only 32%. Ischemic stroke occurred in 15 patients receiving warfarin (6.8%), 5 patients receiving 110 mg of dabigatran (5.2%), and 1 patient receiving 150 mg of dabigatran (0.8%; Table 2). Both doses of dabigatran were superior to warfarin (P = .015). The 150-mg dose of dabigatran was superior to the 110-mg dose of dabigatran (P = .015). Myocardial infarction occurred in 16 (7.3%) patients receiving warfarin whereas in no patients receiving either dose of dabigatran (P = .0001). Pulmonary embolism occurred in 5 (2.3%) patients receiving warfarin, 2 (2.1%) patients receiving 110 mg of dabigatran, and no patients receiving 150 mg of dabigatran. Death from any cause occurred in 10 (4.6%) patients receiving warfarin and 1 (1.0%) patient receiving 110 mg of dabigatran whereas in no patients receiving 150 mg of dabigatran (P = .005). Occurrence of intracranial bleeding, major extracranial bleeding, and minor bleeding were higher with warfarin than with either the 110-mg dose of dabigatran or the 150-mg dose of dabigatran (Table 2). Intracranial bleeding was not observed with the 110-mg dose. Minor bleeding also occurred less with the 110-mg dose. The most common adverse event was dyspepsia in all groups. The adverse events that were significantly more common with dabigatran than with warfarin were weight gain, fluid retention, and insomnia (Table 3).
Baseline Characteristics According to Treatment Group.a
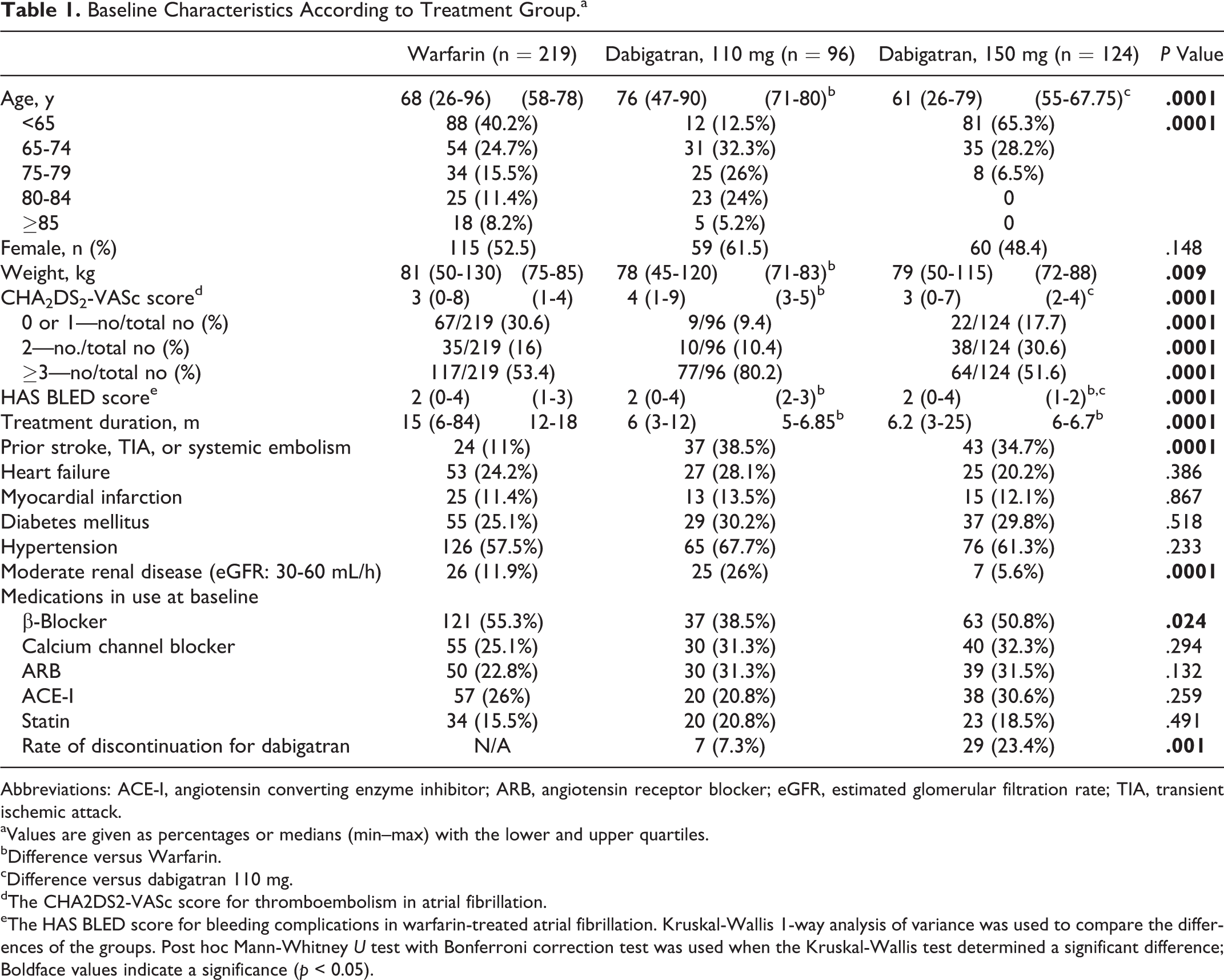
Abbreviations: ACE-I, angiotensin converting enzyme inhibitor; ARB, angiotensin receptor blocker; eGFR, estimated glomerular filtration rate; TIA, transient ischemic attack.
aValues are given as percentages or medians (min–max) with the lower and upper quartiles.
bDifference versus Warfarin.
cDifference versus dabigatran 110 mg.
dThe CHA2DS2-VASc score for thromboembolism in atrial fibrillation.
eThe HAS BLED score for bleeding complications in warfarin-treated atrial fibrillation. Kruskal-Wallis 1-way analysis of variance was used to compare the differences of the groups. Post hoc Mann-Whitney U test with Bonferroni correction test was used when the Kruskal-Wallis test determined a significant difference; Boldface values indicate a significance (p < 0.05).
Efficacy and Safety Outcomes.a
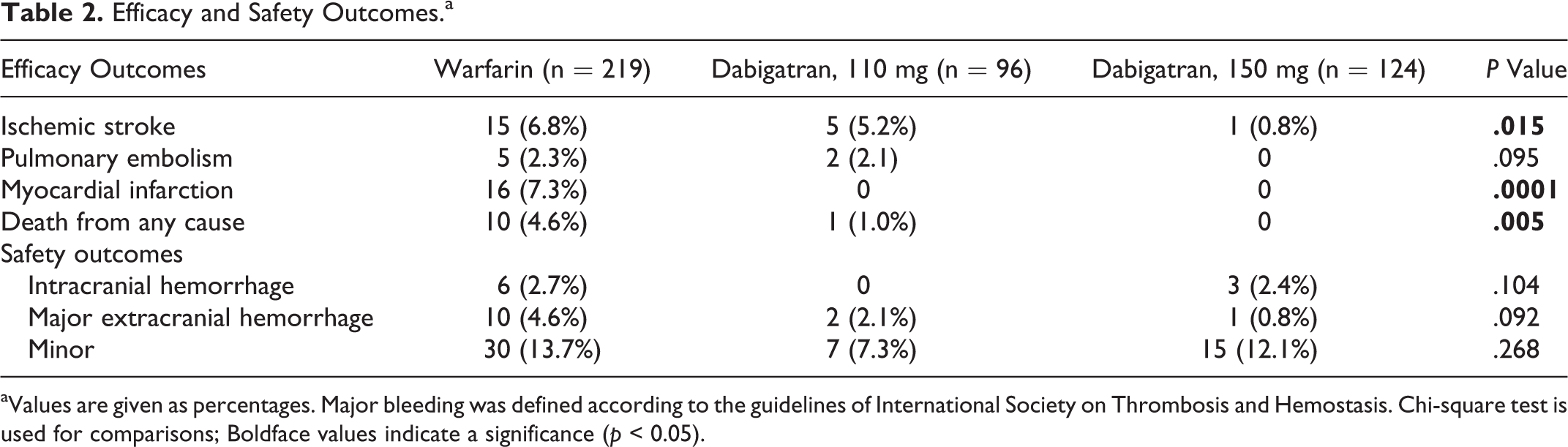
aValues are given as percentages. Major bleeding was defined according to the guidelines of International Society on Thrombosis and Hemostasis. Chi-square test is used for comparisons; Boldface values indicate a significance (p < 0.05).
Adverse Events.a
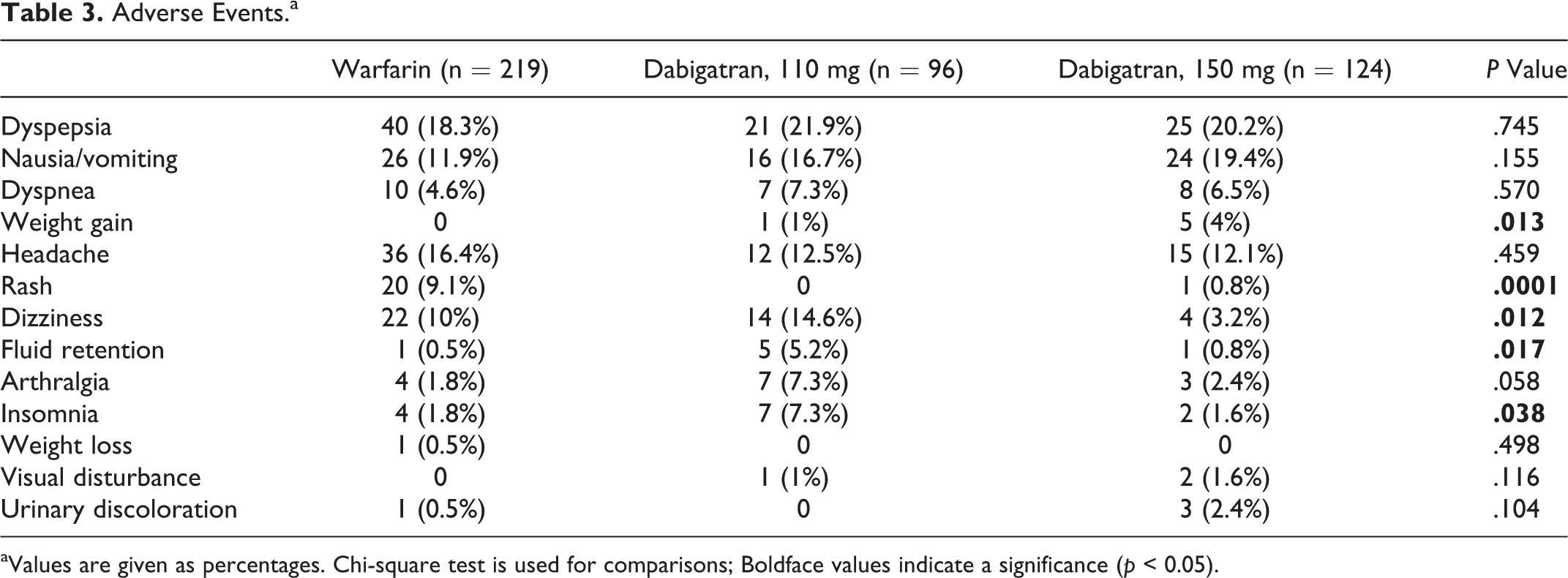
aValues are given as percentages. Chi-square test is used for comparisons; Boldface values indicate a significance (p < 0.05).
Discussion
To our knowledge, from a middle-Eastern country, this is the first report of real-world experience with dabigatran compared to warfarin in patients who had AF and were at risk of stroke. In contrast to the RE-LY study, both dabigatran doses were superior to warfarin in reducing ischemic stroke. The 150-mg dose was superior to the 110-mg dose in reducing ischemic stroke. Less developed countries have not been well represented in the RE-LY trial. 1 Our findings suggest that, in such countries, due to suboptimal prophylaxis with warfarin, dabigatran can be especially preferred.
In this study, we sought to determine efficacy and safety outcomes. This study was designed as a retrospective, open-label, and multicenter study with the limitations inherent to such study designs. However, this observational analysis had a comparison group, warfarin-treated patients, in assessing outcomes. We only included treatment-naive patients with AF to avoid selection bias, and warfarin patients were randomly selected. Our results can be generalized to adults of both genders. Outcome data were limited to 6 months. The median follow-up period was longer for warfarin (15 vs 6 months). The small sample size and short follow-up period precluded a mortality analysis. However, death from any cause occurred in 10 (4.6%) patients receiving warfarin versus 1 (1%) patient receiving 110 mg of dabigatran versus none receiving 150 mg of dabigatran. Reduced mortality with dabigatran has been reported previously. 3,4 Dabigatran etexilate has been approved in Turkey in 2 doses (either 150 or 110 mg twice a day) for stroke prevention in nonvalvular AF. For this indication, its use has been restricted to a cardiologist prescription only. This might be the reason that dabigatran was prescribed on approved indication, in appropriate patients, and according to recommendations concerning renal function in this study similar to previous reports. 5,6
There have been several reports regarding real-world experience with dabigatran. 4,7,8 It is important that additional data on efficacy and safety outcomes be reported so that the efficacy outcomes and frequency of the adverse events can be captured. Compared to patients studied in the RE-LY study, our patient population receiving 150 mg of dabigatran was younger (61 vs 71.5 years) with more patients having CHA2DS2-VASc score of ≥3 (51.6% vs 32.6%). In our analysis, a total of 31 patients with CHA2DS2-VASc score of ≤1 received dabigatran, and 3 of these were surrounding cardioversion. Dabigatran may be useful in this unique situation; however, further studies are needed. Patients receiving 110 mg of dabigatran were older than those receiving both 150 mg of dabigatran and warfarin. The TTR in this study was unacceptably suboptimal. Only 32% of patients receiving warfarin achieved the recommended TTR (>65%), which is consistent with a recent study from this country. 9 This can explain why warfarin was inferior to both doses of dabigatran with respect to ischemic stroke and was not able to reduce the risk of MI, pulmonary embolism, and death. Our findings suggest that in this country, patients with AF receiving warfarin may be largely below the recommended TTR. Thus, dabigatran can be a better choice for those patients. There was a significant difference between treatment groups with respect to ischemic stroke (warfarin: 6.8%, dabigatran 110 mg: 5.2%, and dabigatran 150 mg: 0.8%). This was consistent with the RE-LY study.
Some concerns have been raised regarding fatal bleedings with dabigatran, mostly related to inappropriate prescribing (eg, in renal failure) as well as bleeding related to trauma or MI, drug compliance, and renal impairment. 10,11 In this study, there was no pulmonary embolism in the 150-mg group, and cases with MI were only observed in the warfarin group. The most devastating complication of warfarin therapy is intracerebral hemorrhage. There was no difference in bleeding events between the groups. Of the 9 patients reported to have intracranial bleeding, 6 (66.7%) were on warfarin and 3 (33.3%) were on 150-mg dose of dabigatran. Of the 6 dabigatran-related major bleeding events observed during the 6-month follow-up period, 3 were intracranial and 3 were gastrointestinal (GI) bleeding. At the time of bleeding, no change in renal function was observed for any of the patients. On 6-month follow-up, renal functions had been checked in 90.5% of patients in all groups. Neither age nor sex had any effect on bleeding. Three patients with intracranial bleeding were also using aspirin. We observed more cases of GI bleeding with warfarin than dabigatran. Only 1 patient was found to have eGFR <30 mL/min where use of dabigatran would be inappropriate in Turkey. Patients with higher HAS BLED scores and advanced age appropriately received 110 mg of dabigatran. Our findings show that bleeding occurred similar to warfarin with both doses of dabigatran, without a reduction in the efficacy against ischemic stroke, confirming an important advantage of dabigatran, similar to previous reports. 12 In addition, we did not observe evidence of hepatotoxicity in our dabigatran-treated patients. Dyspepsia was also the most frequent adverse event, similar to previous reports. The adverse events that were significantly more common with 110 mg of dabigatran than with warfarin were weight gain, fluid retention, and insomnia. The reason can be that patients receiving 110 mg of dabigatran were older than the other 2 groups. Patients who discontinued warfarin were excluded from the analysis. More patients receiving 150 mg of dabigatran had discontinued their medication compared to the 110-mg group (Table 1), which was also more than the reported rates in the literature. 13,14 This cannot be explained only by the occurrence of adverse events (Table 3). Patients who had major bleeding had not received any anticoagulant therapy after discharge. One possible explanation for poor adherence can be the minor bleedings, which occurred more in the 150-mg group. Another explanation can be that patients might had not been well informed on the consequences of a cardioembolic stroke and instructions regarding the use of dabigatran at the time of first prescription. Another possible explanation can be the lack of routine monitoring to verify that they were taking and tolerated the medication. This is consistent with the contention that without routine monitoring of the medication, inadequate adherence to the prescribed regimen is not controlled in the same way as with warfarin.
In conclusion, this study showed that the oral thrombin inhibitor dabigatran is more effective than warfarin. In addition, it does not increase bleeding events at both doses. Dabigatran was implemented with appropriate renal function monitoring. Our study demonstrates that dabigatran can be safely used in clinical practice because of suboptimal TTR with warfarin and superiority to warfarin with respect to stroke prevention. Dabigatran may especially provide an alternative in less developed parts of the world or places where resources are limited, where patients may have poor INR control and/or patients or doctors may be reluctant to use warfarin due to its many limitations. 15 Further research looking at effects on mortality and with longer follow-up is needed.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
