Abstract
Generally, recurrent spontaneous abortions (RSAs) have no identifiable cause; yet, vascular alterations during pregnancy may be associated with pregnancy loss. Therefore, we evaluated the association between thrombophilic mutations and RSAs. This case–control study was conducted in 112 patients who had RSAs and 98 health control women. Genomic DNA was extracted from whole blood, and polymorphism genotyping was conducted using polymerase chain reaction. The following 6 genetic variants were analyzed: factor V Leiden, prothrombin mutation, methylenetetrahydrofolate reductase C677T and A1298C, plasminogen activator inhibitor type 1 (4G>5G), and factor XIII G103T (V34L). No correlations were found in any of the investigated polymorphisms. Moreover, 35.0% of cases and 25.5% of controls had at least 2 mutations in combination, and 4.8% of cases and 5.1% of controls had 3, but these combinations were not associated with additional risk. In conclusion, we found no association between the polymorphisms studied and the occurrence of RSAs.
Introduction
Recurrent spontaneous abortions (RSAs) have historically been defined as 3 or more consecutive pregnancy losses. More recently, the accepted clinical definition of RSAs has been recognized as 2 consecutive pregnancy losses. 1,2 This condition affects 0.5% to 2% of couples and can be associated with certain fetal and maternal factors, such as anatomical, hormonal, immunological, and chromosomal abnormalities. 3,4 However, its cause remains unknown in 50% to 60% of cases, 4 thereby making it a frustrating condition for both physicians and patients.
Some evidence has indicated that inherited thrombophilia could influence pregnancy loss at any stage. In addition to thrombotic events in the placenta that would clearly explain the fetal losses, 5 some scholars have suggested that a “nonthrombotic” explanation involving the thrombotic and fibrinolytic system could justify losses at the beginning of gestation. 6 –8 However, the studies found in the literature to date do not unequivocally prove these purported associations.
Thus, there are controversies between studies from all parts of the world, involving genetic variants and its association with RSAs. In Brazil, we found few studies on this subject and none involving the proposed panel of genetic variants.
In this study, we evaluated the possible association between the occurrence of early RSAs and the following gene polymorphisms, either alone or in combination: factor V Leiden ([FVL] G1691A), prothrombin mutation ([PTM] G20210A), methylenetetrahydrofolate reductase (MTHFR) C677T and A1298C, plasminogen activator inhibitor type 1 ([PAI-1] 4G>5G), and factor XIII G103T ([FXIII] V34L).
Materials and Methods
Participant Recruitment and Routine Examinations
This case–control study was approved by the ethics committee of Universidade Federal de São Paulo (UNIFESP) and Colsan. For the extraction of genomic DNA, a total of 210 blood samples drawn from 112 participants in the case group and from 98 participants in the control group were included in the study.
Recruitment for the participants in the study group took place from 2009 to 2012 at the Miscarriage Outpatient Clinic in the Obstetrics Department of UNIFESP in Sao Paulo, Brazil. Participants in the study group included nulliparous women with 3 or more idiopathic miscarriages early in pregnancy (≤12 weeks). All possible participants underwent examinations and tests as part of the research protocol to exclude the known causes of abortion. These procedures included hysteroscopy, hysterosalpingography, transvaginal ultrasound, parental karyotypes, investigation of luteal phase insufficiency (ie, repeated serum progesterone concentration measurements and endometrial biopsy), measurement of prolactin levels, glycemic curve, measurement of thyroid hormone levels, antibody screen for toxoplasmosis, tests to identify infections by cytomegalovirus, rubella, HIV, Group B streptococci, Chlamydia trachomatis, hepatitis B and C, investigation of bacterial vaginosis, and measurement of antiphospholipid antibodies (ie, anticardiolipin antibodies and lupus anticoagulants).
The control group was selected within the same period of time from the Colsan blood bank. Control group and study participants were female blood donors who had at least 2 full-term pregnancies without any obstetric complications. All pregnancies among the women in both groups (ie, the study group and the control group) were naturally conceived. The study and control groups were matched for skin color (ie, white and nonwhite participants).
For the study group, we included only nulliparous women with at least 3 idiopathic miscarriages. In addition, the group was composed predominantly of young women (ie, mean age 30.3 years), thereby decreasing the possibility that the sequence of miscarriages is due to advanced maternal age. For the control group, we chose to include blood donors who had no history of miscarriage and had a history of at least 2 full-term pregnancies. The mean age of the control group was significantly higher (ie, 40.2 years), which decreases the probability of any change in obstetric history. Thus, we believe that we have minimized the possibility of selection bias. The determination of and division based on skin color also present a difficult issue. The Brazilian population is known for extensive miscegenation. However, because the mutations under investigation are more prevalent in caucasian populations, we compiled data for all samples and the impact of ethnicity was studied, including ethnicity as a covariate (ie, whites and nonwhites samples).
All patients who met the inclusion criteria and agreed to participate signed a consent form and were sent to complete the form for the collection of demographic data and to provide a peripheral blood sample.
Search for Polymorphisms
DNA was extracted from the whole blood samples collected from all participants (ie, both study and control participants) using the DNA extraction QIAamp DNA Blood Mini kit (Qiagen Inc., Valencia, CA, USA) and by following the recommendations contained in the manufacturer’s manual.
The measurements of FVL, PTM, MTHFR C677T, MTHFR A1298C, and FXIII V34L were performed via real-time polymerase chain reaction (PCR) using commercial kits (Applied Biosystems, Foster City, CA, USA) designed for each polymorphism. 10 The tests were carried out in the thermocycler of a 7500 Real-Time PCR System (Applied Biosystems, Foster City, CA, USA). The PAI-1 4G>5G polymorphism was measured using the automated DNA sequencing technique. 9 The result was analyzed using the CLC Sequence Viewer software (Qiagen Inc., Valencia, CA, USA).
Statistical Analyses
The statistical analysis involved a comparison of the distribution of heterozygous and/or homozygous genotypes with the normal wild genotype for each alteration using the dominant model for the FVL and PTM polymorphisms and the dominant and codominant models for the MTHFR, PAI-1, and FXIII polymorphisms.
Chi-square, univariate, and multivariate logistic regression tests were used to evaluate the differences between groups. P values of <.05 were considered statistically significant. The odds ratio (OR) was used as a measure of the strength of the associations. The analyses were performed using the statistical package Minitab 16.1.2 for Windows (Minitab Inc., State College, PA, USA).
Results
We analyzed a total of 210 blood samples, that is, 112 participants in the study group and 98 participants in the control group. Because of problems in the quantification of the extracted DNA, we discarded the following number of samples when we analyzed the risk of RSAs associated with each polymorphism: 29 samples from FVL, 7 samples from PTM, 1 sample from MTHFR C677T, 1 sample from MTHFR A1298C, 7 samples from PAI-1 4G>5G, and 1 sample from FXIII V34L. To analyze the risk associated with a combination of mutations, data for all mutations were available for 83 samples from the study group and 98 samples from the control group. All polymorphisms for both groups were found to be in Hardy-Weinberg equilibrium. 11
The patients with recurrent miscarriages were significantly younger than those in the control group (ie, median 31 vs 42, respectively; P < .001). No statistically significant difference was observed (P = 0.11) in the distribution of skin color among the patients and controls (70.5% white and 29.5% nonwhite vs 60.2% white and 39.8% nonwhite, respectively). The mean number of miscarriage in case group was 3.4 (range 3-6). Table 1 shows a comparison of all genotype frequencies among the groups.
Comparison of Genotype Frequencies.
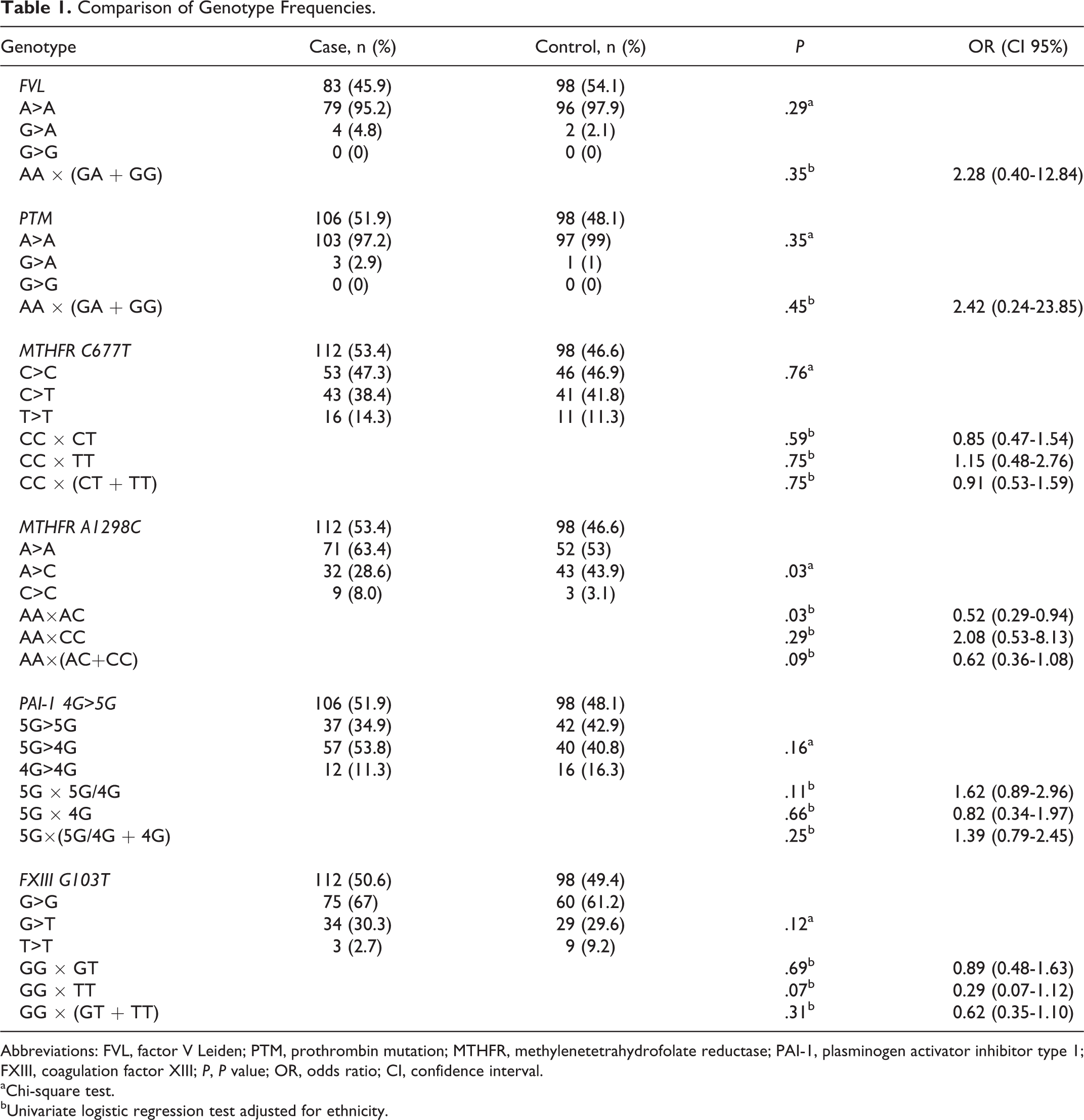
Abbreviations: FVL, factor V Leiden; PTM, prothrombin mutation; MTHFR, methylenetetrahydrofolate reductase; PAI-1, plasminogen activator inhibitor type 1; FXIII, coagulation factor XIII; P, P value; OR, odds ratio; CI, confidence interval.
aChi-square test.
bUnivariate logistic regression test adjusted for ethnicity.
When comparing FVL, we evaluated the samples from 83 patients in the study group and 98 participants in the control group. As for the PTM investigation, we evaluated the samples from 106 patients and 98 controls. We had planned to use the dominant model for the comparative analyses of FVL and PTM, but we did not find any samples with the homozygous mutated genotype for these mutations; therefore, the analyses focused exclusively on the heterozygous genotype, and the observed differences between the groups were not significant.
For the C677T and A1298C polymorphisms of the MTHFR gene, we evaluated a total of 112 blood samples from patients in the study group and 98 samples from participants in the control group. For data analysis, we used the dominant and codominant models. The observed differences in these polymorphisms among the patients and the control participants were not significant except for the heterozygous genotype for the MTHFR A1298C gene, which appears to have a protective effect.
For the 4G>5G polymorphism of the PAI-1 gene, we evaluated a total of 106 samples from patients in the study group and 98 samples from participants in the control group. As for the polymorphism V34L (G103T) of the FXIII gene, we evaluated 112 samples from patients in the study group and 98 samples from the healthy control participants. For the data analysis, we also used the dominant and codominant models, and the observed differences between the groups were not significant.
Moreover, when we performed multivariate logistic regression in order to look for any interactions between the 6 polymorphisms studied as risk factors (predictors) for RSAs and adjusted this model for the influence of ethnicity, none was significantly associated with RSA. Even when we analyzed allelic frequencies for each polymorphism, the observed differences remained nonsignificant (Table 2).
Comparison of Allelic Frequencies.
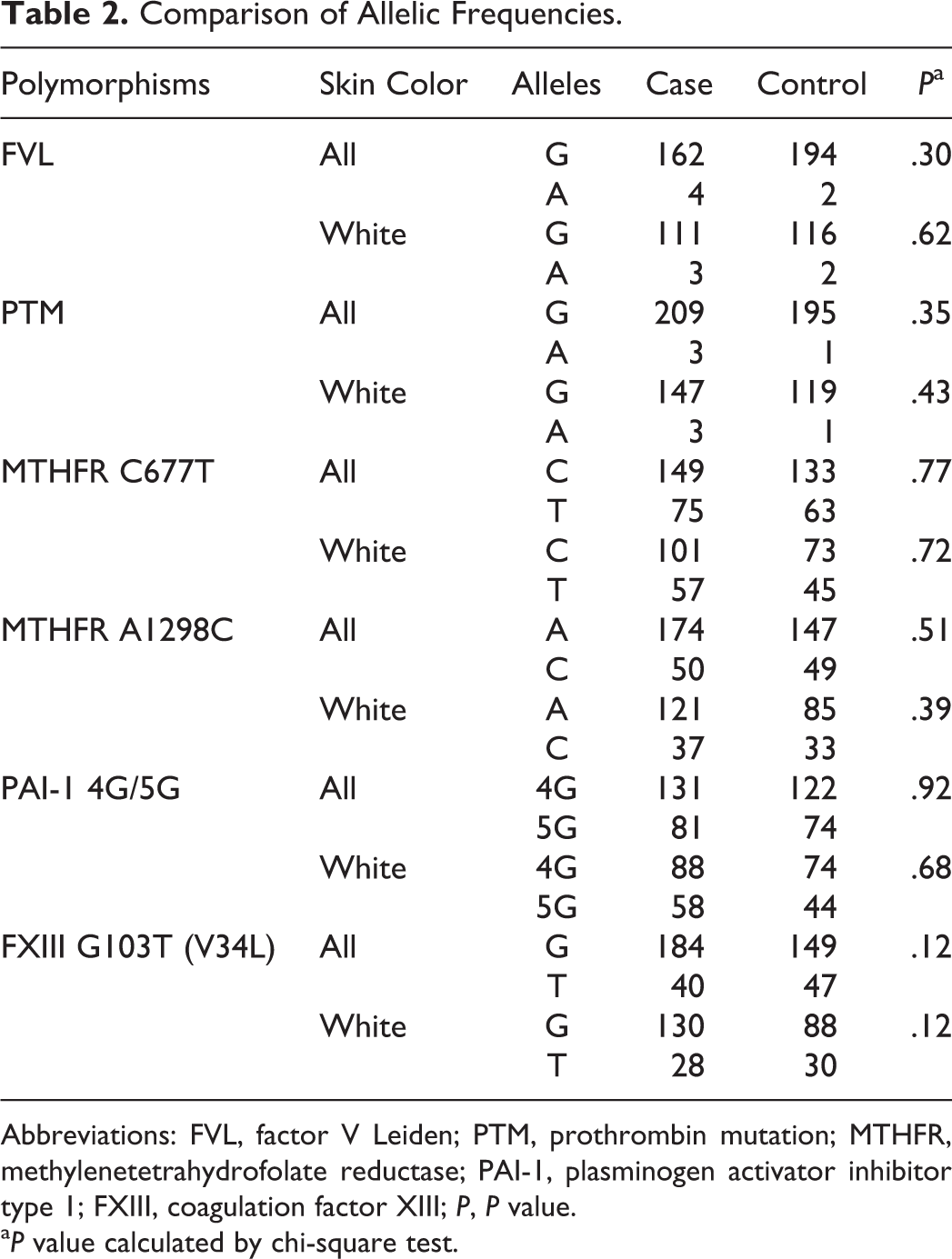
Abbreviations: FVL, factor V Leiden; PTM, prothrombin mutation; MTHFR, methylenetetrahydrofolate reductase; PAI-1, plasminogen activator inhibitor type 1; FXIII, coagulation factor XIII; P, P value.
a P value calculated by chi-square test.
Data for all of the studied polymorphisms were available for 83 patients of the study group and 98 participants of the control group. We considered the risk for RSA associated with the heterozygous or homozygous genotype of FVL, PTM, PAI-1, and FXIII and only the homozygous genotype for both mutations of the MTHFR gene. According to our results, the combined occurrence of these alterations did not increase the RSA risk (Table 3). Even when the total number of mutated alleles was compared (ie, considering the heterozygous form as 1 mutation and the homozygous form as 2 mutations), the difference between groups remained nonsignificant (Table 4).
Comparison of Mutations in Combination.
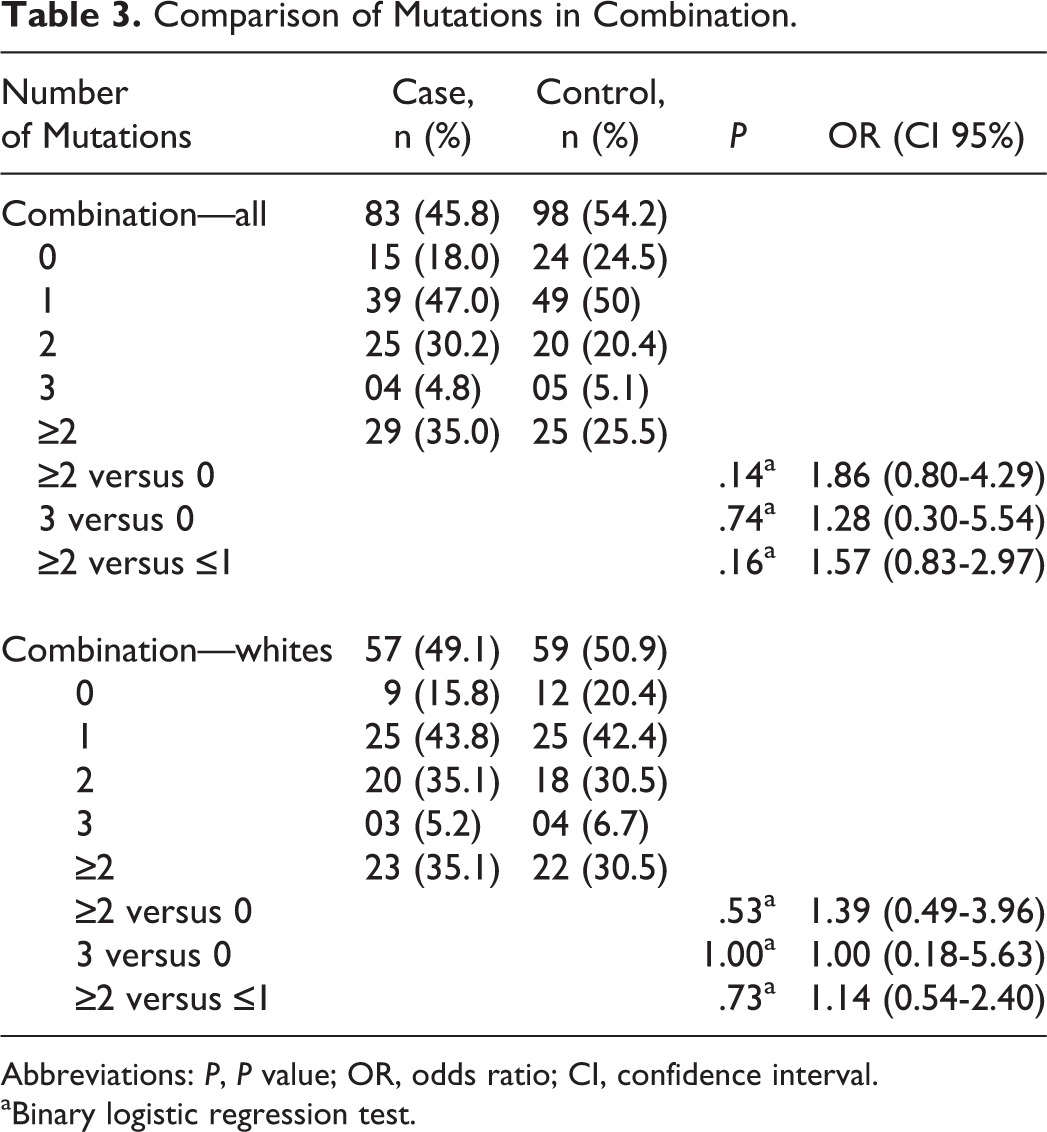
Abbreviations: P, P value; OR, odds ratio; CI, confidence interval.
aBinary logistic regression test.
Comparison of Total Alleles in ≥2 Combination Samples.
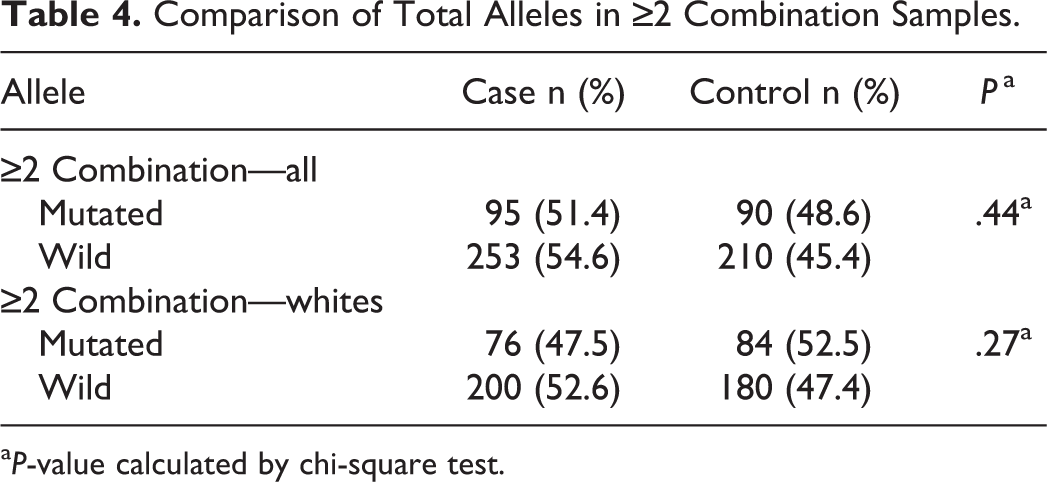
a P-value calculated by chi-square test.
Regarding the possible combination of mutations in the MTHFR gene, we found only 1 participant with the homozygous genotype for both mutations (ie, 4 mutated alleles) and 3 participants with a combination of 1 heterozygous genotype for one gene and 1 homozygous genotype for another gene (ie, 3 mutated alleles). In both cases, the participants were part of the study group. We found the heterozygous combination in 10.7% (12 of 112) of the patients in the case group and in 10.5% (12 of 98) of participants in the control group, and observed no statistical significance in this difference (P = .81).
Regarding the combination of polymorphisms involving the fibrinolytic system (ie, PAI-1 and FXIII), we found that 24 of 106 participants in the study group and 25 of 98 participants in the control group had homozygous (ie, PAI-1 4G/4G and FXIII TT) or heterozygous combination for both polymorphisms or homozygous and heterozygous genotypes for either polymorphism; again, no statistical significance was observed (P = .63).
Discussion
In this study, we evaluated the possible association between 6 thrombophilic alterations, both alone and in combination, and the occurrence of early RSA. Despite the fact that thrombophilic diseases are gaining attention regarding their role in obstetric problems, we found no association.
Considering the hypothesis that thrombophilia could be the cause of RSAs, the main pathophysiology that justifies the repetition of such episodes would be the occurrence of thrombotic events in the placenta, 5 which implies the involvement of thrombophilia from the 10th week of gestation through delivery (ie, the fetal period). However, even 2 days postimplantation, lacunar spaces develop in the trophoblast, thereby forming an intercommunicating network with the endometrium and supporting the growth and development of the embryo through the maternal blood supply. 12 Furthermore, some scholars have postulated a “nonthrombotic” theory 6 –8 based on experimental evidence indicating that the coagulation system influences the initial stage of gestation. 6 Thus, we might suppose that the inherited thrombophilia can also influence the early stages of pregnancy.
A comparison of the frequency of FVL and PTM mutations found in our study with that of other Brazilian studies 13,14 reveals that our findings were similar with exception to a higher prevalence of FVL in the study group of Souza et al. 13 Our study showed only conflicting results regarding the risk of RSA in relation to the occurrence of FVL; indeed, the other study showed a positive association between RSA and the occurrence of FVL. 13 This can be explained by the difference in the inclusion criteria regarding the time at which the abortion occurred (ie, ≤12 weeks in our study vs ≤20 weeks in the study group of Souza et al 13 ). Our result is also consistent with older and more recent international publications. 15 –17 However, 2 meta-analyses 18,19 showed a significant association between FVL and early RSA, but the confidence interval was very close to 1. Also note that the studies were very heterogeneous in terms of the composition of the study population and the inclusion criteria, including the time at which the miscarriage took place.
The literature highlights the relationship between the FVL mutation with pregnancy loss during the fetal period, 17,18,20,21 including a meta-analysis study. 22 However, we did not demonstrate an association between FVL and RSA. The inclusion of patients with only early RSA possibly limited our ability to find this association in our study.
A systematic review 19 reported an OR of 2.71 (95% confidence interval [CI] of 1.32-5.58) for early pregnancy loss in patients homozygous for FVL. Yet, in our study, we did not find any patient with homozygous FVL mutation.
The results in the literature for the association between RSA and PTM are also controversial. Some authors found no association between this mutation and pregnancy loss, 21 –23 while others have shown a higher prevalence of the mutation in the group with RSA. 24 –26 Moreover, as previously mentioned, a meta-analysis 19 showed a possible association between PTM and early RSA, but the confidence interval was very close to 1.
Recognizably, hyperhomocysteinemia is a risk factor associated with the development of thrombosis. 27 Individuals with homozygous and heterozygous genotypes for the C677T polymorphism demonstrate 30% and 65% of the MTHFR activity, respectively, which can lead to the development of hyperhomocysteinemia. However, even in patients with the A1298C mutation, which leads to reduced MTHFR enzyme activity, no significant hyperhomocysteinemia was seen. 28
For the MTHFR C677T mutation, 38.4% of the study group in this research and 41.8% of the control group had the heterozygous genotype. The prevalence of the homozygous genotype was slightly higher in the patient group (ie, 14.3% vs 11.3% in the control group). Similar to our results, other studies, including 3 meta-analyzes, 18,29,30 have not shown this mutation as a risk factor for RSA. 17,27,31 Some international publications have published results that differ from our results, 28,32 –34 including 2 Indian studies 35,36 that cited the heterozygous and homozygous genotypes as risk factors for RSA. A significant association with early RSA, even for the heterozygous genotype, is seemingly explained by the local prevalence of this mutation in India and is also justified by nutritional factors that influence homocysteine metabolism in the Indian population.
A few studies in the literature have evaluated the A1298C mutation, and our results agree with almost all of them. 32,37 –41 An Asian study 27 also did not associate the homozygous mutation solely with the subgroup of patients with early RSA (ie, at 5-10 weeks’ gestation), but they did find a higher and significant prevalence for the homozygous genotype in those with early to late RSA (ie, at 11-30 weeks’ gestation). On the other hand, Iranian women 34 demonstrated a greater risk of RSA prior to 20 weeks’ gestation when carrying the homozygous or heterozygous mutation.
The PAI-1 mutation could favor RSAs by promoting fibrin deposition in early placental circulation or through limitation of trophoblastic invasion or both. 40 Yet, interestingly, we found a higher percentage of 4G homozygote genotype in the control group than the study group (ie, 16.3% vs 11.3%, respectively). Thus, our results differ from certain studies in the literature, particularly those studies that report an association between this mutation and RSA, with this genotype posing highest risk. 32,33,40,42 –44 However, our results do not differ from some other publications. 45 –48 More currently, in a prospective cohort study, 49 the PAI-1 polymorphism was not associated with obstetrical complications, including fetal loss.
The FXIII G103T polymorphism (ie, V34L) is the most common mutation to affect this gene. It does not lead to changes in plasma FXIII, but it modifies its activity. Its activation is increased by a factor of 2 to 3 in the presence of thrombin. 34 This response leads to a dysfunction in the homeostasis since it affects the stability of the clot. 34 We did not see a significantly high presence of the FXIII mutation in women with early RSA. In fact, we found a higher frequency of the mutated allele in the control group compared to the study group, that is, 38.8% and 33%, respectively. Another Brazilian study 50 and 3 Iranian studies 34,43,51 are in agreement with our results. However, in contrast with our results, in a larger study of 550 samples, an American group 32,40 found that this mutant allele is a risk factor for RSA occurring at less than 20 weeks’ gestation. The influence of this polymorphism on early RSA (ie, 5-12 weeks gestation) has also been described. 33 On the other hand, a meta-analysis 39 highlighted the low impact of fibrinolytic defects on RSA.
Assuming that the presence of more than 1 thrombophilic mutation in the same patient would have an additive effect on the risk of miscarriage, some publications have evaluated certain alterations in combination. 32,38,52 Thus, we considered FVL, PTM, PAI-1, and FXIII in the form of heterozygous and homozygous genotypes and both MTHFR mutations only in the homozygous form as potential risk factors for RSA, and we found that 35% of the study population (ie, 29 of 83) and 25.5% of the control population (ie, 25 of 98) have 2 or more mutations combined. However, only 4.8% of the study population (ie, 4 of 83) and 5.1% of the control population (ie, 5 of 98) had 3 mutations combined. These differences were not significant, even in the analysis of the caucasian subgroup (Table 3). In fact, we did not find significant differences even when evaluating the number of mutated alleles in the group with 2 or more mutations (Table 4). However, the small sample size clearly limits the interpretation of this evidence. Other authors also evaluated some specific combinations but found no evidence of an increased risk for RSA. 43,45,49,50,52
Despite the possibility that inherited thrombophilia could pose a risk for RSA, published studies still differ in terms of their results for the most frequent and/or known mutations. Part of this discrepancy can be explained by the varying ethnic and geographic prevalence of these mutations and the inclusion criteria used for the studies, particularly the definition of RSA and the lack of consensus on the division of losses according to the gestational period.
Considering the small differences in frequency observed between wild genotypes and genotypes with mutation in our population, the number of samples included in this study may have limited the statistical power to find a real difference. However, a larger sample is naturally difficult to achieve because the prevalence of inherited thrombophilia is small in general population.
Persistent doubts about the real influence of inherited thrombophilia on episodes of RSA have led to the following recommendations by the American College of Chest Physicians 53 : (1) doctors should not search for inherited thrombophilia in women with obstetric complications and (2) they should not use prophylactic antithrombotic therapy in women with inherited thrombophilia and a history of obstetric complications.
Multicenter studies are required in order to achieve a larger sample and to reduce the heterogeneity of the studies, which would thus help to identify the real impact of inherited thrombophilia on pregnancy loss. Nevertheless, negative results must be considered and may have substantial value, especially when performing meta-analyses.
Conclusions
In this study, we found no association between the occurrence of RSA and the gene polymorphisms of PAI-1 (4G/5G), FXIII (V34L), and MTHFR (C677T and A1298C), and the gene mutations of FV (G1691A) and prothrombin (G20210A), either alone or in combination.
Footnotes
Acknowledgments
We would like to thank Colsan Associação Beneficente de Coleta de Sangue by this research partnership.
Authors’ Note
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
