Abstract
Anticoagulants have been used in clinical practice for more than 50 years. Their indications expand, as more people are diagnosed each year with atrial fibrillation and venous thromboembolism. Vitamin K antagonists have been the most popular choice due to their effectiveness and their ability to reverse bleeding using a known antidote; oral and intravenous vitamin K have long been known to reverse the effects of warfarin. With new classes of anticoagulants making their way onto the market, such as factor Xa inhibitors (rivaroxaban, apixaban) and direct thrombin inhibitors (dabigatran), the need for new reversal agents is paramount. Patients tend to be more receptive to these medications because they do not require routine blood monitoring, can be used at fixed doses, and do not have major drug or food interactions. Antidotes for these medications have shown promise in animal models and are currently in clinical trials.
Keywords
Introduction
Oral anticoagulants have been used in clinical practice to prevent or treat venous thromboembolism (VTE) in patients with atrial fibrillation (AF) and in other thromboembolic disorders. The average annual incidence of VTE among caucasians is 108 per 100 000 person-years. This corresponds to approximately 250 000 cases occurring annually in the United States among caucasians. 1 It is believed that the incidence of VTE is similar or even higher among African Americans and slightly lower among Asian Americans and Native Americans. 1 The incidence of AF is expected to increase during the next decade. It is estimated that the current prevalence of AF is between 3 and 6 million Americans. This number is expected to rise to between 6 and 12 million by 2050. 2,3
There are currently 4 oral anticoagulants available in the United States, comprising 3 unique classes of medications. Figure 1 shows a timeline of historical advances in the development of oral anticoagulants. It has been estimated by the Food and Drug Administration (FDA) that 2 million patients initiate warfarin therapy annually in the United States. 3,4 Dabigatran, the first novel oral anticoagulant, is a direct thrombin inhibitor, and it was approved in October 2010 by the FDA. The final class is the factor Xa (FXa) inhibitors, which consist of rivaroxaban and apixaban, both approved by the FDA in late 2012. As a whole, oral anticoagulants are effective treatment options, but they also come with some inherent risk. Shifting the clotting cascade too far in either direction can lead to significant problems in patients. There is evidence to suggest that clinically relevant nonmajor bleeding occurs at a rate of 18 per 100 patient-years, in a study comparing rivaroxaban and warfarin. 5 Major bleeds also occur in approximately 0.5% to 3% of patients depending on definition and medication used. 6,7
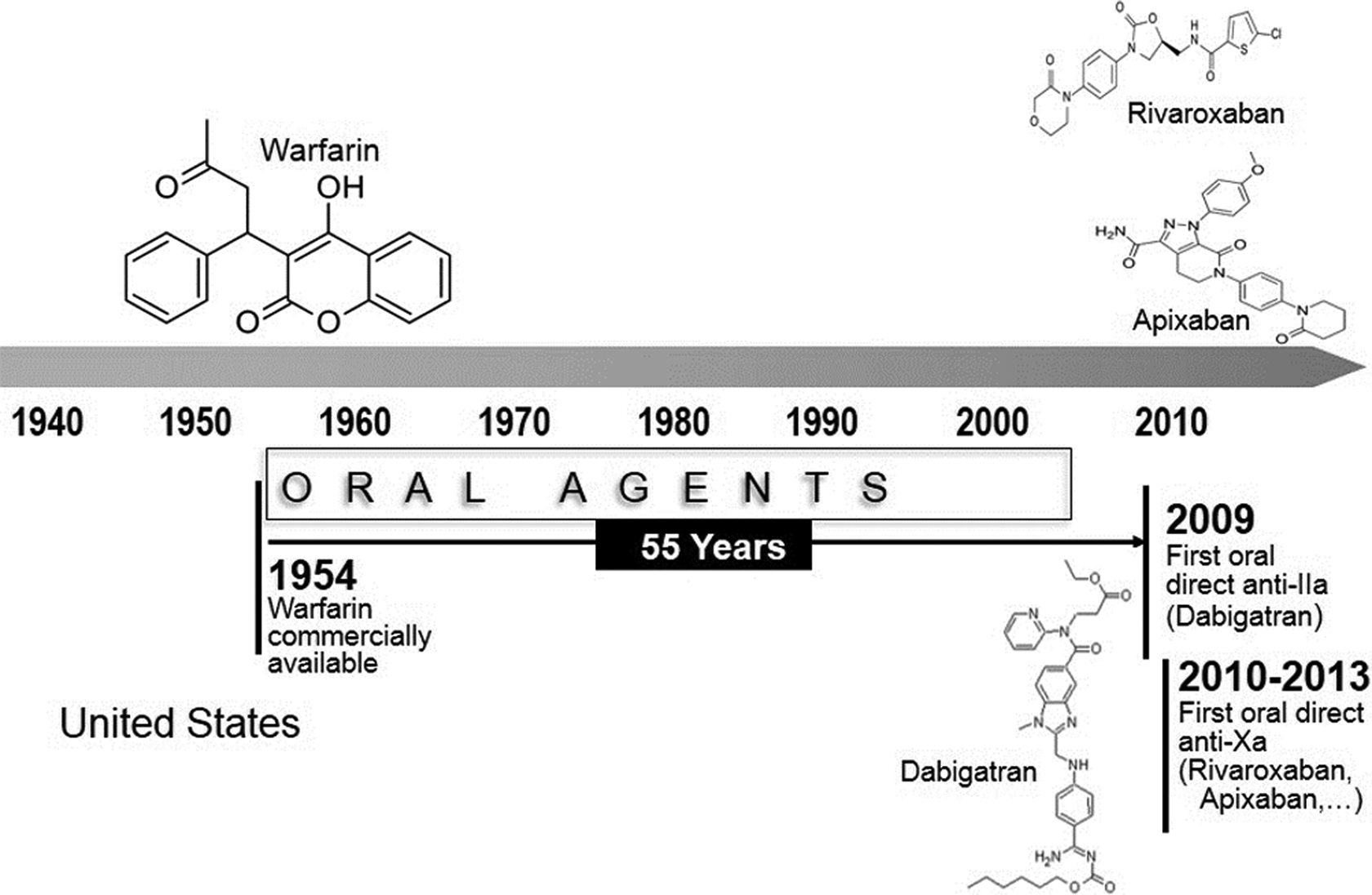
Advances in oral anticoagulation from 1940 to present. There are currently 3 broad classes (vitamin K antagonists, anti-IIa, and anti-Xa) of oral anticoagulants available in the United States. Individual agents are warfarin, dabigatran, rivaroxaban, and apixaban.
Warfarin
Warfarin is an oral vitamin K antagonist (VKA) that works by blocking the regeneration of vitamin K epoxide, thus inhibiting the syntheses of vitamin K-dependent clotting factors, which include factors II, VII, IX, and X as well as the anticoagulant proteins C and S. 8 It is indicated for a number of conditions, including prophylaxis and treatment of VTE, including pulmonary embolism (PE), prophylaxis, and treatment of thromboembolic complications associated with AF and/or cardiac valve replacement, and reducing the risk of death, recurrent myocardial infarction, and thromboembolic events such as stroke or systemic embolization after myocardial infarction. 7 Although effective, warfarin comes with a number of limitations. Warfarin is a narrow therapeutic medication that requires the individualization of doses for each patient according to their international normalized ratio (INR) and indication, although a general range is 2.0 to 3.0. 9 It typically takes 4 to 5 days for a patient to become therapeutic on warfarin. In addition, due to the drug’s mechanism of action, it is susceptible to a number of food and drug interactions. For example, green leafy vegetables contain a significant amount of vitamin K that can alter the clotting cascade in patients. It is always important to counsel all patients on the importance of eating a balanced, consistent diet.
Warfarin is highly water soluble, is rapidly absorbed from the gastrointestinal tract, has high bioavailability, and reaches maximal blood concentrations about 90 minutes after oral administration. 10,11 It is highly protein bound with a half-life of 36 to 42 hours 12 and is highly metabolized by cytochrome P (CYP) 2C9 and CYP3A4. 13
The risk of bleeding is a major concern in all warfarin-treated patients. According to a meta-analysis, patients were in therapeutic range only 63.6% of the time. 14 According to the package insert from Bristol-Myers Squibb, the rate of major bleeding in warfarin-treated patients is between 0.6% and 2.7%. 15 There are a number of factors that predispose patients to bleeding episodes. The most important factor is the intensity of anticoagulant therapy. 13,16,17 Patients with a higher goal INR are more likely to develop a bleed than those with a conservative number. It is important to set a reasonable target range depending on the indication trying to be reached (not discussed here) and the specific patient. There are a number of other risk factors for developing a bleed including advanced age, cancer, renal or hepatic dysfunction, alcohol abuse, stroke, a history of bleeding, or concomitant use of other therapies including antiplatelet drugs.
If a patient presents requiring anticoagulant reversal, there are a few medications and strategies that can be used. The American College of Chest Physicians guidelines (Table 1) outline the treatment options depending on the patient who presents. 18 Withholding 1 or more doses is an effective option in patients who are not actively bleeding. These patients may present for an elective procedure or only with an elevated INR and low risk of bleeding. It is important for patients to know that their INR will remain elevated for a couple of days, and during that time there will be an increased risk of bleeding. Reporting any signs and symptoms of bleeding immediately is important.
2012 American College of Chest Physicians (ACCP) Evidence-Based Clinical Practice Guidelines. 18

Abbreviations: INR, international normalized ratio; IV, intravenous; PCC, prothrombin complex concentrate; 2B, weak recommendation, moderate-quality evidence; 2C, weak recommendation, low- or very low-quality evidence.
If withholding doses is not enough, the patient can be supplemented with oral or intravenous (IV) vitamin K. Other products available include fresh frozen plasma (FFP), 3-factor prothrombin complex concentrate (3-F PCC), and 4-factor prothrombin complex concentrate (4-F PCC). There are a number of differences between these 3 products. Fresh frozen plasma is obtained by either separation of supernatant plasma from a whole blood donation or an apheresis technique. The plasma must be frozen within 8 hours to preserve factors V and VIII. Fresh frozen plasma must be stored at −18°C in order to maintain its effectiveness. Prior to administration, FFP must be thawed at 37°C. 19 The main difference between the 3-F PCC and 4-F PCC is the addition of factor VII in meaningful amounts. Factor VII has the shortest half-life, 4 to 6 hours, and when replaced is able to return a patient’s INR to near-normal levels.
Vitamin K
Vitamin K or phytonadione has been used for warfarin reversal for many years. It is available orally, subcutaneously, and intravenously. It is currently not recommended to use subcutaneous vitamin K because the response is rather unpredictable and may be delayed. Oral vitamin K is an effective treatment option for patients who do not require rapid reversal. It is safe, effective, and more convenient than parenteral routes. The limitation for oral vitamin K is that it lowers INR within 24 to 48 hours. 20 A randomized, placebo-controlled trial evaluated withholding the dose versus oral phytonadione + withholding the dose for patients with excessive anticoagulation. This study looked at 30 nonbleeding patients with INRs of 6 to 10. The mean calculated time to reach an INR of 4.0 was significantly greater in the placebo group than in the phytonadione group (2.6 vs 1.4 days, P = .006). Adding oral vitamin K to patients with elevated INRs is able to reduce the patients’ INR level more rapidly than withholding the dose alone. 21 It is important to continue to monitor the patient’s INR because warfarin has a much longer duration of action than vitamin K. In cases of extreme overdose or ongoing bleeds, it may be necessary to readminister vitamin K. 20
Vitamin K administered intravenously can effectively lower the patient’s INR more rapidly than oral vitamin K, within 12 to 14 hours. There have been head-to-head comparison trials between the oral and the IV agents. In one study of patients presenting with a baseline INR of 6 to 10, the decline was significantly more rapid in the IV group. At 12 hours, the IV group reached a mean INR of 3.8 ± 1.4, while the oral group had an INR of 4.4 ± 1.1 at 12 hours. At 24 hours, there was no longer a statistical difference; the IV group had a mean INR of 2.6 ± 0.8 compared with 2.9 ± 0.8 for the oral group. This suggests that IV vitamin K is able to provide a more rapid reversal, but over time, the results do not differ. 22
Fresh Frozen Plasma and PCCs
For patients presenting with acute bleeds, it will not be possible to wait for oral or even IV vitamin K to start working. These patients require immediate supplementation with coagulation factors to assist in stopping the bleed. Fresh frozen plasma remains the most widely used coagulation factor replacement product for urgent reversal of warfarin anticoagulation. 23 It contains all coagulation factors in their normal amounts, which limit the risk of thrombosis. However, there are a number of limitations with this product as outlined in Table 2. There is an administration delay due to the need to thaw it and the need for FFP to be ABO matched to the patient, it has a slow onset (13-48 hours), and patients are at risk of fluid overload, infectious disease, allergic reactions, and transfusion-related acute lung injury. 24,25
Vitamin K Antagonist Reversal Strategies.
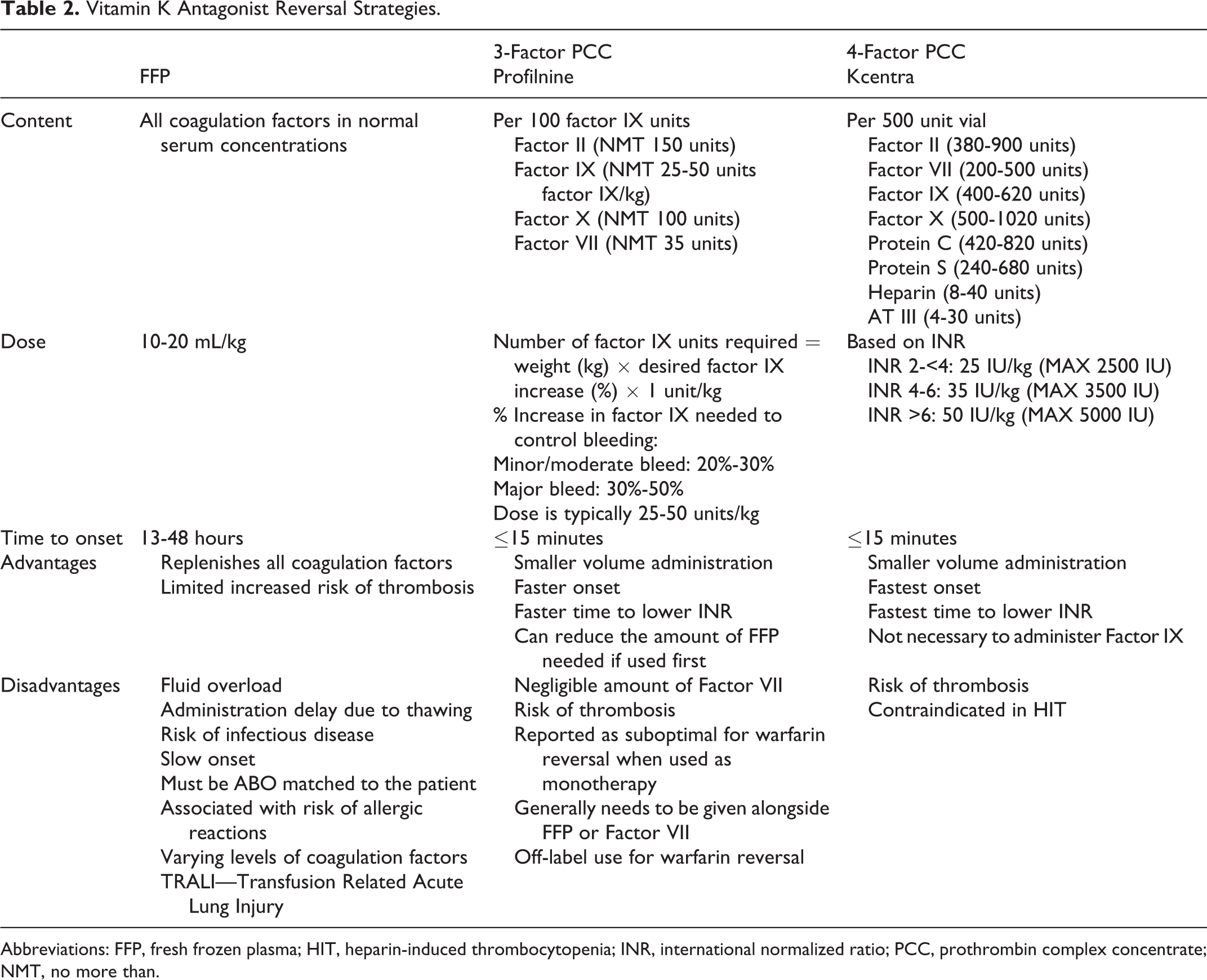
Abbreviations: FFP, fresh frozen plasma; HIT, heparin-induced thrombocytopenia; INR, international normalized ratio; PCC, prothrombin complex concentrate; NMT, no more than.
Prothrombin complex concentrates do not require ABO matching, and they have a smaller volume of administration, faster onset, faster time to lower INR, and are probably more effective. These products do come with an increased risk of thrombosis, and 4-F PCC is contraindicated in patients with recent heparin-induced thrombocytopenia. The 4-F PCC was approved in the United States in April 2013, although it has been used in Europe for years. 25 –29
Although 3-F PCC is not approved for use in this patient population, there is some evidence to suggest it can be an effective treatment option for patients requiring rapid INR reversal during an acute bleeding episode. One trial conducted by Chapman et al compared 3-F PCC + FFP + vitamin K versus FFP + vitamin K to determine whether the addition of 3-F PCC could rapidly correct the INR. The percentage of patients who reached goal INR (<1.5) was similar in each group; however, the time it took to reach goal INR was significantly shorter in the PCC group. It took 16:59 hours to achieve INR <1.5 in the PCC group versus 30:03 hours in the no PCC group (P = .048). There were no statistical differences in FFP units received or the number of patients who received vitamin K. Conclusions from this study were that the addition of PCC to conventional treatment methods did result in a more rapid reversal of INR. 26
Holland et al assessed the efficacy of a low- and high-dose 3-F PCC for urgent warfarin reversal in 40 patients presenting with elevated INR >5, with bleeding, or at a high risk of bleeding. These patients were compared to historical controls, who received FFP alone, to assess the rate of INR correction. Fresh frozen plasma alone, an average of 3.6 units, was able to lower the INR to less than 3.0 in 63% of patients. This was compared to a 50% success rate in the low-dose PCC group and 43% in the high-dose group. However, when a small amount of FFP, 2.1 units, was added to the PCC patients, 89% in the low-dose group and 88% in the high-dose group reached the goal INR. They concluded that 3-F PCC does not satisfactorily lower INR due to its low factor VII content and that the addition of a small infusion of plasma increased the chance of obtaining a satisfactory INR. 27
The addition of recombinant factor VIIa (rFVIIa) to 3-F PCC has also been analyzed by Sarode et al in patients with the most serious bleeding complication of VKA, intracerebral hemorrhage. The authors developed a Trauma Coumadin Protocol (TCP) that consisted of a 3-F PCC and a low-dose rFVIIa. Forty-six patients were analyzed with a mean pre-TCP INR of 3.4 and compared to historical controls. After administration, the mean INR level in the TCP group was 1.0 compared to 1.6 in the control FFP group and 1.4 in the control FFP + PCC group. At 24-hour post-TCP, the survival rate was 89.1%, and 3 days post-TCP, the survival rate was 82.6%. The addition of rFVIIa comes with an increased risk of thrombosis and should be used cautiously in patients who are at high risk. Two patients developed thrombotic complications after administration of TCP. 19
The phase 3 trial that led to the approval of a 4-F PCC (Kcentra in the United States) was conducted in 184 patients ≥18 years of age, receiving VKA therapy with an INR ≥2, and experiencing an acute major bleeding event, defined as a life-threatening or potentially life-threatening bleed, acute bleeding with a fall in hemoglobin ≥2 g/dL, or a bleed requiring blood product transfusion. 28 These patients were randomly assigned (1:1) to receive either 4-F PCC or FFP in addition to slow IV vitamin K. Due to the inherent characteristics of each treatment option, the need for thawing, matching, and the volume of FFP, the study staff were not blinded. The primary end points of the study were hemostatic efficacy that was assessed by a blinded Endpoint Adjudication Board based on predefined clinical criteria over a 24-hour period and rapid INR reduction ≤1.3 over a half hour after the end of infusion. The study also considered treatment-related and serious adverse events. One of the primary end points was effective hemostasis, defined as a rating of excellent or good by the blinded adjudication board. In the 4-F PCC group, 72.4% of patients had effective hemostasis compared with 65.4% in the FFP group; this was not statistically significant. The other primary end point was rapid INR reduction to ≤1.3. The 4-F PCC group was successful 62.2% of the time compared to only 9.6% in the FFP group. This was significant with a difference of 52.6 and a 95% confidence interval of 39.4 to 65.9. The safety profile was also favorable for 4-F PCC when looking at related significant adverse events. In the 4-F PCC group, 1 patient did experience a deep vein thrombosis (DVT) on day 13, and 1 patient had an ischemic stroke on day 43 compared to the FFP group in which 2 patients had myocardial ischemia, 1 with fluid overload and 1 with respiratory failure. This study indicated that 4-F PCC is an effective and safe treatment option for patients on VKA and experiencing an acute bleed. 28
Another prospective trial was conducted by Pabinger et al and had only a single-treatment arm. 29 Patients had to be ≥18 years of age, on oral anticoagulant therapy with an INR ≥2, and requiring an emergency surgical or urgent invasive diagnostic test or INR normalization as a result of acute bleeding. Forty-four patients were enrolled in the trial with INR values ranging from 1.9 to 17.4. The primary end point was normalization of INR ≤1.3 at 30 minutes after the end of PCC infusion. Thirty minutes postinfusion, INR declined to ≤1.3 in 93% of patients and remained stable for the following 48 hours. This trial also looked at hemostatic efficacy, which was deemed to be very good in 93% of patients, satisfactory in 2 patients, and questionable in 1 patient. In 58% of patients, an adverse event occurred; 6 patients had a serious adverse event, and 3 of these patients died as a result. One serious adverse event, a PE resulting in the death of a 70-year-old man, was considered treatment-related, while the others were judged to be unrelated. This study demonstrated that prompt INR reduction to ≤1.3 was accompanied in most cases by clinical hemostatic efficacy. The 4-F PCC was able to successfully return INR to normal levels and lead to clinically significant improvements in hemostatic efficacy. Rapid reduction in INR is not possible with FFP due to the thawing, matching, and its factor content.
Factor Xa Inhibitors
There are currently 2 oral FXa inhibitors approved for use in the United States, along with other novel oral anti-Xa agents to follow. The mechanisms of action of the 2 agents are very similar. They are both selective inhibitors of FXa and do not require a cofactor (such as antithrombin III) for activity. Rivaroxaban inhibits free FXa and prothrombinase activity, while apixaban is able to inhibit free and clot-bound FXa as well as prothrombinase. Neither agent has a direct effect on platelet aggregation, but they indirectly inhibit platelet aggregation induced by thrombin. By inhibiting FXa, both agents decrease thrombin generation and thrombus development. 30,31
The first approved agent in this class was rivaroxaban, which is indicated for use in reducing the risk of stroke and systemic embolism in patients with nonvalvular AF, treatment of DVT including PE, reduction in the risk of recurrent DVT and/or PE following initial 6-month treatment, or prophylaxis of DVT following hip or knee replacement surgery. Doses vary based on indication ranging from 10 to 20 mg once daily or 15 mg twice daily. It is recommended that the 15- and 20-mg tablets, sold as Xarelto, be taken with food, while the 10-mg tablet can be taken with or without food. 32 Use of rivaroxaban requires consideration of a patient’s renal status and should be avoided in patients with a creatinine clearance (CrCl) <30 mL/min. In addition, rivaroxaban should be avoided with combined P-glycoprotein (P-gp) and strong CYP3A4 inhibitors. Rivaroxaban has a t 1/2 of 5 to 9 hours in healthy patients and can be increased to 11 to 19 hours in the elderly patients, 10.1 to 10.4 hours in the patients with hepatic impairment, and 8.7 to 9.5 hours in the patients with renal impairment. Rivaroxaban is highly protein bound to albumin, about 92% to 95%.
Apixaban is only indicated to reduce the risk of stroke and systemic embolism in patients with nonvalvular AF. The recommended dose of apixaban for most patients is 5 mg taken orally twice daily with or without food. Dose adjustments are required in certain patient populations. It is recommended that the dose be adjusted to 2.5 mg twice daily if a patient has 2 of the following characteristics: ≥80 years of age, body weight ≤60 kg, or with a serum creatinine ≥1.5 mg/dL. In addition, if administered with combined inhibitors of CYP3A4 and P-gp, it is recommended that the dose be 2.5 mg twice daily. Apixaban has a t 1/2 of 15.2 hours following the 5-mg dose and 6.8 hours following a 2.5-mg dose. In patients with low body weight, the t 1/2 can be increased to 15.8 hours. Apixaban is 87% protein bound.
Even with these newer agents, this risk of bleeding is still an important consideration. In the Rivaroxaban Once Daily Oral Direct Factor Xa Inhibition Compared with Vitamin K Antagonism for Prevention of Stroke and Embolism Trial (ROCKET) AF trial, which compared rivaroxaban to warfarin for patients with nonvalvular AF, the most frequent adverse reactions associated with permanent drug discontinuation were bleeding events with incidence rates of 4.3% for rivaroxaban versus 3.1% for warfarin. 30 Table 3 shows that 5.6% of patients in the rivaroxaban group experienced a major bleed while on the medication compared to 5.4% in the warfarin group. This worked out to an event rate of 3.6 per 100 patient-years in the rivaroxaban group and 3.5 per 100 patient-years in the warfarin-treated group.
ROCKET AF Bleeding Trial Results.a
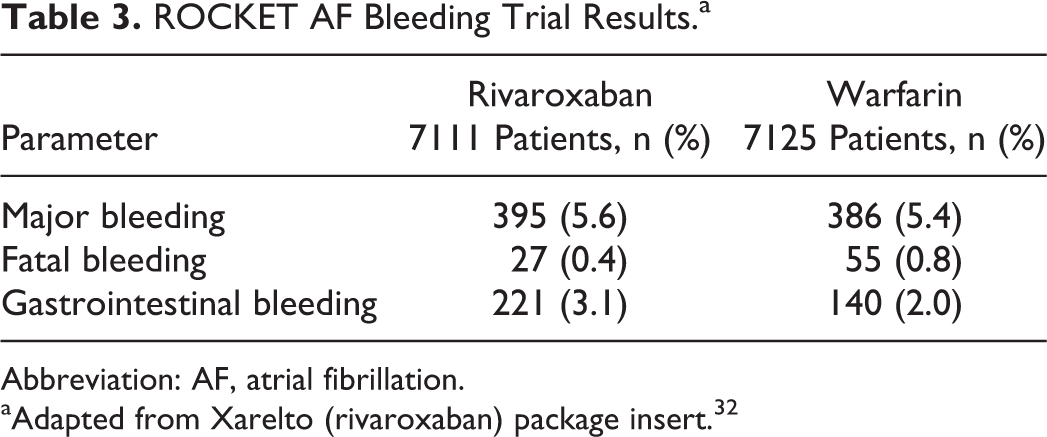
Abbreviation: AF, atrial fibrillation.
aAdapted from Xarelto (rivaroxaban) package insert. 32
For apixaban, the most common reason for treatment discontinuation in its landmark studies was once again bleeding-related adverse reactions. In the Apixaban for Reduction In Stroke and Other ThromboemboLic Events in Atrial Fibrillation (ARISTOTLE) study, this occurred in 1.7% of patients treated with apixaban and 2.5% treated with warfarin. 33 In the Apixaban Versus Acetylsalicylic Acid to Prevent Stroke in Atrial Fibrillation Patients Who Have Failed or Are Unsuitable for Vitamin K Antagonist Treatment (AVERROES) trial, the rate was 1.5% for apixaban compared to 1.3% for aspirin-treated patients. 34 As shown in Table 4, both major bleeding rates and clinically relevant nonmajor bleeding rates with apixaban were significantly lower than that of warfarin.
ARISTOLE Trial Bleeding Results.a
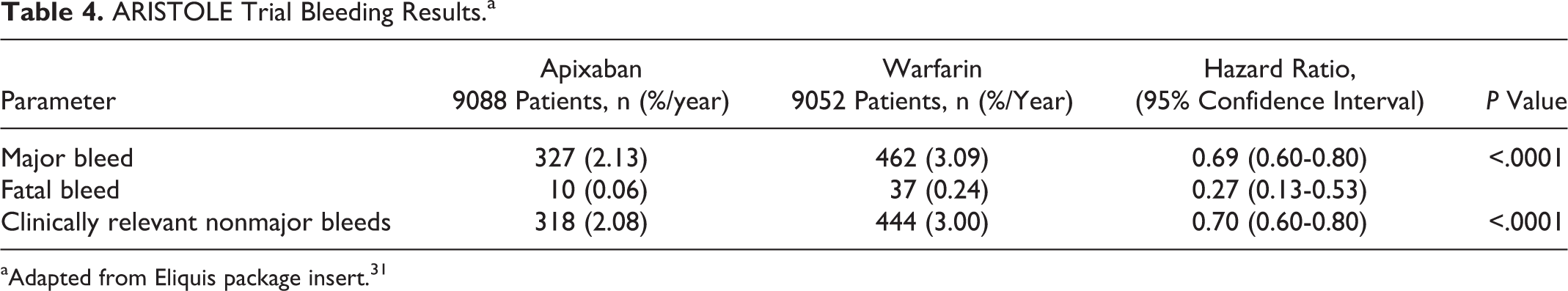
aAdapted from Eliquis package insert.31
According to the package insert for apixaban, marketed as Eliquis, “there is no established way to reverse the anticoagulant effect, which can be expected to persist for at least 24 hours after the last dose, that is, for about 2 half-lives.” 31 Because both agents are highly protein bound, they are not expected to be dialyzable. In addition, other conventional approaches for anticoagulant reversal including protamine sulfate and vitamin K are not expected to be effective due to the mechanism of action of this class. Currently, the use of supportive care and PCCs has been shown to partially reverse prothrombin time prolongation in healthy volunteers, although it has not been adequately evaluated in powered clinical studies. In 1 randomized, double-blind, placebo-controlled crossover study, 12 healthy volunteers received rivaroxaban 20 mg twice daily for 2.5 days, followed by either a single bolus of 50 IU/kg PCC or a similar volume of saline. Prior to PCC reversal, rivaroxaban had caused a significant prolongation of the prothrombin time versus baseline (15.8 ± 1.3 vs 12.3 ± 0.7 second P < .001). After administration of PCC, this prolonged prothrombin time was completely reversed 12.8 ± 1.0; P < .001 compared to the saline washout, which did not reverse the prothrombin time (16.2 ± 0.8 seconds; P = .4). 35 Prothrombin complex concentrate was also evaluated as a reversal for dabigatran in this study, which will be discussed later.
It is anticipated that these FXa inhibitors will be a mainstay in anticoagulant activity for years to come. Currently in development is a specific antidote for these medications that would be instrumental in assuring the safety of this class of medications when significant bleeds occur. The FDA has recognized that the lack of an effective reversal agent for FXa inhibitors is a significant unmet medical need. The FDA has designated andexanet alfa as a breakthrough therapy, which will potentially speed its approval process.
This recombinant protein called r-antidote, or andexanet alfa (PRT4445), is catalytically inactive and lacks the membrane-binding γ-carboxyglutamic acid domain of native FXa but retains the ability to bind to direct FXa inhibitors as well as activated antithrombin III. This protein has been able to dose dependently reverse the inhibition of FXa by these direct FXa inhibitors. When given alone, the r-antidote did not interfere with TF-initiated thrombin generation in human plasma. In early phase studies, it has been demonstrated both in vitro and in vivo that r-antidote does not interfere with normal FXa function in hemostasis and does not have anticoagulant activity. The r-Antidote is capable of binding to direct FXa inhibitors with affinities comparable to those of native FXa.
In a mouse model of blood loss, mice were treated with aspirin for 5 days followed by oral rivaroxaban that was administered at a high dose and produced variable blood loss in mice. These mice were then treated with an IV injection of r-antidote and compared to controls. The r-antidote was given 2 hours after rivaroxaban and reduced the blood loss by 84%. 36 In a liver laceration rabbit model, r-antidote was able to reduce blood loss by >85%, decrease peak anti-FXa activity by 98%, prothrombin time by 74%, and activated partial thromboplastin time (aPTT) by 66%. The nonprotein-bound fraction of rivaroxaban in plasma (active drug able to exhibit an effect) was decreased from 26% to 0.5%. 36 The r-antidote has the potential to reverse anticoagulant activity and restore hemostasis in patients treated with both direct and indirect FXa inhibitors.
In a phase 2 randomized, placebo-controlled, double-blind, cohort dose-escalation study, 54 healthy volunteers were treated with apixaban 5 mg twice a day for 6 days. 37,38 These patients were then randomized into 6 different cohorts each receiving varying doses of andexanet alfa. The first 3 cohorts used a single IV bolus at 90, 210, and 420 mg. The last 3 cohorts were 420 mg IV bolus plus a 45-minute infusion, a 2-hour infusion, or a repeat bolus at 45 minutes. Two minutes after administration of 420 mg of andexanet alfa, the anticoagulant activity of apixaban had decreased 95% as measured by anti-FXa activity. Safety data for over 65 healthy volunteers dosed with andexanet alfa has not shown any thrombotic events or the formation of any antibodies against andexanet alfa. Safety will continue to be monitored throughout the phase 2 and 3 trials; 1 serious adverse event was reported, a case of pneumonia in the phase 1 trial.
Direct Thrombin Inhibitors
Dabigatran etexilate is currently the only oral direct thrombin inhibitor available in the United States. Dabigatran etexilate is available as a prodrug that is rapidly converted to dabigatran, the active compound, in the body. Dabigatran is able to provide an anticoagulant effect by competitively inhibiting thrombin directly, which prevents thrombus development. Dabigatran and its acyl glucornides inhibit both free and clot-bound thrombin as well as thrombin-induced platelet aggregation. At recommended doses, dabigatran prolongs the aPTT, ecarin clotting time (ECT), and thrombin time (TT) but does not require routine blood testing for dose adjustments. 39
Dabigatran, marketed as Pradaxa, is indicated for use in the prevention of stroke and systemic embolism in adult patients with nonvalvular AF. It is primarily excreted renally with a half-life of 12 to 17 hours; this can be increased in patients with renal impairment to 15 to 34.1 hours. 40 For this reason, there are dose adjustments recommended with dabigatran. For patients with a CrCl greater than 30 mL/min the recommendation is 150 mg twice daily. With severe renal impairment, a CrCl 15 to 30 mL/min, the dose should be decreased to 75 mg twice daily. There are currently no recommendations available for patients with a CrCl less than 15 mL/min or in dialysis patients. However, dabigatran is dialyzable, and 48.8% to 68% of the drug can be removed at 4 hours. Thirty-five percent of the drug is protein bound in the body. It is recommended to avoid concomitant use of P-gp inducers. It is important to consider a patient’s bleeding risk while they are on dabigatran. Some situations are associated with a high bleeding risk including renal failure, advanced age, extremes in body weight, and drug–drug interactions.
For patients presenting with a bleed and currently on dabigatran, there are a few options that can be used to decrease blood loss. Blood levels should be drawn to determine the patient’s aPTT and TT levels. Activated charcoal can be used to reduce absorption from the gastrointestinal tract. This method is only recommended if the ingestion was within the last 2 hours since dabigatran is rapidly absorbed from the gastrointestinal tract. For patients with a mild bleed, tranexamic acid can be used to inhibit the binding of plasmin to fibrin. Tranexamic acid has been shown to reduce bleeding after tissue injury associated with surgery. 41 In addition, general supportive measures should be utilized to maintain renal perfusion and urine output—both aid in the elimination of dabigatran. These methods include compression, tamponade, surgery, and radiological interventions. Due to the short half-life of dabigatran for many patients with minor bleeds, these interventions may be sufficient.
For some patients, dialysis may be a valuable option. Due to dabigatran’s low protein binding, it is believed to be highly dialyzable. There has been some success in certain case studies with using hemodialysis to reverse dabigatran-related bleeds and prior to surgeries. In 1 such case, a 59-year-old female with heart failure and AF required an orthotropic heart transplant. Her last dose of dabigatran was taken approximately 36 hours prior to admission. When the patient presented, she had normal aPTT, an INR of 1.2, and an elevated TT of 90.6 seconds. After 1 hour of dialysis, her TT was 65.5 seconds, and after 2.5 hours, it was decreased to 60.2 seconds. The patient then underwent transplantation with no abnormal bleeding. 42
In another case report, a 94-year-old man with normal renal function developed an intracranial hemorrhage after a fall. The patient was started on hemodialysis to remove dabigatran from the body. After 3 hours of hemodialysis, the patient’s dabigatran concentrations decreased dramatically; however, an increase in concentration was observed after hemodialysis was stopped. This most likely reflects the redistribution of dabigatran from inside tissues to the plasma given the large volume of distribution of dabigatran in the terminal phase of elimination and may be evidence for increasing hemodialysis time. 43
In certain animal models, PCC has been found to result in hemostasis and the reversal of blood loss. Although dabigatran ultimately inhibits thrombin, it is believed that with enough factors present, dabigatran may not be able to inhibit all the thrombin present. This was tested in the rivaroxaban and dabigatran crossover study by Eerenberg et al. 35 Dabigatran was evaluated for its ability to be reversed by PCC. Twelve patients received dabigatran for 2.5 days, their aPTT was significantly increased after administration with dabigatran from 33.6 ± 3.3 to 59.4 ± 15.8 seconds P < .001. Endogenous thrombin potential (ETP) lag time was also prolonged with dabigatran use from 2.9 ± 0.4 to 7.5 ± 2.5 minutes P < .001. Thrombin time was >120 seconds, the upper limit for TT, in all patients receiving dabigatran. Ecarin clotting time was significantly prolonged as well, from 33 ± 1 seconds at baseline to 69 ± 26 seconds P = .002. The administration of PCC or saline had no effect on reducing any of these levels (aPTT, ETP, TT, or ECT). 35 Based on the findings in this study, there is limited evidence to support the use of PCC to neutralize the anticoagulant effect of dabigatran.
Factor eight inhibitor bypassing activity (FEIBA) contains activated factor X and prothrombin, which appear to be the main components responsible for its hemostatic action. The FEIBA has been shown to significantly reduce the bleeding time associated with dabigatran in a rat tail bleeding model. Because FEIBA contains activated factors, it is believed that this will lead to a greater procoagulant effect since activation is not required in the body and may help to reverse bleeding quicker than PCC. 44
Recombinant factor VIIa is able to directly activate factors IX and X, which results in an increase in thrombin production. This increase may be able to overcome the effect of dabigatran and lead to the formation of a fibrin clot. The rFVIIa has also been shown to reduce dabigatran-induced bleeding in a rat tail bleeding model; however, rFVIIa failed to reduce hematoma expansion or have an impact on mortality in a murine intracranial hemorrhage model. 37 In addition, rFVIIa has been associated with an increased risk of thromboembolic complications in other studies. Following the algorithm shown in Figure 2, rFVIIa can ultimately reduce life-threatening bleeding complications of dabigatran. 44
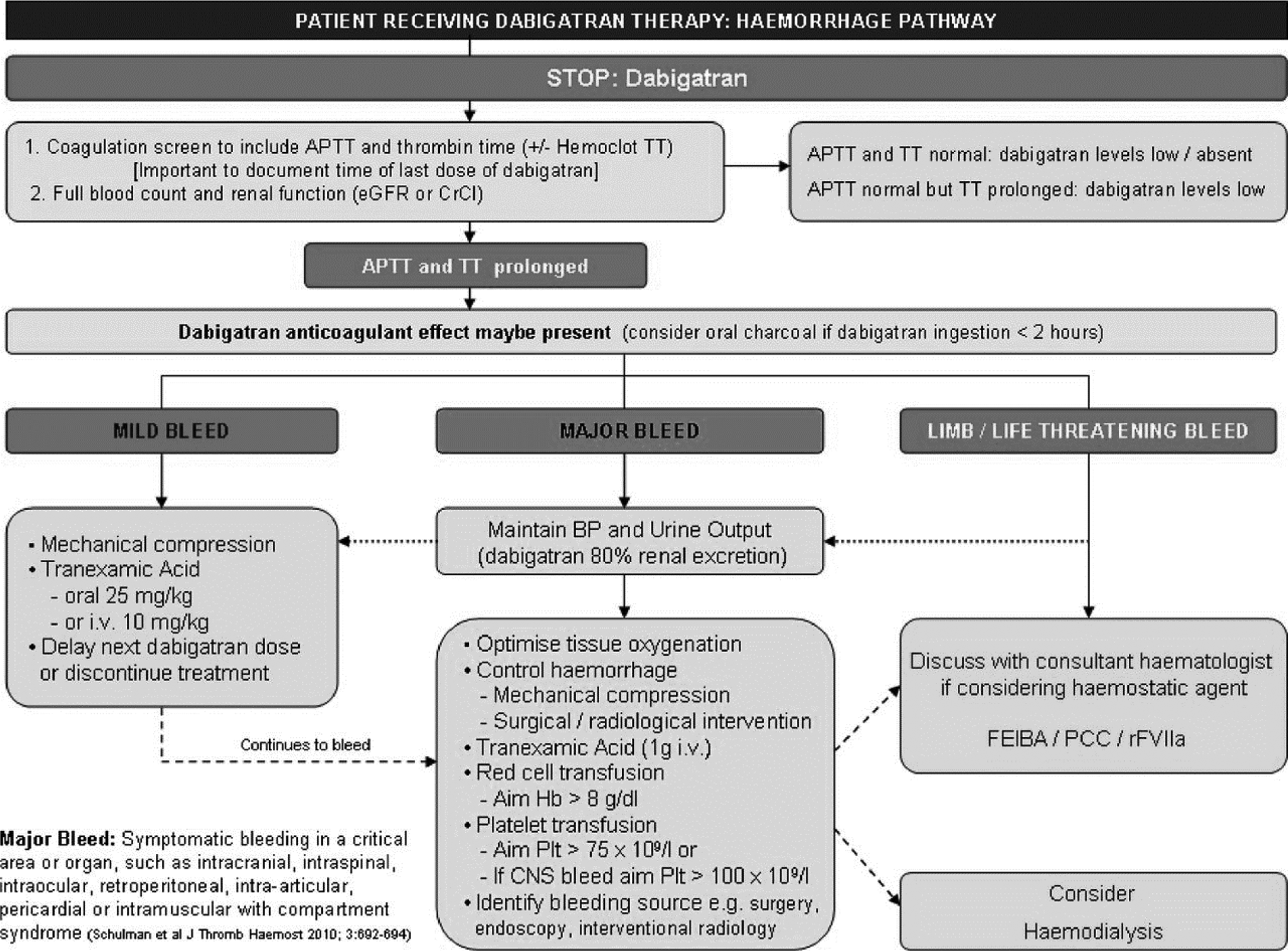
Treatment algorithm for dabigatran-related bleeds. Reproduced under open access from Alikhan et al, figure 1. 44 APTT indicates activated partial thromboplastin time; TT, thrombin time; eGFR, estimated glomerular filtration rate; CrCl, creatinine clearance; BP, blood pressure; IV, intravenous; Hb, hemoglobin; Plt, platelet; CNS, central nervous system; FEIBA, factor eight inhibitor bypassing activity; PCC, prothrombin complex concentrate; rFVIIa, recombinant factor VIIa.
Algorithms have also been published regarding how to handle patients who present requiring emergency surgery. Depending on the current APTT/TT, the time since administration, and how long the surgery can be delayed, there are treatment options outlined. 44 Additional algorithms will emerge to further simplify other clinical conditions as we learn more about the new, novel oral agents.
Conclusion
Oral anticoagulants are a mainstay in current clinical practice. Over the last few years, there has been an influx of novel oral agents gaining FDA approval. With a projected increase in AF and VTE cases and other thromboembolic disorders, these medications will remain an important part of treatment plans. Like most medications, these drugs come with some potential adverse events, including an increased risk of bleeding. Handling these situations quickly and effectively will ultimately lead to safer outcomes for patients. Utilizing current clinical treatment guidelines and staying up to date on newly approved reversal agents will decrease the risk associated with this class of medications.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
