Abstract
To review the diagnostic difficulties of a prolonged activated partial thromboplastin time (aPTT) when 2 inhibitors with opposite clinical presentations coexist, we searched MEDLINE from January 1970 to November 2013 using acquired, factor VIII (FVIII), factor IX, hemophilia A and B, inhibitor, lupus anticoagulant (LA), antiphospholipid, anticardiolipin, anti-β2-glycoprotein I, antibodies, syndrome, bleeding, and thrombosis. We identified 13 articles for a total of 15 cases of possible coexistence of FVIII inhibitor and LA. The presenting clinical manifestation was thrombosis in 6 cases and bleeding in 9 cases. Activated partial thromboplastin time was the presenting laboratory abnormality in all cases, and first-line investigations suggested the coexistence of LA and acquired FVIII inhibitor. None of the articles addressed the diagnostic accuracy of the screening tests by performing “second line” assays. We reviewed the diagnostic pitfalls of the cases under study and provide some guidance for alternative tests when facing a prolonged aPTT that may have a double meaning.
Introduction
The differential diagnosis of a prolonged activated partial thromboplastin time (aPTT) is quite extensive and necessitates further investigations following an algorithm to identify factor deficiencies and inhibitors. 1 However, once the laboratory differential diagnosis has narrowed down to the presence of inhibitors, their identification is of the utmost importance to avoid significant delays 2 in the correct therapeutic approach 3 that could be otherwise fatal. 4 Things may be further complicated by the coexistence of different inhibitors such as an acquired factor VIII (FVIII) and a lupus anticoagulant (LA) that underscore completely opposite clinical scenarios. Both inhibitors prolong the aPTT and may affect each other in clotting assays hence their discrimination may be quite challenging 5 regardless of whether the presenting clinical presentation is bleeding or thrombosis. To address this rare but important laboratory and clinical scenario, we performed a review of case reports and case series where an LA coexisted with acquired FVIII inhibitor, and we critically reviewed the laboratory methodology employed to differentiate the coexistent inhibitors and the management of the clinical phenotypes.
Materials and Methods
We performed a PubMed literature search from January 1970 to November 2013 using the following search terms: acquired, FVIII, factor IX (FIX), hemophilia A and B, inhibitor, LA, antiphospholipid, anticardiolipin, anti-β2-glycoprotein I, antibodies, and syndrome. The minimal inclusion criteria required that the title or the abstract described the coexistence of an LA with acquired FVIII inhibitor. This search yielded 379 articles; allowing for replication and after checking titles and abstracts for the coexistence of LA with acquired hemophilia A (AHA) and B (AHB), we ended up with 13 articles: of these 11 were individual cases and 2 case series of LA coexistent with AH for a total of 15 cases.
Results
We identified 15 cases of an LA coexisting with AHA (n = 14) and AHB (n = 1) where the laboratory presentation was a prolonged aPTT: the initial clinical presentation was vascular occlusion in 6 cases 6 –11 and bleeding in 9 cases. 9,12 –18 The different tests to diagnose LA, FVIII levels, and its inhibitors are shown in Table 1. The subsequent paragraphs comment on the cases according to the presenting clinical phenotype leaving in the discussion-specific remarks on the tests employed for the diagnosis.
Cases of Coexistent Lupus Anticoagulant and Acquired Factor VIII (Factor IX) Inhibitor: Test Patterns.a
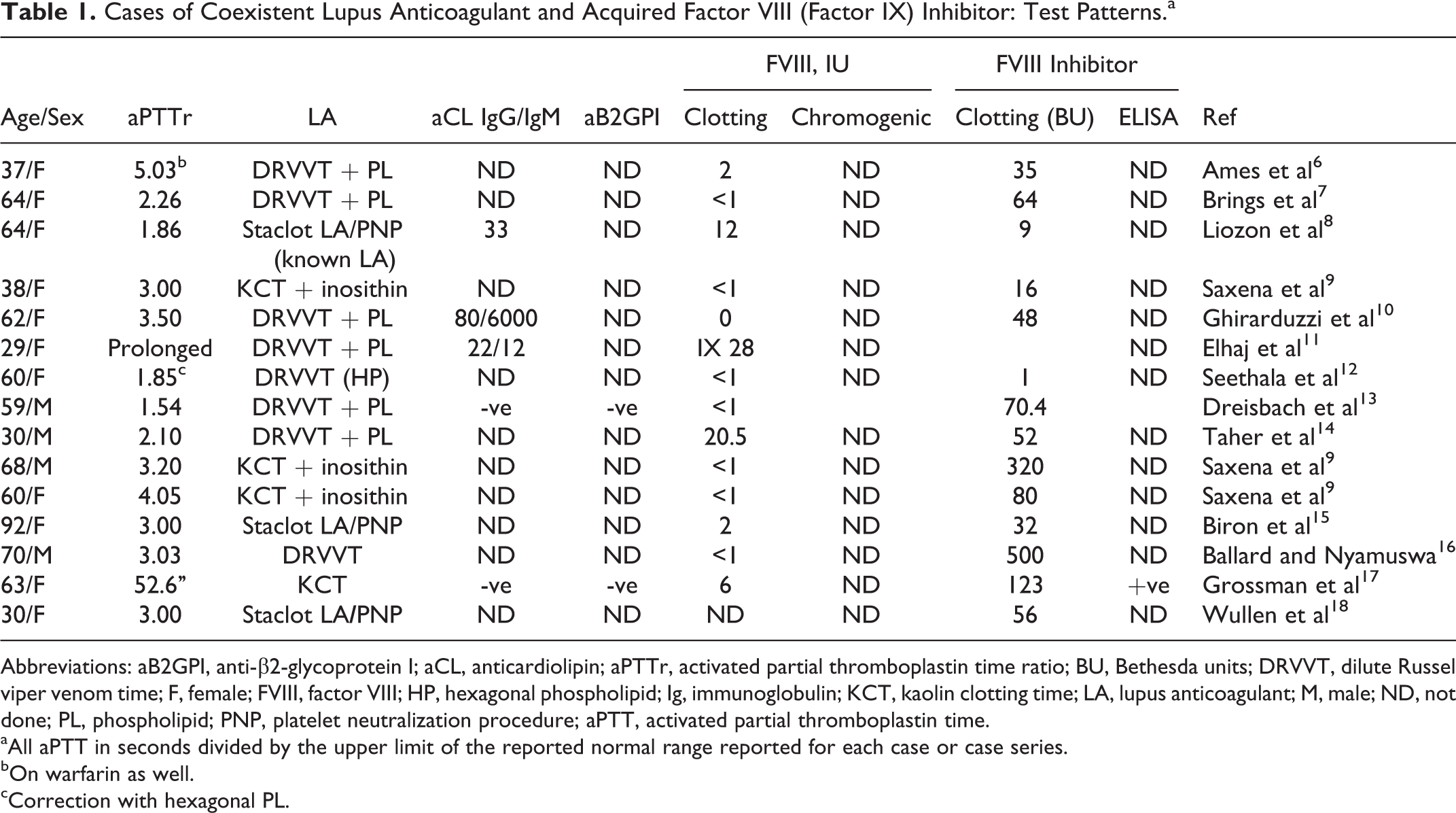
Abbreviations: aB2GPI, anti-β2-glycoprotein I; aCL, anticardiolipin; aPTTr, activated partial thromboplastin time ratio; BU, Bethesda units; DRVVT, dilute Russel viper venom time; F, female; FVIII, factor VIII; HP, hexagonal phospholipid; Ig, immunoglobulin; KCT, kaolin clotting time; LA, lupus anticoagulant; M, male; ND, not done; PL, phospholipid; PNP, platelet neutralization procedure; aPTT, activated partial thromboplastin time.
aAll aPTT in seconds divided by the upper limit of the reported normal range reported for each case or case series.
bOn warfarin as well.
cCorrection with hexagonal PL.
Coexistent LA and AH Presenting as Thrombosis
Vessel occlusion was the clinical presentation in 6 of the 15 cases of which 5 were deep vein thrombosis (DVT) 6 –10 and 1 was ischemic stroke. 11 Of the 5 DVT cases, 2 developed postpartum, 6,9 1 in the absence of an underlying disorder, 7 1 during the course of non-Hodgkin lymphoma, 8 and 1 during treatment for chronic lymphoid leukemia. 10 Ischemic stroke developed in 1 patient with AHB having underlying insulin-dependent diabetes mellitus. 11 These underlying conditions may have contributed to the development of thrombosis. This was accompanied by bleeding in 3 cases where the FVIII level was 2 IU or less, 6,7,9 whereas it was the only clinical manifestation in the patients with lymphoma where FVIII was 12 IU and FIX was 28 IU, 8 although the patients with chronic lymphatic leukemia developed a fatal pulmonary embolism despite an unmeasurable FVIII level. 10 Conversely, warfarin may have contributed to the development of the hematoma in the first patient listed in Table 1 6 who presented a week earlier with DVT. The hematomas or ecchymoses of the 3 patients with bleeding were variably but effectively managed with recombinant activated factor VII (rVIIa), FVIII, or FVIII by-passing agents 6,7,9 whereas the FIX inhibitor was ameliorated by plasmapheresis. 11 The use of procoagulants to correct the bleeding tendency was not associated with recurrent vascular occlusions.
The LA was detected by a dilute Russell viper venom time (DRVVT) in 3 instances, 7,10,11 by kaolin clotting time (KCT) with inosithin neutralization in 1 patient 9 and by the Staclot LA in 1 case where it had been presented before the appearance of the FVIII inhibitor. 8 In the first patient listed in Table 2, 6 LA was suspected at presentation by comparing the ratios of the aPTT performed with a sensitive and an insensitive reagent to the LA, whereas the DRVVT tested positive during follow-up. Immune suppression with cyclophosphamide and steroids leads to clearance of the FVIII inhibitor in 4 cases (including the FIX inhibitor) 6 –9,11 of which only 1 reported disappearance of the LA. 6 In the remaining 2 cases, the follow-up of the patient did not report on the FVIII inhibitor status after immune suppression 7 and in 1 case the patient died preventing further testing. 10
Cases of Coexistent Lupus Anticoagulant and Acquired Factor VIII (factor IX) Inhibitor: Clinical Presentations and Outcomes.
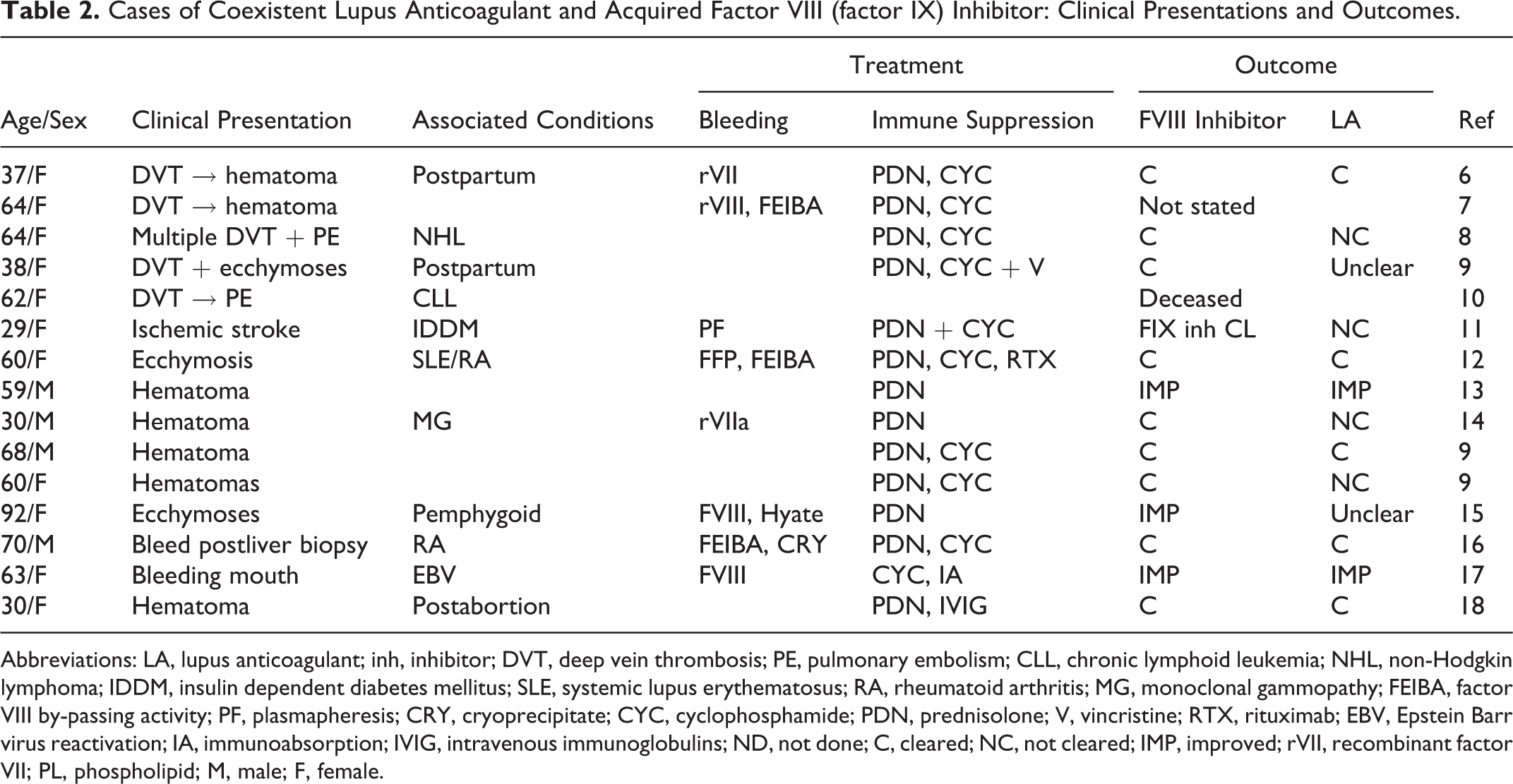
Abbreviations: LA, lupus anticoagulant; inh, inhibitor; DVT, deep vein thrombosis; PE, pulmonary embolism; CLL, chronic lymphoid leukemia; NHL, non-Hodgkin lymphoma; IDDM, insulin dependent diabetes mellitus; SLE, systemic lupus erythematosus; RA, rheumatoid arthritis; MG, monoclonal gammopathy; FEIBA, factor VIII by-passing activity; PF, plasmapheresis; CRY, cryoprecipitate; CYC, cyclophosphamide; PDN, prednisolone; V, vincristine; RTX, rituximab; EBV, Epstein Barr virus reactivation; IA, immunoabsorption; IVIG, intravenous immunoglobulins; ND, not done; C, cleared; NC, not cleared; IMP, improved; rVII, recombinant factor VII; PL, phospholipid; M, male; F, female.
Coexistent LA and AH Presenting as Bleeding
Of the 9 patients with bleeding, 9,12 –18 an underlying autoimmune disorder was present in 3 patients, 12,15–16 a monoclonal gammopathy in 1 patient, 14 reactivation of Epstein Barr virus in 1 patient, 17 and ovarian hyperstimulation followed by an abortion in a further one. 18 In the latter case, it is interesting that bleeding occurred despite 2 thrombogenic risk factors (ovarian hyperstimulation and abortion). 18 Control of bleeding was obtained with rVIIa in 1 patient, 14 FVIII by-passing activity in 2 patients, 12,16 and FVIII in 2 patients, 15,17 followed by immune suppression. This was with cyclophosphamide and prednisolone in 4 cases 9,12,16 (aided by rituximab in 1 case), 12 with cyclophosphamide and immune adsorption in 1 case, 17 by prednisolone alone in 3 cases, 13 –15 and by prednisolone and intravenous immunoglobulins (Igs) in 1 case. 18 Lupus anticoagulant was detected by a DRVVT in 4 instances, 12 –14,16 by the Staclot LA in 2 cases, 15,18 and by KCT in 3 cases. 9,17 The FVIII inhibitor disappeared or improved in all cases, but the LA disappeared or improved only in 6 cases, 9,12,13,16 –18 did not disappear in 2 cases 9,14 and was not documented in 1 case. 15
Discussion
Clinical Approach to the Coexistence of Acquired FVIII (FIX) Inhibitor and LA
Acquired hemophilia A has an annual incidence of 1.5/million and affects prevalently people between 75 and 80 years of age, 19 whereas thrombosis associated with LA occurs at a much younger age. 20 A history of connective tissue disorders, autoimmune disorders, and cancer may not necessarily provide useful clues as both inhibitors may be associated with these underlying conditions, although the presenting clinical manifestation should guide the discerning clinician. In AHA, the clinical presentation is prevalently hemorrhage according to the level of FVIII but attention should be paid to concomitant anticoagulation and/or antiplatelet agents 21 as AHA is not exempt from rare vessel occlusions not due to LA. 22 Conversely, bleeding in LA is rare and mostly due to the LA-hypoprothrombinemia syndrome where hemorrhage was present in 89% (66 of 74) of a specific series of patients with the syndrome. 23 Thrombocytopenic bleeding associated with LA in APS is uncommon 24 : a prolonged bleeding time was reported in 78% (21 of 27) of patients with LA despite normal platelet counts in the absence of clinical bleeding, 25 although Primary Antiphospholipid Syndrome (PAPS) patients on oral anticoagulation showed a greater rate of mucosal bleeding at international normalized ratio (INR) 3.0 to 4.0 compared to patients with inherited thrombophilia within a similar INR range. 26
Conventional Laboratory Approach to the Diagnosis of Acquired FVIII (FIX) Inhibitor and LA
The clinical manifestations mentioned previously provide hints toward the interpretation of an isolated aPTT prolongation. The sequential steps to diagnose the LA and acquired FVIII inhibitor are summarized in Table 3. 29,30 A thrombin time will exclude the presence of heparin (whether as an inhibitor or as a treatment contaminant); protamine sulfate and heparanase may normalize the test via heparin neutralization or digestion, respectively. With a normal thrombin time, the mixing stage provides clues to the presence of inhibitors.
Diagnostic Pathway for Coexistent Lupus Anticoagulant and Factor VIII Inhibitors.

Abbbreviations: aPTT, activated partial thromboplastin time; DRVVT, dilute Russel venom viper time; FVIII, factor VIII.
aAdapted from Pengo et al. 29
bAdapted from Hay et al. 30
The interpretation of mixing studies must take into account the factor sensitivity of the aPTT reagent. A specimen containing 1% FVIII mixed 1:1 with normal plasma containing 100% FVIII will yield a plasma mix with 50.5% of FVIII. If the aPTT reagent is sensitive to a FVIII level below 40%, the mixing study will correct into the normal range. If the aPTT reagent is sensitive to a FVIII level below 55%, the mixing study (FVIII at 50.5%) may not correct the aPTT into the normal range. In this case, factor sensitivity to the aPTT reagents plays a role modulating the correction of the mixing study. Nevertheless, correction of the prolonged aPTT into the normal range should not necessarily exclude an LA, as a weak LA might correct upon mixing despite its presence. In addition, the aPTT should be tested immediately after 1:1 (1:4) mixing and after 60 and 120 minutes of incubation as 5% of LA may show time dependency. 31
If the aPTT correction suggests a factor deficiency, the 1-stage aPTT FVIII is generally employed to measure the level of FVIII, but the result may not show parallelism in the presence of an LA, and the level of FVIII may be less reliable. All the cases of the current review used a 1-stage clotting assay for FVIII measurement and did not take this aspect into account (Table 1). In the presence of anti-FVIII autoantibodies, the Bethesda assay should present with a type II inhibitor pattern but also this assay may be confounded by the LA. 32 Again all the cases bar one of the current review measured FVIII inhibitor by the Bethesda assay (Table 1).
With regard to the LA, once suspected it must be confirmed by a phospholipid neutralization assay (platelet neutralization procedure [PNP]): exogenous platelets and hexagonal phospholipids (HPs) yield aPTTs that are shorter than the aPTTs done with reagents sensitive to the LA. The seminal PNP was reported to be specific for the LA as it discriminated LA from acquired factor inhibitors 33 : the study had 1 acquired FVIII inhibitor sample that showed a slight shortening with the PNP that was always greater than that obtained with the aPPT performed with saline control. 33 Hexagonal phospholipid is used in the commercially available Staclot LA, but this confirmatory test can give a false-positive result in the presence of a FVIII inhibitor, although it performs better than the PNP. 34 The same confirmatory effect can be achieved with inosithin (soyabean phosphatides). 27 The present review shows that confirmatory LA assays relying on aPTT systems were used in 7 of the 15 patients: Staclot LA was used in 3 of the 15 patients, the KCT with inosithin correction in 3 patients, and KCT without phospholipid neutralization in 1 patient (Table 1).
Reciprocal Influences on the Detection of FVIII and LA
Factor VIII promotes FIX activation together with activated FIX, calcium ions, and phospholipids clearly explaining why LA systems based on the aPTT and KCT may be influenced by the presence of the FVIII inhibitor and by the decreased availability of FVIII. Therefore, the prolongation of the KCT and the Staclot LA of the 7 cases in our review may be partly accounted for by low FVIII and FVIII inhibitor. Only 5 cases reported the results of a solid-phase immune assay for antiphospholipid antibodies in support of the LA 8,10,11,13,17 with only 3 positive results. 8,10,11
In this scenario, the DRVVT is more reliable in that it directly activates factor X downstream of FVIII and should not be influenced by low FVIII and its inhibitor but it must be followed by a mandatory phospholipid neutralization test. However, a FVIII concentration below 0.15 IU/mL may still influence the DRVVT result according to the sensitivity of reagents, and the PNPs may not correct the prolonged DRVVT. 28 The DRVVT was used in 8 of the 16 cases for the diagnosis of LA in the cases we reviewed and in 5 of the 8 cases the 1-stage clotting assay for FVIII was <1 IU (Table 1).
An alternative way to avoid the interference of FVIII deficiency or inhibitor is to resort to the Textarin–Ecarin ratio based on direct prothrombin activation. Textarin is derived from the venom of the Australian Eastern brown snake called Pseudonaja textilis, whereas Ecarin is derived from the venom of the saw-scaled viper called Echis carinatus. Textarin requires factor V, calcium ions, and phospholipid to directly activate prothrombin, whereas Ecarin activates prothrombin to form meizothrombin without phospholipids. Individual Textarin and Ecarin clotting times are derived for a plasma sample and the ratio is then calculated: in the presence of an LA, the Textarin clotting time will be prolonged due to its phospholipid-dependency and will yield a greater Textarin–Ecarin ratio. 35 The Textarin venom can be substituted by the Taipan venom from Oxyuranus scutellatus that also activates prothrombin in the presence of phospholipid and calcium. 36 The Textarin–Ecarin 35 or the Taipan–Ecarin ratios 36 are confirmatory test for LA, but they have not been fully evaluated to be incorporated in the LA guidelines 29
With regard to FVIII measurements, LA may affect the results of coagulation-based assays hence chromogenic or immune assays for FVIII measurement should be performed to establish a proper FVIII level 37 : none of the cases we reviewed employed these assays. Lupus anticoagulant may also influence the Bethesda assay therefore a solid-phase immune assay for FVIII inhibitor would be mandatory but it was performed only in one of the cases presented here. 17 But even the solid phase for FVIII inhibitor may yield a false-positive signal in the presence of LA: it was found positive in 13% of samples for LA (n = 143), in 18% samples for IgG anticardiolipin antibodies (n = 84), and in 10% of samples for IgG anti-β2-glycoprotein 1 antibodies (n = 62). 38 This phenomenon may be explained by the fact that antiphospholipid antibodies cross-react with phospholipid binding sites in the C2 domain of FVIII. 39
Suggestions for Diagnosing Coexistent LA and FVIII Inhibitor
All the points mentioned previously need to be considered when there is a suspicion of coexistent LA and acquired FVIII inhibitor. With regard to the LA: (1) aPTT systems become unreliable for the diagnosis of LA, because of the interference by the FVIII deficiency and its inhibitor in the intrinsic pathway upstream of factor X activation; (2) DRVVT directly activates factor X but may be affected by very low FVIII levels; and (3) the Textarin–Ecarin ratio acts on prothrombin, downstream of factor X and may be the test of choice in this very context. 35 With regard to FVIII and its inhibitor: (1) FVIII should be measured by a chromogenic assay and (2) FVIII inhibitor titer should be performed by a solid-phase immune assay.
Conclusion
We have reviewed the cases of coexistent LA and FVIII inhibitor, a useful exercise that highlighted several diagnostic shortfalls on the accuracy of the measurements employed by the authors of the cases reviewed herein. Factor VIII and its inhibitor were always measured by coagulation assay that can be influenced by the presence of an LA: none of the studies made an effort of measuring FVIII by chromogenic or immune assays, in one instance, it had not been measured at all 18 and only 1 study measured a FVIII inhibitor by immune assay. 17 In cases where the coexistence of a FVIII inhibitor and LA is suspected, appropriate second-line diagnostic tests should be performed, such as the DRVVT with phospholipid neutralization, or the Textarin–Ecarin or Taipan–Ecarin ratios for LA, and a chromogenic assay plus antibody titration by a solid-phase immune assay for FVIII inhibiting antibodies.
When confronted with a prolonged aPTT, the laboratory has the duty to provide a diagnostic interpretation to the clinician: knowledge of the bleeding/thrombotic risk in the preoperative and predelivery period 40 will minimize the risk of hemorrhage and counsel on adequate thromboprophylaxis. This knowledge becomes of the utmost importance in situations such as acute trauma where guidance on life-saving antihemorrhagic measures will be urgently required. Due clinician diligence must be exercised in extracting all relevant pieces of personal and family history information from patients and relatives and in examining patients on signs or symptoms that may inform on a bleeding or thrombosis history. Due laboratory diligence also means that doctors and laboratory personnel dealing with hemostasis must remind themselves of the reciprocal influences that LA and anti-FVIII may have on each other. Due editorial diligence also means that reviewers of such articles should also be aware of the above-mentioned concepts in order to provide the right comments that will improve the quality of the cases to be published. The expertise of clinical and laboratory personnel represents the cornerstone of a successful thrombosis and hemostasis service. It is hoped that this review may provide some diagnostic guidance to those confronted with prolonged aPTT where there is a suspicion of coexistent LA and acquired FVIII inhibitor.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
