Abstract
Direct oral factor Xa inhibitors (DXIs) are approved for use in adult patients with non-valvular heart disease for stroke prevention, and the treatment/prevention of venous thromboembolism (VTE). Recent pediatric clinical trials have demonstrated safety and efficacy of direct oral anticoagulants (DOACs) in the treatment of VTE. However, there is a lack of evidence regarding the use of andexanet alfa and prothrombin complex concentrates (PCC) for anticoagulation reversal of DXIs in the pediatric population. To better understand current DXI reversal strategies in this age group, a 10-question survey was conducted among pediatric hematology members of the Hemostasis and Thrombosis Research Society. Seventeen percent completed the survey (27 of 163 pediatric hematology members) with 74% (n = 20) reporting their use of DXIs for the treatment and prevention of VTE. Forty-four percent (n = 12) would choose andexanet alfa as first-line treatment for life-threatening bleeding while 52% (n = 14) would use PCC first with one individual choosing recombinant factor VII for DXI reversal. Clinical studies of andexanet alfa and PCC for the management of DXI-associated life-threatening bleeding require further investigation in the pediatric population.
Introduction
Warfarin was the first oral anticoagulant approved by the United States Food and Drug Administration (FDA) in 1954. 1 Direct oral anticoagulants (DOACs) were introduced to the market in 2010 with the approval of dabigatran, an oral direct thrombin inhibitor.2,3 Four other DOACs have been released and approved by the FDA for adult stroke prevention in atrial fibrillation, the treatment and secondary prevention of deep vein thrombosis (DVT) and pulmonary embolism: rivaroxaban (2011), apixaban (2012), edoxaban (2015) and betrixaban (2017).2,3 These agents are classified as direct oral factor X a inhibitors (DXIs). The stable pharmacokinetic profile of these agents eliminates the need for frequent pharmacodynamic monitoring that was a necessity for pediatric patients taking warfarin (International Normalized Ratio-INR) or low molecular weight heparin (LMWH; anti-Xa levels).2–8 The most recently published rivaroxaban trial in pediatric patients showed no increased bleeding when compared to standard anticoagulation [3% non-major bleeding in the rivaroxaban arm and 2% (major and non-major in the standard arm [HR 1.58, 95% CI 0.51-6.27]]. 9 The use of DOACs in pediatrics will obviate the need for anticoagulation monitoring and provide an alternative route of administration [oral], preferable for patients and families. With increasing use of these agents in the pediatric population, specific dosing recommendations and guidelines for anticoagulation reversal are needed for management of bleeding complications.
Andexanet alfa received FDA approval in 2018 for reversal of DXIs in adults with life-threatening bleeding. However, dosing recommendations for the pediatric population are lacking.10,11 Our own anticoagulation practice experience has shown an increased use of DOACs, particularly rivaroxaban. The recently published Einstein Jr studies and the ongoing Children's Oncology Group study PREVAPIX, have perhaps stimulated the use of rivaroxaban and apixaban in pediatric patients, particularly in teenagers.9,12–14 Because there is a lack of pediatric evidence-based literature regarding the use of andexanet alfa or PCC therapy for DXI reversal in children, we conducted a brief survey among pediatric hematologists, to better understand prescribing practices with apixaban and rivaroxaban and which agents are preferred when DXI reversal is needed for the management of bleeding complications.
Materials and Methods
A brief survey (10 questions) was designed by the authors (AD, VR, BK) to ascertain the prescribing practices of pediatric providers regarding apixaban and rivaroxaban, and preferred reversal agent depending upon the severity of bleeding. The specific brand of PCC preferred by respondents was not requested in the survey. Activated prothrombin complex concentrates (aPCC) such as FEIBA® were not included in the survey due to the authors’ institutional clinical guidelines recommendations of PCC treatment (instead of aPCC) for anticoagulation reversal if andexanet alfa is not available. Guidance from the Anticoagulation Forum recommends aPCC for dabigatran reversal in the absence of idarucizumab. 15 Other agents such as blood products and recombinant factor VII were added as potential anticoagulation reversal choices as per the author's experience, these agents are commonly used in the management of bleeding complications related to pediatric anticoagulation. The survey was reviewed and approved for distribution by the Hemostasis and Thrombosis Research Society (HTRS) publications committee. The authors decided to use HTRS members as they felt that this pediatric hematology group specializes in thrombosis and regularly prescribes anticoagulants in their practices more frequently than the general pediatric hematology-oncology medical community. A link to the final survey version (see appendix) was sent via email blast to 163 pediatric hematologists who were active trainee or core members of HTRS between January 28 and April 1, 2021.
Survey Monkey© was used as the platform. Responses were anonymous and results of the survey were shared with participants who provided their electronic addresses. Responses were summarized with frequency and percentage. Comparisons of responses between groups were done with chi-square or Fisher's exact tests. P-values were two-sided and those <.05 were considered statistically significant. Analyzes were completed using the base R statistical package (R Foundation for Statistical Computing, Vienna, Austria).
Results
Of 163 eligible HTRS pediatric members, 27 completed the survey (17% response rate). Only one respondent was a trainee, and the rest of the participants were practicing pediatric hematologists with a similar distribution of years in practice (Table 1). The majority of participants (89%; n = 24) practiced primarily in an academic center with 52% (n = 14) reporting care for an average of 5-10 pediatric patients with thrombosis per month (Table 1).
Respondents Demographics.

In terms of anticoagulant practices, 89% (n = 24) reported the use of LMWH for prepubertal children and apixaban/rivaroxaban for adolescents. When asked which clinical scenarios will you prescribe rivaroxaban or apixaban? Seventy-four percent prescribed both DXIs (n = 20), 26% either apixaban or rivaroxaban (n = 7). Eighty-nine percent (n = 24) reported prophylaxis and therapy of VTE as the main indications for choosing DXI therapy. Forty-one percent (n = 11) prescribed apixaban or rivaroxaban regardless of the patients’ age, while 15% (n = 4) reported the use of either DXI specifically in patients weighing greater than 50 kg (Table 2).
Summary of Survey Responses.
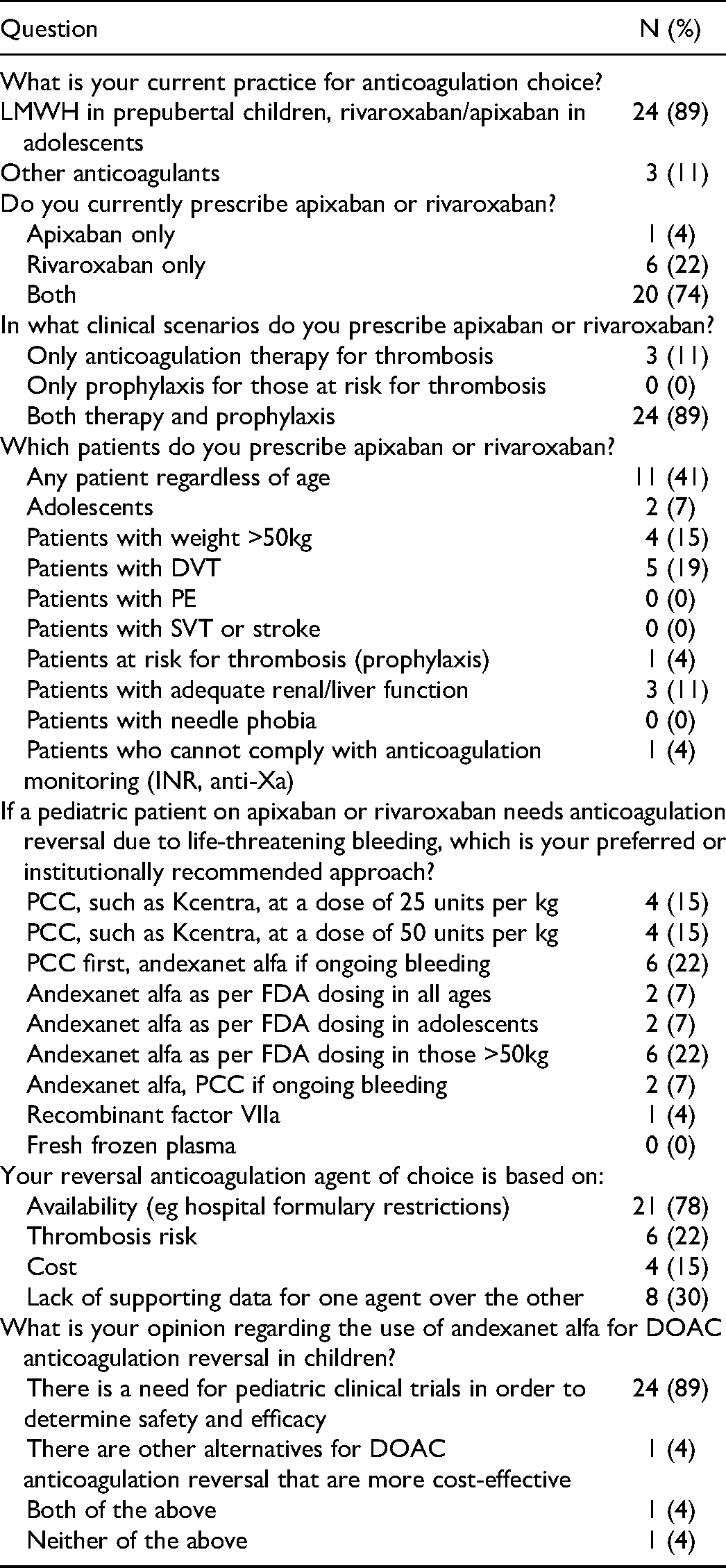
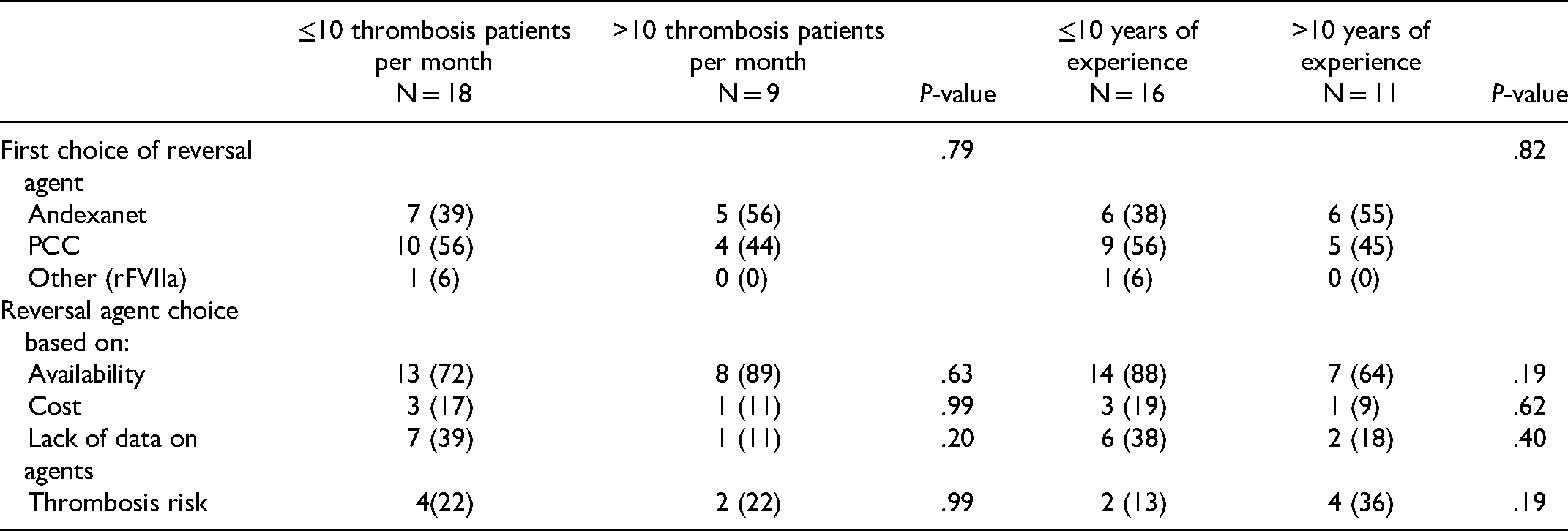
When asked about their drug of choice for anticoagulation reversal of apixaban and rivaroxaban, 44% (n = 12) prefer andexanet alfa while 52% (n = 14) would prescribe a PCC for the reversal of life-threatening bleeding as first-line therapy (one respondent preferred the use of recombinant factor VII). Seventy-eight percent (n = 21) reported formulary availability as the primary reason for choosing a particular reversal agent, followed by 30% (n = 8) indicating the lack of data to recommend one reversal agent over the other, 22% (n = 6) were concerned about thrombotic risk with reversal and 15% (n = 4) noted the cost of medication as other reasons for choosing a particular reversal agent. Eighty-nine percent of respondents (n = 24) agreed that there is a need for clinical studies on the use of andexanet alfa in children needing reversal (Table 2). Potential factors that might have influenced respondents’ choice and reasons for DXI reversal agent preference, such as the number of patients with thrombosis seen per month and years of practice were analyzed and compared (Table 3). The volume of patients with thrombosis (≤ or > 10 per month) and years in practice (≤ or >10 years) were not significantly associated with reversal agent choice nor the reason for choosing a particular agent in this study (Table 3).
Comparative Analysis of Choice of Reversal Agents Based on Number of Patients Seen per Month in Physicians’ Practices and by the Number of Years of Experience.
Discussion
Direct oral anticoagulants (DOACs) are increasingly used in adult patients due to their efficacy and safety profile demonstrating a reduction in the risk of fatal bleeding when compared to warfarin.16,17 However, major or clinically relevant non-major bleeding (CRNMB) have been reported in adult patients receiving rivaroxaban [8.1%] and apixaban [4.3%].4,5,7 Recent studies of DOAC agents in children have demonstrated good efficacy and safety for pediatric VTE therapy.9,12,13 Apixaban is currently being studied in two pediatric clinical trials.14,18 When this survey was conducted, no DXIs were FDA approved for pediatric use but pediatric hematologists were already using DXIs with increasing frequency for childhood VTE management, as illustrated by this survey, perhaps due to the recently published safety and efficacy of rivaroxaban in pediatric patients. 9 In the interval between survey administration and publication of this report, rivaroxaban received FDA approval for the management of childhood VTE. 19 Which is expected to accelerate the prescribing frequency of DXIs to children with VTE.
Choosing the right anticoagulant agent involves consideration of multiple factors: age of the patient, route of administration, frequency of pharmacokinetic monitoring, drug interactions, organ function, bleeding risk, and compliance. Standard anticoagulation agents have reversal options such as vitamin K, fresh frozen plasma, and PCCs for warfarin and protamine sulfate for heparin. 20 The degree of reversal depends on the anticoagulant used, dose and pharmacokinetics. With the increased adoption of DXIs, there will be increasing need for reversal of these agents, even though the overall anticoagulant-related bleeding rate may decrease in pediatric patients receiving these agents. The recommended dose and indication for the use of the different DXI reversal agents, are thus needed for the pediatric population.
Andexanet alfa received FDA approval for use in adults in need of DXI reversal in 2018. 10 It is a recombinant enzymatically inactive factor Xa decoy protein that has higher binding affinity for DXIs than does natural factor Xa. 11 Prospective and retrospective studies in adults have been completed using andexanet alfa and PCC for the reversal of DXIs. Prothrombin complex concentrates contains concentrated vitamin K-dependent coagulation factors (factors II, VII, IX, X) extracted from large pools of donor plasma and stored in the form of a lyophilized powder. Currently PCC is available in two forms: four-factor PCC (4-PCC) and three factor PCC (3-PCC) with 4-PCC having higher concentrations of factor VII in addition to some anticoagulant proteins (Protein C, Protein S, antithrombin). 15 A recent meta-analysis of andexanet alfa and PCC use for the management of DXI-related major bleeding in adult patients, demonstrated similar hemostatic efficacy for both agents: pooled proportion of 0.85 [95% CI 0.80-0.90] for PCC and 0.82 [95% CI 0.78-0.87] for andexanet alfa, respectively. 11 The pooled proportion of patients with thromboembolism was 0.03 [95% CI 0.02-0.04] in the PCC treated group and 0.11 [95% CI 0.04-0.18] in the andexanet alfa studies. 11 So far, the data on PCC for hemostatic control for DXI-related bleeding showed variable results in terms of hemostatic efficacy and thrombosis risk with the previous analyzes limited by sample size, patient comorbidity heterogeneity, and variable study design.21–27 Our survey illustrated a divided opinion among pediatric hematologists regarding the preferred DXI reversal strategy with nearly 50% choosing andexanet alfa versus PCC (44% [n = 12] andexanet alfa vs 52% [n = 14] PCC).
The cost of andexanet alfa is another potential factor that may influence formulary availability in hospitals. The price of PCC (eg Kcentra®, a 4-PCC) is $1.62 per unit contrasted with andexanet alfa ($3300/100 mg) which for an adult size patient can translate into $50 000 compared to $3240 for the recommended 2000 units of Kcentra. 26 Our survey showed that 78% (n = 21) of the respondents’ hospitals lack andexanet alfa formulary approval as the main reason dictating what kind of reversal agent would be used for DXI-related bleeding (15% report cost as a reason). Thirty percent (n = 8) reported a lack of data supporting one agent over the other. Slightly over half of the respondents would use a PCC first while 44% would use andexanet alfa. Based on the adult literature, there is no conclusive evidence that one agent is superior to the other as there are no concurrent parallel comparisons of PCC versus andexanet alfa, in terms of efficacy and safety.11,21–27 Meanwhile guidance from the Anticoagulation Forum recommends supportive measures such as discontinuation of DOAC and medications that can interfere with hemostasis such as antiplatelet agents.28,29 Administration of a reversal agent is advised only if bleeding is life-threatening or involves a critical organ and is not controlled despite maximum supportive measures. The reversal agent suggested in the Anticoagulation Forum Guidance is andexanet alfa dosed according to the FDA label. If andexanet alfa is not available, treatment with 4-factor-PCC at a dose of 2000 units is recommended. In a patient who requires an invasive procedure that cannot be safely performed while the patient is anticoagulated and cannot be delayed, a reversal agent is suggested. 29 These guidelines are for adult patients only and no pediatric guidance has been formulated to address the management of pediatric DXI-related bleeding complications.
Our study has several limitations. The response rate was low (17%) and, as a result, the sample size may not be a true representation of pediatric hematologists who routinely diagnose and manage children and adolescents with thrombotic disease. This small sample size limited subgroup analysis and comparisons. Questions addressing the number of DXI-related bleeding episodes treated by each participant were not included in the survey as these complications were no different compared to standard of care in previous pediatric trials. However, the inclusion of this survey question might have been informative in trying to compare anticoagulant reversal agent preference. With the recent FDA approval of dabigatran (an oral direct thrombin inhibitor) for pediatric use, studies to assess the efficacy and safety of idarucizimab reversal in children are also needed. 30 Our survey did not include questions directed to the use of dabigatran reversal since idarucizumab is the only evidence-based option.
In conclusion, our survey illustrates that DXIs use was increasing in the pediatric setting prior to the recent FDA-approval of rivaroxaban for pediatric use (which may be expected to accelerate this trend). The preference of reversal agent for DXI-related bleeding management essentially equally divided between PCC or andexanet alfa among survey participants. The adult literature does not clearly support the effectiveness and safety of one DXI reversal agent over the other. There is a lack of published data on DXI reversal strategies for children and adolescents. It is reasonable to expect that DXI use will continue to become more common in pediatric patients thus clinical research is needed to develop evidence-based protocols for DXI reversal in this patient population.
Supplemental Material
sj-docx-1-cath-10.1177_10760296221078842 - Supplemental material for Andexanet Alfa Versus Prothrombin Complex Concentrates/Blood Products as Apixaban/Rivaroxaban Reversal Agents: A Survey Among Pediatric Hematologists
Supplemental material, sj-docx-1-cath-10.1177_10760296221078842 for Andexanet Alfa Versus Prothrombin Complex Concentrates/Blood Products as Apixaban/Rivaroxaban Reversal Agents: A Survey Among Pediatric Hematologists by Vilmarie Rodriguez, Joseph Stanek, Bryce A. Kerlin and Amy L. Dunn in Clinical and Applied Thrombosis/Hemostasis
Footnotes
Acknowledgements
We would like to thank Hemostasis and Thrombosis Research Society (HTRS) for facilitating the conduct of this study.
Author Contributions
AD conceived idea of the survey, AD, VR, BK designed survey questions. VR and JS: data collection, data analysis. VR, AD, BK, JS: manuscript writing and review.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
Ethics Approval
Our institution does not require ethical approval for reporting individual cases or case series. Survey was exempt.
Informed Consent
Informed consent for patient information to be published in this article was not obtained because: not applicable.
Relationship Disclosure
Dr Dunn:
Baxalta/Shire/Takeda: clinical trial support Biomarin: clinical trial support CSL Behring: Advisory board, DSMB Freeline: clinical trial support Kedrion: advisory board Genetech: Advisory board Novo Nordisk: clinical trial support, advisory board World Federation of Hemophilia USA: Board member Ohio Bleeding Disorder Council: Board member Uniqure: DSMB Sanofi: Clinical Trial Support
Dr Kerlin:
HEMA Biologics: Advisory Board Catalyst Biosciences: DSMB Novo Nordisk: Advisory Board, Steering Committee, Research Support HTRS and Genentech: Research Support
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
