Abstract
Objective:
We investigated atorvastatin reloading effects on endothelial progenitor cell (EPC) count and inflammatory cytokine expression after percutaneous coronary intervention (PCI) in patients with stable angina pectoris who had previously received long-term statin treatments.
Methods:
Patients with stable angina pectoris were treated with 80 mg atorvastatin 12 hours and 40 mg atorvastatin 2 hours before coronary angioplasty (n = 15) or preoperatively with 40 mg/d atorvastatin for 7 days (n = 15) or did not receive atorvastatin (n = 15). CD45−/133+/34+, CD45−/CD34+/kinase insert domain receptor (KDR)+, and CD45−/CD144+/KDR+ EPCs in the peripheral blood were determined by flow cytometry 1 hour before as well as 1 hour, 6 hours, and 24 hours after PCI. Soluble intercellular adhesion molecule 1 (sICAM-1), hypersensitive C-reactive protein (hCRP), and troponin-I (TnI) serum concentrations were analyzed immediately prior to and 24 hours after PCI.
Results:
In the 40mg Atorvastatin and control groups, none of the analyzed EPC blood concentrations changed significantly from 1h before operation to 1h and 6 h postoperative values. In contrast, the number of circulating early differentiation stage EPCs CD45-/133+/34+ and CD45-/CD34+/ KDR+ raised significantly from 1 h preoperative values (57.3±9.3; 57.3 ± 10.7) to 1 h postoperative ((74.4 ± 11.4; 78.8 ± 16.2), (p < 0.05)) and 6 h postoperative ((93 ± 16.9; 99.7 ± 11.9), (p < 0.05)) concentrations after coronary angioplasty in the 80mg Atorvastatin medication patients. In the control group, the sICAM-1 (174.55 ± 38.91 vs 204.11 ± 58.24) and hCRP (1.89 ± 1.93 vs 9.0 ± 11.1) serum concentrations at 24 hours after PCI were significantly elevated (P < .05) compared to preoperative values, whereas the increases in the 2 groups treated with atorvastatin were not significant. In addition, the rise in serum TnI concentration level from pre- to postoperative in the 80-mg (0.02 ± 0.02 vs 0.09 ± 0.08) and the 40-mg (0.01 ± 0.03 vs 1.2 ± 2.59) reloading groups was less than that of the controls (0.01 ± 0.02 vs 1.75 ± 3.09) (p < 0.05).
Conclusion:
Our results suggested that high-dose atorvastatin application before PCI triggered early EPC circulation. Furthermore, postoperative inflammatory cytokine sICAM-1 as well as hCRP serum levels were reduced, while postinterventional myocardial injury marker TnI elevations were inversely correlated with statin reloadings.
Introduction
Endothelial progenitor cells (EPCs) can proliferate and differentiate into vascular endothelium and eventually into blood vessels. Recent studies have shown that EPCs not only participate in angiogenesis but also play a role in blood vessel development after birth and in repair of endothelial damages. 1,2 Coronary heart disease is essentially a vascular deformation and because EPCs are involved in endothelium repair, a lack of EPCs may play a role in its initiation and development. 3 Statins are a class of drugs inhibiting 3-hydroxyl-3-methyl-glutaryl-coenzyme A (HMG-CoA) reductase, which thereby upregulate low-density lipoprotein (LDL) receptors on cell membranes and consequently reduce LDL in plasma. In addition, statins inhibit smooth muscle cell proliferation and platelet aggregation and have anti-inflammatory as well as plaque-stabilizing properties. 4 –7 Statins are currently widely used clinically as a basic therapy against atherosclerosis. Studies on the effects of statins in perioperative interventions such as percutaneous coronary intervention (PCI) also known as coronary angioplasty, of which the Atorvastatin for Reduction of Myocardial Damage During Angioplasty (ARMYDA) series are representative, suggest that major adverse cardiac events (MACEs) such as myocardial infarctions can be significantly reduced by increasing statin medications before surgery, or, for patients with long-term statin use, by drug reloading before surgery. 8 The statin reloading methods, however, have differed in various experiments, and studies are lacking regarding the influence of loading strategies on pleiotropic statin effects. We therefore compared different pre-PCI atorvastatin reloading in 3 treatment groups who had long-term statin use before the PCI procedure and investigated the effects of atorvastatin reloading strategies on circulating EPCs and inflammatory cytokine expressions during perioperative PCI periods.
Patients and Methods
Patient Information
In this study, 45 patients with stable angina pectoris who had previously received long-term statin treatments and underwent elective PCIs were included. Stable angina was defined as short-term chest pain, induced by exercise or other conditions that increase myocardial oxygen demand that disappeared rapidly after rest or sublingual administration of glyceryl trinitrate, which had occurred for 1 month or more. 9 Other inclusion criteria were men between the age of 45 and 80 and postmenopausal women of less than 80 years who have received long-term statin therapies (>8 weeks) and had elective PCIs. Exclusion criteria were patients with acute coronary syndrome, diabetes, abnormal high ALT/AST levels, creatinine >3 mg/dL, or left ventricular ejection fraction <30%, those with clear infection foci, whether chronic or acute and those with blood diseases, tumors, or systemic autoimmune diseases. All patients gave their written informed consent and the study was approved by the ethical committee of Ningbo First Hospital, the Affiliated Hospital of School of Medcine of Ningbo University.
Study Design and Clinical Parameter
The 45 patients meeting the inclusion criteria were randomized into 3 groups, each group comprising 15 participants. The first group received no atorvastatin reloading before PCI (Control group); the second group received 40 mg atorvastatin reloading for 7 continuous days preceding the PCI operation (40 mg reloading group); and the third group received 80 mg atorvastatin 12 hours before PCI, with an additional dose of 40 mg 2 hours prior to PCI (80 mg reloading group). All patients’ EPC blood concentrations were tested at 1 hour before PCI and 1 hour, 6 hours, and 24 hours after PCI. Based on the definition of PCI-related myocardial infarction set forward by the third edition of international myocardial infarction definitions published 10 in 2012, myocardial injury was defined as a condition when the cardiac troponin (cTn) values were >5 × 99th percentile upper reference limit in the absence of ischemia, angiographic, or imaging findings. The serum concentrations of soluble intercellular adhesion molecule 1 (sICAM-1) and C-reactive protein (CRP), the inflammatory cytokines involved in coronary heart diseases, were measured upon hospital admittance and at 24 hours after PCI. Troponin-I (TnI) serum concentrations were determined with Access AccuTnI assays (Beckman Coulter Inc., Brae, California), CRP concentrations were measured with SYNCHRON System (Beckman Coulter Inc., Brae, California), and sICAM-1 was detected with enzyme-linked immunosorbent assay kits (Bender, Germany). Clinical follow-up examinations were conducted for 30 days following PCI and MACEs, including death, nonlethal myocardial infarction, and target vessel revascularization.
Detection of EPCs
Venous blood of 3 mL was collected and 100 µL was transferred into each of the 4 flow cytometry tubes (BD, Franklin Lakes, New Jersey); 5 µL of fetal bovine serum was added to each tube, and the samples were incubated for 15 minutes at room temperature for Fc-receptor blocking. One tube was randomly chosen as a negative control; 10 µL of PE-labeled CD133+/AC133+ antibodies (Miltenyi Biotec, Germany) and 10 µL of fluorescein isothiocyanate (FITC)-labeled vascular endothelial growth factor receptor 2 (VEGFR-2+)/kinase insert domain receptor (KDR) antibodies (R&D, Minneapolis) were added to the second tube; 10 µL of FITC-labeled VEGFR-2+/KDR antibodies and APC-labeled CD34+ antibodies (Miltenyi Biotec) were added to the third tube; 10 µL of FITC-labeled VEGFR-2+/KDR antibodies and PE-labeled CD144+ antibodies (Miltenyi Biotec) were added to the last tube and 5 µL of PC5-labeled CD45 antibodies (Beckman Coulter, Brae, California) were added to all the tubes. The tubes were placed in the dark and rocked at 4°C for 45 minutes. After thorough rocking, flow cytometry red blood cell lysis buffer (R&D, Minneapolis) was added to lyse the red blood cells and the tubes were rocked again and placed in a 37°C incubator in the dark for 10 minutes, then centrifuged at 1500 rpm for 5 minutes, and the supernatant was discarded. Cells were then washed with 3 mL phosphate-buffered saline (PBS) and centrifuged at 1500 rpm for 5 minutes and the supernatant was discarded. PBS of 500 µL was added to each tube and after mixing 100 µL of samples were run through an FACS Calibur Flow Cytometer (BD, New Jersey) to count the number of the labeled cells. The flow cytometry pattern results are illustrated in Figure 1, which shows CD45−/CD133+/CD34+ progenitor cells, CD45−/CD34+/KDR+ early EPCs, and CD45−/CD144+/KDR+ late EPCs.

CD45-/CD133+/CD34+, CD45-/CD34+/kinase insert domain receptor (KDR)+ and CD45-/CD144+/KDR+ endothelial progenitor cell (EPC) flow cytometry analyses. A) EPCs labeled as CD45-/CD133+/CD34+. (Y-axis: APC-A=APC-labeled CD34 antibodies; X-axis: 133 PE-A=PE labeled CD133 antibodies). B) EPCs labeled as CD45-/CD34+/KDR+. (Y-axis: APC-A=APC-labeled CD34 antibodies; X-axis: KDR-FITC-A= FITC labeled KDR antibodies). C) EPCs labeled as CD45-/CD144+/KDR+. (Y-axis: KDR-FITC-A= FITC labeled KDR antibodies; X-axis: 144 PE-A = PE labeled CD144 antibodies).
Statistical Analyses
All statistical analyses were performed with SPSS 16.0. Quantitative data are shown as mean ± standard deviation (
Results
There was no significant difference between the groups in terms of baseline characteristics, PCI treatments, or postoperative MACEs, thereby allowing reliable difference between comparisons of perioperative atorvastatin effects (Tables 1 and 2).
Patient Baseline Clinical Data and PCI Treatments.

Abbreviations: ACEI, angiotensin-converting enzyme inhibitors; ADP, adenosine diphosphate; ARB, angiotensin receptor blocker; LDL, low-density lipoprotein; PCI, percutaneous coronary intervention; TC, total cholesterol; TG, triglyceride; CCB, calcium channel blocker (amlodipine, nifedipine, felodipine).
Treatment and MACEs of the 3 Groups.a

Abbreviations: DES, drug-eluting stent; MACEs, major adverse cardiac events; PCI, percutaneous coronary intervention.
aMACE included death in 30 days after PCI, nonfatal myocardial infarction, and target vessel revascularization.
Changes in Peripheral Blood EPC Cell Count Before and After PCI in the Different Groups
The CD45−/CD133+/CD34+- and CD45−/CD34+/KDR+-labeled EPCs in the 80-mg reloading group at 1 hour and 6 hours postoperation time points were significantly elevated compared to their values before the operation (P < .05; Table 3). The number of CD45−/CD133+/CD34+-labeled EPCs in the 80-mg reloading group at 1 hour and 6 hours after PCI were 1.3 and 1.6 times higher than the preoperation values and the number of CD45−/CD34+/KDR+-labeled EPCs at 1 hour and 6 hours after PCI were 1.4 and 1.7 times higher than the preoperation values. There was no significant difference in CD45−/CD144+/KDR+-labeled EPC counts in the peripheral blood in the 80-mg group. In the control and 40-mg reloading groups, no significant changes were detected in any of the labeled EPCs in the patients’ peripheral blood EPCs before and after PCI (Table 3). In the 80-mg reloading group, the levels of CD45−/CD133+/CD34+ and CD45-/CD34+/KDR+ EPCs reached a maximum at 6 hours after operation and returned to control levels by 24 hours after operation. In the control and the 40-mg reloading groups, most patients reached maximum EPC levels 1 hour after operation and maintained those levels.
Changes in Circulating EPCs Marked With Different Antibodies at Different Time Points Before and After Operation in Different Groups (cells/100 µL).

Abbreviations: EPCs, endothelial progenitor cells; KDR, kinase insert domain receptor.
aCompared to preoperation, P < .05.
Changes in sICAM-1 and CRP Levels Before and After PCI in the Different Groups
In all 3 groups of patients, sICAM-1 and CRP levels increased after PCI intervention. Levels increased most in the control group, reaching a statistically significant difference compared to levels before PCI (P < .05); in the 40-mg and 80-mg reloading groups sICAM-1 and CRP levels were significantly higher than that in the controls, but when compared to levels before PCI, the differences were not statistically significant (Table 4). Furthermore, statin concentration was inversely correlated with cytokine expression, in that the higher the statin concentration was, the less were the increases after PCI, indicating that high-dose statin treatment before PCI can reduce the activation of inflammatory cytokines.
Atorvastatin Treatment-Related Changes in sICAM-1 and CRP Serum Concentrations Before and After Operation (ng/mL).
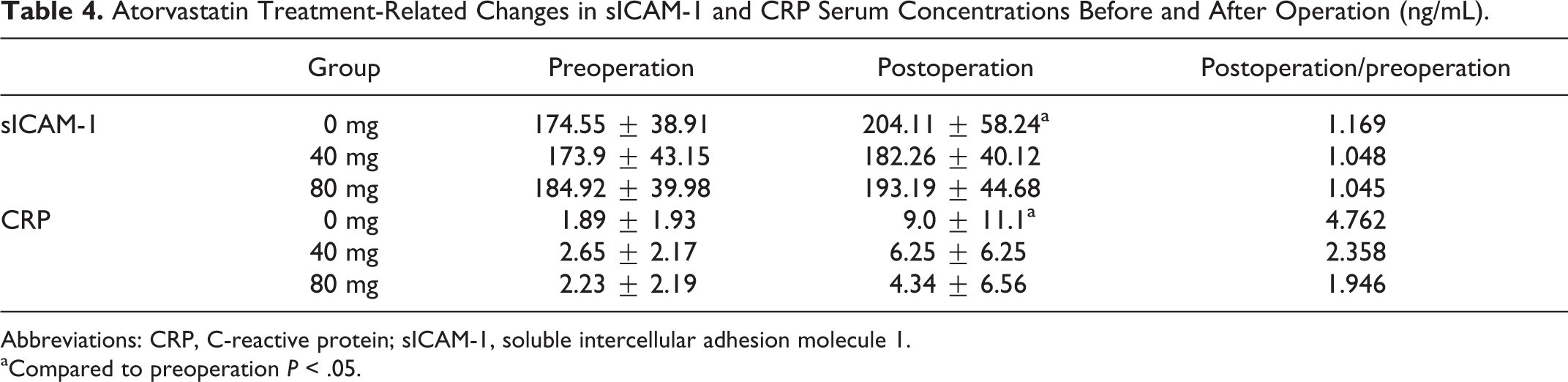
Abbreviations: CRP, C-reactive protein; sICAM-1, soluble intercellular adhesion molecule 1.
aCompared to preoperation P < .05.
Changes in Serum TnI Concentrations Before and After PCI in the Different Groups
In all 3 groups, the serum TnI levels increased significantly after PCI. All statin reloading groups, however, showed a trend of less perioperative myocardial injury than that of the control group. The postoperation serum TnI increase in the 80-mg group was significantly less than that in the control group (Table 5), and the difference between the 2 groups (postoperation vs preoperation) was statistically significant (0.06 ± 0.08 ng/mL vs 1.74 ± 3.09 ng/mL, P < .05). Based on the definition of PCI-related myocardial infarction set forward by the third edition of international myocardial infarction definitions published in 2012 (post-PCI TnI levels 5 times or more than the normal 99% upper limit), there were 8 PCI-related myocardial infarctions in the control group, 6 cases in the 40-mg/7 days statin reloading group, and 3 cases in the 80-mg reloading group. Overall, there was a trend of less perioperative myocardial injury marker expression in the statin reloading groups compared to the control group.
Atorvastatin Treatment-Related Pre- and Postoperative Serum TnI Concentrations (ng/mL).
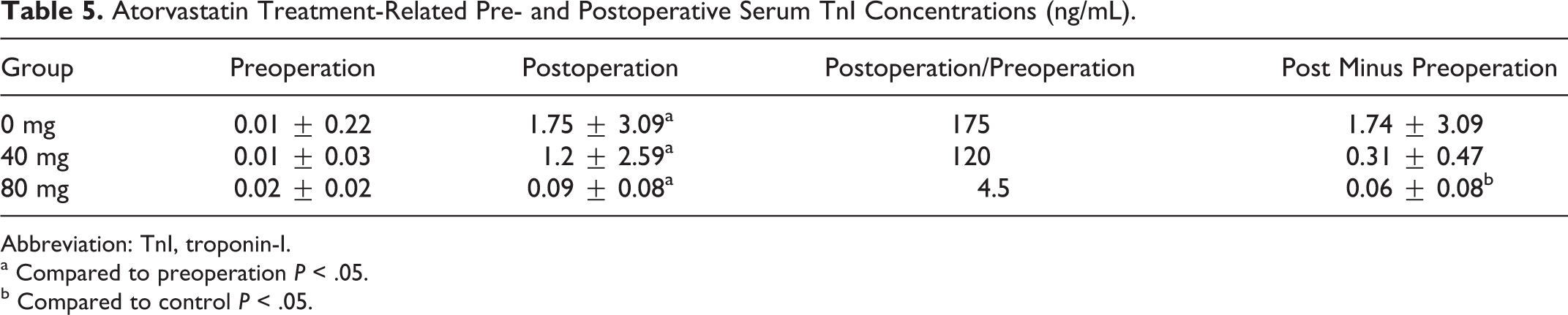
Abbreviation: TnI, troponin-I.
a Compared to preoperation P < .05.
b Compared to control P < .05.
Discussion
Coronary heart disease is often associated with reduced EPC bone marrow mobilization and function. 11 –14 The ARMYDA-RECAPTURE study 15 has confirmed that prePCI statin reloading can reduce the occurrence of myocardial infarctions during the perioperative period. Furthermore, in patients with coronary heart disease, Vasa et al showed that 4 weeks of atorvastatin treatment (40 mg/d) led to significantly increased peripheral blood EPC levels. 16 In contrast, however, a study done by Hristov et al indicated that long-term statin treatment (longer than 8 weeks) led to a reduced number of peripheral blood EPCs. 17 These divergent findings raise several questions. For example, does long-term statin treatment desensitize the EPCs to normal doses of statins? Does long-term statin treatment upregulate integrins associated with EPCs and as a result higher statin doses might be required to induce EPC mobilization? Finally, which preoperative atorvastatin application mode is most beneficial for perioperative PCI medication?
Differentiation of Circulating EPCs
Morphologies of EPCs change at different stages of differentiation. Right after adhering to the vessel surface, they have an oval shape that gradually changes into a fusiform or spindle shape, during which they are known as early EPCs. As they differentiate, they become cobblestone shaped, known as late EPCs. Because EPCs cannot be accurately distinguished based on their cell morphology alone, however, cell surface markers are necessary for their identification. As EPCs differentiate and mature, they begin to express specific endothelial cell markers that allow them to be distinguished from other various stem and mature endothelial cell types in bone marrow. CD34+/CD133+/KDR+ is currently the major label of EPCs. CD133+ cells in the peripheral blood can be induced to differentiate into endothelial cells in vitro, at which point they become CD133 negative; thus, CD133 expression is a specific label of EPCs and distinguishes EPCs from mature endothelial cells. CD34 is believed to be a marker of hematopoietic stem/progenitor cells, but CD34− EPC subpopulations have also been reported. 18 The KDR/VEGFR-2 is a tyrosine kinase receptor expressed on the cell membranes of mature endothelial cells and EPCs. However, EPCs do not express von Willebrand factor (vWF) and vascular endothelial cadherin (CD144). 11 As EPCs differentiate and mature, they exhibit enhanced expression of vWF and VE-cadherin and reduced expression of CD133. Once the cell is completely differentiated into a mature endothelial cell, CD133 expression is absent. Finally, CD45 is a marker for leukocytes that EPCs lack. Thus, to distinguish different stages of differentiation, a combination of markers must be used to accurately identify EPC subsets. In our study, we distinguished between progenitor cells (CD45−/CD133+/CD34+), early-stage differentiated EPCs (CD45−/CD34+/KDR+), and late-stage differentiated EPCs (CD45−/CD144+/KDR+) in order to accurately reflect the EPC pattern during the PCI perioperative period.
Correlation Between Atorvastatin Medication, Myocardial Injury Marker TnI, and EPCs
Both methods of atorvastatin reloading reduced the expression of perioperative myocardial injury TnI marker, although only the application of 80 mg atorvastatin 12 hours before PCI with an additional dose of 40 mg 2 hours before PCI was statistically significant (P < .05). The group that received only 40 mg atorvastatin for 7 days before PCI showed a similar trend compared to the control group. Our study also showed that the number of circulating EPCs did not rise during the preoperative period in patients who used statin for a long term and who were not medicated with a preoperative statin reloading before PCI, whereas patients who received short-term, high-dose atorvastatin, reloading before PCI had increased circulatory and particularly early differentiated EPCs. These findings suggest that atorvastatin reloading activates EPCs by mobilizing bone marrow cells. One of the limitations of our study was a relatively small sample size and a lack of functional EPC studies. In addition, during PCIs myocardial ischemia might have triggered EPC activation via VEGF stimulation, and other PCI pathophysiological changes, particularly in the control group, might have influenced the sICAM-1 and CRP serum levels.
In summary, our results suggest that short-term, high-dose preoperative atorvastatin application can improve PCI treatments of patients with stable angina pectoris who have been receiving long-term statin treatments before the surgical intervention. Early-stage differentiated EPCs were particularly enhanced in circulating blood plasma, and expression of the myocardial injury marker TnI was significantly reduced in patients with stable angina pectoris who received a 1-day preoperatively high dose of atorvastatin prior to PCI treatment. Because atorvastatin was the only statin used in this study and the patient sample size was small, further research is required to substantiate our preliminary findings and expand them to other statins.
Footnotes
Acknowledgments
We particularly thank Ying Zhou and Ying Chen from Ningbo First Hospital, the Affiliated Hospital of School of Medicine of Ningbo University for assisting in preparing the laboratory assays.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
