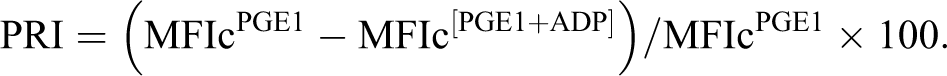Abstract
Introduction:
Dual antiplatelet treatment (DAPT) with clopidogrel and aspirin represents common approach in prevention of thromboembolic events in patients with acute coronary syndrome undergoing percutaneous coronary intervention (PCI). The drawback of clopidogrel treatment is large interindividual variability in response.
Aim:
Our article aims to suggesting the most convenient method in monitoring the DAPT of post-PCI patients.
Methods:
We analyzed the on-treatment platelet reactivity by light transmission aggregometry and vasodilator-stimulated phosphoprotein (VASP) flow cytometric assay. Samples were obtained in 3 intervals: first prior to PCI, then 1, and 30 days after PCI.
Results:
Based on VASP-platelet reactivity index (PRI), we observed 100% response rate in prasugrel-treated patients and 62% to 73 % in the clopidogrel group. Overall, only 2 (7%) patients with the VASP-PRI value in therapeutic range had major adverse cardiovascular events.
Conclusion:
Our results hint VASP-phosphorylation assay as a relevant method for guiding and tailoring DAPT.
Keywords
Introduction
Proaggregatory effect of adenosine diphosphate (ADP) is mediated through interaction between ADP and G-protein coupled platelet surface receptors, P2Y1 and P2Y12. The crucial receptor in the process of platelet activation is P2Y12 whose activation is required for complete platelet response to ADP as well as response to other inducers (such as thrombin, thromboxane A2, collagen, etc). P2Y12 also maintains a role in stimulating platelet secretion and activation of GPIIb/IIIa. Presence of P2Y12 is limited to platelets, endothelial cells, and smooth muscle cells. This fact along with importance of receptors in platelet activation makes it an attractive target for prevention of thromboembolism. 1 Currently, the ADP inhibitors represent the cornerstone in treatment of patients with acute coronary syndrome (ACS) and/or undergoing percutaneous coronary intervention (PCI). Clopidogrel along with aspirin is still commonly used, while ADP inhibitors of newer generation (prasugrel and ticagrelor) are now recommended as the new golden standard. 2,3 In case of insufficient response, patients maintain high platelet reactivity (PR), leading to occurrence of cardiovascular events in spite of receiving antiplatelet treatment. High PR was confirmed as independent predictor of myocardial necrosis, myocardial reperfusion, and clinical outcome of patients post-PCI. 4 Despite high effectivity, thromboembolic events occur even when patients are on antiplatelet therapy. A significant number of patients show and maintain a high PR, similar to that observed before the start of antiplatelet therapy. 2 Low response to clopidogrel can be attributed to various extrinsic and intrinsic factors.
Genetic polymporphisms are one of the predominant factors. Low response to clopidogrel is often caused by polymorphism in genes encoding hepatic cytochromes (CYP)3A4, 2C19, 2B6, 1A2, and glycoprotein paraoxonase 1 (PON1). These enzymes regulate conversion of clopidogrel prodrug into active metabolite (2-oxoclopidogrel). Genetic defects can also result in limited intestinal absorption of prodrug itself (efflux pump P-glycoprotein 1). 4 Several studies have clearly associated these gene polymorphisms to both ischemic and bleeding complications in patients receiving dual antiplatelet therapy. 4,5 Polymorphism in platelet surface receptors (GPIIIa) is another of proposed inherited mechanisms.
Clinical Causes
Needless to say, noncompliance and underdosing are one of the frequent causes of inadequate clopidogrel treatment. Other variables associated with decreased response include advanced age, high body mass index, diabetes mellitus, and renal insufficiency in diabetes mellitus. It is necessary to note that PR changes overtime. The PR in acute phase of ACS is higher and decreases afterward, which was confirmed in prospective study by Campo et al. 6 Another mechanism may be increased platelet turnover. 5 Variability often results from drug interactions with CYP450, for instance some of the proton pump inhibitors, lipophilic statins, and calcium channel blockers. On the other hand, tobacco smoking is associated with increased response via stimulation of CYP1A2 activity. 7
Studies conclude that poor responders to clopidogrel represent between 10% and 40% of patients. 7 –10 Thienopyridine resistance results in inadequate platelet inhibition and high on-treatment PR, which is an established risk factor for occurrence of subacute in-stent thrombosis in post-PCI patients. A prospective study by Buonamici et al monitoring a group of 804 patients demonstrates that those with impaired response to clopidogrel were in 3-fold increased risk of stent thrombosis and cardiac death. 11,12
Recent data have shown that titrating the dose of clopidogrel in patients undergoing PCI significantly reduces the incidence of major adverse cardiovascular events (MACEs), and large-scale clinical trials are currently underway to investigate whether individually tailored treatment based on results of platelet function testing leads to improved clinical outcome. The ideal platelet function test would be rapid, simple, reproducible, and appropriate for use at the point of patient contact. 13
Historically, turbidimetric light transmittance aggregometry (LTA) was considered the “gold standard” platelet function assay. The LTA measures the increase in light transmission crossing a platelet suspension that occurs when platelets aggregate in response to an agonist. Using LTA, 4% to 30% of patients treated with clopidogrel do not have adequate antiplatelet response. Poor response to clopidogrel assessed by LTA correlates with an increased risk of adverse events. 4 Although LTA is the routinely used method for platelet function testing, it does not target primarily P2Y12-linked cascade and requires considerably large volume of blood sample. Despite being regarded as “gold standard,” major drawback lies in standardizing the method between laboratories and patients.
Flow cytometric VASP-phosphorylation assay
Vasodilator-stimulated phosphoprotein is an intracellular platelet protein whose phosphorylation (VASP-P) specifically reflects inhibition of P2Y12 receptor (for further details see Methods section). Therefore, VASP assay accurately detects clopidogrel response. The assay is performed in vitro on flow cytometer using fluorescein isothiocyanate (FITC) staining to detect activated platelets. The VASP/P2Y12 test is strongly correlated with inhibition of ADP-induced platelet aggregation in vitro by specific P2Y12 blockade (r = .72; P < .0001). 14,15 Results of aggregometry with induction by 10 μmol/L ADP strongly correlate with VASP-P values. 12 The VASP-P is considered the most sensitive platelet function testing method and is often used as reference for validation of emerging assays. Cutoff value of ≥50 platelet reactivity index (PRI) demonstrated a 100% sensitivity in predicting the stent thrombosis in high-risk PCI. 5
Point-of-care assays
Point-of-care methods are applied in a near patient setting, in environments other than classical laboratories (eg, intensive care units). Advantages lie in availability, cost, and labor effectiveness. On the other hand, its sensitivity, specificity, and predictive value are questionable and inferior in comparison to standard laboratory assays.
Platelet function analyzer 100/200
In platelet function analyzer (PFA), citrated blood is aspirated through a small aperture in a collagen-coated membrane. Platelets then adhere to the collagen surface and lead to a cessation of blood flow. The measured parameter is closure time. The PFA can use 3 different cartridges (epinephrine, ADP, and P2Y). The results were not satisfying so far in the only clinical study on the new PFA-100 P2Y. Moreover, the Kaplan-Meier curves divided only after 190 days, which makes any use of this cartridge for the clinical management highly questionable. There was no predictivity of the PFA-100 tests for bleeding events found in this study. 16
VerifyNow P2Y12
The VerifyNow system is a test that measures the agglutination of fibrinogen-coated beads by platelets stimulated by the agonist in citrated whole blood. The device specifically detects the degree of the P2Y12 inhibition by clopidogrel. The system consists of 2 assay channels. Clopidogrel metabolite inhibits aggregation in the ADP-containing channel but not in the second channel with thrombin receptor-activating protein. Aggregation in both channels is measured as the increase in light transmission and is reported in platelet response units (PRU). Analysis by Patti et al showed that PRU value above 240 is the optimal cutoff point to predict the 30-day outcome, with sensitivity of 81% and specificity of 53%. 5
Multiple electrode platelet aggregometry multiplate analyzer
Multiple electrode platelet aggregometry (MEA) multiplate analyzer is a device based on MEA, which works with whole blood sample. The MEA implements the principle of impedance aggregometry without the need for blood centrifugation. The device is able to assess platelet function in approximately 10 minutes. Impedance with MEA is measured in arbitrary aggregation units (AU), where patients with AU ≥416 at ADP test were considered low responders. In contrast to LTA, VASP, VerifyNow, and others, in MEA, the aggregation takes place on surfaces which is more similar to in vivo conditions. 5
Patients
In our prospective, single-center study, we analyzed patients with ST-segment elevation myocardial infarction (STEMI) admitted to University Hospital in Martin who underwent PCI. A total of 48 post-PCI patients with STEMI were primarily enrolled, while 40 were able to meet the inclusion criteria. We experienced dropout rate, mostly due to patient noncompliance (n = 4) as well as logistic and laboratory errors (n = 4). Patients were treated by loading dose of antiplatelet agents and underwent short follow-up period of 2 days. Decision about initiating either prasugrel or clopidogrel treatment was based on the choice of the physician. Patients were administered either 600 mg clopidogrel (n = 26) or 60 mg prasugrel (n = 14). The study was performed in accordance with Declaration of Helsinki. Institutional review board approval was given by Ethics Committee of Jessenius Faculty of Comenius University. Informed consent was obtained from all the participants.
Methods
Sample Processing and Assays
Venous blood samples were drawn by a traumatic venipuncture from antecubital vein. Samples were obtained in 3 intervals: first prior to PCI, then in follow-up period within 1 day after PCI, and administration of loading dose of antiplatelet agent. Patients then underwent follow-up until 30 days since PCI; however, these data are too scarce to be included in this brief report. Blood was drawn into 3.8% citrate vacutainer syringe, then gently inverted 3 to 4 times. Further, samples underwent analysis by LTA using ADP induction. In flow cytometric analysis, we used PLT VASP/P2Y12 assay kits (Diagnostica Stago, France). Sample of citrate blood was incubated with prostaglandin E1 (PGE1) and PGE1 + ADP (activated platelets). After cellular permeabilization by nonionic detergent, VASP-P is labeled by indirect no-wash immunofluorescence using a specific monoclonal antibody (FITC, 16C2). Dual color flow cytometry analysis then allowed comparison of the 2 tested conditions. Analysis was carried out on FACSCalibur flow cytometer (BD Biosciences, San Jose, California). In the final step, PRI was calculated using corrected mean VASP fluorescence intensities (MFIc) in the presence of PGE1 alone (resting platelets) or PGE1 + ADP simultaneously (activated platelets). Index represented the ratio of activated/resting platelets and was calculated according to the following equation:
The resulting value described PRI to ADP treatment in the range of 0% to 100 %. Values of PRI above 50% were considered as determinant of high on-treatment PR and inadequate response to treatment. 17
Statistical Analysis
Data were checked for normality with the Shapiro-Wilk test. Normally distributed continuous or interval-scaled variables are presented as mean ± standard deviation (SD); otherwise median (M) and quartile ranges from the lower quartile Q1 to the upper quartile Q3 (Q1-Q3) were used. Group effects (ie, differences between clopidogrel and prasugrel groups) were tested with t test in the case of normally distributed data or with Mann-Whitney U test when data distribution was asymmetrical. Differences between proportions (eg, number of patients in C and P groups) were tested with binominal tests. Categorical variables grouped in 2-way contingency tables were analyzed using chi-square tests. Associations between continuous variables were assessed by Pearson correlation coefficient; for categorical data, Kendall’s rank correlation coefficient “τ” was used. The significance of P < .05 was considered as a criterion for comparison between data sets with equal and unequal variances All statistical analyses were performed with StatsDirect 2.7.8 software (Cheshire, United Kingdom).
Results
A total of 48 patients were enrolled, while 40 have met the inclusion criteria. Mean age was 68.25 ± 10.49 years (range 48-84 years, median: 68), with predominance of male gender (62.5%). In all, 26 patients were administered loading dose of 600 mg clopidogrel, while 14 received 60 mg of prasugrel. Choice of treatment was based on attending cardiologists’ decision. Further baseline characteristics are included in Table 1. Four patients died during the follow-up; however, those were not excluded since we focus on monitoring of adverse events while on treatment. Three patients had in-stent thrombosis of coronary vessels and total number of patients having MACEs was 6. In the group of post-PCI patients with STEMI, prasugrel induced more potent platelet inhibition than that of clopidogrel while avoiding bleeding side effects. Loading dose of 60 mg prasugrel induced significantly stronger platelet inhibition compared to 600 mg of clopidogrel (21.50 vs 49.87, respectively; P = .0012). This is also depicted in spread plot in Figure 1. All patients treated with prasugrel responded by decrease in PR below cutoff value, while 38% of patients with clopidogrel did not respond adequately. No bleeding side effects were observed in either of the groups. Results of VASP-P assay correlated with LTA using induction by 10 μmol ADP (r = .4866, r 2 = .2367, P = .0047). No significant differences were noted in the risk of developing in-stent thrombosis or cerebral stroke.
Baseline Characteristics of Studied Population Groups.
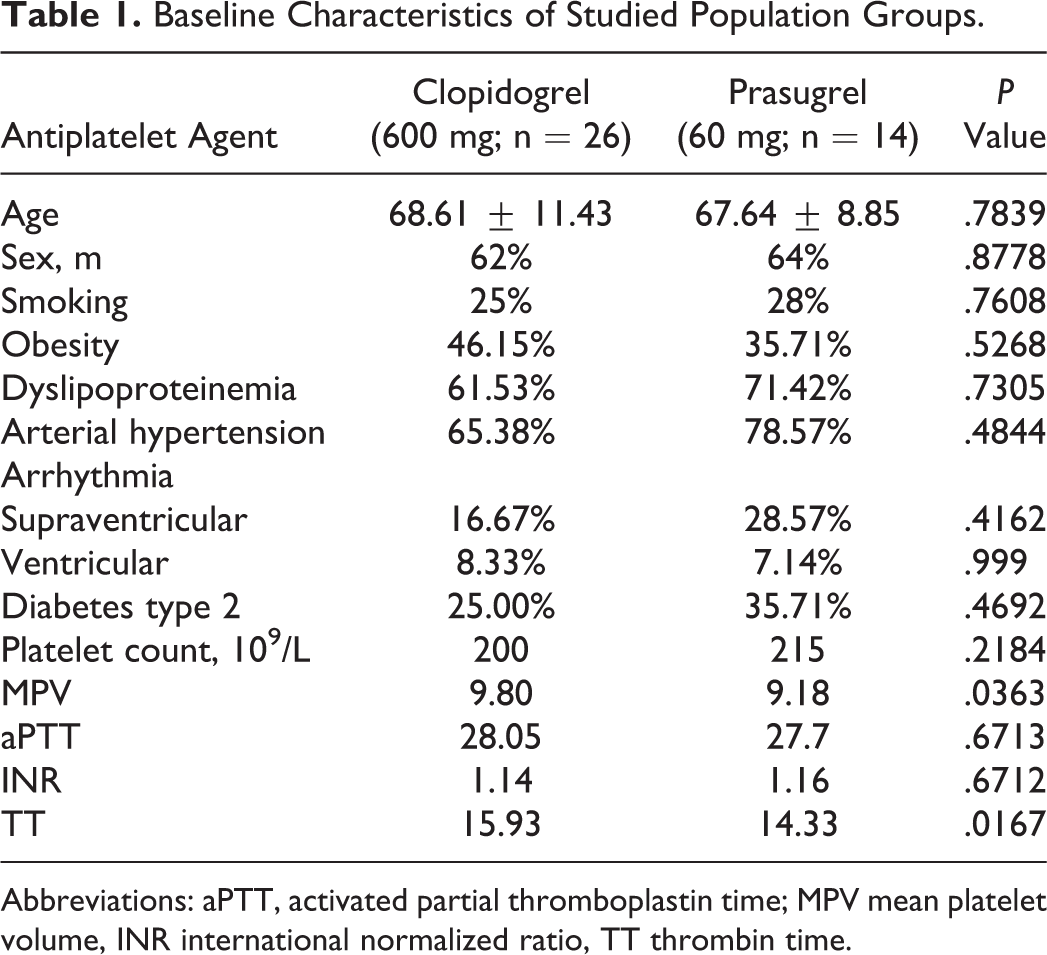
Abbreviations: aPTT, activated partial thromboplastin time; MPV mean platelet volume, INR international normalized ratio, TT thrombin time.
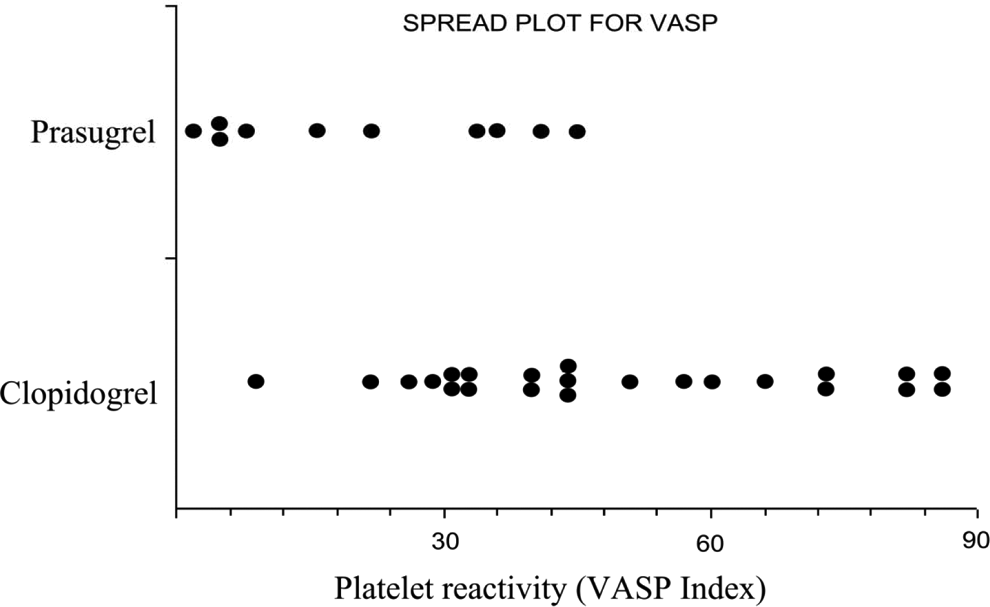
Spread plot for VASP index on ADP-inhibitor treatment. Response measured by platelet reactivity (VASP index in %) for clopidogrel and prasugrel group. VASP indicates vasodilator-stimulated phosphoprotein; ADP, adenosine diphosphate.
Discussion
Our study discusses relevance of VASP-P flow cytometric method in monitoring of the efficacy and side effects of antiplatelet treatment in post-PCI patients with STEMI. Patients were divided into 2 groups, both treated with ADP inhibitors routinely used in clinical practice (clopidogrel or prasugrel).Safety profile of both treatment regimens (clopidogrel + aspirin and prasugrel + aspirin) showed to be feasible. No bleeding episodes were observed. Prasugrel was shown to increase noncoronary artery bypass graft-related bleedings in patients with ACS undergoing PCI. Recent studies have suggested that a VASP index <16% after P2Y12-ADP blockers LD (loading dose) was predictive of nonrelated major bleedings. 18 In our study, 4 patients in the prasugrel group responded to treatment with such intensity. No episodes of major bleeding were recorded. On the other hand in the clopidogrel group, only 1 patient exhibited such value also without record of bleeding. Overall, 6 patients experienced MACEs while on treatment. Of these 6 patients, 4 would be classified as nonresponders by VASP-P assay, yet only 2 patients would be revealed based on LTA measures. This fact suggests positive predictive value of VASP-P assay in targeting the adverse effects of treatment and importance of the tailored ADP inhibitor treatment. Studies report that further laboratory investigations in post-PCI patients on clopidogrel treatment revealed suboptimal response in 15% to 40%. 9,10 Our study displays 100% response rate in prasugrel-treated patients, while in the clopidogrel group response varies from 62% to 73%, depending on chosen VASP index benchmark (PRI = 60 or 50, respectively) (displayed in Table 2). Our data are in accordance with major published studies. It is necessary to note that PR changes overtime. The PR in acute phase of ACSs is higher and decreases afterward. These conclusions were also confirmed in our study. In the follow-up period of 30 days, we noticed slight increase in PR (from 44 up to 45.5 in the clopidogrel group and from 19 up to 25 in the prasugrel group). However, the percentage of poor responders in the clopidogrel group declined in a nonsignificant manner from 27% to 38% to 14%, in correspondence with study by Campo et al (35% to 13%). 19 High on-treatment PR is a known risk factor for occurrence of MACEs, subacute stent thrombosis, and periprocedural myonecrosis during PCI. 6 Although our data are not extensive, we recorded 3-fold higher increase in prevalence of MACEs in group of patients with high PR, which is in concordance with published major studies. Prasugrel induces more potent platelet inhibition, potentially exposing patients to higher bleeding risk. 20 In our prasugrel-treated group, we recorded significantly stronger inhibition of platelet response to ADP compared to patients on clopidogrel (21.50 vs 49.87, respectively; P = .0012). We did not notice any major bleeding episodes, yet data on minor bleeding episodes were not recorded.
Platelet Reactivity (PR) and Clinical Features of Patients Treated With Clopidogrel and Prasugrel.
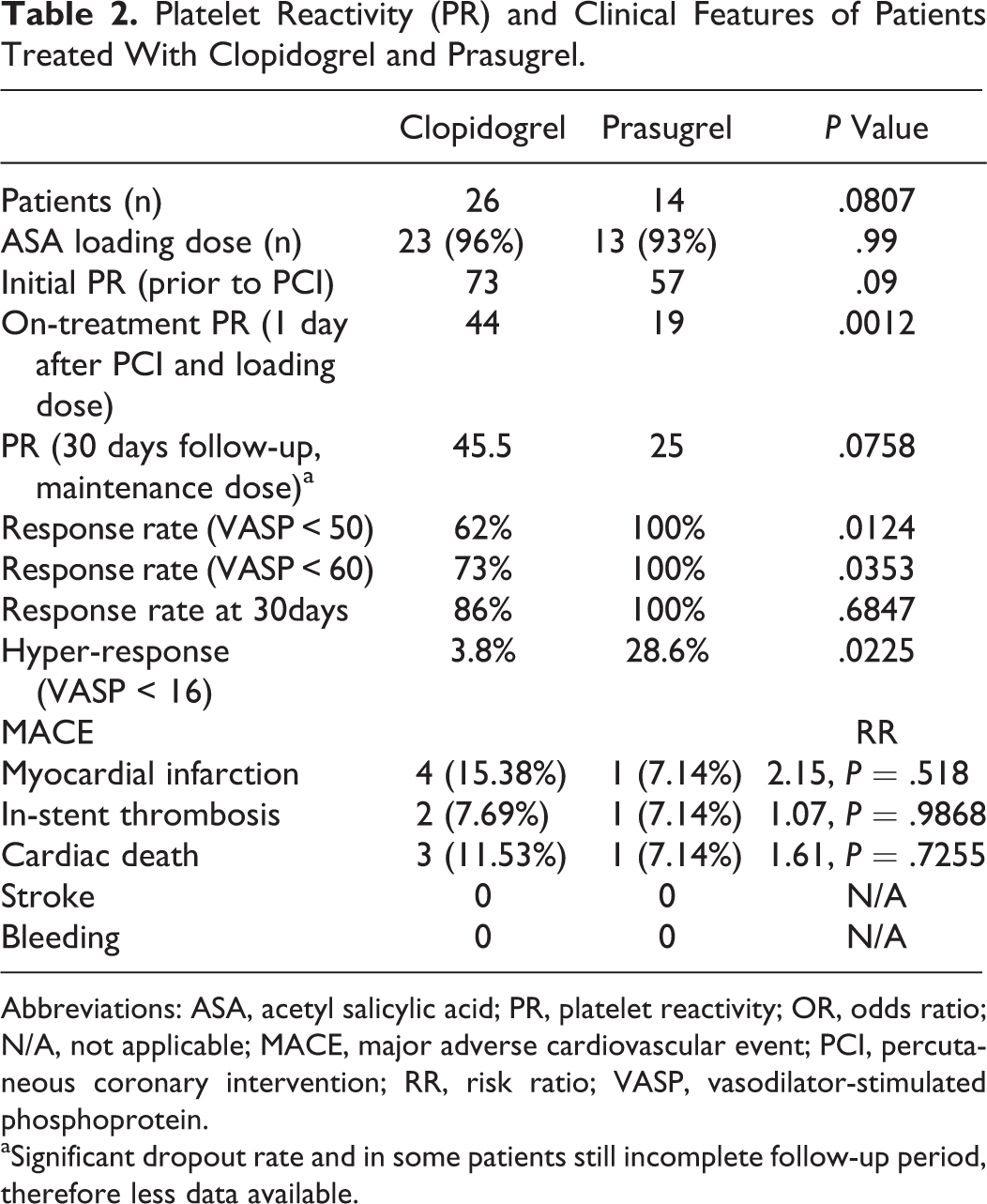
Abbreviations: ASA, acetyl salicylic acid; PR, platelet reactivity; OR, odds ratio; N/A, not applicable; MACE, major adverse cardiovascular event; PCI, percutaneous coronary intervention; RR, risk ratio; VASP, vasodilator-stimulated phosphoprotein.
aSignificant dropout rate and in some patients still incomplete follow-up period, therefore less data available.
Among both groups, only 2 (7%) patients with VASP-PRI value in therapeutic range had MACEs. Such result hints at VASP-P assay as a relevant method for guiding and maintaining ADP inhibitor dosage. Limitations of our study include scarce relevance of clinical end point data due to the rather small group of selected patients. In comparison to the study by Campo et al, 21 our recorded incidence of in-stent thrombosis (7.5%) is high which could be attributed to “small-study effect” as well as severe stenosis of coronary vessels and concurrent polymorbidities in these patients. Cumulative incidence of death was 8.3% during 6-month follow-up, which is comparable to other study outcomes.
According to our results, the VASP-P flow cytometric assay can be recommended for monitoring response to antiplatelet therapy and tailoring treatment in routine clinical practice. In comparison with LTA, VASP-P assay is more valuable for monitoring treatment with ADP inhibitors since it reflects specific activity of P2Y12 receptor. 15 In addition, VASP-P also gives stable results for more than 24 hours after blood sampling (in contrast to 2 hours for LTA) while maintaining advantages in specificity and less sample manipulation. The VASP-P results strongly correlate with LTA methods. Challenge remains in implementing standards to guidance of the effective tailored treatment with individual approach.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by project APVV 0222-11 and Research project of Slovak Society of Cardiology 2012-2015.