Abstract
We aimed to study the endothelial dysfunction among children and adolescents with transfusion-dependent β-thalassemia using von Willebrand factor antigen (VWF:Ag) and flow cytometric analysis of circulating CD144+ endothelial microparticles (EMPs) and endothelial progenitor cells (CD34+VEGFR2+) and assess their relation to iron overload, erythropoietin and chelation therapy as well as echocardiographic parameters and carotid intima–media thickness. The VWF:Ag, EMPs, and CD34+VEGFR2+ cells were significantly higher among patients with β-thalassemia than controls (P < .001). The type of chelation and patients’ compliance did not influence the results. No significant correlations were found between the studied vascular markers. Patients with evident heart disease had higher VWF: Ag, EMPs, and CD34+VEGFR2+ cells than those without. Carotid intima–media thickness was increased among patients but not correlated with vascular markers. We suggest that procoagulant EMPs and VWF: Ag are involved in cardiovascular complications in patients with young β-thalassemia. CD34+VEGFR2+ cells were further increased in response to tissue injury contributing to reendothelialization and neovascularization.
Keywords
Introduction
Transfusion-related iron overload in β-thalassemia (β-TM) has been associated with the onset of cardiovascular complications, including cardiac dysfunction and vascular anomalies. 1 Heart failure always represented and still remains the leading cause of mortality in β-TM, despite the therapeutic advances and the considerable amelioration of prognosis accomplished over the last decades. 2 The endothelium plays a key role in vascular homeostasis through the release of a variety of paracrine factors that interact with platelets, inflammatory cells, and the vessel wall. 3 Patients receiving regular blood transfusions have increased iron load which has an impact on the thrombotic response to arterial injury, vascular production of reactive oxygen species, and endothelium-dependent vasoreactivity. 4
The importance of arterial dysfunction in patients with β-TM major is increasingly recognized as evidenced by reduced brachial flow-mediated dilation and increased stiffness of the carotid artery. 5 Systemic arterial involvement in thalassemia major plays a role in the development of cardiac dysfunction by affecting heart afterload. Vascular involvement starts early in life and becomes obvious in the older patients. 6 –8 Also, carotid atherosclerosis is positively associated with serum ferritin independent of traditional cardiovascular risk factors. 9
The von Willebrand factor antigen (VWF:Ag) has been used as a marker of endothelial dysfunction in vascular diseases. 10 Because VWF is important in the aggregation of platelets and their adhesion to subendothelial cells, increased levels of circulating VWF per se may promote atherosclerosis and contribute to cardiovascular events. 11
Endothelial microparticles (EMPs) are small vesicles released from disturbed endothelial cells characterized by their expression of different cell surface antigens including CD31, CD34, CD54, CD62E, CD51, CD105, CD106, CD144, and CD146, 12 and have recently been reported as a marker of endothelial injury and systemic vascular remodeling. 13,14 Endothelial microparticles can promote a prothrombogenic and proinflammatory vicious circle leading to vascular dysfunction. 15
Repair of the denuded endothelial cells might be crucial for the restoration of endothelial function. 16 Endothelial progenitor cells (EPCs) are a heterogeneous group that exists in a variety of developmental stages. The immature EPCs can be characterized by the expression of CD133, CD34, and vascular endothelial growth factor receptor 2 (VEGFR2). 5,17,18 These EPCs play essential roles in maintenance of vascular integrity, angiogenesis, and wound repair. 19 Erythropoietin (EPO) stimulates proliferation and maturation of erythroid precursors and increases peripheral blood EPCs. 20
The aim of this work was to study the endothelial dysfunction among children and adolescents with transfusion-dependent β-TM using VWF:Ag and flow cytometric analysis of circulating EMPs and EPCs (CD34+VEGFR2+ cells) and assess their relation to the clinicopathological and radiological characteristics of patients including iron overload, markers of hemolysis, EPO levels, and the efficacy of chelation therapy as well as echocardiographic parameters and carotid intima–media thickness (CIMT).
Materials and Methods
This study included 40 patients with transfusion-dependent β-TM (19 males and 21 females with a male-to-female ratio of 1:1.1) who were randomly recruited from the regular attendance of the Pediatric Hematology Unit, Children’s Hospital, Ain Shams University. There were 33 patients with β-TM major and 7 with β-TM intermedia, and they were compared to 40 age- and sex-matched healthy individuals enrolled as controls. The age of the patients ranged from 11 to 20 years with a mean age of 15.4 ± 3.9 years, while the control group had a mean age of 14.7 ± 2.1 years. An informed consent was obtained from the guardian of each patient or control before participation. The procedures applied in this study were approved by the Ethical Committee of Human Experimentation of Ain Shams University and are in accordance with the Helsinki Declaration of 1975.
All included patients were subjected to detailed medical history and thorough clinical examination with special emphasis on disease duration, anthropometric measures, pubertal stage determined according to Tanner classification, 21 blood pressure, evidence of cardiac disease, history of splenectomy, viral hepatitis, transfusion history (frequency of transfusion with calculation of transfusion index), and chelation therapy. Patients with α-thalassemia, sickle β-TM, renal disease, diabetes mellitus, or hypothyroidism were excluded.
Patients with β-TM received either mono or combined chelation therapy. Monochelation was in the form of subcutaneous deferoxamine in a dose that ranged from 30 to 45 mg/kg/d given 5 days/week, oral deferiprone in a daily dose that ranged from 50 to 100 mg/kg/d, or oral deferasirox (dose, 40 mg/kg/d once daily). For combined chelation therapy, deferoxamine was given for 5 days/week with either daily deferiprone or deferasirox.
Sample Collection
Peripheral blood samples were collected on EDTA (1.2 mg/mL) for complete blood count (CBC). Two blood samples were collected on 0.2 mL 3.8% trisodium citrate in a ratio of 9 volumes of blood to 1 volume of citrate for flow cytometry and VWF:Ag assay. For chemical analysis, clotted samples were obtained and serum was separated by centrifugation for 15 minutes at 1000g.
Diagnostic Testing
Laboratory investigations included CBC using Sysmex XT-1800i (Sysmex, Japan), examination of Leishman-stained smears for differential white blood cell (WBC) count, kidney function tests, total and indirect bilirubin as well as serum ferritin on Cobas Integra 800 (Roche Diagnostics, Mannheim, Germany). The mean value of serum ferritin was calculated in the last 2 years prior to the study.
Quantitative VWF:Ag assay (Siemens Healthcare Diagnostics, Marburg, Germany) was performed on a Sysmex CA-1500 coagulation analyzer (Siemens Healthcare Diagnostics, Marburg, Germany) with a normal range between 50% and 150%. 22 Serum EPO concentrations were determined by the DRG EPO (DRG International Inc, Springfield, New Jersey, USA) which is a 2-site enzyme-linked immunosorbent assay for the measurement of the biologically active 165 amino acid chain of EPO.
Flow Cytometric Analysis of EMPs and Circulating CD34+VEGFR2+ Cells
Microparticles (MPs) were isolated as previously described. 23 Citrated blood samples were centrifuged for 10 minutes at 160g to prepare platelet-rich plasma then centrifuged for 10 minutes at 1000g to prepare platelet-poor plasma. A volume of 50 μL of platelet-poor plasma was added to 5 μL of phycoerythrin (PE)-labeled anti-CD144 (anti-human VE-Cadherin; R&D systems; Minneapolis, Minnesota). Isotype-matched control antibodies were obtained from Beckman Coulter, California. Samples were protected from light and incubated at room temperature for 20 minutes until data acquisition.
The circulating EPCs were quantified by flow cytometry as previously reported. 5,24 A volume of 50 μL of whole blood was added to 5 μL of fluorescein isothiocyanate-labeled anti-CD34 (Miltenyi Biotec Inc, Bergisch Gladbach, Germany) and PE-labeled anti-VEGFR2 (R&D systems; Minneapolis, Minnesota). Samples were protected from light and incubated at room temperature for 15 minutes then 1.5 mL lyse solution (Beckman Coulter) was added followed by 2 minutes of incubation for red blood cell lysis.
Data were analyzed using EPICS-XL PROFILE II Coulter flow cytometer (Beckman Coulter, Inc., Fullerton, California). For quantification of CD34+VEGFR2+ cells, each analysis included all the events in the lymphocyte region. The percentage of positive cells for the aforementioned immunophenotypes was determined by a 2-dimensional side scatter fluorescence dot-plot analysis. For evaluation of MPs, EMPs were identified by their characteristic forward scatter and their ability to bind the cell-specific monoclonal antibody CD144. The forward scatter cutoff was set using 1-μm standard beads (Sigma, St. Louis, Missouri) to define the upper limit of the MPs population. 25 Nonspecific binding of the control mouse monoclonal antibodies set the lower fluorescence margin. 26 Data were expressed as the percentage of total MPs. 27 Flow cytometric analysis of circulating EMPs and EPCs is presented in Figure 1.
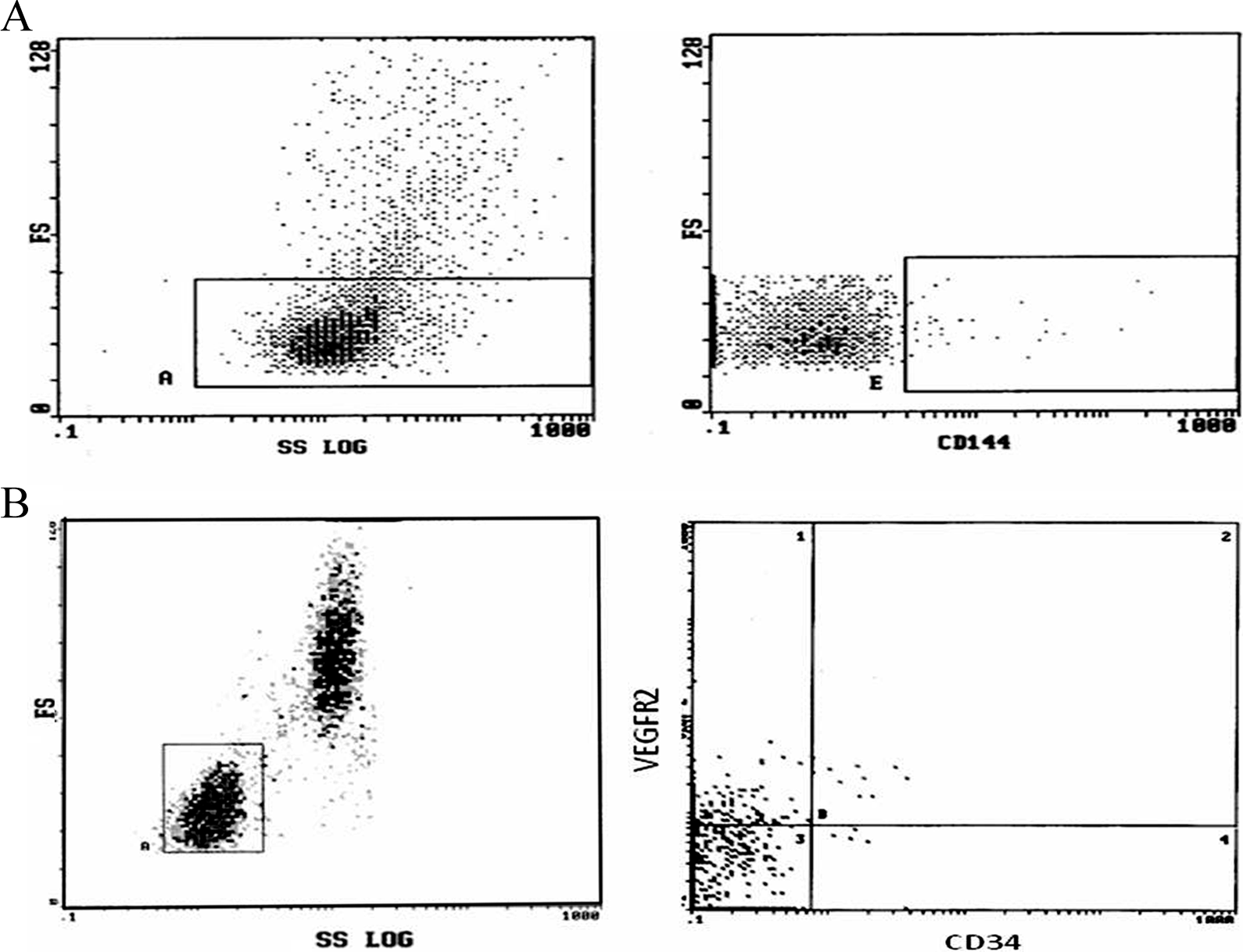
Flow cytometric analysis of circulating CD144+ endothelial microparticles (A) and CD34+VEGFR2+ cells (B) among patients with transfusion-dependent β-thalassemia.
Radiological Examination
All studied patients were clinically asymptomatic for pulmonary hypertension and cardiovascular abnormalities. Screening for pulmonary hypertension was performed by the noninvasive Doppler echocardiography with different modalities using Vivid E9 (GE Healthcare, Norway) to evaluate dimensions of cardiac chambers, pulmonary artery pressure, and tricuspid regurgitant jet velocity (TRV). 28
All of the carotid scans were done using carotid Doppler ultrasound scanner (Toshiba Ultrasonography machine [Xario], Japan) with a 10.0-MHz linear array transducer following a predetermined standardized scanning protocol. 29 All ultrasound scans were performed by an experienced vascular operator who was unaware of children’s clinical details. Patients were placed in supine position having the neck in hyperextension. The location of measurement was standardized in every study at the common carotid artery at 1 cm distance from the carotid bulb. The mean value of CIMT for the right and left carotid arteries was calculated.
Diagnostic Criteria
The definition of β-TM was based on complete blood picture, reticulocyte count, and markers of hemolysis as well as hemoglobin (Hb) analysis using high-performance liquid chromatography. 30 Evident heart disease was defined by at least one of the following: congestive heart failure, systolic left ventricle (LV) dysfunction (LV shortening fraction <30% or LV ejection fraction <55%), and considerable pulmonary hypertension (peak systolic tricuspid pressure gradient of at least 30 mm Hg corresponding to a peak TRV of ≥2.5 m/s). 28,31,32 Compliance to chelation therapy was assessed by reviewing patient self-report of dose-taking, and the appropriate number of doses taken during each day was checked by prescription refills and pill count; a cutoff point below 70% was considered as poor compliance to the regimen. 33
Statistical Analysis
Analysis of data was done using SPSS version 15 (SPSS Inc, Chicago, Illinois). Quantitative variables were described in the form of range, mean and standard deviation, or median and interquartile range. Qualitative variables were described as number and percentage. In order to compare quantitative parametric variables between the 2 groups, Student t test was applied, while Mann-Whitney was used for nonparametric variables. Comparison between categorical variables was performed using chi-square (χ2) test or Fischer exact test when frequencies were below 5. Pearson correlation coefficient and multiple regression analysis were employed to determine the relation between VWF:Ag, circulating CD144+ EMPs, and CD34+VEGFR2+ cells and other clinical, laboratory, and radiological variables. Receiver–operating characteristic (ROC) curve was used to determine the best cutoff value of the studied vascular markers to detect cardiovascular complications that best combined sensitivity and specificity. The area under the curve (AUC) and 95% confidence interval (CI) were calculated for each plot. A P value <.05 was considered significant in all analyses.
Results
The clinical and laboratory and radiological data of all patients and controls are illustrated in Table 1.
Clinical, Laboratory, and Radiological Data of Patients With Transfusion-Dependent β-Thalassemia and Healthy Controls.a
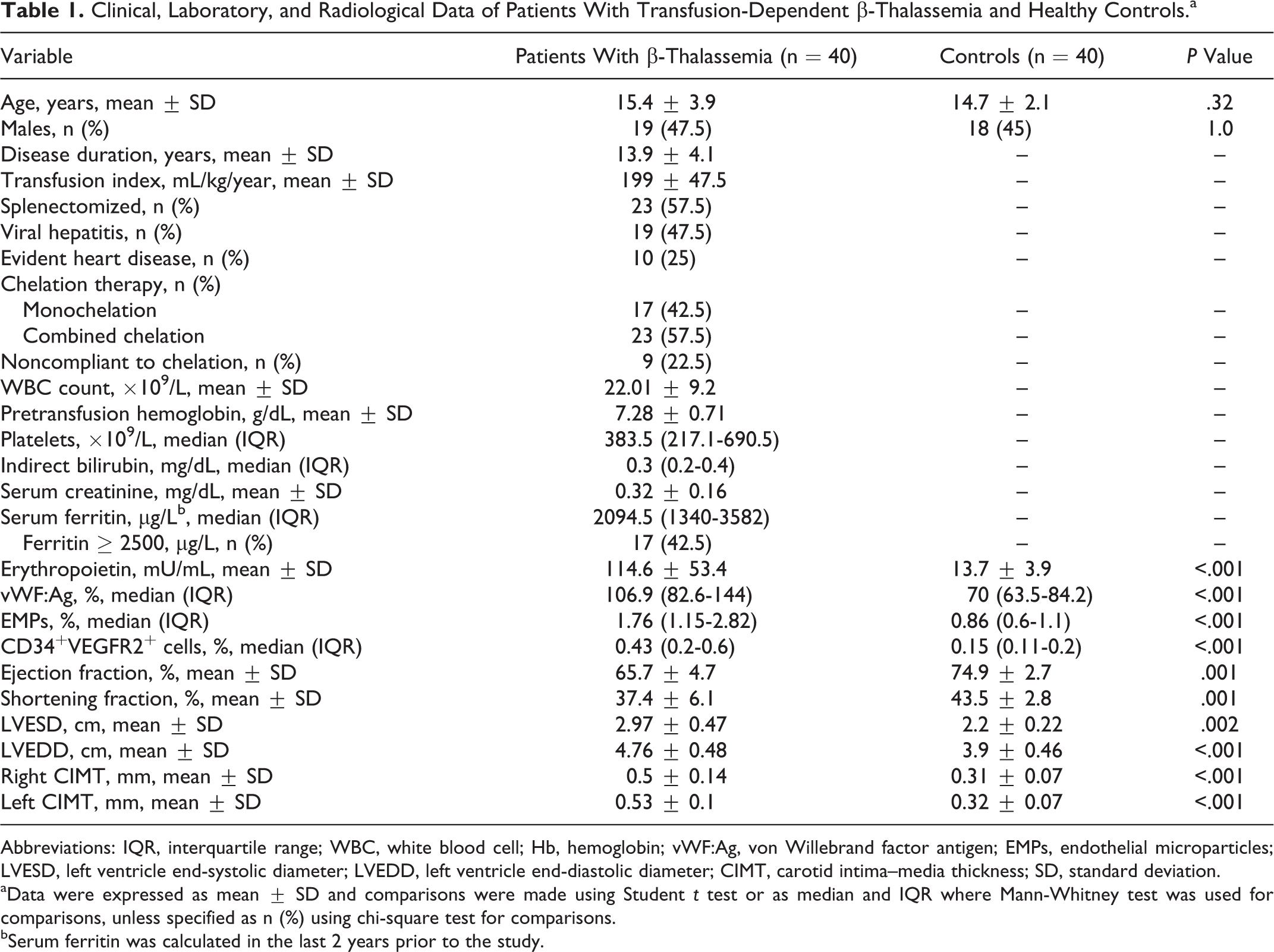
Abbreviations: IQR, interquartile range; WBC, white blood cell; Hb, hemoglobin; vWF:Ag, von Willebrand factor antigen; EMPs, endothelial microparticles; LVESD, left ventricle end-systolic diameter; LVEDD, left ventricle end-diastolic diameter; CIMT, carotid intima–media thickness; SD, standard deviation.
aData were expressed as mean ± SD and comparisons were made using Student t test or as median and IQR where Mann-Whitney test was used for comparisons, unless specified as n (%) using chi-square test for comparisons.
bSerum ferritin was calculated in the last 2 years prior to the study.
Plasma VWF:Ag, EMPs, CD34+VEGFR2+ Cells, and EPO Among Patients With β-TM and Healthy Controls
Patients with β-TM had significantly higher VWF:Ag levels, EMPs, and circulating CD34+VEGFR2+ cells than those that of controls (P < .001; Table 1 and Figure 2). The levels of EPO were significantly increased among patients compared to controls (Table 1).
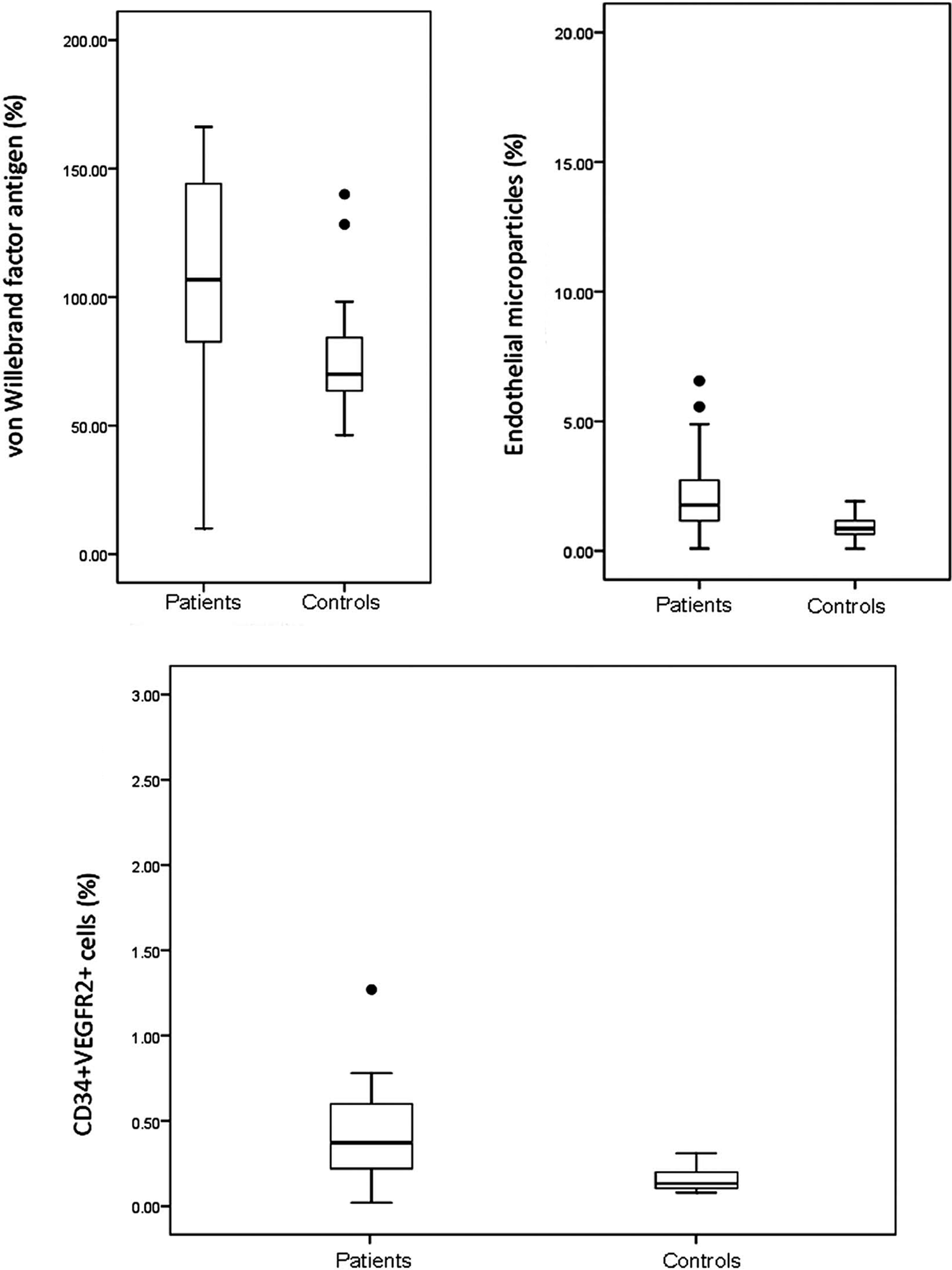
Serum von Willebrand factor antigen, endothelial microparticles, and CD34+VEGFR2+ cells among patients with β-thalassemia and healthy controls.
Relation Between Markers of Vascular Dysfunction and Different Clinicopathological Characteristics of Patients With Thalassemia
No significant difference was found between patients with β-TM major and β-TM intermedia as regards the levels of VWF:Ag, EMPs, and CD34+VEGFR2+ cells (P > .05). Both patients with splenectomized and nonsplenectomized β-TM had comparable levels of VWF:Ag, EMPs, and CD34+VEGFR2+ cells (P > .05). The levels of the studied markers were also consistent among patients with and without viral hepatitis (P > .05; Table 2).
Relation Between Markers of Vascular Dysfunction and Clinicopathological Characteristics of Patients With Transfusion-Dependent β-Thalassemia.
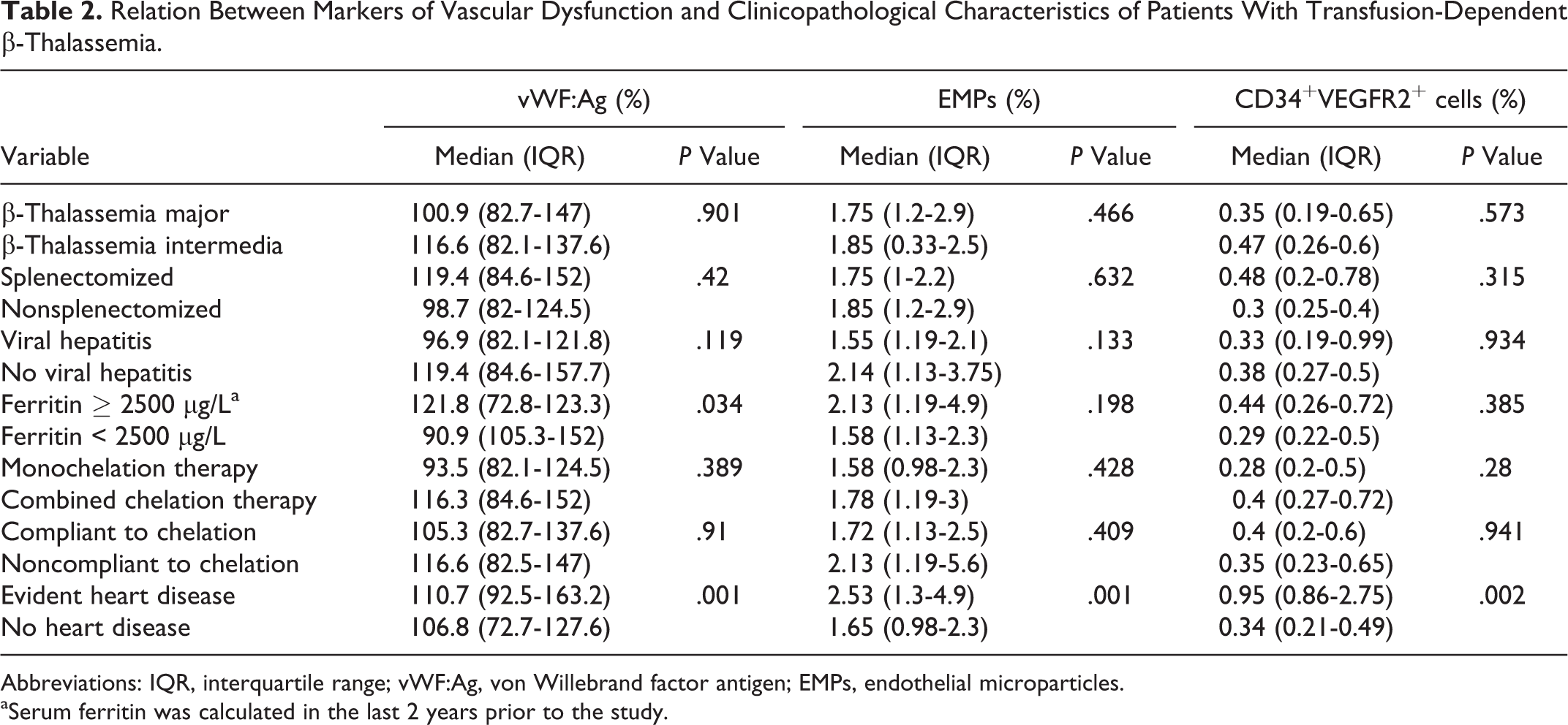
Abbreviations: IQR, interquartile range; vWF:Ag, von Willebrand factor antigen; EMPs, endothelial microparticles.
aSerum ferritin was calculated in the last 2 years prior to the study.
Patients with serum ferritin ≥2500 µg/L in last 2 years prior to the study had higher levels of VWF:Ag, EMPs, and CD34+VEGFR2+ cells than those with lower serum ferritin; however, the difference only reached significant levels with respect to VWF: Ag (P = .034; Table 2).
Notably, the type of chelation and patients’ compliance did not influence the results where patients with thalassemia on monochelation therapy and combined therapy did not show differences as regards any of the 3 studied markers (P > .05; Table 2).
No significant correlations were found between VWF:Ag, EMPs, and CD34+VEGFR2+ cells. However, a significant positive correlation was observed between CD34+VEGFR2+ cells and indirect bilirubin (r = .383, P = .03). Other variables as disease duration, transfusion index, mean pretransfusion Hb level, WBC count, platelets, serum ferritin in last 2 years prior to the study, and EPO were not correlated with the studied vascular markers (P > .05).
Relation Between Vascular Markers and Echocardiographic Features and CIMT in Patients With Thalassemia
Patients with thalassemia had significantly lower ejection fraction and shortening fraction, while LV diameters were increased compared with controls (Table 1). Evident heart disease was found in 25% of patients with β-TM. Patients with cardiac disease had higher VWF:Ag, EMPs, and CD34+VEGFR2+ cells than those without (P = .001, .001, and .002, respectively; Table 2). Moreover, significant positive correlations were observed between CD34+VEGFR2+ cells and LV end-systolic diameter (r = .39, P = .043) and LV end-diastolic diameter (r = .59, P < .001; Figure 3).
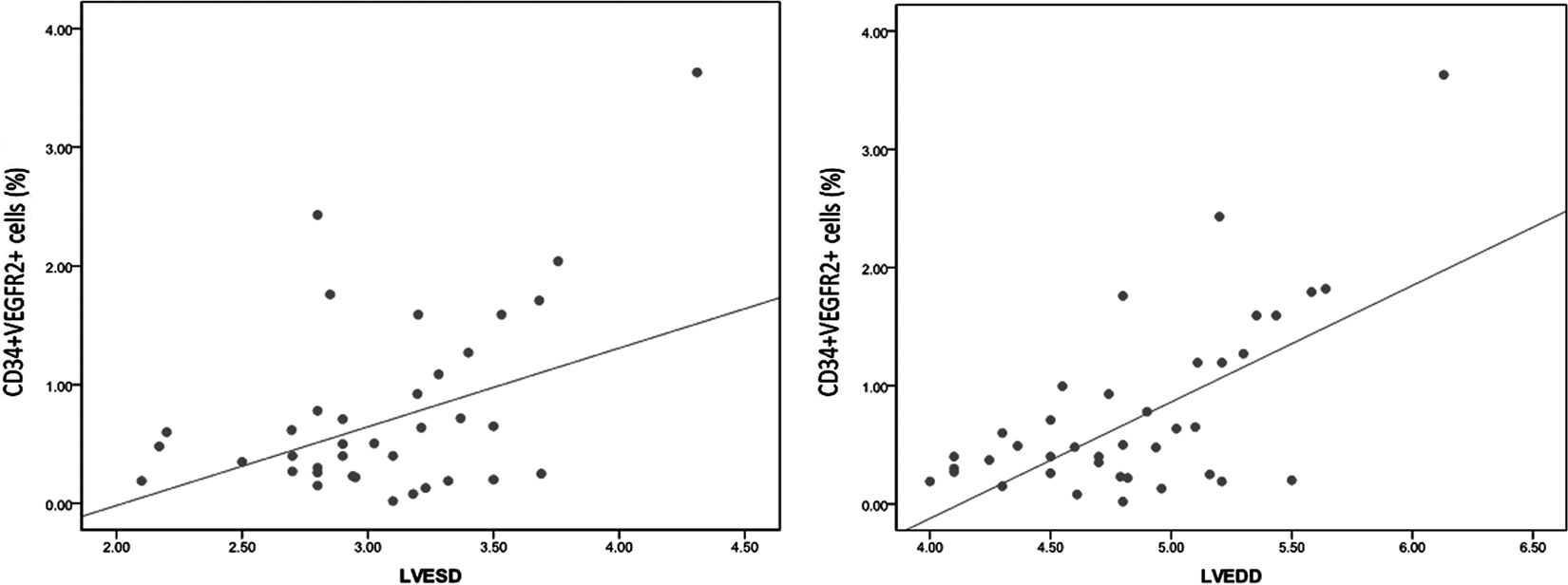
Correlation between circulating CD34+VEGFR2+ cells and echocardiographic parameters. LVESD indicates left ventricle end-systolic diameter; LVEDD, left ventricle end-diastolic diameter.
The ROC curve analysis revealed that VWF:Ag cutoff value of 120% was able to differentiate patients having β-TM with and without evident heart disease with a sensitivity of 100% and specificity of 67% (AUC, 0.857; CI, 0.76-1.0; P < .001). Additionally, EMPs cutoff value of 2.13% could detect patients having β-TM with evident heart disease with a sensitivity of 100% and specificity of 70% (AUC, 0.85; CI, 0.75-0.95; P < .001), while the best cutoff value of CD34+VEGFR2+ cells to detect cardiovascular complications was 0.71% with a sensitivity of 100% and specificity of 87% (AUC, 0.917; CI, 0.86-1.52; P < .001).
Both right and left CIMT were significantly increased in patients with thalassemia compared to controls (0.5 ± 0.14 vs 0.31 ± 0.07 mm and 0.53 ± 0.1 vs 0.32 ± 0.07 mm, respectively; P < .001); however, no correlations were found between CIMT and any of VWF:Ag, EMPs, or CD34+VEGFR2+ cells (data not shown).
Discussion
The study of vascular biomarkers has greatly enhanced our understanding of the underlying pathophysiology of atherosclerosis. This includes soluble biomarkers and physiological parameters such as measures of arterial stiffness and endothelial function. 34 To our knowledge, this study was the first to assess VWF:Ag, EMPs, and EPCs in children and adolescents with β-TM as surrogate vascular biomarkers and a noninvasive way for assessing disease progression. We showed the presence of endothelial damage and subclinical atherosclerosis in those patients before symptomatic vascular impairment as characterized by high levels of vascular markers, together with an evidence of cardiovascular abnormalities by echocardiography and increased CIMT.
Patients with β-TM major demonstrate an increased incidence of vascular complications as a result of a procoagulant/proinflammatory environment. 35 Our findings of endothelial dysfunction and carotid arterial stiffening in patients with iron overloaded transfusion-dependent β-TM concur with other reports in adults 5,36 –38 and support the few studies that investigated subclinical atherosclerosis in children and adolescents. 39,40
Plasma levels of VWF have been considered as a marker of endothelial dysfunction and damage, 41 although this is controversial 42 and results from studies that have correlated plasma levels of VWF, severity of inflammation, and patient outcome are inconsistent. 42 In our study, we found higher plasma VWF: Ag levels in patients with β-thalassaemia, particularly those with evident heart disease. This was in line with other reports. 41,43
We also found elevated EMPs in young patients with β-thalassaemia, suggesting the presence of endothelium activation or injury. Likewise, Habib et al 44 showed that the levels of procoagulant MPs of RBC, leukocytic, and endothelial origins are high in patients with β-TM intermedia, but their possible deleterious role in endothelial dysfunction remains to be further investigated. Recently, increased levels of platelets and erythrocytes-derived MPs have been reported in thalassemia and may be implicated in vascular dysfunction, pulmonary hypertension risk, and aortic wall stiffness observed in those patients. Their quantification could provide utility for early detection of cardiovascular abnormalities and monitoring the biological efficacy of chelation therapy. 40
On the other hand, Pattanapanyasat et al 45 described an increase in MPs of platelet but not endothelial origin in patients with β-thalassaemia/HbE. In another hemolytic condition, van Beers et al 46 found that the majority of MPs in sickle cell disease (SCD) originated from platelets and erythrocytes while neither MPs originating from monocytes nor endothelial cells were detectable. Their data contrast the previous findings of Shet et al 47 who found endothelial- and monocyte-derived MPs exposing tissue factor to be responsible for coagulation activation in SCD. van Beers et al 46 attributed the lack of detection of this small subset of MPs to differences in the centrifugation forces used in isolation. Therefore, further characterization of MPs is needed for more accurate assessment of the endothelial status. 44
Endothelial progenitor cells move toward injured endothelium or inflamed tissues and incorporate into foci of neovascularization, thereby improving blood flow and tissue repair. 48 This may explain elevated circulating CD34+VEGFR2+ cells in our study. In agreement, Cheung et al 5 demonstrated significant vascular dysfunction in adult patients with transfusion-dependent β-TM major with impaired proliferation, despite an increase in the number of circulating CD34+VEGFR2+ cells. Their data suggested a dose-dependent effect of iron load on the proliferation of these cells. Similarly, despite Ofori-Acquah et al 49 found higher CD34+VEGFR2+cells in the Hb SS group than in controls, an abnormal reparative function has been suggested. It is worth to note that although the number of these circulating cells has been once regarded as a surrogate of function, discordance between their quantity and function is increasingly recognized. 50,51 This discrepancy has also been shown in patients with β-TM 5 probably due to increased oxidative stress secondary to iron loading, 52 although this speculation remains to be proven. 5
Indeed, endothelial integrity is a fine balance between endothelial damage and repair. 53 Experimental and clinical studies have shown that atherosclerosis is associated with reduced numbers and dysfunction of EPCs 54 and inverse associations between the quantity of EPCs and the occurrence of cardiovascular events, 55 and risk of developing subclinical atherosclerosis 24 and vasculopathy in SCD 56 has been widely reported. However, the presence of high EPCs in transfusion-dependent patients with significant vascular dysfunction was explained by Cheung et al 5 by increased serum EPO in patients with thalassaemia, despite repeated blood transfusion, 57 which has been shown to increase the mobilization of circulating EPCs in both experimental models 58 and humans. 59 Erythropoietin stimulates postnatal neovascularization at least in part by enhancing EPCs mobilization from the bone marrow. 58 This might be considered an intrinsic homeostatic mechanism to restore endothelial function. 5 Additionally, induction of heme oxygenase 1 secondary to increased oxidative stress and iron overload in thalassaemia may also play a role. 5 These data were further supported by the presence of high EPO levels among our patients with thalassemia and increased numbers of CD34+VEGFR2+ cells among patients with serum ferritin ≥2500 µg/L in last 2 years prior to the study.
Although high serum EPO was found in our patients with thalassemia, possibly accounting for high EPCs in those patients, we could not find a significant correlation between CD34+VEGFR2+ cells and EPO levels. This implies that the elevation in CD34+VEGFR2+ cells may be due to their mobilization by EPO, but the number of these cells was independent of elevated EPO levels. This was in agreement with Ofori-Acquah et al 49 who found that EPO was associated with increase in number of CD34 and CD34/CXCR4 cells but not all types of circulating progenitor cells (CPCs) in children with sickle cell anemia. Meanwhile, stromal-derived growth factor was consistently associated with the number of all CPC types. In contrast, serum levels of EPO were significantly associated with the number of stem and progenitor cells in the bone marrow as well as with the number and function of circulating EPCs in patients with ischemic heart disease. 58 The relation between EPO and number of EPCs seems different in patients with thalassemia or other hemolytic conditions than patients with ischemic heart disease, probably due to the observed discordance between the quantity and function of these cells.
In our study, evident heart disease was found in 25% of young patients with β-TM probably due to regular transfusion and even in patients with thalassemia intermedia. This was in line with other reports. 60,61 Cardiac structure and function in thalassemia major are mainly affected by 2 competing factors: iron load and increased cardiac output. The cardiac iron deposition results in a decrease in LV function. The anemia together with marrow expansion leads to volume overload and increased cardiac output that then demands increased contractility adding additional stress to the heart. 2,7
Importantly, we found higher levels of VWF:Ag, EMPs, and CD34+VEGFR2+ cells in patients having thalassemia with evidence of cardiac disease, and the best cutoff value for detection of cardiovascular complications was determined. Moreover, significant positive correlations were observed between CD34+VEGFR2+ cells and LV diameters. Plasma VWF levels have been proposed as a risk factor for cardiovascular events. 62 In agreement with our results, Lip et al 63 found that mean plasma VWF was higher among patients with atrial fibrillation and congestive heart failure. Patients with severe LV dysfunction and low fractional shortening also had significantly higher VWF concentrations than those with no dysfunction. Although the cutoff value of VWF:Ag 120%, differentiating patients with and without heart disease, lies within normal range, but this may be because the studied children and adolescents with thalassemia were clinically asymptomatic for pulmonary hypertension and cardiovascular abnormalities, and the definition for heart disease in our study involved a high cutoff for LV ejection fraction <55%. Given the high sensitivity of vWF:Ag (100%) and taken into consideration the aforementioned observations, it could be considered as a noninvasive early indicator in young patients with thalassemia. Since it has only 67% specificity, we suggest performing echocardiographic analysis for patients above this cutoff to exclude the presence of heart disease.
In addition, EMPs are known to be elevated in acute coronary syndromes, severe hypertension with end-organ damage, thrombotic thrombocytopenic purpura, and all conditions associated with endothelial injury and prothrombotic state. 64 Moreover, circulating concentrations of EPCs have been correlated with the occurrence of cardiovascular disease and events. 55 So far, EPCs have been implicated in the whole cardiovascular disease process, and many conventional therapies have been shown to alter EPCs number and function. 65 With the aforementioned observations taken into account, our findings support the biological role of these vascular markers in inflammation, vascular injury, angiogenesis, and thrombosis. Their role in defining cardiovascular risk may have important therapeutic implications.
Evidence in thalassemia supports the presence of a hypercoagulable state greatly exacerbated by splenectomy, which is the result of platelet activation, enhanced red blood cell adherence to the endothelium, reduced levels of the natural anticoagulants protein C and protein S, and increased thrombin generation. 66 In the current study, the lack of significant difference between patients with splenectomized and nonsplenectomized β-TM as regards the EMPs implies that the role of splenectomy in the pathophysiology of vascular disorder may be related to total MPs, particularly those originated from platelet and RBCs. In agreement, Habib et al 44 only reported higher platelet MPs in patients with splenectomized β-TM. Furthermore, Tantawy et al 40 found a significant increase in both platelet- and erythrocyte-derived MPs in patients with splenectomized thalassemia compared to patients with nonsplenectomized thalassemia which may be due to the hypercoagulability and postsplenectomy vascular complications including pulmonary hypertension.
Patients with chronic hepatitis C present a proangiogenic profile of angiogenic markers 67 ; however, we could not find any association between vascular markers and viral hepatitis. It has been reported that in the patients with chronic hepatitis C, the found elevated blood sVCAM-1 and VWF:Ag levels related to the biochemical activity of hepatitis and, to a greater extent, with the degree of hepatic fibrosis can judge the implication of endothelial dysfunction in the pathogenesis of chronic HCV infection. 68 In a study by Roed et al, 69 although patients with chronic hepatitis C had increased CIMT and elevation in several circulating endothelial biomarkers, no correlation was found. Thus, it seems that the mere presence of viral infection may not be necessarily associated with elevated vascular markers, but it depends on the severity of the disease and extent of fibrosis. High levels of vascular markers in our young patients with β-TM may reflect an early state of endothelial dysfunction, and the relation between these markers and viral hepatitis remains to be elucidated later in adult life.
Furthermore, patients with serum ferritin ≥2500 µg/L in last 2 years prior to the study had higher plasma VWF:Ag, circulating EMPs, and CD34+VEGFR2
In agreement with our results, Cheung et al 5 reported a lack of association between ferritin and the proliferative capacity of EPCs and suggested that nontransferrin bound iron may be a better reflection of the free iron potentially acting on these cells rather than serum ferritin. Although serum ferritin is a convenient way to monitor iron overload and a significant correlation between serum ferritin and liver iron concentration has been established in regularly transfused patients with thalassemia major, there is tremendous scatter in the relation. These observations have important implications for patient management, as assessment of serum ferritin alone may result in a delay in initiating chelation therapy and may, therefore, prolong patient exposure to high iron levels and the associated morbidity and mortality risks. 73 On the other hand, Tantawy et al 40 found positive correlations between MPs of platelet and erythrocyte origin and serum ferritin, while MPs were decreased among compliant patients with adequate chelation. Elsayh et al 74 found that the percentage of total MPs correlated positively with serum ferritin, suggesting that both hypoxia and oxidative stress are responsible for increasing MPs in thalassemia.
In this study, no significant correlations were found between VWF:Ag, EMPs, and CD34+VEGFR2+ cells or between any of these vascular markers and other variables, yet a significant positive correlation was found between CD34+VEGFR2+ cells and indirect bilirubin supporting the role of chronic hemolysis in endothelial dysfunction. Tantawy et al 40 found that both platelet and erythrocyte MPs positively correlated with transfusion index, lactate dehydrogenase, and indirect bilirubin levels. Clinical studies also showed the existence of a human syndrome of hemolysis-associated nitric oxide dysregulation, which may contribute to the vasculopathy of hereditary, acquired, and iatrogenic hemolytic states. 75,76
The relation between vascular markers and WBC counts and platelets was of importance because it is well known that all the peripheral blood cells, including WBCs and platelets, are derived from bone marrow and mobilized into the circulation to play their individual roles. Circulating EPCs, a subset of the hematopoietic stem cell population, have been shown to be derived from bone marrow and released into the circulation for the maintenance of endothelial function and vascular integrity. 77 In healthy Taiwanese population, circulating level of EPCs was significantly associated with an increase in platelet count (an index of hematopoietic activity in bone marrow) and inversely associated with WBC count (an indicator of inflammatory activity). This suggests that the circulating number of EPCs is not only related to coronary artery disease risk factors, but it may also be related to the hematopoietic activity and functional reserve of bone marrow. 78
Moreover, total WBC counts play an important role in inflammation and are associated with the presence, severity, and extent of coronary atherosclerosis. 79 Leukocytosis has also been associated with reduced epicardial blood flow, thromboresistance, and an adverse outcome in patients treated with thrombolytic therapy for acute myocardial infarction. 80 In particular, peripheral monocytosis is related to LV dysfunction. 81 In our study, there was no correlation between vascular markers and WBC counts. In agreement, the number of CD34+VEGFR2+ cells was higher in children with sickle cell anemia (SCA) after correction for WBC count, while there were no differences in the other CPC populations. This implies that the elevation in most of the CPC populations in SCA was a secondary effect of WBC mobilization but that the number of circulating CD34+VEGFR2+cells was independent of elevated WBC. 49
Noninvasive assessment of vascular dysfunction in the pediatric population has taken advantage of the development of high-resolution ultrasound techniques. The most frequently used methods are the quantification of flow-mediated, endothelium-dependent dilation of the brachial artery and measurement of the intima–media thickening of the carotid artery. Both reduced flow-mediated dilation and increased intima–media thickness have been proven to correlate with late cardiovascular events and/or mortality in adults. 82 In line with other reports, 38,39 CIMT was significantly increased in the studied patients with thalassemia compared with controls suggesting the occurrence of premature atherosclerosis among those patients. However, no correlations were found between CIMT and vascular markers. These results may not support their involvement in subclinical atherosclerosis in thalassemia. However, the relation between vascular markers and CIMT may be evident later in adult life so larger prospective studies are needed. Papasavvas et al 83 reported no association between EPCs and CIMT in chronic HIV-positive patients. Although Dursun et al 84 found significant positive correlations between EMPs and each of CIMT and pulse wave velocity in children with chronic kidney disease, multiple regression analysis showed that EMPs was independent predictor of pulse wave velocity only.
Finally, elevated levels of vascular markers in children and adolescents with β-TM may have clinical implications on treatment besides defining patients at risk of cardiac complications. Improved knowledge of EMPs composition, their biological effects, and the mechanisms leading to their clearance will probably open new therapeutic approaches in the treatment of atherothrombosis. 64 Moreover, novel EPC-based therapies may provide enhancement in restoring EPCs’ population and improvement of vascular function. 54
Study Limitations
One limitation of this study includes the presence of a relatively small number of patients which may have implications upon the significance of some data. It is also worth to mention that we only investigated VWF:Ag and the quantitative value of circulating CD34+VEGFR2+ cells to correlate the levels of vascular markers to each other and to clinical, hematological, and radiological variables. Therefore, further larger longitudinal studies are warranted to assess VWF activity and determine the qualitative function of EPCs and their pathophysiological role in patients with β-TM.
In conclusion, procoagulant EMPs and plasma VWF:Ag are involved in cardiovascular complications in young patients with β-TM. The quantity of CD34+VEGFR2+ cells was further increased in response to tissue ischemia or injury contributing to reendothelialization and neovascularization. Quantification of VWF:Ag, EMPs, and EPCs in those patients could provide important research tools to further our understanding of vascular biology and a noninvasive way for early detection of patients at risk of cardiovascular abnormalities. Larger longitudinal studies are further recommended to verify these results and the cutoff of these vascular markers as predictors of cardiovascular complications in young patients with thalassemia; thereafter, these vascular markers can be used in risk stratification to guide future therapies.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
