Abstract
Background:
The relationships between the endothelial progenitor cells (EPCs)-CD34+ and CD14+ and coronary artery disease (CAD) were reported and the association of CD34+ cells with renal function was studied previously. Another kind EPC-CD14+ cell and its association with renal function in patients with CAD have not been reported yet. Our aim was to assess CD14+ cell counts versus renal function in CAD.
Methods and Results:
We studied 242 patients with severe angiographic CAD and 30 healthy control participants. The CD14+ cells were enumerated by flow cytometry. With lowering glomerular filtration rate (GFR), CD14+ cell numbers (percentage of lymphocytes, median and interquartile range) decreased: 0.04 (0.03-0.06), 0.03 (0.02-0.05), 0.02 (0.01-0.03) for estimated glomerular filtration rate (eGFR) ≥90, 60 to 89, and 30 to 89 mL/min per 1.73 m2, respectively (P < .001 for trend
Conclusions:
The CD14+ cell depletion is associated with renal dysfunction in CAD.
Introduction
Atherosclerotic heart disease remains one of the major causes of morbidity and mortality worldwide. Chronic kidney disease (CKD) is associated with excessive cardiovascular morbidity 1 and increases the risk of cardiovascular event independently and additively to preexistent cardiovascular disease. 2 It has been noted that a high prevalence of CAD in patients with CKD and this prevalence increases monotonically with a decrease in the estimated glomerular filtration rate (eGFR). 3,4 The prognostic relevance of mild renal derangement in stable CAD has recently been confirmed in 7311 patients with stable angina as serum creatinine contributed to a combined risk score beginning from a cutoff value of 1.15 mg/dL, 5 which is equivalent to a respective GFR cutoff value higher than 60 mL/min per 1.73 m.2, 6
Indeed, growing experimental and clinical evidence underscores the critical role that circulating cells play a role in healing the endothelium when the intrinsic system is unable to adequately support tissue repair. Targeted delivery of endothelial progenitor cells (EPCs) has been shown to improve the function of the infarcted myocardium, decrease hindlimb ischemia, rescue the kidney from acute ischemia injury, and participate in glomerular endothelial repair in glomerulonephritis. 7 Additionally, in advanced CKD EPC numbers were depressed in nondialyzed participants, 8 whereas either decreased, 9,10 increased, or unchanged in patients who undergo hemodialysis. Previous studies indicated that the functional activity of EPCs is impaired in patients with CAD. 11 In vivo data revealed that the number of circulating progenitor cells inversely correlates with risk factors for CAD, such as diabetes, hypertension, or smoking. 11 Although the decreased eGFR was associated with the prevalence of CAD, the factor, as well as the traditional Framingham risk factors such as hypertension, diabetes, and obesity, can only partly explain the increased vascular disease burden of patients with CKD. The EPCs have been proposed to display vasculoprotective properties, while their depletion may predict CAD risk in patients with normal renal function. 12 –14 Nonetheless, the exact role and regulation of EPCs with CAD in patients with CKD remains unclear. The relationship between EPC-CD34+ and CAD was reported previously, 11 and the association of CD34+ with renal function was also studied in this study. 11 Therefore, we raised the hypothesis that EPC-CD14+ (another kind of EPCs) maybe associated with the renal function in patients with CAD. In the present study, we aimed to assess CD14+ cell counts versus renal function in patients with CAD.
Participants and Methods
Patients
The protocol was approved by the Ethics Committee of the 2nd Affiliated Hospital of Chongqing Medical University. We studied 242 patients who underwent angiography between May 2009 and March 2010. Inclusion criteria were stable angina (angina class II-III according to the Canadian Cardiovascular Society), a standard therapy (low-dose aspirin, angiotensin-converting enzyme inhibitors [ACEI] and statins) for ≥3 preceding months, and angiographically significant CAD defined as the presence of a diameter stenosis of ≥70% of at least one major epicardial artery segment according to the American College of Cardiology/American Heart Association guidelines on stable angina. 15 A wide set of exclusion criteria and requirement concerning therapy was adopted in order to limit the previously reported effects of statins, 16 ACEI, 17 acute coronary syndrome (ACS), 12 heart failure, 13 and depressed left-ventricular ejection fraction (EF) on EPC.
Of the 322 eligible patients, 80 had been eliminated by the exclusion criteria that included the following: age >75 years, ACS within past 3 months, percutaneous revascularization in past 1 month, any surgery within past 6 months, GFR <30 mL/min per 1.73 m2 (calculated according to the simplified Modification of Diet in Renal Disease study equation), significant valvular heart disease, congenital heart disease, EF <50%, heart failure, arterial hypertension uncontrolled adequately by drugs, significant extracoronary atherosclerosis, hemoglobin <11 g/dL, thrombocytopenia (<105/μL), thyroid or liver function abnormalities, malignant or inflammatory diseases, infections within past 2 months, and any chronic noncardiovascular medication.
A healthy control group was defined as patients with the same age but normal renal function and where CAD had been excluded by angiography. Normal renal function was defined as having a normal serum creatinine and an eGFR >90 mL/min.
Blood samples for fluorescence-activated cell sorter analysis and extended biochemical assays were taken after an overnight fast from an antecubital vein on the occasion of routine blood sampling 0 to 2 days prior to the planned angiography. The Bioethical Committee of our university approved the protocol and the patients gave informed consent.
Quantification of CD14+ cells in blood
Blood collection and cell isolation
Late and early EPCs were obtained as described previously. 14 Late EPCs were cultured from peripheral mononuclear cells collected 21 days before administration, and early EPCs were obtained from cells collected and cultured 7 days before in vivo computed tomography studies. All cells were cultured in endothelial growth medium.
Colony-forming units
Colony-forming units (CFUs) were counted to assess the availability of circulating EPCs. The EPC colonies consisting of multiple thin, flat cells emanating from a central cluster of rounded cells were counted after 7 days of culture in 10 random (×20) microscope fields per participant and expressed as CFU/cm.
Flow cytometry
After placement of an arterial sheath, 3 to 5 mL blood was wasted and then 1 mL whole blood was taken in an evacuated tube containing EDTA as the anticoagulant. A volume of 100 μL peripheral blood was evaluated by flow cytometry (fluorescence-activated cell sorter; Calibur, Becton Dickinson Biosciences, Franklin Lakes, New Jersey) within 3 hours after drawing. All procedures were similar to those previously described. 18 In brief, before being stained with specific monoclonal antibodies, cells were treated with fetal calf serum for 10 minutes, and the samples were then washed with buffer containing phosphate buffered saline (PBS) and 0.5% bovine albumin. This approach is used to saturate sites for nonspecific binding. Blood cells were then stained with fluorescein isothiocyanate-conjugated anti-human CD14 monoclonal antibody (Jingmei Biotech, Shenzhen, China) and phycoerythrin-conjugated anti-human kinase insert domain receptor (KDR) monoclonal antibody (R&D Systems, Minneapolis, Minnesota). Appropriate isotype controls were used for each staining procedure. The frequency of peripheral blood cells positive for the aforementioned reagents was determined by a 2-dimensional side-scatter fluorescence dot-plot analysis. The CD14/KDR double-positive cells were identified by the expression of KDR in the CD14 gate. After incubation, cells were lysed and washed with PBS. Data were processed with use of the CellQuest software program (Becton Dickinson, Franklin Lakes, NJ, USA). The same trained operator, who was blinded to the participants’ characteristics, performed all the tests throughout the study.
Biochemical Analysis
Serum was separated from the samples within 30 minutes and stored at −80°C until analysis. We measured the serum concentration of creatinine, triglyceride, total cholesterol, high-density lipoprotein and low-density lipoprotein cholesterol, and fasting glucose using the equipment for chemical analysis (Dimension AR/AVL Clinical Chemistry System, Newark, New Jersey) employed by the Clinical Laboratory Department of the 2nd Affiliated Hospital of Chongqing Medical University. Glomerular filtration rate was estimated by the following equation: eGFR = 0.741 × 175 × Cr−1.154 × Age−0.203 × 0.742 (if female). 19,20
Statistical Analysis
Data are presented as means ± standard deviation for continuous variables with normal distribution, medians and interquartile ranges (25th to 75th percentile) for not normally distributed parameters (CD14+ cell counts, levels of high-sensitivity C-reactive protein, homocysteine, soluble vascular cell adhesion molecule 1, and erythropoietin), and counts ([n] proportions) for categorical variables. The participants were divided into 3 groups on the basis of the GFR value according to the National Kidney Foundation. Trend effects across the GFR categories were analyzed by Spearman rank-order correlation coefficients. Intergroup differences in CD14+ cell numbers were also assessed by nonparametric tests. For bivariate correlations, Spearman or Pearson correlation coefficients (r) were computed.
Multivariate determinants of the CD14+ cell count were identified by backward stepwise multiple linear regression with logarithmic derivatives of CD14+ cell numbers as a dependent variable. Only the variables presenting the P value of ≤.20 in the univariate analysis were taken into account in the multiple regression. The F-to-remove value was set at 4.0, which is an approximate variance ratio (F; variability due to regression divided by variability about the regression line) for >30 degrees of freedom at a P value of .05. As in 4 patients the cell counts equaled 0, the logarithmic transformation to obtain a normal distribution consisted in the computation of log (raw value [%] + 0.01). A P value ≤.05 was considered significant.
Results
Characteristics of the study group stratified by GFR categories are shown in Table 1. Across decreasing GFR categories, we observed a gradual increase in CAD extension score (Table 1). Patients with CAD displayed a significant reduction in absolute EPC numbers (P< .01) when compared with control participants (n = 30) without CAD (Figure 1).
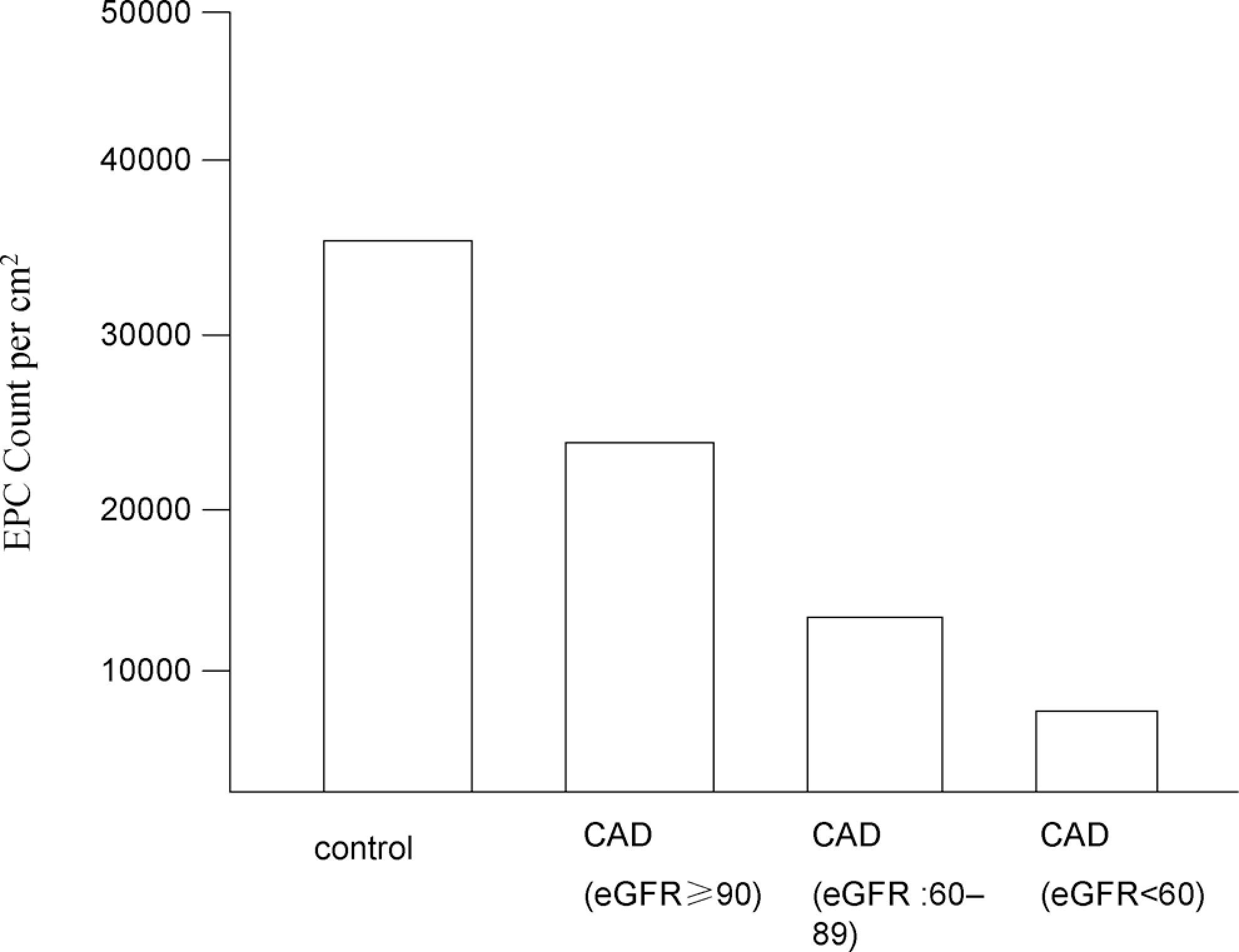
Absolute number of isolated EPCs in healthy control participants (n= 30) and patients with CAD. CAD indicates coronary artery disease; EPCs, endothelial progenitor cells.
Clinical and Angiographic Data of the Participants.
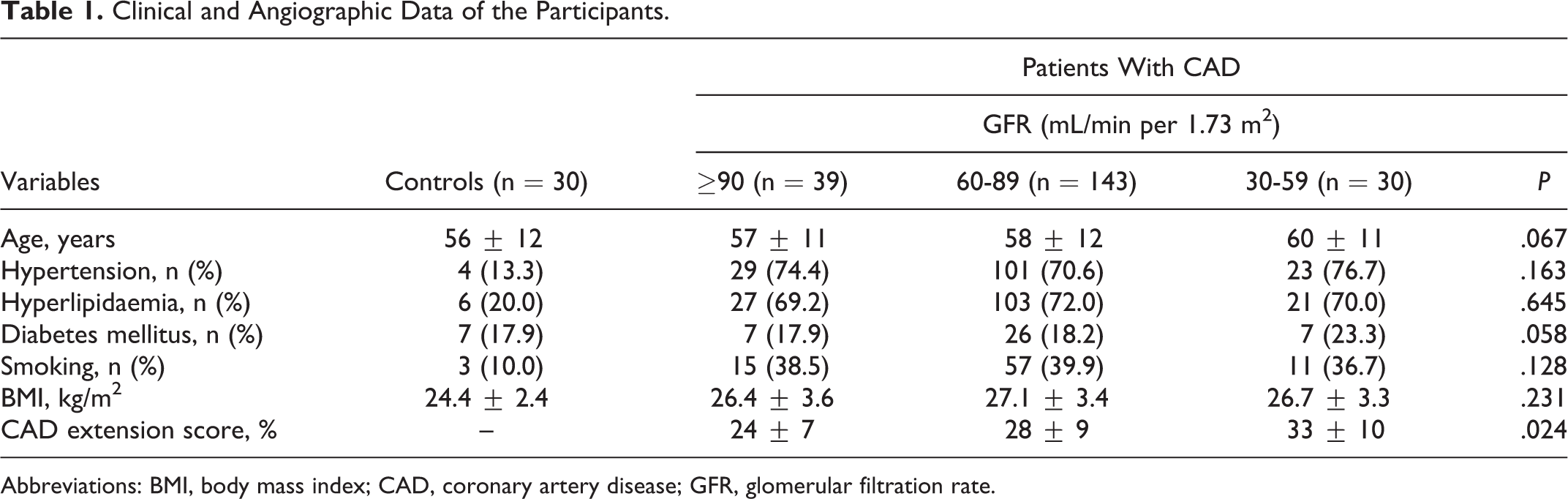
Abbreviations: BMI, body mass index; CAD, coronary artery disease; GFR, glomerular filtration rate.
With lowering GFR, CD14+ cell numbers (percentage of lymphocytes, median and interquartile range) decreased: 0.04 (0.02-0.06), 0.03 (0.02-0.05), 0.02 (0.01-0.04) for eGFR ≥90, 60 to 89, and 30 to 89 mL/min per 1.73 m2, respectively (P < .001 for trend). The CD14+ cell counts correlated with eGFR (r = −.27, P = .03) and with CAD extension score (r = −0.22, P = .02; Table 2). However, the age, smoking, hyperlipidemia, hypertension, and diabetes were not associated with CD14+ counts (all P > .05). By multivariate linear regression analysis, only GFR, diabetes mellitus, and CAD extension score entered the final regression equation describing logarithmic derivatives of CD14+ cell counts (Table 3).
Univariate Correlates of CD14+ Cell Counts.
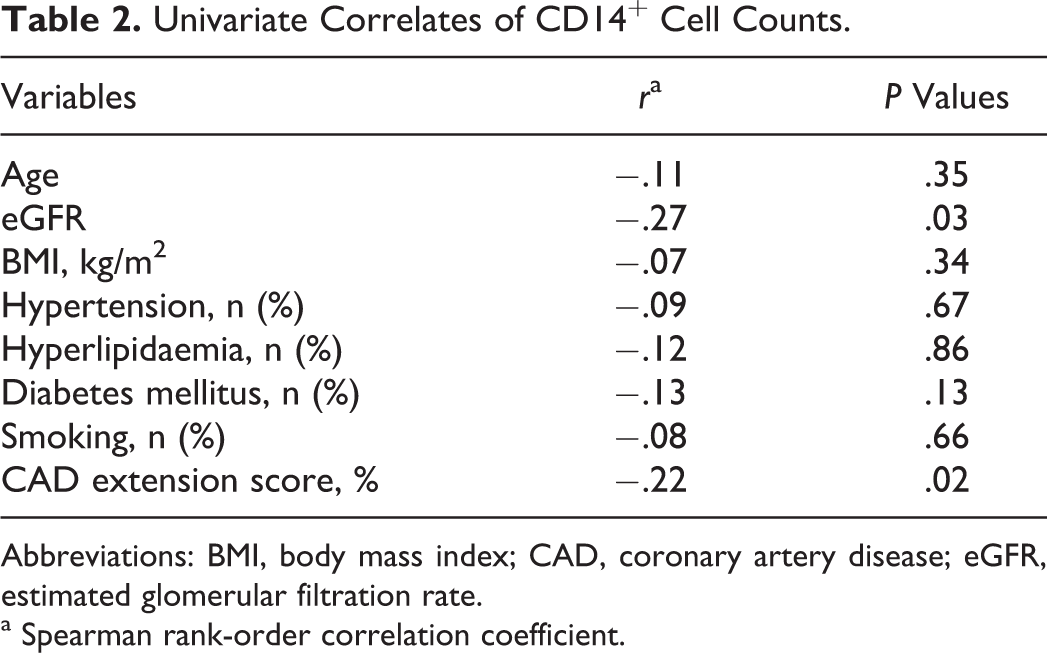
Abbreviations: BMI, body mass index; CAD, coronary artery disease; eGFR, estimated glomerular filtration rate.
a Spearman rank-order correlation coefficient.
Multivariate Analysis of CD14+ Cell Counts.
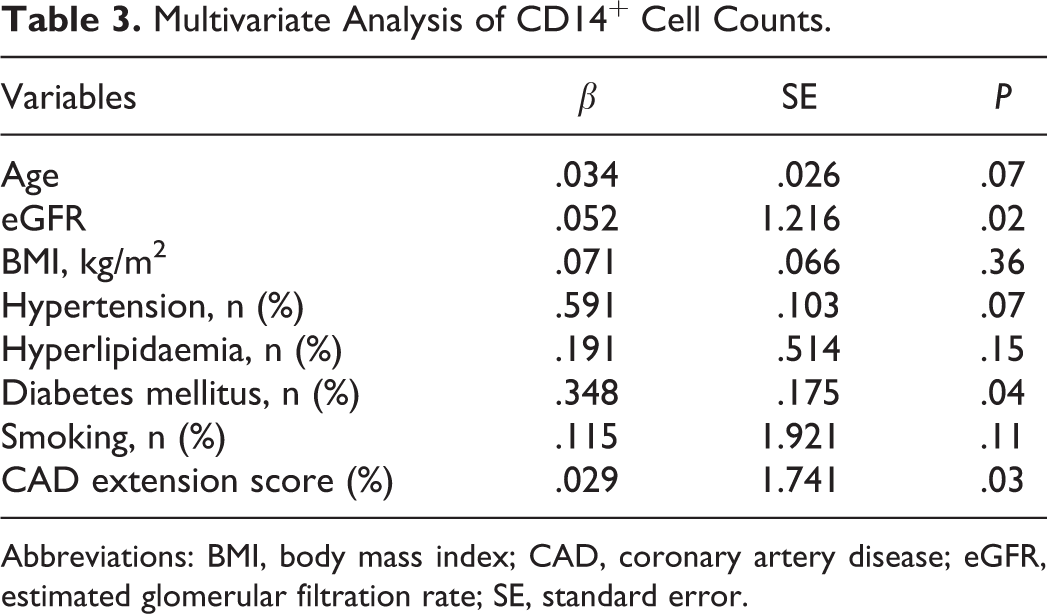
Abbreviations: BMI, body mass index; CAD, coronary artery disease; eGFR, estimated glomerular filtration rate; SE, standard error.
Discussion
Previous reports have indicated a correlation between circulating CD34+KDR+ cells and eGFR in CAD. 21 However, to the best of our knowledge, this is the first study to address the relationship between circulating CD14+ cells and eGFR in CAD. The principal finding of this study is CD14+ cell depletion is associated with renal dysfunction in coronary artery disease (CAD).
In many ischemic and injured organs, mobilization, homing, and transdifferentiation of EPCs play an important role in augmenting neovascularization and endothelial replacement after vascular injury. The EPCs augment angiogenesis both by stimulating the secretion of angiogenic growth factors and by providing a source of progenitor cells that can differentiate into mature vascular endothelial cells. Endothelial dysfunction is a typical finding in CKD. It contributes to accelerated arteriosclerosis and impaired angiogenesis and, therefore, to high cardiovascular morbidity and mortality in these patients. 22 Previous studies demonstrated reduced EPC levels in CKD, 23 whereas graft function seems to influence EPC number and function in kidney transplantation recipients. 24 –26
In the present study, the correlation between GFR and CD14+ cell count was not due to different characteristics of patients with decreased GFR. Although many risk factors such as hypertension, diabetes, and smoking were associated with decreased CD14+ cell count, we found CD14+ cell depletion was independently associated with renal dysfunction in CAD by multivariate analysis. The mechanisms of the GFR-CD14+ cell count relationship remains unclear. In the present study, the correlation between GFR and CD14+ cell count was inversely. However, we did not found other traditional risk factors besides diabetes were associated with CD14+ cell counts Several previous studies revealed altered number and impaired function of EPC in hyperlipidemia, hypertension, diabetes, and obesity. The reduced circulating concentration of EPC has become a surrogate marker of endothelial function and has been implicated in the pathogenesis of many vascular diseases. 27 In addition, as the previous study indicated that both lower CD34+/KDR+ cell count and excessive cardiovascular risk accompanying renal insufficiency might be due to common factors other than the CAD extent, inflammatory activation, or homocysteine accumulation. 28 However, the relationship between CD14+ count and GFR can be explained by the same mechanism that merits further investigation.
The present study has a limitation; due to methodological limitations, we have not studied functional properties of cultured EPC—their migratory capacity, adhesive properties, and differentiation—all of which may be impaired in CKD. Age-related endothelial dysfunction was demonstrated to be linked to the depressed capacity of culture-enriched EPC to migrate and proliferate despite comparable CD14+ cell counts in young and elderly participants, which points to the relevance of EPC activity independently of their number.
Conclusion
Our results suggest that CD14+ cell depletion is associated with renal dysfunction in CAD.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: the National Science Foundation, China (30970826).
